INTRODUCTION
Polycystic ovary syndrome (PCOS) is the most widespread form of female infertility and affects an estimated 10–20% of women of reproductive age [1]. A 1990 National Institutes of Health (NIH) conference defined PCOS as hyperandrogenism and/or hyperandrogenemia (HA) with oligoanovulation, excluding other endocrine diseases [2]. In 2003, the Rotterdam consensus expanded the diagnostic criteria to include at least two of the following features 1) clinical hyperandrogenism 2) oligo-anovulation and 3) polycystic ovaries (PCO). Recently, an NIH expert panel suggested the broader Rotterdam criteria is more suitable for the diagnosis PCOS [1].
Although the etiology of PCOS remains largely unknown, it is believed to be a complex polygenic disorder heavily influenced by environment risk factors (for example, high-fat diet) [3, 4]. Previous evidence has suggested that the disturbed cycle and homocysteine-methionine cycle, two closely intertwined processes folate cycle in ovarian dysfunction. Deficiency or excess of folates inhibits ovulation in immature superovulated rats [5]. Folate deprived monkeys have degenerated Graffian follicles and an increase in atretic and cystic follicles [6]. Clinical evidence has also suggested that there are homocysteine levels in PCOS cases and homocysteine levels returned to normal levels following folic acid supplementation [7–9]. This makes Methylenetetrahydrofolate reductase (MTHFR) an essential gene to investigate in PCOS, as it has important roles in the processing of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, a reaction required to convert homocysteine to methionine [10–12]. Some of its variants have show decreased efficiency of the folate/homocysteine pathway and have been linked to the susceptibility of neural tube defects, Alzheimer’s disease, colon cancer, acute leukemia, and PCOS [13].
Experimental data have suggested that the functional substitution C677T (alanine substituted by valine, rs1801133) in the MTHFR gene reduces the activity of the folate pathway by 50% [10, 14, 15]. However, clinical studies thus far have found conflicting results regarding the association of MTHFR and PCOS [16–29]. To clarify the in-conflict findings reported so far as well as heterogeneity and publication bias that exists between studies, we have conducted a meta-analysis of genetic association studies of the MTHFR C677T polymorphism to assess its effect on the risk of PCOS.
RESULTS
Study characteristics
The search yielded a combined 272 references. Study selection process was shown in Figure 1. The final meta-analysis included a total of 14 articles of 16 data sets [16–29]. The 16 data sets included 1628 controls and 1478 PCOS cases. The detailed characteristics of included studies are shown in Table 1. Of the PCOS cases, 439 were Asian, 517 were Caucasian, 522 were Middle Eastern.
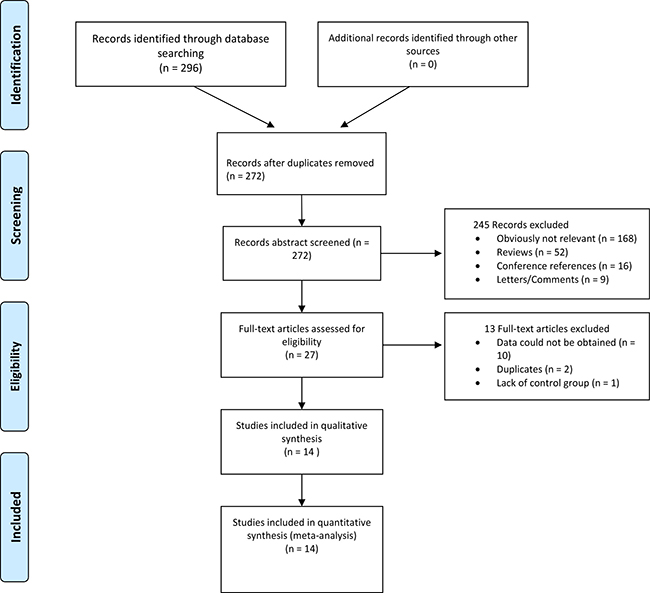
Figure 1: Flowchart of study selection.
Table 1: Characteristics of included studies
Meta-analysis results
Overall, there was no evidence of an association between the T allele variant and increased risks of PCOS when all data sets were pooled together. The per-allele OR of Pro using the random effects models was 1.08 [95% CI: 0.96–1.21; P(Z) = 0.000; P(Q) = 0.001; Figure 2]. The main results of the meta-analysis were listed in Table 2.
