INTRODUCTION
Lung cancer is a daunting malady victimizing millions globally, on an annual basis. Being the most common form of tumor worldwide, 1.82 million lung cancer cases were diagnosed and 1.59 million deaths occurred, in 2012 [1]. The widespread nature and high global incidence of lung cancer calls for urgent advances in treatment regimes.
The two main subtypes of lung cancer are non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). NSCLC is a heterogeneous group of tumors comprising adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Whilst occurrence of histological type differs among countries, NSCLC is more prevalent than SCLC [1]. Of lung cancer diagnoses globally, adenocarcinoma has the highest incidence (29–69%), followed by squamous cell carcinoma (28–46%), and then SCLC (9–22%), with the lowest frequency [2].
Existing first-line and conventional therapies for lung cancer include chemotherapy, radiation therapy, surgery, and most recently, immunotherapy. With the toxicities and health hazards associated with existing treatments, novel therapies are increasingly gaining attention. In recent years, meticulous studies coupled with technological innovations have enabled the scientific community to better understand intricate intracellular and extracellular events at the genomic, molecular and cellular levels within neoplasms [3]. This has led to the evolution of a novel form of cancer treatment, called targeted therapy, that has maximum toxicity for cancer cells and minimal toxicity for healthy cells. It is directed towards exploiting the abnormal composition of cell surface receptors on cancer cells. Agents disrupting angiogenesis through vascular endothelial growth factor receptor (VEGFR) signaling and those targeting the epidermal growth factor receptor (EGFR) have enhanced outcome in NSCLC in large and randomized trials, thus, paving the entry to clinical practice [3]. The agents involved, typically monoclonal antibodies or small molecules, are directed towards the ectopically expressed proteins/receptors in various intracellular processes essential for disease/tumor perpetuation and growth [3]. The therapeutic potential of one of the most important ectopically expressed receptors in cancer, the G-protein coupled receptors (GPCRs), has yet to be explored.
GPCRs, the largest family of cell surface receptors, play crucial roles in both normal and diseased body states [4]. They have been implicated to play vital roles in the formation and progression of tumor, and have been shown to be ectopically expressed on the cell surface of cancer cells [5, 6]. GPCRs have served, for over a millennium, as targets for drug intervention in the treatment of disease; medicinal drugs used by the Romans and Egyptians contained alkaloids and opioids, derived from foxglove and mandrake, which moderate their action through GPCRs [7]. Due to their significance in the many physiological processes, GPCRs are now a major focus of current pharmaceutical research: about 50% of drugs on market target the G-protein coupled receptor family [6].
With over 800 members [8], GPCRs are categorized into five major classifications, namely Rhodopsin (class A); Secretin and Adhesion (class B); Glutamate (class C); Adhesion (Class D) and Frizzled/smoothened (Class E) (TAS2) receptor families. These families are further divided into many subfamilies based on sequence similarity and ligand classes, including peptide, opsin, prostaglandin, MECA, Melatonin receptors, etc. The peptide receptor subfamily is the pivot of the current article owing to its significant association with the neoplastic state and its strict selectivity and high specificity to its cognate ligands. Research has proven the high specificity and potency of peptide-based therapeutics, which has intensified the efficacy of rather nonspecific drugs and reduced drug-associated toxic side-effects. Furthermore, peptide-based therapeutics have facilitated diagnosis and noninvasive gross morphological evaluation of solid tumors. Radiotherapy utilizing peptide-based compounds has been proven effective in the treatment of certain forms of solid tumors. [9]. For these reasons, peptide-based therapeutics such as peptide-drug conjugates [10–12] and peptide ligand functionalized nanoparticles [13, 14], among others, are popular compounds undergoing scrutiny and evaluation in ongoing oncological research.
This review highlights some recent GPCR-targeted therapeutics tested as potential candidates for lung cancer treatment. The article begins with the introduction of various overexpressed peptide GPCRs in lung cancer and their significance in neoplastic processes in tumor development. The next two sections describe how these processes may be altered or ‘corrected’ by targeting the overexpressed GPCRs through treatment system involving agonists and antagonists, as well as through Nano-scaled delivery platforms that enclose chemotherapeutic drug(s) and are decorated with peptide ligand of the peptide GPCR to be targeted, on their external surface. Finally, future directions and perspectives will be discussed.
Overexpressed peptide receptors in lung cancer
Common overexpressed peptide GPCRs in lung cancer include the Cholecystokinin B/Gastrin receptor, the Bombesin receptor family, Bradykinin B1 and B2 receptors, Arginine Vasopressin Receptors 1a, 1b and 2, and Somatostatin receptor type 2. All these receptors belong to the rhodopsin-like class A family of the GPCR superfamily [15–18]. Almost all of them are overexpressed in both, NSCLC and SCLC, with Table 1 showing the extent of over- or ectopic expression in different subtypes of lung cancer. All the cognate peptide ligands are classed as neuropeptides. Cholecystokinin (CCK), gastrin, Gastrin releasing peptide (GRP), Neuromedin-B (NMB), bradykinin and vasopressin are considered growth-stimulatory neuropeptides as they act as autocrine/paracrine growth factors [19, 20] to promote cellular proliferation [21] in lung cancer and other cancers, whereas somatostatin is classified as a growth-inhibitory neuropeptide.
Table 1: Ectopically expressed peptide GPCRs in lung cancer
Receptor |
Gene |
Lung cancer subtype |
Patient tissue |
|---|---|---|---|
Cholecystokinin-B/ Gastrin receptor |
CCKBR/ CCK2 |
SCLC |
mRNA overexpressed in 10/10 (100%) [24] and protein overexpressed (57%) [34] |
Squamous cell carcinoma |
mRNA overexpressed in 1/13 (7.7%) [24] |
||
Adenocarcinoma |
mRNA overexpressed in 1/21 (4.76%) [24] |
||
Bombesin receptor subtype 3 |
BRS3/ BB3/ BB3R |
SCLC |
mRNA overexpressed in 4/9 (44.4%) [50] |
NSCLC |
N/A |
||
Bronchial carcinoid |
mRNA overexpressed in 9/26 (34.6%) [50] |
||
LCNEC |
mRNA overexpressed in 1/1 (100%) [50] |
||
Gastrin Releasing Peptide Receptor (GRPR) |
GRPR/ BB2/ BB2R |
SCLC |
mRNA overexpressed in 3/9 (33.3%) [50] |
NSCLC |
N/A |
||
Neuromedin-B Receptor |
NMBR/ BB1/ BB1R |
SCLC |
N/A |
NSCLC |
N/A |
||
Bradykinin Receptor B1 |
BDKRB1/ B1BKR/ BKR1/ bradyb1 |
Adenocarcinoma |
Protein overexpressed in 6/6 (100%) [70] |
Squamous cell carcinoma |
Protein overexpressed in 5/6 (83.3%) [70] |
||
Large cell carcinoma |
Protein overexpressed in 5/6 (83.3%) [70] |
||
Small cell carcinoma |
Protein overexpressed in 6/6 (100%) [70] |
||
Carcinoid tumors |
Protein overexpressed in 4/6 (66.6%) [70] |
||
Bradykinin Receptor B2 |
BDKRB2/ BK-2 |
Adenocarcinoma |
Protein overexpressed in 6/6 (100%) [70] |
Squamous cell carcinoma |
Protein overexpressed in 6/6 (100%) [70] |
||
Large cell carcinoma |
Protein overexpressed in 4/6 (66.6%) [70] |
||
Small cell carcinoma |
Protein overexpressed in 3/6 (50%) [70] |
||
Carcinoid tumors |
Protein overexpressed in 5/6 (83.3%) [70] |
||
Arginine Vasopressin Receptor 1a |
AVPR1A/ V1aR |
SCLC |
mRNA overexpressed in 5/7 (71.4%) [96] |
NSCLC |
mRNA overexpressed in 17/22 (77.3%) [96] |
||
Arginine Vasopressin Receptor 1b |
AVPR1b/ V3R/V1bR |
SCLC |
mRNA overexpressed in 2/7 (29%) [96] |
NSCLC |
mRNA overexpressed in 4/22 (18%) [96] |
||
Arginine Vasopressin Receptor 2 |
AVPR2/ V2R |
SCLC |
mRNA overexpressed in 7/7 (100%) [96] |
NSCLC |
mRNA overexpressed in 18/22 (82%) [96] |
||
Somatostatin receptor (type 2A) |
SSTR2 |
SCLC |
Protein overexpressed in 23/61 (37.7%) [128] |
Typical carcinoid |
Protein overexpressed in 17/24 (70.8%) [128] |
||
Atypical carcinoid |
Protein overexpressed in 37/73 (50.7%) [128] |
||
LCNEC |
Protein overexpressed in 20/60 (33.3%) [128] |
Cholecystokinin-B/Gastrin receptor (CCKBR)
The cholecystokinin-B/gastrin receptor (CCKBR) is activated by its endogenous ligands, cholecystokinin and gastrin. Under physiological conditions, the CCKBR is commonly expressed in the stomach, pancreas and in particular areas of the human brain [22–24]. The receptor exerts a growth-stimulating effect in peripheral tissues [25, 26] and is involved in gastric acid secretion [27]. In the central nervous system, the receptor mediates emotional behaviors such as pain, anxiety and panic [28–31].
The CCKBR is, however, not expressed in the normal lung [24]. The mRNA expression was below the detectable level in all 12 normal lung tissues [24]. However, CCKBR mRNA was detectable in lung cancer, including SCLC, adenocarcinoma and squamous cell carcinoma [24]. CCKBR has been proposed as an attractive therapeutic target specifically for SCLC, whose prognosis persists to be disappointing despite initial response to chemotherapy [24]. Receptor autoradiographic studies revealed that CCKBR is expressed in high percentages in lung [24] and pancreatic [32] cancers, medullary thyroid carcinomas, some ovarian cancers, astrocytomas, gastrointestinal tumor, and colorectal cancer [33–35]. CCKBR was either not expressed or rarely expressed in differentiated thyroid cancers, meningiomas, lymphomas, renal cell cancers, and prostate carcinomas [34]. In regards to lung cancer, CCKBR is expressed in both SCLC and NSCLC [24, 34] (Table 1). Recently, Tripathi et al. created a comprehensive, literature-based map elucidating intracellular signaling cascades mediated by CCKBR (and CCKAR) [36]. The map may assist in the formulation of novel hypotheses on molecular mechanisms [36] and aid in the discovery and identification of novel molecular markers for CCKBR-based cancer therapeutics.
The paralog of CCKBR, CCKAR, is rarely or negligibly expressed in SCLC [37, 38] and other tumors [34, 39], with its expression level being significantly less than that of CCKBR. Thus, CCKAR is not covered in the current review.
Bombesin receptor family (GRPR, NMBR, and BRS-3)
Three receptors have been identified to belong to the bombesin (BN) receptor family, namely Gastrin Releasing Peptide receptor (GRPR), the Neuromedin-B receptor (NMBR), and the Bombesin receptor subtype 3 (BRS-3). High-affinity endogenous ligands for GRPR and NMBR are gastrin releasing peptide (GRP) and neuromedin-B (NMB), respectively; meanwhile, BRS-3 remains an orphan receptor, having low affinity for all natural-occurring bombesin type peptides [40, 41]. Human GRP is the mammalian analog of bombesin (BN), a 14 amino-acid peptide primarily discovered in the skin of the frog Bombina bombina [42].
The BN receptor family is a family of brain-gut peptide receptors [42, 43]. BRS-3 has high homology, 47–51% to GRPR/NMBR. Under physiological conditions, the BN-like peptides act on the central nervous system to regulate food intake, body temperature and glucose levels and certain behavioral responses [44]. In the periphery, GRP and NMB are involved in a spectrum of actions including smooth muscle contraction and endocrine/exocrine secretion. GRP is named for its property to induce gastrin release from gastrin (G) cells in the antral mucosa. GRPR has been fully characterized in the gastrointestinal epithelial cells [45, 46] and plays a role in gastrointestinal functions [47]. BRS-3 and its undefined ligand mediate vital metabolic and endocrine processes [48]. The activation of the BN receptor family causes numerous intracellular signaling cascades, which are chiefly moderated by phospholipase-C activation causing stimulation of protein-kinase C and cellular calcium changes [40, 42, 49]. Nevertheless, much is yet to be discovered on the BN receptor family.
Overexpression of the BN receptors has been evidenced in several tumor types, including lung, breast, prostate, intestinal, pancreatic, and colon carcinomas, gastrinomas, neuroblastomas, pituitary adenomas, head and neck cancers, and tumors of the CNS (gliomas, meningiomas) [42, 50–55]. Lung cancer has been the principal experimental model for discovering possible treatments curing through BN receptor family targeting. This is because SCLC has long been known to produce and release BN-related peptides [53, 54]: in 1985, SCLC was the first human tumor in which an autocrine growth effect was discovered [56], revealing fundamental information on the indispensability of this group of peptides and their cognate receptors. Overexpression of the BN receptor family was shown in different subtypes of lung cancer [50] (Table 1).
Generally, human tumors preferentially express the individual receptor subtypes of the BN receptor family, especially more frequently GRPR and less frequently NMBR [57, 58]. The significance of the bombesin/GRP-R in promoting cancer cell growth through the induction of autocrine loops and the high density of these receptors on the cell surface of various human tumors renders the receptor family a popular focus of nuclear oncology and extremely attractive targets for developing innovative therapeutic strategies, particularly for life-threatening neuroendocrine tumors such as SCLC [59–62].
Bradykinin receptors B1 and B2 (B1R and B2R)
Two pharmacologically distinct kinin receptor subtypes exist, namely bradykinin receptors B1 (B1R) and B2 (B2R) which are mainly involved in pain and inflammatory pathways [63]. The endogenous ligands for B2R are bradykinin (BK) and lysyl-bradykinin (Lys-BK) [17]; those for B1R are metabolites lacking the C-terminal arginyl residue, [des-Arg9] BK and [Lys-des-Arg9] BK [17, 64]. B2R is ubiquitously and constitutively expressed, whereas the physiological expression of B1R is extremely low. However, the expression of B1R surges in stressful situations [65], such as various pathological conditions, in several cell types including neurons, endothelial and blood cells, and smooth muscles. B1R is induced in various models of cancer, angiogenesis, inflammation, pain syndromes, diabetes mellitus, multiple sclerosis, epilepsy, and Alzheimer’s disease [17, 66–69].
B2R and B1R are highly expressed in cancers of the lung [70], breast [71], prostate [72], gall bladder [73], head and neck squamous cell carcinoma (HNSCC) [74], chondrosarcomas [75], colorectal adenomas [76], clear cell renal carcinoma [77], esophageal squamous cell carcinomas, and astrocytic tumors [78]. Table 1 shows the upregulation of B1R and B2R in lung cancer subtypes [70].
Whereas B1R and B2R may have a high expression profile in numerous tumor types, their relative expression levels may differ in different tumors. For instance, Chee et al. reported a somewhat similar expression pattern of the two bradykinin receptor subtypes in lung cancer [70], whereas a study by Nicoletti et al. on glioma cells showed a higher expression of B1R relative to B2R [79]. The authors of the glioma study attribute the higher expression level of B1R to the inducible nature of the receptor [79].