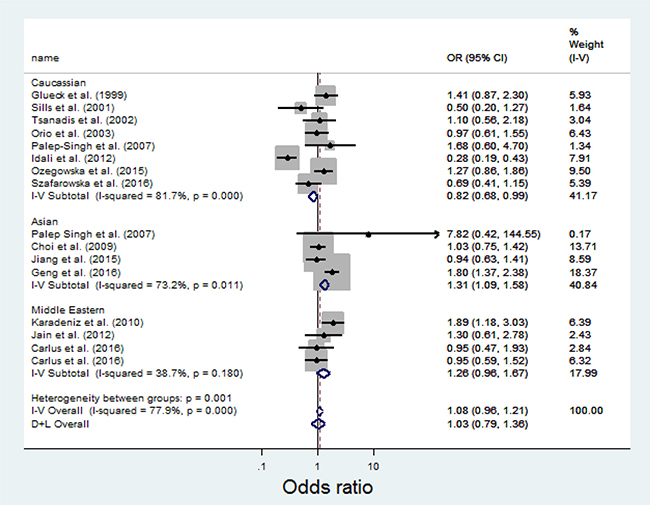
Figure 2: Meta-analysis of the association between MTHFR C667T and PCOS.
Table 2: Meta analysis of the MTHFR C667T polymorphism and the risks of PCOS
Total/Subgroup |
Number of data sets |
Number of cases/controls |
C Allele |
||
|---|---|---|---|---|---|
OR (95% CI) |
P(Z) |
P(Q) |
|||
Total |
16 |
2980/7054 |
1.08 (0.96–1.21) |
0.229 |
0.000 |
Caucasian |
8 |
517/482 |
0.82 (0.68–0.99) |
0.040 |
0.000 |
Asian |
4 |
522/721 |
1.31 (1.09–1.58) |
0.004 |
0.011 |
MiddleEastern |
4 |
439/425 |
1.26 (0.96–1.67) |
0.101 |
0.180 |
In the stratified analysis by ethnicity, the T allele significantly increases risks for the Asian [OR = 1.31; 95% CI: 1.09–1.58] population (See Figure 2). No significant associations were detected for the Middle Eastern [OR = 1.26; 95% CI: 0.96–1.67]. The T allele was found to be protective in the Caucasian population [OR = 0.82; 95% CI: 0.68–0.99].
In the dominant model, The CT + TT genotypes are significantly associated with increased risks of PCOS in the Asian [OR = 1.37; 95% CI: 1.02–1.83] and Middle Eastern [OR = 1.48; 95% CI: 1.06–2.07] populations. No significant associations were detected for the Middle Eastern [OR = 0.94; 95% CI: 0.73–1.21].
Sensitivity analysis
Sensitivity analyses using single-study omission demonstrated that this meta-analysis was stable (Figure 3). Statistical significance of the summary ORs was not modified (data not shown). A cumulative meta-analysis (Figure 4) also show that the results of this study are stable.
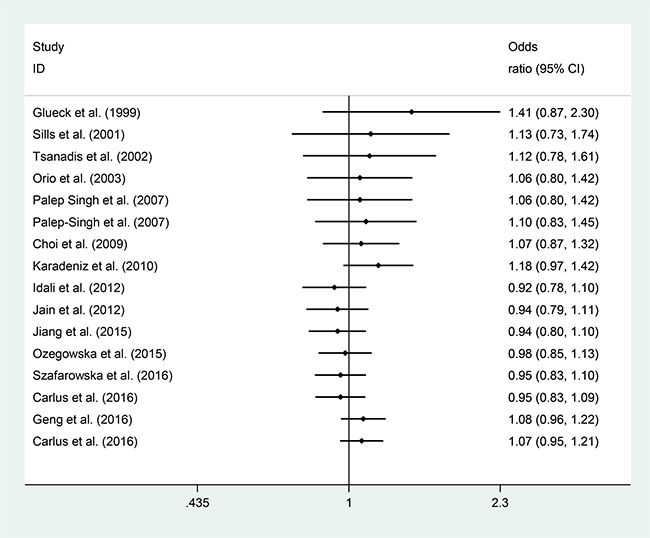
Figure 3: Cumulative meta-analysis of the studies included.