Studies on mice lacking each receptor gene and different specific peptidic and non-peptidic antagonists have implied that both B1R and B2R are potential therapeutic targets in various pathophysiological events in the aforementioned diseases [17]. Due to negligible expression of B1R in healthy tissues, the receptor is very appealing as an imaging marker for tumor diagnosis and for the development of anticancer therapeutic agents [80, 81].
Arginine Vasopressin Receptors 1a, 1b and 2 (V1a, V1b, and V2)
Three arginine vasopressin receptors (AVPRs) have been identified via molecular cloning techniques, V1a (vascular), V1b (pituitary) and V2 (renal) [82]. The endogenous ligand for the AVPRs is the nonapeptide amine, vasopressin (or antidiuretic hormone), produced by magnocellular neurons of the hypothalamus [83]. This hormone is essential for survival as it is involved in the fundamental physiological processes of osmotic and cardiovascular homeostasis [84].
The expression of each subtype of AVPR is distinct and tissue-specific. V1a receptors are normally expressed in the heart, brain, testis, liver, superior cervical ganglion, vascular smooth muscle cells and renal medulla [85, 86]. V1a receptors in the brain moderate anxiety producing responses to arginine vasopressin, whilst their presence in the vasculature help moderate the pressor actions of arginine vasopressin through a phospholipase C-mediated pathway [87, 88]. The V1b receptor is involved in mediating anxiety and stress in humans and rats [89]. Their presence in the anterior pituitary helps mediate the ACTH-releasing effects of AVP, through a phospholipase C-mediated pathway [90]. V1b receptors are also present in extra-pituitary tissues such as the adrenal medulla, kidney and brain [91]. On the other hand, the V2R is mostly expressed in the renal medulla, where it is involved in mediating the antidiuretic effect of AVP [85, 92].
In normal cells, the vasopressin gene is transcriptionally restricted, while in SCLC, it is activated concomitantly with expression of all three of its receptors (V1aR, V1bR, and V2R) [93–95]. Vasopressin receptors are overexpressed only in lung cancer [96, 97] and ACTH-secreting pituitary tumors [98, 99]. Table 1 shows the pattern of overexpression of AVPRs in lung cancer subtypes [96, 97].
Somatostatin receptor type 2 (SSTR2)
The past 30 years saw the identification and characterization of the somatostatin receptor family, which includes five different subtypes, SSTR1- SSTR5 [100–102], that show 40–60% structure homology [103]. The endogenous high-affinity ligand for the SSTRs is the acid polypeptide, somatostatin, which is native to the central nervous system and various peripheral tissues and organs. Somatostatin has several biological functions including the potent inhibition of basal and stimulated secretions from a wide range of exocrine and endocrine cells [102, 104], and acts as a neurotransmitter (exerting both stimulatory and inhibitory effects [105]) and neuromodulator in the central nervous system, and as an antiproliferative agent for cell proliferation and differentiation [106], and as an autocrine/paracrine agent in the immune system [107]. SSTRs are differentially expressed in the immune and central nervous systems, pituitary, thyroid and adrenal glands, gut, pancreas and kidney [108, 109]. Multiple receptor subtypes may be co-expressed in a tissue-specific pattern, having distinct physiological roles [108]. In the peripheral nervous system, SSTRs are implicated to play roles in nociception [110]. In the pituitary gland, SSTR2 is involved in the release of ACTH, GH, and TSH [102].
SSTRs are highly expressed in a variety of tumors, including cancers of the lung [111, 112], breast [113, 114], prostate [115], brain [116, 117], and pheochromocytomas [118], gastric carcinomas [119, 120], meningiomas [121], hepatocellular carcinoma [122], endocrine pancreatic tumors, gastrointestinal carcinoids, and thyroid medullary cancer [123]. Out of the five subtypes, the SSTR type 2 is generally the most commonly overexpressed receptor in cancer [108, 124–127]. Thus, SSTR2 is the subtype covered in the present article. The extent of SSTR2 overexpression in lung cancer subtypes [128] may be viewed in Table 1.
Carcinogenesis pathways mediated by overexpressed peptide GPCRS
Cancer cells produce and secrete the neuropeptides CCK/gastrin [34, 129], GRP and NMB [130, 131], bradykinin [132], and vasopressin [133, 134]. A plethora of evidence exists elucidating the autocrine growth and pro-proliferative effects exerted by CCK/gastrin [135, 136], GRP and NMB [137, 138], bradykinin [139, 140] and vasopressin [21, 94, 95, 141, 142] through their respective receptors. In the process, they increase the colony number of cancer cells, including lung cancer [21, 135, 143]. Weber et al. reported that even activation of orphan receptor BRS-3 may contribute to cancer cell proliferation, since the analogue [d-Phe6, β-Ala11, Phe13, Nle14] bombesin (6–14) caused enhanced nuclear oncogene expression, MAPK phosphorylation, and Elk-1 activation in lung cancer cells [144]. Research has indicated that GPCR transactivation of epidermal growth factor receptor (EGFR) is significantly involved in cancer cell proliferation [135, 145]. For instance, the BRS-3 agonist (DTyr6, βAla11, Phe13, Nle14)bombesin6–14 (BA1) caused Tyr1068 phosphorylation of EGFR in NCI-H727 or NCI-H1299 cells stably transfected with BRS-3 (NCI-H1299-BRS-3), and significantly increased the clonal growth of NCI-H1299-BRS-3 cells. Other BRS-3 agonists such as (DTyr6, R-Apa11, Phe13, Nle14)bombesin6–14 (BA2) and (DTyr6, R-Apa11, 4-Cl,Phe13, Nle14)bombesin6–14 (BA3) also caused EGFR transactivation in NCI-H1299-BRS-3 cells [146]. Furthermore, it has been found that CCK/ gastrin [135, 147–150], GRP [53, 54, 131, 138, 151, 152], bradykinin [153, 154] and vasopressin [21, 155, 156] bind with high affinity to their cognate receptors to promote DNA synthesis [157], increase intracellular calcium levels [37, 132, 143, 147–150, 153–155, 158], promote cellular growth, proliferation, survival [139, 140, 159–165], cause loss of cell adhesion, and stimulate tumor progression, invasion, migration and metastasis [166–169]. Moreover, it is thought that CCK/gastrin [170], GRP and NMB [130, 131, 171], and bradykinin [172, 173], by acting on their cognate receptors, promote angiogenesis and suppress apoptosis.
Somatostatin stands out from the crowd of receptors included in this review. While all ligands whose cognate receptors included in this paper act as growth factors, somatostatin is inherently a broad inhibitory neuropeptide and has anti-secretory, anti-proliferative and anti-angiogenic activities [174]. It mediates its effects through five receptors but SSTR2 subtype is generally the most commonly expressed in neoplastic cells [123]. Somatostatin and its analogs/agonists inhibit tumor growth and metastatic spread through activation of SSTRs on both cancer and microenvironment cells, such as endothelial cells of tumor vessels that are responsible for the neovascularization of the tumor. SST exerts its effects through direct antiproliferative (inhibition of mitogenic stimuli of growth factors such as IGF-1 and cell cycle arrest) and proapoptotic signals, as well as, indirectly, by inhibiting the secretion of proliferative and angiogenic growth factors and hormones, or suppressing neoangiogenesis at the endothelial cell level or regulating an immune response [175, 176].
Therapeutics targeting overexpressed peptide receptors for lung cancer
Standard therapies have encountered a plateau in improving patient overall health and survival and quality of life [177]. In addition, despite initial responsiveness to existing standard cancer treatments, like chemotherapy and radiotherapy, some cancer types like SCLC are highly aggressive and commonly relapse within months [178]. Cancer therapeutics targeting overexpressed and/or ectopically expressed peptide GPCRs in cancer is one novel and promising treatment option which has gained considerable interest over the past two decades. The ectopic and over-expression of GPCRs such as CCKBR, GRPR, B1R, and other receptors, drives tumor growth. Therefore, interference in receptor signaling and inhibition of signaling pathways driving tumor growth and perpetuation are envisioned as ideal targets for cancer therapeutics. This may be achieved using monoclonal antibodies, agonists and antagonists. This section will include some of the recent targeted therapeutics, for each peptide GPCR, that have been discovered to show positive results in vitro and in vivo to ultimately contribute to tumor shrinkage.
Cholecystokinin-B/Gastrin receptor (CCKBR)
A plethora of studies have shown the significant role that upregulation of CCKBR and its endogenous ligands play in the regulation of tumor growth and maintenance [25, 26]. Antagonists for CCKBR help downregulate expression of the receptor resulting in decreased DNA synthesis, cell cycle arrest through inhibition of G(1) to S phase progression, reduced cancer cell proliferation, mobility and invasiveness. In addition, downregulation of CCKBR increased caspase-3 activity and TUNEL-positive cells, suggesting apoptotic activity [179].
CCKBR therapeutics include antagonists such as CI-988 (Figure 1A, [180]) and L365, 260 (Figure 1A, [181]). CI-988 inhibited the abilities of CCK-8 to elevate cytosolic Ca2+, to stimulate EGFR, ERK and FAK tyrosine phosphorylation as well as VEGF expression, and so cell growth and proliferation in NCI-H727 lung cancer cells [135]. L365, 260 inhibited the proliferative capacity of human medullary thyroid carcinoma cells, resulting in a marked attenuation in growth [182].
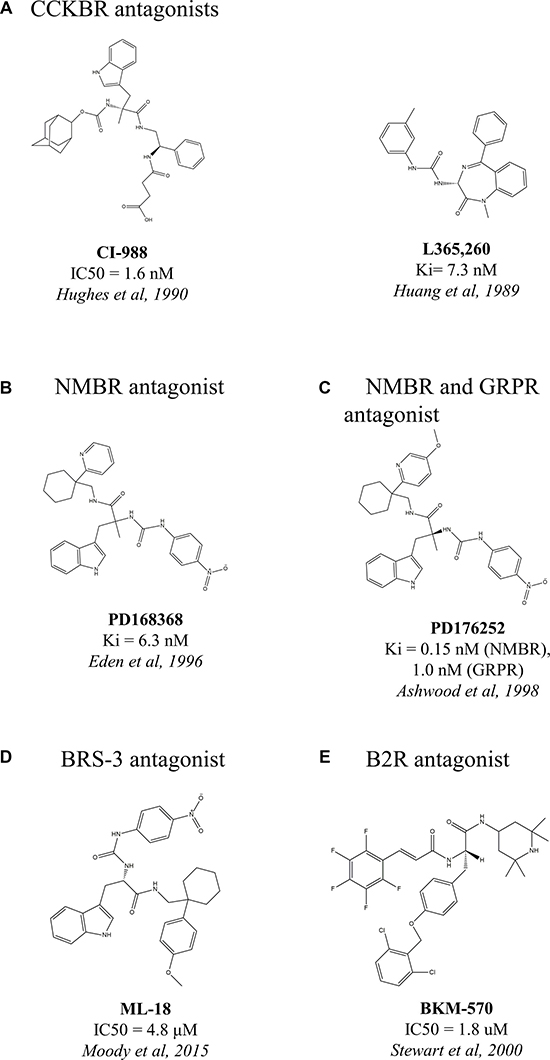
Figure 1: Chemical structures of non-peptide antagonists of overexpressed peptide GPCRs in lung cancer.
Bombesin Receptors (GRPR, NMBR, and BRS-3)
Bombesin related peptides are synthesized and secreted by cancer cells and are reported to cause autocrine-growth effects in human SCLC [56] and other cancers [50, 183, 184]. Some studies demonstrated that monoclonal BN antibodies inhibit the growth of these tumors both in vitro and in vivo xenografts [56]. Many studies demonstrate the potential therapeutic significance of BN receptor antagonists and other agents that inhibit the growth-stimulatory effect of BN receptor agonists on tumors [61].
High-affinity NMB receptor non-peptide antagonist PD168368 (Figure 1B, [185]) inhibited lung neoplastic cell growth by inhibiting the transactivation of EGFR and the tyrosine phosphorylation of ERK caused by NMB-like peptides [152].
High affinity NMBR (Ki = 0.15 nM) and moderate affinity GRPR (Ki = 1.0 nM) non-peptide antagonist PD176252 [186] (Figure 1C, [186]) significantly inhibited NCI-H1299 proliferation (and was more potent than PD168368) and considerably inhibited lung cancer colony number in vitro [187]. PD176252 inhibited the binding of GRP/bombesin to GRPR and blocked the ability of the ligand to elevate cytosolic calcium levels and c-fos mRNA in lung cancer cells NCI-H1299, NCI-H345 and H1299. It also blocked the ability of bombesin to cause tyrosine phosphorylation of focal adhesion kinase in NCI-H1299 cells [187]. In vivo studies revealed that the antagonist dose-dependently inhibited NCI-H1299 xenograft growth in nude mice [187].
Both the afore-mentioned antagonists, PD168368 and PD176252, do not bind BRS-3 with high affinity [188]. For BRS-3, however, the novel BRS-3 antagonist ML-18 (Figure 1D, [189]), had anti proliferative effects in lung cancer [189]. ML-18 inhibited specific (125)I-BA1 (DTyr-Gln-Trp-Ala-Val-βAla-His-Phe-Nle-NH2)BB(6–14) binding, with an IC50 value of 4.8 μM, to lung cancer cells NCI-H1299 stably transfected with BRS-3. ML-18 bound to GRPR and NMBR with lower affinity with IC50 values of 16 and > 100 μM, respectively. ML-18 inhibited the ability of BA1 to increase cytosolic calcium and tyrosine phosphorylation of EGFR and ERK in lung cancer cells [189].
Another BRS-3 antagonist, BRS-3 ant. (DNal-Cys-Tyr-DTrp-Lys-Val-Cys-Nal)NH2, blocked BA1-induced EGFR or ERK tyrosine phosphorylation in lung cancer cells and diminished clonal growth of NCI-H1299-BRS-3 cells [146]. In addition, BA1, BA2, BA3 (BRS-3 agonists) and BRS-3 ant. (BRS-3 antagonist) blocked specific 125I-BA1 binding to NCI-H1299-BRS-3 cells with IC50 values of 1.1, 21, 15 and 750 nM, respectively [146].
An additional antagonist of BN/GRP is the powerful inhibitor RC-3940-II. It significantly inhibited growth of H460 and A549 NSCLC xenografts and caused the upregulation of tumor suppressor gene p53, which may contribute to the anti-tumor effects of the antagonist [190], and an increase in several angiogenesis inhibitors and a reduction in proangiogenic genes [191]. There was also an escalation in the number of cells with lower G(0)/G(1) DNA content and in those blocked in S and G2/M phases [192]. Zhou et al.. reported on antibody-mediated therapy for SCLC. They conjugated a monoclonal antibody OKT3 (anti-CD3) with a bombesin/GRP antagonist (Antag2) to create a bispecific molecule, OKT3xAntag2. The molecule was shown to mediate growth inhibition and apoptosis of SCLC cells by activated T cells via the activation and cleavage of Poly (ADP-ribose) polymerase (PARP) and caspase-3 in vitro and in vivo [193].
Bradykinin receptors B1 and B2 (B1R and B2R)
In 1984, the Stewart laboratory discovered the first antagonists for bradykinin (BK) and ongoing investigations by this group and others resulted in the present generation of extremely potent peptide antagonists that are orally active and have proven resistant to all tested degrading enzymes [194, 195]. It is noteworthy that prolonged administration of bradykinin antagonists to humans has not caused adverse effects [196].
CU201 (also called B9870) is a highly potent, metabolism-resistant bradykinin antagonist peptide dimer. CU201 is a growth inhibitor for SCLC both in vitro [197, 198] and, in vivo for SCLC SHP-77 and NSCLC A549 tumor growth [199], giving significant inhibition (65%) of tumor growth in athymic nude mice models upon daily intratumoral administration at a dose of 5 mg/kg/day [195]. The antagonist inhibited proliferation signals but induced apoptotic signals [197] by a novel “biased agonist” action. It blocked the Gαq, G11 signaling pathway that induces intracellular free Ca2+ in response to bradykinin but stimulated the Gα12, G13 pathway associated to c-JUN kinase activation [44], inducing caspase-3 activity and causing unique changes in apparent nuclear DNA binding, ultimately resulting in cell death. Therefore, CU201 offers great promise in being branded as a new form of targeted therapy for neoplasms with neuroendocrine properties, owing to its unique mechanism of action [197].
BKM-570 (Figure 1E, [200]) is the non-peptide mimetic of CU201 [198]. BKM-570 strongly inhibited extracellular signal-regulated kinases 1/2 (ERK1/2) and protein kinase B (AKT) [201], effectively suppressing tumor cell growth [202]. BKM-570 showed strong anticancer activity against SCLC and prostate cancer in vivo [198] and in vitro [195] and various other cancer cell lines of NSCLC, cervix and colon cancers in vitro, where the antagonist did not harm the growth of normal pulmonary fibroblasts [195].
Another bradykinin antagonist is R954. It is a stable, selective and potent peptide antagonist of the inducible B1R. It exhibits favorable preclinical pharmacological, pharmacokinetic characteristics and toxicological profile [64] and has shown antitumoral activity on ascitic and solid tumors induced by Ehrlich cell inoculation in rats and mice [203]. However, more research is warranted for this antagonist as current evidence is greatly limited. R954 has, nevertheless, been tested in some other conditions with success, including acute lung injury [204], osteoarthritis [205], inflammatory edema [206] and more. The testing of this antagonist in cancer models is relatively recent when compared to its testing in other pathological states, hence the limited data on its effects in cancer. This antagonist may potentially emerge as a new anti-cancer drug.
Arginine vasopressin receptors 1a, 1b and 2 (V1a, V1b, and V2)
Vasopressin agonists with anticancer effects exert their action through V2 receptor, as it is involved in antiproliferative effects; the V1 receptors are related to cellular proliferative effects [207]. The synthetic nonapeptide 1desamino8Darginine vasopressin, desmopressin (dDAVP), is an agonist on the V2 receptor. dDAVP decreased expression levels of neuroendocrine markers chromogranin A (CgA) and specific neuronal enolase (NSE) in aggressive SCLC cell line NCI-H82 [208]. dDAVP also displayed dose-dependent anti-metastatic effects with maximum impact at clinically relevant doses of 1–2 μg/kg [209] and a 70% reduction in the number of pulmonary nodules in experimental lung metastatic disease [210]. dDAVP seemed to stimulate a dual angiostatic and antimetastatic effect, breaking the cooperative function between cancerous and endothelial cells during residual tumor progression [210, 211]. Pastrian et al. showed that the amino acids present at the loop of dDAVP are crucial for the antiproliferative activity of dDAVP, accentuating the significant role of the N terminal region of the peptide in the interaction with the cancer cell surface receptor. These findings help show novel strategies for designing and developing improved compounds with augmented stability for tumor therapy [212]. An analog of dDAVP, [V4Q5]dDAVP , decreased tumor growth and angiogenesis in F3II mammary tumour-bearing immunocompetent mice, and exhibited higher antimetastatic efficacy than dDAVP on experimental lung colonisation by sarcomatoid mammary carcinoma F3II cells [213]. Preliminary acute toxicology studies revealed that [V4Q5]dDAVP was well-tolerated at doses ≥ 300-fold above those needed for anti-angiogenic/antimetastatic effects [213]. DDAVP and [V4Q5]dDAVP markedly decreased proliferation, doubling time, and migration in NCI-H82 cells. [V4Q5]dDAVP demonstrated a greater cytostatic effect than dDAVP, on cellular proliferation in the NCI-H82 cell line [208].
Somatostatin receptor type 2 (SSTR2)
Somatostatin is of limited clinical utility owing to its extremely short half-life of 2–3 minutes, resulting in the development of many synthetic somatostatin analogs (SSAs) such as octreotide, lanreotide and pasireotide, by attenuation of the polypeptide chain while preserving binding affinity to the SST receptors [214]. Octreotide and lanreotide bind to SSTR-2 with high affinity (0.32 and 0.5 nM) [215], whilst pasireotide binds SSTR2 with 2.5 times lower binding affinity than octreotide [216]. Subsequently, long-acting release (LAR) octreotide [217] and long-acting release (LAR) lanreotide [218] were also developed.
Octreotide and lanreotide are registered in many countries [219] and have proven to be successful therapeutics in the management of tumors. These SSAs have similar effects, in neuroendocrine tumors, to native endogenous somatostatin in that they decrease cellular proliferation, induce apoptosis, inhibit protein synthesis and cell signaling [220], and inhibit secretory processes [221].
SSAs exert antiproliferative effects through direct and indirect mechanisms. Direct mechanisms entail the activation of SSTRs in tumor cell surfaces [222]. Upon activation, SSTRs induce cell cycle inhibitors such as p21, p27 and p130/Rb, thus resulting in cell cycle arrest [222]. Somatostatin and SSAs can directly induce apoptosis in cancer cells, with a p53- dependent or independent mechanism [223]. SSTR2, along with SSTR1, SSTR3 and SSTR4, may be involved in inhibiting cell invasion by impacting the PI3K pathway. SSTR2 is also important in restoring gap junctions, which are essential for contact inhibition and maintenance of a differentiated condition [224, 225]. Indirect mechanisms by which SSAs exert antiproliferative effects involve the inhibition of circulating growth factors such as insulin-growth factor (IGF), vascular endothelial growth factor (VEGF), growth hormone (GH), platelet-derived growth factor (PDGF), basic fibroblast growth factor (bFGF), as well as inhibition of tumor angiogenesis by inhibiting the proliferation and migration of endothelial cells and monocytes, which secrete proangiogenic factors [226–229]. It is thought that the main mechanism of angiogenesis inhibition may revolve around interference in endothelial NO release. [230].
Whilst the antiproliferative and anti-tumor effects of SSA were shown in vitro and in vivo years ago, their use as anti-cancer agents has only recently been recognized [224]. Evidence of SSA success in lung neuroendocrines is very limited; a mere handful studies have analyzed the antiproliferative effects of SSAs in bronchial carcinoids. A retrospective study involving 48 patients with lung NET discovered that the most frequently administered first-line therapy in patients with advanced disease, was administration of SSAs [231]. A phase III RADIANT-2 trial randomized 429 patients with hormonally active carcinoid tumors, including bronchial NETs, to treatment with octreotide LAR plus placebo or octreotide LAR plus anticancer agent, everolimus. Analysis of the subgroups revealed that, for the 44 patients with lung carcinoid who received only the SSA as active drug, the median PFS was 5.6 months [232]. Furthermore, tumor shrinkage, although not satisfying RECIST criteria for partial response, was seen in 27% of participants from the octreotide LAR monotherapy cohort [233]. The results of a randomized phase III trial (PROMID) demonstrated that the median time to progression in patients with midgut carcinoid tumors treated with octreotide LAR was 14.3 months versus 6 months in patients treated with placebo [234]. Recently, a retrospective study analyzed the efficacy of octreotide in 15 patients with advanced pulmonary carcinoids, and reported a median PFS of 15 months and a 70% disease control rate [235]. A prospective, randomized, open-label, 3-arm, phase II study evaluating the effectiveness and safety of pasireotide, everolimus, or both, in patients with advanced neuroendocrine carcinoma of the lung and thymus (LUNA trial) is currently being conducted in Europe [236].
Targeting overexpressed peptide receptors through delivery systems for lung cancer
Targeted drug delivery (TDD) platforms are based on the success and advantages of using peptides to successfully target over- or ectopically expressed receptors in cancer cells. Specific tumor receptor interaction is instrumental in mediating the ability of peptide ligand-cytotoxic constructs to induce cytotoxicity [237]. TDD platforms may involve peptide-drug conjugates (PDCs) that generally comprise a therapeutic moiety, a linker moiety, and a peptide as a targeting moiety [238]. Also, external surfaces of Nano-sized drug delivery systems, including liposomes and micelles, are decorated with peptide receptor ligands. The use of peptides as targeting moiety is advantageous because they cause the cytotoxic drug-loaded delivery platform to be targeted specifically to tumor cells, inflicting no/negligible damage to healthy cells. This section focuses on some recent works incorporating receptor peptide ligands within Nano-scale delivery systems for targeted and effective therapeutic intervention in neoplasms. A summary of the works covered in this section may be viewed in Table 2.
Table 2: Ligands of ectopically expressed peptide receptors in targeted delivery/imaging systems for lung cancer
Receptor |
Ligand |
Ligand sequence |
Used in targeted delivery/ imaging system |
|---|---|---|---|
Cholecystokinin-B/Gastrin receptor |
CCK-33 |
KAPSGRMSIVKNLQNLDPSHRISDRDYMGWMDF-NH2 [http://www.uniprot.org] |
N/A |
CCK-8 |
DYMGWMDF-NH2 [241] |
N/A |
|
CCK-5 |
GWMDF-NH2 [http://www.uniprot.org] |
N/A |
|
Gastrin |
pEGPWLEEEEEAY(SO3H)GWMDF-NH2 [268] |
N/A |
|
Bombesin Receptor Subtype 3 (BRS-3) |
Orphan receptor (has synthetic ligand) |
[D-Tyr, β-Ala, Phe,Nle] BB(6–14) [269] |
N/A |
Gastrin Releasing Peptide (GRPR) |
Gastrin releasing peptide (GRP) |
VPLPAGGGTVLTKMYPRGNHWAVGHLM-NH2[268] |
N/A |
Bombesin peptide |
pEQRLGNQWAVGHLM-NH2 [268] |
||
Neuromedin-B receptor (NMBR) |
Neuromedin B (NMB) |
GNLWATGHFM-NH2 [268] |
N/A |
Bombesin Receptor family (BRS-3, GRPR, NMBR) |
Pan-BBN ligand (binds to all 3 receptors) |
D-Tyr, β-Ala, Phe, Nle] BBN [6–14] [246] |
CPT-L2-BA3 |
Bradykinin Receptor B1 |
Bradykinin |
RPPGFSPFR- NH2 [268] |
N/A |
Bradykinin Receptor B2 |
Bradykinin Potentiating Peptide (BPP) |
EWPRPQIPP- NH2 [251] |
Pt-CS-BPP [251] |
Arginine Vasopressin Receptor 1a |
Vasopressin |
CYFQNCPRG-NH2 [268] |
99mTc(NS3)(CN-AVP) [257] |
Arginine Vasopressin Receptor 2 |
99mTc(NS3)(CN-AVP(an)) [257] |
||
Somatostatin receptor (type 2) |
Somatostatin |
AGCKNFFWKTFTSC-NH2 [268] |
N/A |
Octreotide (analog of somatostatin) |
(D)FCF(D)WKTCT-ol [270] |
1.[OCT(Phe)-PEG-ss-PTX] [259] |
|
2. SSTR2-3207-86 [261] |
Cholecystokinin-B/Gastrin receptor (CCKBR)
The Cholecystokinin-B/gastrin receptor binds CCK-33, CCK-8, CCK-5 and gastrin. However, it is CCK-8 that is most commonly used as a targeting moiety in targeted delivery systems. There is a lack of research and classical literature focusing on CCK-8 labelled/loaded delivery systems tested specifically in lung cancer. They were however, tested in other cell types including HuVEC cells and A431 epidermoid carcinoma cell line, with positive results [239–241].
Bombesin receptors (GRPR, NMBR, and BRS-3)
Due to the high frequency of overexpression of BN receptor subtypes in many tumors, increasing interest has led to massive investments of resources and time towards investigating the Bombesin receptors as potential therapeutic targets. In vitro autoradiographic studies reveal that GRPR, out of the three human BN receptors, is the most commonly expressed in human tumors [242]. Hence the analogue of GRP, the Bombesin peptide (QQRLGNQWAVGHLM) is a popular targeting ligand used in targeted therapy directed at GRP receptors. The eight-residue C-terminal peptide sequence (BN[7–14]), may be used to selectively target GRPR. Several studies show that the BN (7–14) fragment, modified with radiometal complexes on its N-terminus, preserves its affinity for these receptors, hence proving vital for diagnostic or therapeutic nuclear medicine applications [243–245].
Moody et al. discovered a potent BN agonist drug conjugate (CPT-L2-BA3) that had cytotoxic effect towards cells overexpressing all mammalian BN receptor subtypes [237, 246]. MTT and clonal growth assays revealed that CPT-L2-BA3 inhibited the growth of NSCLC NCI-H1299 cells and H1299 xenograft growth in nude mice, showing a high cytotoxic effect [237, 246]. On cells lacking BN receptors, CPT-L2-BA3 had only a very small effect. CPT-L2-BA3 also inhibited growth of several other tumor cell lines, including lung cancer (NCI-H69), neuroblastoma (IMR32, SKNSH), glioblastoma (U-87MG), leukemia (MOLT-4), breast cancer (MCF-7), prostate cancer (PC-3, DU-145, LNCaP), gastric cancer (Hs746T) and colon cancer (HT-29) cell lines with IC50 values ranging from 33 to 2269 nM [237]. Safavy et al. showed that the cytotoxicity of PTXPEGBBN[7–13], a tumor directed derivative of paclitaxel, was improved by a factor of 17.3 for 24 h and 10 for 96 h exposure times, in comparison to paclitaxel alone in NCI-H1299 human NSCLC cells [247]. The IC50 of the conjugate was lower than that of the free drug by a factor of 2.5 for exposure times of both 24 h and 96 h [247]. Moreover, the order of addition of peptide ligands to nano-sized delivery systems is an appealing and noteworthy perspective since the order may affect the size, stability and cytotoxicity exhibited by the final product. Post-bombesin decorated nanostructured lipid carriers (NLC) exhibited more stability and markedly higher transfection efficiency and better anti-tumor activity than pre-bombesin decorated NLC for lung cancer therapy, both in vitro and in vivo [248].