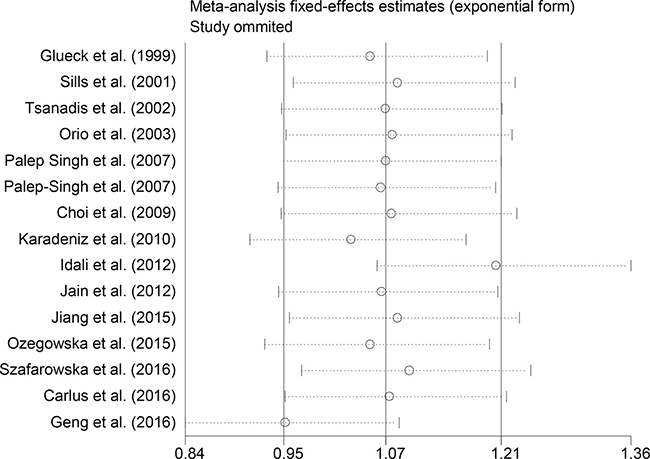
Figure 4: Sensitivity analysis excluding each included study one at a time.
Publication bias
Begger’s and Eggar’s funnel plots were constructed using the standard error and compared against the OR of each study (Figures 5 and 6). The plots do not suggest the existence of publication bias towards positive findings in smaller studies. Thus, this indicates that this meta-analysis is statistically robust.
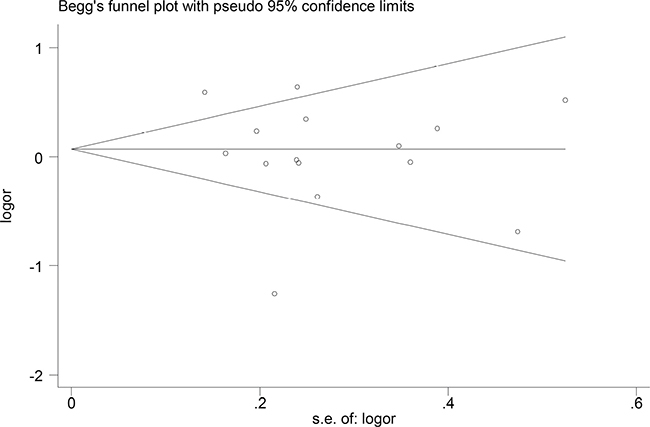
Figure 5: Begg’s funnel plot of the included studies.
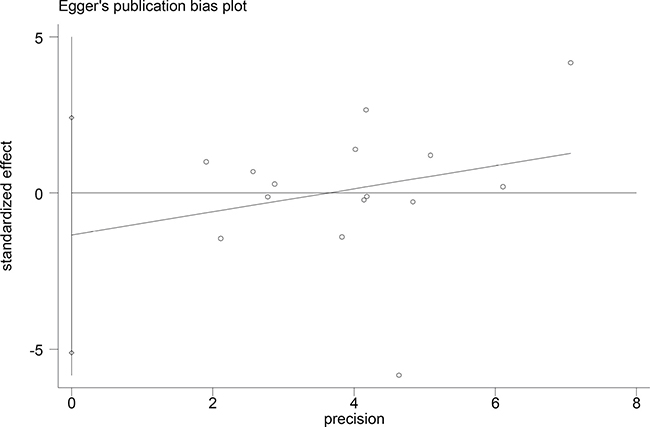
Figure 6: Egger’s plot of the association of the included studies.
DISCUSSION
MTHFR is an enzyme that catalyzes the production of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate that is critical to the remethylation to methionine. Recently, the C677T variant has been linked with increased risks of PCOS, although to inconsistent results [16–29]. Due to small sample sizes, data generated by the above studies lacked the power of independent representation.
Previous meta-analyses analyzing the relationship between MTHFR C667T polymorphism and PCOS have yielded conflicting results. Bagos analyzed six data sets published before 2009 and found no correlation between C667T and the risk of PCOS [30]. However, the credibility of the study should be re-examined as the some of the values of homozygous rare and homozygous common genotypes have been interchanged. Fu and his colleagues performed a meta-analysis in 2014 analyzing 638 cases and 759 controls and reported that C667T as a risk factor for PCOS [31]. More recently, in 2015 Carlus and colleagues studied 960 cases and 1028 controls and found no correlation between C667T and the risks of PCOS [28].
Our up-to-date meta-analysis summarizes the evidence to date regarding the association between the MTHFR C677T variant and PCOS using a total of 1628 controls and 1478 PCOS cases, the largest to date by far. Overall, our results suggest that C677T is not associated with the risk of PCOS.
In our stratified analysis by ethnicity, a strong association was observed for the Asian population [OR: 1.31, 95% CI: 1.09–1.58] population but not for the Middle Eastern [OR = 1.26, 95% CI = 0.96–1.67] populations. Interestingly, risk T allele has protective effects for the Caucasian population [OR = 0.82, 95% CI = 0.68–0.99]. These results indicate that the association of the MTHFR C677T polymorphism and PCOS pathogenesis has deep genetic and possibly environmental background factors. Other factors such as differences in selection bias and matching criteria could also play a role in the difference between ethnic groups. It should also be noted that many studies included have very small study size. This suggests the possibility that the observed differences may be due to chance. Thus, additional studies are required to increase the statistical power and validate the racial difference of the MTHFR C677T polymorphism and PCOS risk.
The preferential publication of studies with positive results is a significant source of bias in many meta-analyses. However, the included studies in our meta-analysis also consist of studies with negative conclusions. Furthermore, our funnel plots do not show asymmetry, suggesting the lack of publication bias in our meta-analysis.
Several limitations should be noted in interpreting our results. We were not able to adjust for potential confounding effects conferred by gender, environmental factors, and lifestyle due to the lack of data. Our results were based on unadjusted estimates - a more precise analysis could be conducted if all raw data were available. The lack of individual health and metabolic data, such as drug history, diet, and body weight, also forbid us from performing a more sensitive analysis.
In conclusion, the pooled results of our meta-analysis indicate that C677T is not associated with the risks of PCOS. However, in the Middle Eastern populations, the T allele is strongly associated with the risks for PCOS while protective in Caucasian populations. Larger association studies with strict selection criteria are required to validate this result.
MATERIALS AND METHODS
Search strategy and inclusion criteria
We searched the literature hosted on PubMed, Web of Science, EMBASE, Ovid, Chinese National Knowledge Databases and WanFang with keywords related to disease (e.g. “Polycystic ovary syndrome,” “PCOS”) and the gene of interest (e.g. “Methylenetetrahydrofolate reductase”, “MTHFR”). Genetic association studies published before Dec 2016 were retrieved. The last search was performed on the Dec 28th, 2016. For each study, we checked their references to identify other relevant publications. No earlier publication date limit was applied. We did not define a minimum number of patients as a criterion for a study’s inclusion in this meta-analysis. The search was conducted without any restrictions on the language used and focused on human studies.
All retrieved study were screened, and all eligible studies included needed to satisfy each point of the following criteria: 1) original papers containing independent trials, 2) case-control or cohort study, 3) confirmation of PCOS according to either the NIH or Rotterdam diagnostic criteria, and 4) genotype distribution information or odds ratio (OR) with its 95% confidence interval (CI) and P value. The major reasons for exclusion of studies were overlapping data, review articles, case-only studies, and insufficient data for analyses.
Data extraction
Data extraction was performed independently by two reviewers using a standard extraction form. All data were checked for internal consistency and disagreements were resolved through thorough discussion between all authors. If there doubts about the result of studies, the corresponding author of the study was calculated. For each study, the following were extracted from each article: first author’s name, publication year, diagnostic criterion, definition and numbers of cases and controls, frequency of genotypes, genotyping method, source of controls, Hardy–Weinberg equilibrium (HWE), age, body-mass index (BMI), and ethnicity. Studies with different ethnic groups within the same study were considered as individual studies for our analyses.
Statistical analysis
The association strength between MTHFR C677T polymorphism and PCOS was assessed by calculating OR with 95% CI. The chi-square (χ2) test was used to evaluate whether there is a significant deviation from HWE among the control subjects of the study. The per-allele OR of risk allele T was compared between cases and controls in each study. The ORs were pooled using both the random-effects model (the DerSimonian and Laird method) and the fixed effects model (the Mantel-Haenszel method) as previously described [32, 33]. The Woolf’s method was used to calculate 95% CI [34]. The results of the random effects model were reported in this article because it takes into consideration the variation between studies. A prespecified stratified analysis was conducted to explain the heterogeneity between each study and to investigate the relationship present in a subgroup. Stratified analysis was performed for ethnicity (Caucasian, Asian, and Middle Eastern).
Heterogeneity across individual studies was examined using Cochran’s χ2 Q test [35]. Q test was also performed to detect the heterogeneity within each subgroup. Publication bias was assessed using linear regression to measure funnel plot asymmetry on the natural logarithm of OR using Egger’s method [36]. All statistical analysis was carried out with Stata Version 13.0 (Stata Corporation, College Station, Texas, USA. All P values were for two-sided analysis. Type I error rate was set at 0.05.