Bradykinin B1 and B2 receptors (B1R and B2R)
Bradykinin has a short plasma half-life of about 15 seconds [249] and is rapidly inactivated in pulmonary circulation [250]. Therefore, bradykinin potentiating peptide (BPP) has been used as targeting moiety for targeted delivery [251]. BPPs have the ability to inhibit bradykinin inactivation in lung [252] and potentiate bradykinin action [253–255]. BPP has a relative selectivity for tumor vasculature and exerts potent effects through B2R, which is commonly overexpressed in various types of tumors [70, 256]. Wang et al. speculated that BPP can promote drug accumulation in primary tumor and lung metastasis by facilitating an increase in vascular permeability and enhancing drug penetration [251]. They used a 9 amino acid residues-long BPP (EWPRPQIPP) and a drug which was bioreductive sensitive platinum (IV) compound which became cisplatin in the intracellular reductive environments. Both the drug and the BPP were covalently attached to 120 nm-diameter chitosan nanoparticles. In vivo biodistribution and tumor inhibition investigations revealed that, compared with the free drug and the peptide-free nanoparticle formulation, the BPP-studded nanoparticle formulation boasted superior efficacy in promoting drug accumulation in tumor, thus confining tumor growth and prolonging the lives of tumor-bearing mice. In addition, drug accumulation in lung metastasis was about 17% and 20% injected dose/gram of lung for the chitosan nanoparticles without and with BPP, respectively. This was 10-fold greater than that of free cisplatin, which was about 1.6% injected dose/gram of lung. Thus with improved drug accumulation in lung metastasis tissue, the BPP-studded chitosan nanoparticle formulations effectively inhibited metastasis to lungs [251].
Arginine vasopressin receptors 1a, 1b and 2 (V1a, V1b, and V2)
To our knowledge, the arginine vasopressin peptide has not been used as a targeting ligand in delivery systems carrying cytotoxic chemotherapeutic agents. The nonapeptide has, however, been conjugated to radionuclides for tumor imaging/diagnosis. Gniazdowska et al. investigated the conjugation of vasopressin (AVP) (CYFQNCPRG) and its analogue (d(CH2)5[D-Tyr(Et2), Ile4, Eda9 ]AVP (AVP(an)) to technetium-99m radionuclide to test its potential as a diagnostic radiopharmaceutical for SCLC patients [257]. In serum, the 99mTc(NS3)(CN-AVP) was enzymatically degraded into two species, whilst the 99mTc(NS3)(CN-AVP(an)) conjugate proved to be highly stable. In addition, the AVP (an) is one of the many effective antagonists to V2 receptor [258] and conjugates containing the AVP (an) demonstrated specific and high binding ability to V2 receptors on the SCLC cell line H69 [257].
Somatostatin receptor type 2 (SSTR2)
Recently, Yin et al. developed a redox-sensitive prodrug, octreotide(Phe)-polyethylene glycol-disulfide bond-paclitaxel [OCT(Phe)-PEG-ss-PTX] for the targeted intracellular delivery of paclitaxel [259]. The conjugate demonstrated approximately 23,000-fold increase in water solubility than the parent paclitaxel [259], which has extremely low water solubility [260]. The OCT(Phe)-PEG-ss-PTX was selectively internalized into tumor cells through SSTR-mediated endocytosis and showed a high degree of cytotoxicity and apoptosis-inducing ability against NCI-H466 SCLC cells that ectopically expressed SSTR. Furthermore, in vivo studies on NCI-H466 tumor-bearing nude mice showed that the OCT(Phe)-PEG-ss-PTX had superior tumor-targeting ability and antitumor activity along with minimal collateral damage, compared with free paclitaxel [259]. Also, recently, Redko et al. developed five novel PDCs by separately linking the SSTR2 specific backbone cyclic peptide 3207–86 with five different anticancer drugs. The PDCs exhibited selective and significant cytotoxic effects in the human NSCLC cell line H1299 and various other human cancerous cell lines overexpressing SSTR2 [261].
Shen and coworkers analyzed the anti-tumor effects of a conjugate developed by coupling two molecules of paclitaxel to octreotide in A549 human NSCLC cells xenografted into nude mice [262]. 2paclitaxel-octreotide caused significant tumor growth inhibition at 150 nM/kg and 300 nM/kg, and significantly lengthened the tumor doubling time and considerably decreased tumor micro vessel density at these doses. Increased amount of fragmented DNA was seen in the 2paclitaxel-octreotide single and double dose groups relative to the controls [262]. Similarly, Sun et al. developed paclitaxel-octreotide conjugates which dose- and time-dependently inhibited the growth of NSCLC cells A549 and Calu-6. Paclitaxel and the conjugates could stimulate the increase of G(2)/M phase ratio in A549 cells [263]. A noteworthy point is that the conjugates had less cytotoxicity than paclitaxel-alone in SSTR-negative fibroblasts.
Conclusions and Perspectives
Overexpressed peptide GPCRs are valuable biomarkers for cancer diagnosis, imaging, and treatment. While CCKBR [24], and B1R [264] are not expressed in normal lungs but only in cancerous lungs, all the other receptors included in the present review are expressed in both normal lungs as well as cancerous lungs; however they are upregulated in cancerous lungs. In this regard, CCKBR and B1R are ideal therapeutic targets for lung cancer because minimal side-effects can be expected. In contrast, the expression of other receptors in normal tissues must be considered when targeting ligands are assessed for use as imaging or therapeutic agents. In addition, normal tissue samples from the primary tumor organ site should be scrutinized prior to a receptor’s designation as an overexpressed or upregulated entity, and normal tissues from multiple organ sites for concern for toxicity should be examined prior to the determination of a marker as an appropriate entity for the ligand targeting of therapeutic agents, or for concern for background signal interference prior to the targeting of imaging agents [265].
It is likely that, except somatostatin, all neuropeptides mentioned in this article bind to their overexpressed cognate receptors on cancer cell surfaces to establish an autocrine loop by inducing growth of the tumor cells they originate from, promoting neoplastic growth. Using peptide and nonpeptide antagonists to interrupt this autocrine loop has been proven an effective approach for the inhibition of tumor growth in vivo as well as in vitro in preclinical studies. In the coming years, these anti-autocrine therapies may be tested alone in clinical trials to determine the optimal dose for humans and evaluate the safety profile, or with immunotherapies to generate synergistic effects. For peptide antagonists, due to the inherent serum instability and renal infiltration, modifications such as cyclization or PEGlyation could be applied to improve their pharmacokinetics profile.
With high affinity and specificity, peptide ligands of overexpressed GPCRS are good carriers to deliver cancer-toxic agents. Since most studies involving the targeted delivery of cytotoxic agents were performed in various tumor types excluding lung cancer, future studies could investigate peptide-conjugated nanocarriers/targeted delivery systems in lung neoplasm [266]. For example, besides using vasopressin as targeting peptide in radiopharmaceuticals for tumor imaging and diagnosis, the use of the peptide in targeted delivery system for therapeutic purposes is deemed scientifically viable and appropriate for tumors overexpressing vasopressin receptors [267]. Therefore, the incorporation of vasopressin and/or its agonists to targeted delivery systems could be a future step for research and development of targeted therapy for cancer.
With the highest mortality rate worldwide and the various concerns associated with existing treatments, lung cancer requires urgent attention from the scientific community. Targeting peptide GPCRs that are ectopically expressed in lung tumor, qualify as promising candidates for lung cancer treatment in the near future. The successful results obtained from the use of SSAs in growth hormone-secreting tumors bears witness to this insight.
Abbreviations
SCLC: Small cell lung cancer; NSCLC: Non-small cell lung cancer; GPCR: G-protein coupled receptor; CCK: Cholecystokinin; CCKBR: Cholecystokinin-B/Gastrin receptor; GRP: Gastrin releasing peptide ; GRPR: Gastrin Releasing Peptide receptor; NMB: Neuromedin-B; NMBR: Neuromedin-B receptor; BRS-3: Bombesin receptor type 3 ; BPP: Bradykinin potentiating peptide; B1R: Bradykinin receptor 1; B2R: Bradykinin receptor 2; AVPRs: Arginine vasopressin receptors; V1aR: Vasopressin receptor 1a; V1bR: Vasopressin receptor 1b; V2R: Vasopressin receptor 2; SSTR2: Somatostatin receptor type 2; SSAs: Somatostatin analogs; VEGFR: Vascular endothelial growth factor receptor; EGFR: Epidermal growth factor receptor ; IGF: Insulin-growth factor; GH: Growth hormone; PDGF: Platelet-derived growth factor; bFGF: basic fibroblast growth factor; LAR: (Octreotide/Lanreotide) long acting release; TDD: Targeted drug delivery; PDC: Peptide drug conjugate.
Authors’ contributions
ZXB designed the study. MK and TH researched literatures and analyzed data. MK, TH, and ZXB contributed in preparing the draft of this manuscript. CYL, JW, and BMF made contributions to content. All authors reviewed and approved the final manuscript. MK and TH made equal contributions to this work.
ACKNOWLEDGMENTS
The authors would like to thank all members in YMU-HKBU-PDCC Group, for fruitful suggestions and discussions.
CONFLICTS OF INTEREST
The authors declare no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
FUNDING
The authors have received funding from RGC, HKSAR (HKBU12104415).
REFERENCES
1. Cheng TY, Cramb SM, Baade PD, Youlden DR, Nwogu C, Reid ME. The International Epidemiology of Lung Cancer: Latest Trends, Disparities, and Tumor Characteristics. J Thorac Oncol. 2016; 11:1653–71.
2. Curado MP, Edwards B, Shin HR, Storm H, Ferlay J, Heanue M, Boyle P. Cancer incidence in five continents. Volume IX. IARC Sci Publ. 2008; 160:1–837.
3. Francis H, Solomon B. The current status of targeted therapy for non-small cell lung cancer. Intern Med J. 2010; 40:611–18.
4. Burns KE, Thévenin D. Down-regulation of PAR1 activity with a pHLIP-based allosteric antagonist induces cancer cell death. Biochem J. 2015; 472:287–95.
5. Li S, Huang S, Peng SB. Overexpression of G protein-coupled receptors in cancer cells: involvement in tumor progression. Int J Oncol. 2005; 27:1329–39.
6. Dorsam RT, Gutkind JS. G-protein-coupled receptors and cancer. Nat Rev Cancer. 2007; 7:79–94.
7. Hutchings CJ, Koglin M, Marshall FH. Therapeutic antibodies directed at G protein-coupled receptors. MAbs. 2010; 2:594–606.
8. Stevens RC, Cherezov V, Katritch V, Abagyan R, Kuhn P, Rosen H, Wüthrich K. The GPCR Network: a large-scale collaboration to determine human GPCR structure and function. Nat Rev Drug Discov. 2013; 12:25–34.
9. Sun L, Luo J, Mackey LV, Morris LM, Franko-Tobin LG, LePage KT, Coy DH. Investigation of cancer cell lines for peptide receptor-targeted drug development. J Drug Target. 2011; 19:719–30.
10. Kurzrock R, Gabrail N, Chandhasin C, Moulder S, Smith C, Brenner A, Sankhala K, Mita A, Elian K, Bouchard D, Sarantopoulos J. Safety, pharmacokinetics, and activity of GRN1005, a novel conjugate of angiopep-2, a peptide facilitating brain penetration, and paclitaxel, in patients with advanced solid tumors. Mol Cancer Ther. 2012; 11:308–16.
11. Wang Y, Cheetham AG, Angacian G, Su H, Xie L, Cui H. Peptide-drug conjugates as effective prodrug strategies for targeted delivery. Adv Drug Deliv Rev. 2017; 110–111:112–26.
12. Böhme D, Beck-Sickinger AG. Drug delivery and release systems for targeted tumor therapy. J Pept Sci. 2015; 21:186–200.
13. Lee WH, Loo CY, Leong CR, Young PM, Traini D, Rohanizadeh R. The achievement of ligand-functionalized organic/polymeric nanoparticles for treating multidrug resistant cancer. Expert Opin Drug Deliv. 2017; 14:937–57.
14. Accardo A, Salsano G, Morisco A, Aurilio M, Parisi A, Maione F, Cicala C, Tesauro D, Aloj L, De Rosa G, Morelli G. Peptide-modified liposomes for selective targeting of bombesin receptors overexpressed by cancer cells: a potential theranostic agent. Int J Nanomedicine. 2012; 7:2007–17.
15. Peeters MC, van Westen GJ, Li Q, IJzerman AP. Importance of the extracellular loops in G protein-coupled receptors for ligand recognition and receptor activation. Trends Pharmacol Sci. 2011; 32:35–42.
16. Møller LN, Stidsen CE, Hartmann B, Holst JJ. Somatostatin receptors. Biochim Biophys Acta. 2003; 1616:1–84.
17. Leeb-Lundberg LM, Marceau F, Müller-Esterl W, Pettibone DJ, Zuraw BL. International union of pharmacology. XLV. Classification of the kinin receptor family: from molecular mechanisms to pathophysiological consequences. Pharmacol Rev. 2005; 57:27–77.
18. Decaux G, Soupart A, Vassart G. Non-peptide arginine-vasopressin antagonists: the vaptans. Lancet. 2008; 371:1624–32.
19. Zachary I, Woll PJ, Rozengurt E. A role for neuropeptides in the control of cell proliferation. Dev Biol. 1987; 124:295–308.
20. Sethi T, Langdon S, Smyth J, Rozengurt E. Growth of small cell lung cancer cells: stimulation by multiple neuropeptides and inhibition by broad spectrum antagonists in vitro and in vivo. Cancer Res. 1992 (Suppl ); 52:2737s–42s.
21. Sethi T, Rozengurt E. Multiple neuropeptides stimulate clonal growth of small cell lung cancer: effects of bradykinin, vasopressin, cholecystokinin, galanin, and neurotensin. Cancer Res. 1991; 51:3621–23.
22. Wank SA, Pisegna JR, de Weerth A. Brain and gastrointestinal cholecystokinin receptor family: structure and functional expression. Proc Natl Acad Sci USA. 1992; 89:8691–95.
23. Pisegna JR, de Weerth A, Huppi K, Wank SA. Molecular cloning of the human brain and gastric cholecystokinin receptor: structure, functional expression and chromosomal localization. Biochem Biophys Res Commun. 1992; 189:296–303.
24. Matsumori Y, Katakami N, Ito M, Taniguchi T, Iwata N, Takaishi T, Chihara K, Matsui T. Cholecystokinin-B/gastrin receptor: a novel molecular probe for human small cell lung cancer. Cancer Res. 1995; 55:276–79.
25. Yassin RR. Signaling pathways mediating gastrin’s growth-promoting effects. Peptides. 1999; 20:885–98.
26. Håkanson R, Sundler F. Trophic effects of gastrin. Scand J Gastroenterol Suppl. 1991; 180:130–36.
27. Tracy HJ, Gregory RA. Physiological Properties of a Series of Synthetic Peptides Structurally Related to Gastrin I. Nature. 1964; 204:935–38.
28. Walsh JH. Role of gastrin as a trophic hormone. Digestion. 1990 (Suppl 1); 47:11–16.
29. Dembinski AB, Johnson LR. Stimulation of pancreatic growth by secretin, caerulein, and pentagastrin. Endocrinology. 1980; 106:323–28.
30. Sandvik AK, Waldum HL. CCK-B (gastrin) receptor regulates gastric histamine release and acid secretion. Am J Physiol. 1991; 260:G925–28.
31. Wank SA. G protein-coupled receptors in gastrointestinal physiology. I. CCK receptors: an exemplary family. Am J Physiol. 1998; 274:G607–13.
32. Smith JP, Hamory MW, Verderame MF, Zagon IS. Quantitative analysis of gastrin mRNA and peptide in normal and cancerous human pancreas. Int J Mol Med. 1998; 2:309–15.
33. Behr TM, Jenner N, Béhé M, Angerstein C, Gratz S, Raue F, Becker W. Radiolabeled peptides for targeting cholecystokinin-B/gastrin receptor-expressing tumors. J Nucl Med. 1999; 40:1029–44.
34. Reubi JC, Schaer JC, Waser B. Cholecystokinin(CCK)-A and CCK-B/gastrin receptors in human tumors. Cancer Res. 1997; 57:1377–86.
35. Schmitz F, Otte JM, Stechele HU, Reimann B, Banasiewicz T, Fölsch UR, Schmidt WE, Herzig KH. CCK-B/gastrin receptors in human colorectal cancer. Eur J Clin Invest. 2001; 31:812–20.
36. Tripathi S, Flobak Å, Chawla K, Baudot A, Bruland T, Thommesen L, Kuiper M, Lægreid A. The gastrin and cholecystokinin receptors mediated signaling network: a scaffold for data analysis and new hypotheses on regulatory mechanisms. BMC Syst Biol. 2015; 9:40.
37. Sethi T, Herget T, Wu SV, Walsh JH, Rozengurt E. CCKA and CCKB receptors are expressed in small cell lung cancer lines and mediate Ca2+ mobilization and clonal growth. Cancer Res. 1993; 53:5208–13.
38. Herget T, Sethi T, Wu SV, Walsh JH, Rozengurt E. Cholecystokinin stimulates Ca2+ mobilization and clonal growth in small cell lung cancer through CCKA and CCKB/gastrin receptors. Ann N Y Acad Sci. 1994; 713:283–97.
39. Schaeffer P, Prabonnaud V, Roux M, Gully D, Herbert JM. CCK-JMV-180 acts as an antagonist of the CCKA receptor in the human IMR-32 neuroblastoma cell line. FEBS Lett. 1994; 354:203–06.
40. Ramos-Álvarez I, Moreno P, Mantey SA, Nakamura T, Nuche-Berenguer B, Moody TW, Coy DH, Jensen RT. Insights into bombesin receptors and ligands: highlighting recent advances. Peptides. 2015; 72:128–44.
41. Uehara H, González N, Sancho V, Mantey SA, Nuche-Berenguer B, Pradhan T, Coy DH, Jensen RT. Pharmacology and selectivity of various natural and synthetic bombesin related peptide agonists for human and rat bombesin receptors differs. Peptides. 2011; 32:1685–99.
42. Jensen RT, Battey JF, Spindel ER, Benya RV. International Union of Pharmacology. LXVIII. Mammalian bombesin receptors: nomenclature, distribution, pharmacology, signaling, and functions in normal and disease states. Pharmacol Rev. 2008; 60:1–42.
43. Pelosi G, Volante M, Papotti M, Sonzogni A, Masullo M, Viale G. Peptide receptors in neuroendocrine tumors of the lung as potential tools for radionuclide diagnosis and therapy. Q J Nucl Med Mol Imaging. 2006; 50:272–87.
44. Heasley LE. Autocrine and paracrine signaling through neuropeptide receptors in human cancer. Oncogene. 2001; 20:1563–69.
45. Rettenbacher M, Reubi JC. Localization and characterization of neuropeptide receptors in human colon. Naunyn Schmiedebergs Arch Pharmacol. 2001; 364:291–304.
46. Ferris HA, Carroll RE, Lorimer DL, Benya RV. Location and characterization of the human GRP receptor expressed by gastrointestinal epithelial cells. Peptides. 1997; 18:663–72.
47. Carrieri A, Lacivita E, Belviso BD, Caliandro R, Mastrorilli P, Gallo V, Niso M, Leopoldo M. Structural determinants in the binding of BB2 receptor ligands: in silico, X-ray and NMR studies in PD176252 analogues. Curr Top Med Chem. 2017; 17:1599-1610.
48. Ohki-Hamazaki H, Watase K, Yamamoto K, Ogura H, Yamano M, Yamada K, Maeno H, Imaki J, Kikuyama S, Wada E, Wada K. Mice lacking bombesin receptor subtype-3 develop metabolic defects and obesity. Nature. 1997; 390:165–69.
49. Weber HC. Regulation and signaling of human bombesin receptors and their biological effects. Curr Opin Endocrinol Diabetes Obes. 2009; 16:66–71.
50. Reubi JC, Wenger S, Schmuckli-Maurer J, Schaer JC, Gugger M. Bombesin receptor subtypes in human cancers: detection with the universal radioligand (125)I-[D-TYR(6), beta-ALA(11), PHE(13), NLE(14)] bombesin(6-14). Clin Cancer Res. 2002; 8:1139–46.
51. Cornelio DB, Roesler R, Schwartsmann G. Gastrin-releasing peptide receptor as a molecular target in experimental anticancer therapy. Ann Oncol. 2007; 18:1457–66.
52. Sancho V, Di Florio A, Moody TW, Jensen RT. Bombesin receptor-mediated imaging and cytotoxicity: review and current status. Curr Drug Deliv. 2011; 8:79–134.
53. Moody TW, Moreno P, Jensen RT. Neuropeptides as lung cancer growth factors. Peptides. 2015; 72:106–11.
54. Moody TW, Chan D, Fahrenkrug J, Jensen RT. Neuropeptides as autocrine growth factors in cancer cells. Curr Pharm Des. 2003; 9:495–509.
55. Schulz S, Röcken C, Schulz S. Immunohistochemical detection of bombesin receptor subtypes GRP-R and BRS-3 in human tumors using novel antipeptide antibodies. Virchows Arch. 2006; 449:421–27.
56. Cuttitta F, Carney DN, Mulshine J, Moody TW, Fedorko J, Fischler A, Minna JD. Bombesin-like peptides can function as autocrine growth factors in human small-cell lung cancer. Nature. 1985; 316:823–26.
57. Zhang H, Chen J, Waldherr C, Hinni K, Waser B, Reubi JC, Maecke HR. Synthesis and evaluation of bombesin derivatives on the basis of pan-bombesin peptides labeled with indium-111, lutetium-177, and yttrium-90 for targeting bombesin receptor-expressing tumors. Cancer Res. 2004; 64:6707–15.
58. Nock BA, Nikolopoulou A, Galanis A, Cordopatis P, Waser B, Reubi JC, Maina T. Potent bombesin-like peptides for GRP-receptor targeting of tumors with 99mTc: a preclinical study. J Med Chem. 2005; 48:100–10.
59. Lozza C, Navarro-Teulon I, Pèlegrin A, Pouget JP, Vivès E. Peptides in receptor-mediated radiotherapy: from design to the clinical application in cancers. Front Oncol. 2013; 3:247.
60. Yu Z, Ananias HJ, Carlucci G, Hoving HD, Helfrich W, Dierckx RA, Wang F, de Jong IJ, Elsinga PH. An update of radiolabeled bombesin analogs for gastrin-releasing peptide receptor targeting. Curr Pharm Des. 2013; 19:3329–41.
61. Hohla F, Schally AV. Targeting gastrin releasing peptide receptors: new options for the therapy and diagnosis of cancer. Cell Cycle. 2010; 9:1738–41.
62. Popp I, Del Pozzo L, Waser B, Reubi JC, Meyer PT, Maecke HR, Gourni E. Approaches to improve metabolic stability of a statine-based GRP receptor antagonist. Nucl Med Biol. 2017; 45:22–29.
63. Campos MM, Leal PC, Yunes RA, Calixto JB. Non-peptide antagonists for kinin B1 receptors: new insights into their therapeutic potential for the management of inflammation and pain. Trends Pharmacol Sci. 2006; 27:646–51.
64. Gobeil F Jr, Sirois P, Regoli D. Preclinical pharmacology, metabolic stability, pharmacokinetics and toxicology of the peptidic kinin B1 receptor antagonist R-954. Peptides. 2014; 52:82–89.
65. Marceau F, Regoli D. Bradykinin receptor ligands: therapeutic perspectives. Nat Rev Drug Discov. 2004; 3:845–52.
66. Lacoste B, Tong XK, Lahjouji K, Couture R, Hamel E. Cognitive and cerebrovascular improvements following kinin B1 receptor blockade in Alzheimer’s disease mice. J Neuroinflammation. 2013; 10:57.
67. Marceau F. Kinin B1 receptors: a review. Immunopharmacology. 1995; 30:1–26.
68. Rodi D, Buzzi A, Barbieri M, Zucchini S, Verlengia G, Binaschi A, Regoli D, Boschi A, Ongali B, Couture R, Simonato M. Bradykinin B2 receptors increase hippocampal excitability and susceptibility to seizures in mice. Neuroscience. 2013; 248:392–402.
69. Rodi D, Couture R, Ongali B, Simonato M. Targeting kinin receptors for the treatment of neurological diseases. Curr Pharm Des. 2005; 11:1313–26.
70. Chee J, Naran A, Misso NL, Thompson PJ, Bhoola KD. Expression of tissue and plasma kallikreins and kinin B1 and B2 receptors in lung cancer. Biol Chem. 2008; 389:1225–33.
71. Molina L, Matus CE, Astroza A, Pavicic F, Tapia E, Toledo C, Perez JA, Nualart F, Gonzalez CB, Burgos RA, Figueroa CD, Ehrenfeld P, Poblete MT. Stimulation of the bradykinin B(1) receptor induces the proliferation of estrogen-sensitive breast cancer cells and activates the ERK1/2 signaling pathway. Breast Cancer Res Treat. 2009; 118:499–510.
72. Taub JS, Guo R, Leeb-Lundberg LM, Madden JF, Daaka Y. Bradykinin receptor subtype 1 expression and function in prostate cancer. Cancer Res. 2003; 63:2037–41.
73. Toledo C, Matus CE, Barraza X, Arroyo P, Ehrenfeld P, Figueroa CD, Bhoola KD, Del Pozo M, Poblete MT. Expression of HER2 and bradykinin B1 receptors in precursor lesions of gallbladder carcinoma. World J Gastroenterol. 2012; 18:1208–15.
74. Beck C, Piontek G, Haug A, Bas M, Knopf A, Stark T, Mißlbeck M, Rudelius M, Reiter R, Brandstetter M, Pickhard A. The kallikrein-kinin-system in head and neck squamous cell carcinoma (HNSCC) and its role in tumour survival, invasion, migration and response to radiotherapy. Oral Oncol. 2012; 48:1208–19.
75. Yang WH, Chang JT, Hsu SF, Li TM, Cho DY, Huang CY, Fong YC, Tang CH. Bradykinin enhances cell migration in human chondrosarcoma cells through BK receptor signaling pathways. J Cell Biochem. 2010; 109:82–92.
76. Zelawski W, Machnik G, Nowaczyk G, Plewka D, Lorenc Z, Sosada K, Stadnicki A. Expression and localisation of kinin receptors in colorectal polyps. Int Immunopharmacol. 2006; 6:997–1002.
77. Moodley R, Snyman C, Odhav B, Bhoola KD. Visualisation of transforming growth factor-beta 1, tissue kallikrein, and kinin and transforming growth factor-beta receptors on human clear-cell renal carcinoma cells. Biol Chem. 2005; 386:375–82.
78. Raidoo DM, Sawant S, Mahabeer R, Bhoola KD. Kinin receptors are expressed in human astrocytic tumour cells. Immunopharmacology. 1999; 43:255–63.
79. Nicoletti NF, Erig TC, Zanin RF, Pereira TC, Bogo MR, Campos MM, Morrone FB. Mechanisms involved in kinin-induced glioma cells proliferation: the role of ERK1/2 and PI3K/Akt pathways. J Neurooncol. 2014; 120:235–44.
80. Lin KS, Amouroux G, Pan J, Zhang Z, Jenni S, Lau J, Liu Z, Hundal-Jabal N, Colpo N, Bénard F. Comparative studies of three 68Ga-labeled [Des-Arg10]kallidin derivatives for imaging bradykinin B1 receptor expression with PET. J Nucl Med. 2015; 56:622–27.
81. Liu Z, Amouroux G, Zhang Z, Pan J, Hundal-Jabal N, Colpo N, Lau J, Perrin DM, Bénard F, Lin KS. (18)F-trifluoroborate derivatives of [des-arg(10)]kallidin for imaging bradykinin b1 receptor expression with positron emission tomography. Mol Pharm. 2015; 12:974–82.
82. Thibonnier M, Berti-Mattera LN, Dulin N, Conarty DM, Mattera R. Signal transduction pathways of the human V1-vascular, V2-renal, V3-pituitary vasopressin and oxytocin receptors. Prog Brain Res. 1998; 119:147–61.
83. Meyer-Lindenberg A, Domes G, Kirsch P, Heinrichs M. Oxytocin and vasopressin in the human brain: social neuropeptides for translational medicine. Nat Rev Neurosci. 2011; 12:524–38.
84. Turner RA, Pierce JG, du Vigneaud V. The purification and the amino acid content of vasopressin preparations. J Biol Chem. 1951; 191:21–28.
85. Barberis C, Morin D, Durroux T, Mouillac B, Guillon G, Seyer R, Hibert M, Tribollet E, Manning M. Molecular pharmacology of AVP and OT receptors and therapeutic potential. Drug News Perspect. 1999; 12:279–92.
86. Phillips PA, Abrahams JM, Kelly JM, Mooser V, Trinder D, Johnston CI. Localization of vasopressin binding sites in rat tissues using specific V1 and V2 selective ligands. Endocrinology. 1990; 126:1478–84.
87. Antoni FA. Characterization of high affinity binding sites for vasopressin in bovine adrenal medulla. Neuropeptides. 1984; 4:413–20.
88. Hanley MR, Benton HP, Lightman SL, Todd K, Bone EA, Fretten P, Palmer S, Kirk CJ, Michell RH. A vasopressin-like peptide in the mammalian sympathetic nervous system. Nature. 1984; 309:258–61.
89. Serradeil-Le Gal C, Wagnon J, Valette G, Garcia G, Pascal M, Maffrand JP, Le Fur G. Nonpeptide vasopressin receptor antagonists: development of selective and orally active V1a, V2 and V1b receptor ligands. Prog Brain Res. 2002; 139:197–210.
90. Jard S. Vasopressin receptors. A historical survey. Adv Exp Med Biol. 1998; 449:1–13.
91. Grazzini E, Lodboerer AM, Perez-Martin A, Joubert D, Guillon G. Molecular and functional characterization of V1b vasopressin receptor in rat adrenal medulla. Endocrinology. 1996; 137:3906–14.
92. Knepper MA, Inoue T. Regulation of aquaporin-2 water channel trafficking by vasopressin. Curr Opin Cell Biol. 1997; 9:560–64.
93. Péqueux C, Keegan BP, Hagelstein MT, Geenen V, Legros JJ, North WG. Oxytocin- and vasopressin-induced growth of human small-cell lung cancer is mediated by the mitogen-activated protein kinase pathway. Endocr Relat Cancer. 2004; 11:871–85.