Abbreviations
BMI = body mass index, CI = confidence Interval, HWE = Hardy–Weinberg equilibrium, MTHFR = methyle netetrahydrofolate reductase, PCOS = polycystic ovary syndrome, OR = odds ratio.
Authors’ contributions
Conceived and designed the experiments: LW. Performed the experiments: LW, WX, CW, and YZ. Analyzed the data: MT, CW. Wrote the paper: LW, WX, and XW.
CONFLICTS OF INTEREST
We declare no conflicts of interest.
FUNDING
This work is supported by the Natural Science Foundation of Jiangsu Province in 2015 (BK20151261), Projects For Science and Technology of Chinese Medicine of Jiangsu Province in 2015 (No.YB2015107), and The Science and Technology Development Plan of SuZhou in 2014 (No.SYSD2014006). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
REFERENCES
1. Dumesic DA, Oberfield SE, Stener-Victorin E, Marshall JC, Laven JS, Legro RS. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr Rev. 2015; 36:487–525.
2. Zawadzki JK, Dunaif A. Diagnostic criteria for polycystic ovary syndrome: towards a rational approach. Blackwell Sci. 1992; 377–84.
3. Diamanti-Kandarakis E, Christakou C, Marinakis E. Phenotypes and Enviromental Factors: Their Influence in PCOS. Curr Pharm Des. 2012; 18:270–82.
4. Diamanti-Kandarakis E, Kandarakis H, Legro RS. The Role of Genes and Environment in the Etiology of PCOS. Endocrine. 2006; 30:19–26.
5. Willmott M, Bartosik DB, Romanoff EB. The effect of folic acid on superovulation in the immature rat. J Endocrinol. 1968; 41:439–45.
6. Mohanty D, Das KC. Effect of folate deficiency on the reproductive organs of female rhesus monkeys: a cytomorphological and cytokinetic study. J Nutr. 1982; 112:1565–76.
7. Badawy A, State O, El Gawad SSA, El Aziz OA. Plasma homocysteine and polycystic ovary syndrome: the missed link. Eur J Obstet Gynecol Reprod Biol. 2007; 131:68–72.
8. Kazerooni T, Asadi N, Dehbashi S, Zolghadri J. Effect of folic acid in women with and without insulin resistance who have hyperhomocysteinemic polycystic ovary syndrome. Int J Gynaecol Obstet. 2008; 101:156–60.
9. Bayraktar F, Dereli D, Ozgen AG, Yilmaz C. Plasma homocysteine levels in polycystic ovary syndrome and congenital adrenal hyperplasia. Endocr J. 2004; 51:601–8.
10. Yamada K, Chen Z, Rozen R, Matthews RG. Effects of common polymorphisms on the properties of recombinant human methylenetetrahydrofolate reductase. Proc Natl Acad Sci. 2001; 98:14853–8.
11. Reilly R, McNulty H, Pentieva K, Strain JJ, Ward M. MTHFR 677TT genotype and disease risk: is there a modulating role for B-vitamins? Proc Nutr Soc. 2014; 73:47–56.
12. Frosst P, Blom HJ, Milos R, Goyette P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuvel LP, Rozen R. A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet. 1995; 10:111–3.
13. E. Trimmer E. Methylenetetrahydrofolate Reductase: Biochemical Characterization and Medical Significance. Curr Pharm Des. 2013; 19:2574–93.
14. McQuillan BM, Beilby JP, Nidorf M, Thompson PL, Hung J. Hyperhomocysteinemia but Not the C677T Mutation of Methylenetetrahydrofolate Reductase Is an Independent Risk Determinant of Carotid Wall Thickening. Circulation. 1999; 99.
15. Cortese C, Motti C. MTHFR gene polymorphism, homocysteine and cardiovascular disease. Public Health Nutr. 2001; 4:493–7.
16. Glueck CJ, Wang P, Fontaine RN, Sieve-Smith L, Tracy T, Moore SK. Plasminogen activator inhibitor activity: an independent risk factor for the high miscarriage rate during pregnancy in women with polycystic ovary syndrome. Metabolism. 1999; 48:1589–95.
17. Sills ES, Genton MG, Perloe M, Schattman GL, Bralley JA, Tucker MJ. Plasma homocysteine, fasting insulin, and androgen patterns among women with polycystic ovaries and infertility. J Obstet Gynaecol Res. 2001; 27:163–8.