94. North WG, Fay MJ, Du J. All three vasopressin receptor sub-types are expressed by small-cell carcinoma. Adv Exp Med Biol. 1998; 449:335–38.
95. North WG, Fay MJ, Longo KA, Du J. Expression of all known vasopressin receptor subtypes by small cell tumors implies a multifaceted role for this neuropeptide. Cancer Res. 1998; 58:1866–71.
96. Péqueux C, Breton C, Hagelstein MT, Geenen V, Legros JJ. Oxytocin receptor pattern of expression in primary lung cancer and in normal human lung. Lung Cancer. 2005; 50:177–88.
97. Ocejo-Garcia M, Ahmed SI, Coulson JM, Woll PJ. Use of RT-PCR to detect co-expression of neuropeptides and their receptors in lung cancer. Lung Cancer. 2001; 33:1–9.
98. de Keyzer Y, René P, Beldjord C, Lenne F, Bertagna X. Overexpression of vasopressin (V3) and corticotrophin-releasing hormone receptor genes in corticotroph tumours. Clin Endocrinol (Oxf). 1998; 49:475–82.
99. Dahia PL, Ahmed-Shuaib A, Jacobs RA, Chew SL, Honegger J, Fahlbusch R, Besser GM, Grossman AB. Vasopressin receptor expression and mutation analysis in corticotropin-secreting tumors. J Clin Endocrinol Metab. 1996; 81:1768–71.
100. Hoyer D, Bell GI, Berelowitz M, Epelbaum J, Feniuk W, Humphrey PP, O’Carroll AM, Patel YC, Schonbrunn A, Taylor JE, Reisine T. Classification and nomenclature of somatostatin receptors. Trends Pharmacol Sci. 1995; 16:86–88.
101. Florio T, Schettini G. Multiple intracellular effectors modulate physiological functions of the cloned somatostatin receptors. J Mol Endocrinol. 1996; 17:89–100.
102. Patel YC. Somatostatin and its receptor family. Front Neuroendocrinol. 1999; 20:157–98.
103. Yamada Y, Kagimoto S, Kubota A, Yasuda K, Masuda K, Someya Y, Ihara Y, Li Q, Imura H, Seino S, Seino Y. Cloning, functional expression and pharmacological characterization of a fourth (hSSTR4) and a fifth (hSSTR5) human somatostatin receptor subtype. Biochem Biophys Res Commun. 1993; 195:844–52.
104. Kumar U. Cross-talk and modulation of signaling between somatostatin and growth factor receptors. Endocrine. 2011; 40:168–80.
105. Epelbaum J, Guillou JL, Gastambide F, Hoyer D, Duron E, Viollet C. Somatostatin, Alzheimer’s disease and cognition: an old story coming of age? Prog Neurobiol. 2009; 89:153–61.
106. Lamberts SW, Krenning EP, Reubi JC. The role of somatostatin and its analogs in the diagnosis and treatment of tumors. Endocr Rev. 1991; 12:450–82.
107. Ferone D, van Hagen PM, Pivonello R, Colao A, Lamberts SW, Hofland LJ. Physiological and pathophysiological role of somatostatin receptors in the human thymus. Eur J Endocrinol. 2000 (Suppl 1); 143:S27–34.
108. Guillermet-Guibert J, Lahlou H, Cordelier P, Bousquet C, Pyronnet S, Susini C. Physiology of somatostatin receptors. J Endocrinol Invest. 2005 (Suppl International); 28:5–9.
109. Florio T, Thellung S, Schettini G. Intracellular transducing mechanisms coupled to brain somatostatin receptors. Pharmacol Res. 1996; 33:297–305.
110. Pintér E, Helyes Z, Szolcsányi J. Inhibitory effect of somatostatin on inflammation and nociception. Pharmacol Ther. 2006; 112:440–56.
111. Callison JC, Walker RC, Massion PP. Somatostatin receptors in lung cancer: from function to molecular imaging and therapeutics. J Lung Cancer. 2011; 10:69-76.
112. Herlin G, Kölbeck KG, Menzel PL, Svensson L, Aspelin P, Capitanio A, Axelsson R. Quantitative assessment of 99mTc-depreotide uptake in patients with non-small-cell lung cancer: immunohistochemical correlations. Acta Radiol. 2009; 50:902–08.
113. Kumar U, Grigorakis SI, Watt HL, Sasi R, Snell L, Watson P, Chaudhari S. Somatostatin receptors in primary human breast cancer: quantitative analysis of mRNA for subtypes 1–5 and correlation with receptor protein expression and tumor pathology. Breast Cancer Res Treat. 2005; 92:175–86.
114. He Y, Yuan XM, Lei P, Wu S, Xing W, Lan XL, Zhu HF, Huang T, Wang GB, An R, Zhang YX, Shen GX. The antiproliferative effects of somatostatin receptor subtype 2 in breast cancer cells. Acta Pharmacol Sin. 2009; 30:1053–59.
115. Mazzucchelli R, Morichetti D, Santinelli A, Scarpelli M, Bono AV, Lopez-Beltran A, Cheng L, Montironi R. Immunohistochemical expression and localization of somatostatin receptor subtypes in androgen ablated prostate cancer. Anal Cell Pathol (Amst). 2010; 33:27–36.
116. Massa A, Barbieri F, Aiello C, Arena S, Pattarozzi A, Pirani P, Corsaro A, Iuliano R, Fusco A, Zona G, Spaziante R, Florio T, Schettini G. The expression of the phosphotyrosine phosphatase DEP-1/PTPeta dictates the responsivity of glioma cells to somatostatin inhibition of cell proliferation. J Biol Chem. 2004; 279:29004–12.
117. Lamszus K, Meyerhof W, Westphal M. Somatostatin and somatostatin receptors in the diagnosis and treatment of gliomas. J Neurooncol. 1997; 35:353–64.
118. Ullrich M, Bergmann R, Peitzsch M, Zenker EF, Cartellieri M, Bachmann M, Ehrhart-Bornstein M, Block NL, Schally AV, Eisenhofer G, Bornstein SR, Pietzsch J, Ziegler CG. Multimodal Somatostatin Receptor Theranostics Using [(64)Cu]Cu-/[(177)Lu]Lu-DOTA-(Tyr(3))octreotate and AN-238 in a Mouse Pheochromocytoma Model. Theranostics. 2016; 6:650–65.
119. Szepeshazi K, Schally AV, Nagy A, Wagner BW, Bajo AM, Halmos G. Preclinical evaluation of therapeutic effects of targeted cytotoxic analogs of somatostatin and bombesin on human gastric carcinomas. Cancer. 2003; 98:1401–10.
120. Hu C, Yi C, Hao Z, Cao S, Li H, Shao X, Zhang J, Qiao T, Fan D. The effect of somatostatin and SSTR3 on proliferation and apoptosis of gastric cancer cells. Cancer Biol Ther. 2004; 3:726–30.
121. Reubi JC, Maurer R, Klijn JG, Stefanko SZ, Foekens JA, Blaauw G, Blankenstein MA, Lamberts SW. High incidence of somatostatin receptors in human meningiomas: biochemical characterization. J Clin Endocrinol Metab. 1986; 63:433–38.
122. Ji XQ, Ruan XJ, Chen H, Chen G, Li SY, Yu B. Somatostatin analogues in advanced hepatocellular carcinoma: an updated systematic review and meta-analysis of randomized controlled trials. Med Sci Monit. 2011; 17:RA169–76.
123. Barbieri F, Bajetto A, Pattarozzi A, Gatti M, Würth R, Thellung S, Corsaro A, Villa V, Nizzari M, Florio T. Peptide receptor targeting in cancer: the somatostatin paradigm. Int J Pept. 2013; 2013:926295.
124. Hofland LJ. Responsiveness to somatostatin analog treatment and potentials of novel somatostatin analog. J Endocrinol Invest. 2003 (Suppl ); 26:8–13.
125. Kanakis G, Grimelius L, Spathis A, Tringidou R, Rassidakis GZ, Öberg K, Kaltsas G, Tsolakis AV. Expression of Somatostatin Receptors 1-5 and Dopamine Receptor 2 in Lung Carcinoids: Implications for a Therapeutic Role. Neuroendocrinology. 2015; 101:211–22.
126. Hofland LJ, Liu Q, Van Koetsveld PM, Zuijderwijk J, Van Der Ham F, De Krijger RR, Schonbrunn A, Lamberts SW. Immunohistochemical detection of somatostatin receptor subtypes sst1 and sst2A in human somatostatin receptor positive tumors. J Clin Endocrinol Metab. 1999; 84:775–80.
127. Vikić-Topić S, Raisch KP, Kvols LK, Vuk-Pavlović S. Expression of somatostatin receptor subtypes in breast carcinoma, carcinoid tumor, and renal cell carcinoma. J Clin Endocrinol Metab. 1995; 80:2974–79.
128. Righi L, Volante M, Tavaglione V, Billè A, Daniele L, Angusti T, Inzani F, Pelosi G, Rindi G, Papotti M. Somatostatin receptor tissue distribution in lung neuroendocrine tumours: a clinicopathologic and immunohistochemical study of 218 ‘clinically aggressive’ cases. Ann Oncol. 2010; 21:548–55.
129. Geijer T, Folkesson R, Rehfeld JF, Monstein HJ. Expression of the cholecystokinin gene in a human (small-cell) lung carcinoma cell-line. FEBS Lett. 1990; 270:30–32.
130. Alexander RW, Upp JR Jr, Poston GJ, Gupta V, Townsend CM Jr, Thompson JC. Effects of bombesin on growth of human small cell lung carcinoma in vivo. Cancer Res. 1988; 48:1439–41.
131. Castellone MD, Laukkanen MO, Teramoto H, Bellelli R, Alì G, Fontanini G, Santoro M, Gutkind JS. Cross talk between the bombesin neuropeptide receptor and Sonic hedgehog pathways in small cell lung carcinoma. Oncogene. 2015; 34:1679–87.
132. Bunn PA Jr, Dienhart DG, Chan D, Tagawa M, Jewett P. Effects of neuropeptides on human lung and breast cancer cells. J Natl Cancer Inst Monogr. 1992; 13:145–51.
133. Sausville E, Carney D, Battey J. The human vasopressin gene is linked to the oxytocin gene and is selectively expressed in a cultured lung cancer cell line. J Biol Chem. 1985; 260:10236–41.
134. Woll PJ, Rozengurt E. Neuropeptides as growth regulators. Br Med Bull. 1989; 45:492–505.
135. Moody TW, Nuche-Berenguer B, Moreno P, Jensen RT. CI-988 Inhibits EGFR Transactivation and Proliferation Caused by Addition of CCK/Gastrin to Lung Cancer Cells. J Mol Neurosci. 2015; 56:663–72.
136. Ishizuka J, Townsend CM Jr, Bold RJ, Martinez J, Rodriguez M, Thompson JC. Effects of gastrin on 3′,5′-cyclic adenosine monophosphate, intracellular calcium, and phosphatidylinositol hydrolysis in human colon cancer cells. Cancer Res. 1994; 54:2129–35.
137. Park HJ, Kim MK, Choi KS, Jeong JW, Bae SK, Kim HJ, Bae MK. Neuromedin B receptor antagonism inhibits migration, invasion, and epithelial-mesenchymal transition of breast cancer cells. Int J Oncol. 2016; 49:934–42.
138. Corjay MH, Dobrzanski DJ, Way JM, Viallet J, Shapira H, Worland P, Sausville EA, Battey JF. Two distinct bombesin receptor subtypes are expressed and functional in human lung carcinoma cells. J Biol Chem. 1991; 266:18771–79.
139. Searovic P, Alonso M, Oses C, Pereira-Flores K, Velarde V, Saez CG. Effect of tamoxifen and retinoic acid on bradykinin induced proliferation in MCF-7 cells. J Cell Biochem. 2009; 106:473–81.
140. Barki-Harrington L, Bookout AL, Wang G, Lamb ME, Leeb-Lundberg LM, Daaka Y. Requirement for direct cross-talk between B1 and B2 kinin receptors for the proliferation of androgen-insensitive prostate cancer PC3 cells. Biochem J. 2003; 371:581–87.
141. Bolignano D, Medici MA, Coppolino G, Sciortino MT, Merlo FM, Campo S, Donato V, Venuti A, Sturiale A, Zaccaria D, Buemi A, Lacquaniti A, Buemi M. Aquaretic inhibits renal cancer proliferation: role of vasopressin receptor-2 (V2-R). Urol Oncol. 2010; 28:642–47.
142. Friedmann AS, Malott KA, Memoli VA, Pai SI, Yu XM, North WG. Products of vasopressin gene expression in small-cell carcinoma of the lung. Br J Cancer. 1994; 69:260–63.
143. Seckl MJ, Higgins T, Widmer F, Rozengurt E. [D-Arg1,D-Trp5,7,9,Leu11]substance P: a novel potent inhibitor of signal transduction and growth in vitro and in vivo in small cell lung cancer cells. Cancer Res. 1997; 57:51–54.
144. Weber HC, Walters J, Leyton J, Casibang M, Purdom S, Jensen RT, Coy DH, Ellis C, Clark G, Moody TW. A bombesin receptor subtype-3 peptide increases nuclear oncogene expression in a MEK-1 dependent manner in human lung cancer cells. Eur J Pharmacol. 2001; 412:13–20.
145. Moody TW, Nuche-Berenguer B, Nakamura T, Jensen RT. EGFR Transactivation by Peptide G Protein-Coupled Receptors in Cancer. Curr Drug Targets. 2016; 17:520–28.
146. Moody TW, Sancho V, di Florio A, Nuche-Berenguer B, Mantey S, Jensen RT. Bombesin receptor subtype-3 agonists stimulate the growth of lung cancer cells and increase EGF receptor tyrosine phosphorylation. Peptides. 2011; 32:1677–84.
147. Yoder DG, Moody TW. High affinity binding of cholecystokinin to small cell lung cancer cells. Peptides. 1987; 8:103–07.
148. Staley J, Fiskum G, Moody TW. Cholecystokinin elevates cytosolic calcium in small cell lung cancer cells. Biochem Biophys Res Commun. 1989; 163:605–10.
149. Yu HG, Schäfer H, Mergler S, Müerköster S, Cramer T, Höcker M, Herzig KH, Schmidt WE, Schmitz F. Valine-286 residue in the third intracellular loop of the cholecystokinin 2 receptor exerts a pivotal role in cholecystokinin 2 receptor mediated intracellular signal transduction in human colon cancer cells. Cell Signal. 2005; 17:1505–15.
150. Piiper A, Elez R, You SJ, Kronenberger B, Loitsch S, Roche S, Zeuzem S. Cholecystokinin stimulates extracellular signal-regulated kinase through activation of the epidermal growth factor receptor, Yes, and protein kinase C. Signal amplification at the level of Raf by activation of protein kinase Cepsilon. J Biol Chem. 2003; 278:7065–72.
151. Lee S, Qiao J, Paul P, Chung DH. Integrin β1 is critical for gastrin-releasing peptide receptor-mediated neuroblastoma cell migration and invasion. Surgery. 2013; 154:369–75.