18. Tsanadis G, Vartholomatos G, Korkontzelos I, Avgoustatos F, Kakosimos G, Sotiriadis A, Tatsioni A, Eleftheriou A, Lolis D. Polycystic ovarian syndrome and thrombophilia. Hum Reprod. 2002; 17:314–9.
19. Orio F, Palomba S, Di Biase S, Colao A, Tauchmanova L, Savastano S, Labella D, Russo T, Zullo F, Lombardi G. Homocysteine levels and C677T polymorphism of methylenetetrahydrofolate reductase in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2003; 88:673–9.
20. Palep-Singh M, Picton HM, Yates ZR, Barth J, Balen AH. Polycystic ovary syndrome and the single nucleotide polymorphisms of methylenetetrahydrofolate reductase: a pilot observational study. Hum Fertil (Camb). 2007; 10:33–41.
21. Choi SW, Gu BH, Ramakrishna S, Park JM, Baek KH. Association between a single nucleotide polymorphism in MTHFR gene and polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol. 2009; 145:85–8.
22. Karadeniz M, Erdogan M, Zengi A, Eroglu Z, Tamsel S, Olukman M, Saygili F, Yilmaz C. Methylenetetrahydrofolate reductase C677T gene polymorphism in turkish patients with polycystic ovary syndrome. Endocrine. 2010; 38:127–33.
23. Idali F, Zareii S, Mohammad-Zadeh A, Reihany-Sabet F, Akbarzadeh-Pasha Z, Khorram-Khorshid HR, Zarnani AH, Jeddi-Tehrani M. Plasminogen activator inhibitor 1 and methylenetetrahydrofolate reductase gene mutations in iranian women with polycystic ovary syndrome. Am J Reprod Immunol. 2012; 68:400–7.
24. Jain M, Pandey P, Tiwary NK, Jain S. MTHFR C677T polymorphism is associated with hyperlipidemia in women with polycystic ovary syndrome. J Hum Reprod Sci. 2012; 5:52–6.
25. Jiang Y, Lu Y, Li Y, Zhu P, Yang Q. Study on the correlation between methylene tetrahydrofolate reductase gene polymorphism and polycystic ovary syndrome. Matern Child Heal Care China. 2015; 30:3831–3.
26. Ożegowska K, Bogacz A, Bartkowiak-Wieczorek J, Seremak-Mrozikiewicz A, Pawelczyk L. Is there an association between the development of metabolic syndrome in PCOS patients and the C677T MTHFR gene polymorphism? Ginekol Pol. 2016; 87:246–53.
27. Szafarowska M, Segiet A, Jerzak MM. Methyle notetrahydrololate reductase A1298C and C677T polymorphisms and adverse pregnancy outcome in women with PCOS. Neuro Endocrinol Lett. 2016; 37:141–6.
28. Carlus SJ, Sarkar S, Bansal SK, Singh V, Singh K, Jha RK, Sadasivam N, Sadasivam SR, Gireesha PS, Thangaraj K, Rajender S. Is MTHFR 677 C>T Polymorphism Clinically Important in Polycystic Ovarian Syndrome (PCOS)? A Case-Control Study, Meta-Analysis and Trial Sequential Analysis. PLoS One. 2016; 11:e0151510.
29. Geng J, Zhang C, Hu S, Zhang Y, Zhang S, Lu J, Xie J, Zhao H. Role of methylenetetrahydrofolate reductase genetic polymorphisms in polycystic ovary syndrome risk. Int J Clin Exp Pathol. 2016; 9:8532–7.
30. Bagos P. Plasminogen activator inhibitor-1 4G/5G and 5, 10-methylene-tetrahydrofolate reductase C677T polymorp-hisms in polycystic ovary syndrome. Mol Hum Reprod. 2009; 15:19–26.
31. Fu L, Dai L, Li X, Zhang K, Bai Y. Association of methylenetetrahydrofolatereductase gene C677T polymorp-hismwith polycystic ovary syndrome risk: a systematic review and meta-analysis update. Eur J Obstet Gynecol Reprod Biol. 2014; 172:56–61.
32. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986; 7:177–88.
33. Mantel N, Haenszel W. Statistical Aspects of the Analysis of Data From Retrospective Studies of Disease. J Natl Cancer Inst. 1959; 22:719–48.
34. Woolf B. On estimating the relation between blood group and disease. Ann Hum Genet. 1955; 19:251–3.
35. Cochran WG. The combination of estimates from different experiments. Biometrics. 1954; 10:101–29.
36. Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34S.