152. Moody TW, Berna MJ, Mantey S, Sancho V, Ridnour L, Wink DA, Chan D, Giaccone G, Jensen RT. Neuromedin B receptors regulate EGF receptor tyrosine phosphorylation in lung cancer cells. Eur J Pharmacol. 2010; 637:38–45.
153. Roelle S, Grosse R, Buech T, Chubanov V, Gudermann T. Essential role of Pyk2 and Src kinase activation in neuropeptide-induced proliferation of small cell lung cancer cells. Oncogene. 2008; 27:1737–48.
154. Moody TW, Cuttitta F. Growth factor and peptide receptors in small cell lung cancer. Life Sci. 1993; 52:1161–73.
155. Woll PJ, Rozengurt E. Multiple neuropeptides mobilise calcium in small cell lung cancer: effects of vasopressin, bradykinin, cholecystokinin, galanin and neurotensin. Biochem Biophys Res Commun. 1989; 164:66–73.
156. MacKinnon AC, Tufail-Hanif U, Wheatley M, Rossi AG, Haslett C, Seckl M, Sethi T. Targeting V1A-vasopressin receptors with [Arg6, D-Trp7,9, NmePhe8]-substance P (6-11) identifies a strategy to develop novel anti-cancer therapies. Br J Pharmacol. 2009; 156:36–47.
157. Rozengurt E, Sinnett-Smith J. Early signals underlying the induction of the c-fos and c-myc genes in quiescent fibroblasts: studies with bombesin and other growth factors. Prog Nucleic Acid Res Mol Biol. 1988; 35:261–95.
158. Ryder NM, Guha S, Hines OJ, Reber HA, Rozengurt E. G protein-coupled receptor signaling in human ductal pancreatic cancer cells: neurotensin responsiveness and mitogenic stimulation. J Cell Physiol. 2001; 186:53–64.
159. Drube S, Liebmann C. In various tumour cell lines the peptide bradykinin B(2) receptor antagonist, Hoe 140 (Icatibant), may act as mitogenic agonist. Br J Pharmacol. 2000; 131:1553–60.
160. Greco S, Storelli C, Marsigliante S. Protein kinase C (PKC)-delta/-epsilon mediate the PKC/Akt-dependent phosphorylation of extracellular signal-regulated kinases 1 and 2 in MCF-7 cells stimulated by bradykinin. J Endocrinol. 2006; 188:79–89.
161. Hong M, Moody TW. Vasopressin elevates cytosolic calcium in small cell lung cancer cells. Peptides. 1991; 12:1315–19.
162. Moore TC, Jepeal LI, Boylan MO, Singh SK, Boyd N, Beer DG, Chang AJ, Wolfe MM. Gastrin stimulates receptor-mediated proliferation of human esophageal adenocarcinoma cells. Regul Pept. 2004; 120:195–203.
163. Rozengurt E. Early signals in the mitogenic response. Science. 1986; 234:161–66.
164. Rozengurt E. Neuropeptides as cellular growth factors: role of multiple signalling pathways. Eur J Clin Invest. 1991; 21:123–34.
165. Rozengurt E. Signal transduction pathways in the mitogenic response to G protein-coupled neuropeptide receptor agonists. J Cell Physiol. 1998; 177:507–17.
166. Kimura A, Kihara T, Ohkura R, Ogiwara K, Takahashi T. Localization of bradykinin B(2) receptor in the follicles of porcine ovary and increased expression of matrix metalloproteinase-3 and -20 in cultured granulosa cells by bradykinin treatment. Biol Reprod. 2001; 65:1462–70.
167. Lu DY, Leung YM, Huang SM, Wong KL. Bradykinin-induced cell migration and COX-2 production mediated by the bradykinin B1 receptor in glioma cells. J Cell Biochem. 2010; 110:141–50.
168. Mishra P, Senthivinayagam S, Rangasamy V, Sondarva G, Rana B. Mixed lineage kinase-3/JNK1 axis promotes migration of human gastric cancer cells following gastrin stimulation. Mol Endocrinol. 2010; 24:598–607.
169. Ding J, Yu JP, Li D, Yu HG, Luo HS, Wei WZ. [Effect of gastrin on invasiveness of human colon cancer cells]. Zhonghua Zhong Liu Za Zhi. 2005; 27:213–15.
170. Watson SA, Grabowska AM, El-Zaatari M, Takhar A. Gastrin - active participant or bystander in gastric carcinogenesis? Nat Rev Cancer. 2006; 6:936–46.
171. Park HJ, Kim SR, Kim MK, Choi KS, Jang HO, Yun I, Bae SK, Bae MK. Neuromedin B receptor antagonist suppresses tumor angiogenesis and tumor growth in vitro and in vivo. Cancer Lett. 2011; 312:117–27.
172. Velarde V, Ullian ME, Morinelli TA, Mayfield RK, Jaffa AA. Mechanisms of MAPK activation by bradykinin in vascular smooth muscle cells. Am J Physiol. 1999; 277:C253–61.
173. Hammerschmidt S, Kuhn H, Gessner C, Seyfarth HJ, Wirtz H. Stretch-induced alveolar type II cell apoptosis: role of endogenous bradykinin and PI3K-Akt signaling. Am J Respir Cell Mol Biol. 2007; 37:699–705.
174. Rai U, Thrimawithana TR, Valery C, Young SA. Therapeutic uses of somatostatin and its analogues: current view and potential applications. Pharmacol Ther. 2015; 152:98–110.
175. Florio T. Molecular mechanisms of the antiproliferative activity of somatostatin receptors (SSTRs) in neuroendocrine tumors. Front Biosci. 2008; 13:822–40.
176. Oberg K. Chemotherapy and biotherapy in the treatment of neuroendocrine tumours. Ann Oncol. 2001 (Suppl 2); 12:S111–14.
177. Tartarone A, Lerose R, Aieta M. Somatostatin Analog Therapy in Small Cell Lung Cancer. Semin Nucl Med. 2016; 46:239–42.
178. Lapa C, Hänscheid H, Wild V, Pelzer T, Schirbel A, Werner RA, Droll S, Herrmann K, Buck AK, Lückerath K. Somatostatin receptor expression in small cell lung cancer as a prognostic marker and a target for peptide receptor radionuclide therapy. Oncotarget. 2016; 7:20033–40. https://doi.org/10.18632/oncotarget.7706.
179. Fino KK, Matters GL, McGovern CO, Gilius EL, Smith JP. Downregulation of the CCK-B receptor in pancreatic cancer cells blocks proliferation and promotes apoptosis. Am J Physiol Gastrointest Liver Physiol. 2012; 302:G1244–52.
180. Hughes J, Boden P, Costall B, Domeney A, Kelly E, Horwell DC, Hunter JC, Pinnock RD, Woodruff GN. Development of a class of selective cholecystokinin type B receptor antagonists having potent anxiolytic activity. Proc Natl Acad Sci USA. 1990; 87:6728–32.
181. Huang SC, Zhang L, Chiang HC, Wank SA, Maton PN, Gardner JD, Jensen RT. Benzodiazepine analogues L365,260 and L364,718 as gastrin and pancreatic CCK receptor antagonists. Am J Physiol. 1989; 257:G169–74.
182. Bläker M, Arrenberg P, Stange I, Schulz M, Burghardt S, Michaelis H, Pace A, Greten H, von Schrenck T, de Weerth A. The cholecystokinin2-receptor mediates calcitonin secretion, gene expression, and proliferation in the human medullary thyroid carcinoma cell line, TT. Regul Pept. 2004; 118:111–17.
183. Lango MN, Dyer KF, Lui VW, Gooding WE, Gubish C, Siegfried JM, Grandis JR. Gastrin-releasing peptide receptor-mediated autocrine growth in squamous cell carcinoma of the head and neck. J Natl Cancer Inst. 2002; 94:375–83.
184. Kim S, Hu W, Kelly DR, Hellmich MR, Evers BM, Chung DH. Gastrin-releasing peptide is a growth factor for human neuroblastomas. Ann Surg. 2002; 235:621–29.
185. Eden JM, Hall MD, Higginbottom M, Horwell DC, Howson W, Hughes J, Jordan RE, Lewthwaite RA, Martin K, McKnight AT, O’Toole JC, Pinnock RD, Pritchard MC, et al. OToole JC, Pinnock RD, Pritchard MC, SumanChauhan N, Williams SC. PD 165929 - the first high affinity non-peptide neuromedin-B (NMB) receptor selective antagonist. Bioorg Med Chem Lett. 1996; 6:2617–22.
186. Ashwood V, Brownhill V, Higginbottom M, Horwell DC, Hughes J, Lewthwaite RA, McKnight AT, Pinnock RD, Pritchard MC, Suman-Chauhan N, Webb C, Williams SC. PD 176252—the first high affinity non-peptide gastrin-releasing peptide (BB2) receptor antagonist. Bioorg Med Chem Lett. 1998; 8:2589–94.
187. Moody TW, Leyton J, Garcia-Marin L, Jensen RT. Nonpeptide gastrin releasing peptide receptor antagonists inhibit the proliferation of lung cancer cells. Eur J Pharmacol. 2003; 474:21–29.
188. González N, Mantey SA, Pradhan TK, Sancho V, Moody TW, Coy DH, Jensen RT. Characterization of putative GRP- and NMB-receptor antagonist’s interaction with human receptors. Peptides. 2009; 30:1473–86.
189. Moody TW, Mantey SA, Moreno P, Nakamura T, Lacivita E, Leopoldo M, Jensen RT. ML-18 is a non-peptide bombesin receptor subtype-3 antagonist which inhibits lung cancer growth. Peptides. 2015; 64:55–61.
190. Hohla F, Schally AV, Kanashiro CA, Buchholz S, Baker B, Kannadka C, Moder A, Aigner E, Datz C, Halmos G. Growth inhibition of non-small-cell lung carcinoma by BN/GRP antagonist is linked with suppression of K-Ras, COX-2, and pAkt. Proc Natl Acad Sci USA. 2007; 104:18671–76.
191. Szepeshazi K, Schally AV, Rick FG, Block NL, Vidaurre I, Halmos G, Szalontay L. Powerful inhibition of in-vivo growth of experimental hepatic cancers by bombesin/gastrin-releasing peptide antagonist RC-3940-II. Anticancer Drugs. 2012; 23:906–13.
192. Rick FG, Buchholz S, Schally AV, Szalontay L, Krishan A, Datz C, Stadlmayr A, Aigner E, Perez R, Seitz S, Block NL, Hohla F. Combination of gastrin-releasing peptide antagonist with cytotoxic agents produces synergistic inhibition of growth of human experimental colon cancers. Cell Cycle. 2012; 11:2518–25.
193. Zhou J, Chen J, Zhong R, Mokotoff M, Shultz LD, Ball ED. Targeting gastrin-releasing peptide receptors on small cell lung cancer cells with a bispecific molecule that activates polyclonal T lymphocytes. Clin Cancer Res. 2006; 12:2224–31.
194. Whalley ET, Hanson WL, Stewart JM, Gera L. Oral activity of peptide bradykinin antagonists following intragastric administration in the rat. Can J Physiol Pharmacol. 1997; 75:629–32.
195. Stewart JM, Gera L, Chan DC, Bunn PA Jr, York EJ, Simkeviciene V, Helfrich B. Bradykinin-related compounds as new drugs for cancer and inflammation. Can J Physiol Pharmacol. 2002; 80:275–80.
196. Rodell TC. The kallikrein/kinin system and kinin antagonists in trauma. Immunopharmacology. 1996; 33:279–83.
197. Chan D, Gera L, Stewart J, Helfrich B, Verella-Garcia M, Johnson G, Baron A, Yang J, Puck T, Bunn P Jr. Bradykinin antagonist dimer, CU201, inhibits the growth of human lung cancer cell lines by a “biased agonist” mechanism. Proc Natl Acad Sci USA. 2002; 99:4608–13.
198. Stewart JM, Gera L, Chan DC, York EJ, Simkeviciene V, Bunn PA Jr, Taraseviciene-Stewart L. Combination cancer chemotherapy with one compound: pluripotent bradykinin antagonists. Peptides. 2005; 26:1288–91.
199. Chan DC, Gera L, Stewart JM, Helfrich B, Zhao TL, Feng WY, Chan KK, Covey JM, Bunn PA Jr. Bradykinin antagonist dimer, CU201, inhibits the growth of human lung cancer cell lines in vitro and in vivo and produces synergistic growth inhibition in combination with other antitumor agents. Clin Cancer Res. 2002; 8:1280–87.
200. Gera LC, Helfrich B, Bunn PA, York EJ, Stewart JM. Bradykinin-related compounds having anticancer activity in vivo superior to cisplatin and SU5416. Proc of the 26th Eur Peptide Symp E. 2000; 637–638.
201. Avdieiev S, Gera L, Havrylyuk D, Hodges RS, Lesyk R, Ribrag V, Vassetzky Y, Kavsan V. Bradykinin antagonists and thiazolidinone derivatives as new potential anti-cancer compounds. Bioorg Med Chem. 2014; 22:3815–23.
202. Jutras S, Bachvarova M, Keita M, Bascands JL, Mes-Masson AM, Stewart JM, Gera L, Bachvarov D. Strong cytotoxic effect of the bradykinin antagonist BKM-570 in ovarian cancer cells—analysis of the molecular mechanisms of its antiproliferative action. FEBS J. 2010; 277:5146–60.
203. Fernandes PD, Gomes NM, Sirois P. The bradykinin B1 receptor antagonist R-954 inhibits Ehrlich tumor growth in rodents. Peptides. 2011; 32:1849–54.
204. Campanholle G, Landgraf RG, Borducchi E, Semedo P, Wang PH, Amano MT, Russo M, Pacheco-Silva A, Jancar S, Camara NO. Bradykinin inducible receptor is essential to lipopolysaccharide-induced acute lung injury in mice. Eur J Pharmacol. 2010; 634:132–37.
205. Warde N. Osteoarthritis: local antagonism of endothelin-1 and bradykinin receptors improves OA pain and joint morphology in rats. Nat Rev Rheumatol. 2011; 7:375.
206. Lawson SR, Gabra BH, Guérin B, Neugebauer W, Nantel F, Battistini B, Sirois P. Enhanced dermal and retinal vascular permeability in streptozotocin-induced type 1 diabetes in Wistar rats: blockade with a selective bradykinin B1 receptor antagonist. Regul Pept. 2005; 124:221–24.
207. Keegan BP, Akerman BL, Péqueux C, North WG. Provasopressin expression by breast cancer cells: implications for growth and novel treatment strategies. Breast Cancer Res Treat. 2006; 95:265–77.
208. Pifano M, Garona J, Capobianco CS, Gonzalez N, Alonso DF, Ripoll GV. Peptide Agonists of Vasopressin V2 Receptor Reduce Expression of Neuroendocrine Markers and Tumor Growth in Human Lung and Prostate Tumor Cells. Front Oncol. 2017; 7:11.
209. Garona J, Pifano M, Scursoni AM, Gomez DE, Alonso DF, Ripoll GV. Insight into the effect of the vasopressin analog desmopressin on lung colonization by mammary carcinoma cells in BALB/c mice. Anticancer Res. 2014; 34:4761–65.
210. Alonso DF, Skilton G, Farías EF, Bal de Kier Joffé E, Gomez DE. Antimetastatic effect of desmopressin in a mouse mammary tumor model. Breast Cancer Res Treat. 1999; 57:271–75.
211. Giron S, Tejera AM, Ripoll GV, Gomez DE, Alonso DF. Desmopressin inhibits lung and lymph node metastasis in a mouse mammary carcinoma model of surgical manipulation. J Surg Oncol. 2002; 81:38–44.
212. Pastrian MB, Guzmán F, Garona J, Pifano M, Ripoll GV, Cascone O, Ciccia GN, Albericio F, Gómez DE, Alonso DF, Iannucci NB. Structure-activity relationship of 1-desamino-8-D-arginine vasopressin as an antiproliferative agent on human vasopressin V2 receptor-expressing cancer cells. Mol Med Rep. 2014; 9:2568–72.
213. Garona J, Pifano M, Orlando UD, Pastrian MB, Iannucci NB, Ortega HH, Podesta EJ, Gomez DE, Ripoll GV, Alonso DF. The novel desmopressin analogue [V4Q5]dDAVP inhibits angiogenesis, tumour growth and metastases in vasopressin type 2 receptor-expressing breast cancer models. Int J Oncol. 2015; 46:2335–45.
214. Bauer W, Briner U, Doepfner W, Haller R, Huguenin R, Marbach P, Petcher TJ, Pless J. SMS 201-995: a very potent and selective octapeptide analogue of somatostatin with prolonged action. Life Sci. 1982; 31:1133–40.
215. Grozinsky-Glasberg S, Shimon I, Korbonits M, Grossman AB. Somatostatin analogues in the control of neuroendocrine tumours: efficacy and mechanisms. Endocr Relat Cancer. 2008; 15:701–20.
216. Schmid HA, Schoeffter P. Functional activity of the multiligand analog SOM230 at human recombinant somatostatin receptor subtypes supports its usefulness in neuroendocrine tumors. Neuroendocrinology. 2004 (Suppl 1); 80:47–50.
217. Rubin J, Ajani J, Schirmer W, Venook AP, Bukowski R, Pommier R, Saltz L, Dandona P, Anthony L. Octreotide acetate long-acting formulation versus open-label subcutaneous octreotide acetate in malignant carcinoid syndrome. J Clin Oncol. 1999; 17:600–06.
218. Caron P, Beckers A, Cullen DR, Goth MI, Gutt B, Laurberg P, Pico AM, Valimaki M, Zgliczynski W. Efficacy of the new long-acting formulation of lanreotide (lanreotide Autogel) in the management of acromegaly. J Clin Endocrinol Metab. 2002; 87:99–104.
219. de Herder WW, Lamberts SW. Somatostatin and somatostatin analogues: diagnostic and therapeutic uses. Curr Opin Oncol. 2002; 14:53–57.
220. de Herder WW, Hofland LJ, van der Lely AJ, Lamberts SW. Somatostatin receptors in gastroentero-pancreatic neuroendocrine tumours. Endocr Relat Cancer. 2003; 10:451–58.
221. Bousquet C, Lasfargues C, Chalabi M, Billah SM, Susini C, Vezzosi D, Caron P, Pyronnet S. Clinical review: current scientific rationale for the use of somatostatin analogs and mTOR inhibitors in neuroendocrine tumor therapy. J Clin Endocrinol Metab. 2012; 97:727–37.
222. Weckbecker G, Lewis I, Albert R, Schmid HA, Hoyer D, Bruns C. Opportunities in somatostatin research: biological, chemical and therapeutic aspects. Nat Rev Drug Discov. 2003; 2:999–1017.
223. Guillermet J, Saint-Laurent N, Rochaix P, Cuvillier O, Levade T, Schally AV, Pradayrol L, Buscail L, Susini C, Bousquet C. Somatostatin receptor subtype 2 sensitizes human pancreatic cancer cells to death ligand-induced apoptosis. Proc Natl Acad Sci USA. 2003; 100:155–60.
224. Pyronnet S, Bousquet C, Najib S, Azar R, Laklai H, Susini C. Antitumor effects of somatostatin. Mol Cell Endocrinol. 2008; 286:230–37.
225. Lahlou H, Fanjul M, Pradayrol L, Susini C, Pyronnet S. Restoration of functional gap junctions through internal ribosome entry site-dependent synthesis of endogenous connexins in density-inhibited cancer cells. Mol Cell Biol. 2005; 25:4034–45.
226. Woltering EA, Watson JC, Alperin-Lea RC, Sharma C, Keenan E, Kurozawa D, Barrie R. Somatostatin analogs: angiogenesis inhibitors with novel mechanisms of action. Invest New Drugs. 1997; 15:77–86.
227. Serri O, Brazeau P, Kachra Z, Posner B. Octreotide inhibits insulin-like growth factor-I hepatic gene expression in the hypophysectomized rat: evidence for a direct and indirect mechanism of action. Endocrinology. 1992; 130:1816–21.
228. Florio T, Morini M, Villa V, Arena S, Corsaro A, Thellung S, Culler MD, Pfeffer U, Noonan DM, Schettini G, Albini A. Somatostatin inhibits tumor angiogenesis and growth via somatostatin receptor-3-mediated regulation of endothelial nitric oxide synthase and mitogen-activated protein kinase activities. Endocrinology. 2003; 144:1574–84.
229. Kumar M, Liu ZR, Thapa L, Qin RY. Anti-angiogenic effects of somatostatin receptor subtype 2 on human pancreatic cancer xenografts. Carcinogenesis. 2004; 25:2075–81.
230. Arena S, Pattarozzi A, Corsaro A, Schettini G, Florio T. Somatostatin receptor subtype-dependent regulation of nitric oxide release: involvement of different intracellular pathways. Mol Endocrinol. 2005; 19:255–67.
231. De Dosso S, Bajetta E, Procopio G, Cortinovis D, Buzzoni R, Catena L, Platania M, Verzoni E. Pulmonary carcinoid tumours: indolent but not benign. Oncology. 2007; 73:162–68.
232. Pavel ME, Hainsworth JD, Baudin E, Peeters M, Hörsch D, Winkler RE, Klimovsky J, Lebwohl D, Jehl V, Wolin EM, Öberg K, Van Cutsem E, Yao JC, and RADIANT-2 Study Group. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): a randomised, placebo-controlled, phase 3 study. Lancet. 2011; 378:2005–12.
233. Fazio N, Granberg D, Grossman A, Saletan S, Klimovsky J, Panneerselvam A, Wolin EM. Everolimus plus octreotide long-acting repeatable in patients with advanced lung neuroendocrine tumors: analysis of the phase 3, randomized, placebo-controlled RADIANT-2 study. Chest. 2013; 143:955–62.
234. Rinke A, Müller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, Mayer C, Aminossadati B, Pape UF, Bläker M, Harder J, Arnold C, Gress T, Arnold R, and PROMID Study Group. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009; 27:4656–63.
235. Chong CR, Wirth LJ, Nishino M, Chen AB, Sholl LM, Kulke MH, McNamee CJ, Jänne PA, Johnson BE. Chemotherapy for locally advanced and metastatic pulmonary carcinoid tumors. Lung Cancer. 2014; 86:241–46.
236. Novartis Pharmaceuticals. 3-arm Trial to Evaluate Pasireotide LAR/Everolimus Alone/in Combination in Patients With Lung/Thymus NET - LUNA Trial (LUNA). 2017. https://clinicaltrials.gov/ct2/show/NCT01563354.
237. Moody TW, Sun LC, Mantey SA, Pradhan T, Mackey LV, Gonzales N, Fuselier JA, Coy DH, Jensen RT. In vitro and in vivo antitumor effects of cytotoxic camptothecin-bombesin conjugates are mediated by specific interaction with cellular bombesin receptors. J Pharmacol Exp Ther. 2006; 318:1265–72.
238. Gilad Y, Firer M, Gellerman G. Recent innovations in peptide based targeted drug delivery to cancer cells. Biomedicines. 2016; 4:11.
239. Nardon C, Boscutti G, Dalla Via L, Ringhieri P, Di Noto V, Morelli G, Accardo A, Fregona D. CCK8 peptide-labeled Pluronic (R) F127 micelles as a targeted vehicle of gold-based anticancer chemotherapeutics. MedChemComm. 2015; 6:155–63.
240. Morisco A, Accardo A, Tesauro D, Palumbo R, Benedetti E, Morelli G. Peptide-labeled supramolecular aggregates as selective doxorubicin carriers for delivery to tumor cells. Biopolymers. 2011; 96:88–96.
241. Accardo A, Tesauro D, Aloj L, Tarallo L, Arra C, Mangiapia G, Vaccaro M, Pedone C, Paduano L, Morelli G. Peptide-containing aggregates as selective nanocarriers for therapeutics. ChemMedChem. 2008; 3:594–602.
242. Reubi JC. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev. 2003; 24:389–427.
243. Smith CJ, Volkert WA, Hoffman TJ. Radiolabeled peptide conjugates for targeting of the bombesin receptor superfamily subtypes. Nucl Med Biol. 2005; 32:733–40.
244. Rogers BE, Bigott HM, McCarthy DW, Della Manna D, Kim J, Sharp TL, Welch MJ. MicroPET imaging of a gastrin-releasing peptide receptor-positive tumor in a mouse model of human prostate cancer using a 64Cu-labeled bombesin analogue. Bioconjug Chem. 2003; 14:756–63.
245. Parry JJ, Kelly TS, Andrews R, Rogers BE. In vitro and in vivo evaluation of 64Cu-labeled DOTA-linker-bombesin(7-14) analogues containing different amino acid linker moieties. Bioconjug Chem. 2007; 18:1110–17.
246. Moody TW, Mantey SA, Pradhan TK, Schumann M, Nakagawa T, Martinez A, Fuselier J, Coy DH, Jensen RT. Development of high affinity camptothecin-bombesin conjugates that have targeted cytotoxicity for bombesin receptor-containing tumor cells. J Biol Chem. 2004; 279:23580–89.
247. Safavy A, Raisch KP, Khazaeli MB, Buchsbaum DJ, Bonner JA. Paclitaxel derivatives for targeted therapy of cancer: toward the development of smart taxanes. J Med Chem. 1999; 42:4919–24.
248. Du J, Li L. Which one performs better for targeted lung cancer combination therapy: pre- or post-bombesin-decorated nanostructured lipid carriers? Drug Deliv. 2016; 23:1799–809.
249. Nussberger J, Cugno M, Cicardi M, Agostoni A. Local bradykinin generation in hereditary angioedema. J Allergy Clin Immunol. 1999; 104:1321–22.
250. Emerich DF, Snodgrass P, Dean RL, Lafreniere D, Agostino M, Wiens T, Xiong H, Hasler B, Marsh J, Pink M, Kim BS, Bartus RT. Bradykinin modulation of tumor vasculature: I. Activation of B2 receptors increases delivery of chemotherapeutic agents into solid peripheral tumors, enhancing their efficacy. J Pharmacol Exp Ther. 2001; 296:623–31.
251. Wang X, Yang C, Zhang Y, Zhen X, Wu W, Jiang X. Delivery of platinum(IV) drug to subcutaneous tumor and lung metastasis using bradykinin-potentiating peptide-decorated chitosan nanoparticles. Biomaterials. 2014; 35:6439–53.
252. Bakhle YS. Conversion of angiotensin I to angiotensin II by cell-free extracts of dog lung. Nature. 1968; 220:919–21.
253. Ferreira SH, Rocha e Silva M. Potentiation of bradykinin and eledoisin by BPF (bradykinin potentiating factor) from Bothrops jararaca venom. Experientia. 1965; 21:347–49.
254. Ferreira SH, Bartelt DC, Greene LJ. Isolation of bradykinin-potentiating peptides from Bothrops jararaca venom. Biochemistry. 1970; 9:2583–93.
255. Soffer RL. Angiotensin-converting enzyme and the regulation of vasoactive peptides. Annu Rev Biochem. 1976; 45:73–94.
256. Wu J, Akaike T, Hayashida K, Miyamoto Y, Nakagawa T, Miyakawa K, Müller-Esterl W, Maeda H. Identification of bradykinin receptors in clinical cancer specimens and murine tumor tissues. Int J Cancer. 2002; 98:29–35.
257. Gniazdowska E, Koźmiński P, Bańkowski K, Ochman P. 99mTc-labeled vasopressin peptide as a radiopharmaceutical for small-cell lung cancer (SCLC) diagnosis. J Med Chem. 2014; 57:5986–94.
258. Manning M, Misicka A, Olma A, Bankowski K, Stoev S, Chini B, Durroux T, Mouillac B, Corbani M, Guillon G. Oxytocin and vasopressin agonists and antagonists as research tools and potential therapeutics. J Neuroendocrinol. 2012; 24:609–28.
259. Yin T, Wu Q, Wang L, Yin L, Zhou J, Huo M. Well-Defined Redox-Sensitive Polyethene Glycol-Paclitaxel Prodrug Conjugate for Tumor-Specific Delivery of Paclitaxel Using Octreotide for Tumor Targeting. Mol Pharm. 2015; 12:3020–31.
260. Richheimer SL, Tinnermeier DM, Timmons DW. High-performance liquid chromatographic assay of taxol. Anal Chem. 1992; 64:2323–26.
261. Redko B, Ragozin E, Andreii B, Helena T, Amnon A, Talia SZ, Mor OH, Genady K, Gary G. Synthesis, drug release, and biological evaluation of new anticancer drug-bioconjugates containing somatostatin backbone cyclic analog as a targeting moiety. Biopolymers. 2015; 104:743–52.
262. Shen H, Hu D, Du J, Wang X, Liu Y, Wang Y, Wei JM, Ma D, Wang P, Li L. Paclitaxel-octreotide conjugates in tumor growth inhibition of A549 human non-small cell lung cancer xenografted into nude mice. Eur J Pharmacol. 2008; 601:23–29.
263. Sun ML, Wei JM, Wang XW, Li L, Wang P, Li M, Yi CH. Paclitaxel-octreotide conjugates inhibit growth of human non-small cell lung cancer cells in vitro. Exp Oncol. 2007; 29:186–91.
264. Kastin A. Handbook of biologically active peptides. Elsevier Science; 2013.
265. Roy J, Putt KS, Coppola D, Leon ME, Khalil FK, Centeno BA, Clark N, Stark VE, Morse DL, Low PS. Assessment of cholecystokinin 2 receptor (CCK2R) in neoplastic tissue. Oncotarget. 2016; 7:14605–15. https://doi.org/10.18632/oncotarget.7522.
266. Kulhari H, Pooja D, Singh MK, Kuncha M, Adams DJ, Sistla R. Bombesin-conjugated nanoparticles improve the cytotoxic efficacy of docetaxel against gastrin-releasing but androgen-independent prostate cancer. Nanomedicine (Lond). 2015; 10:2847–59.
267. Gupta SL, Dhiman V, Jayasekharan T, Sahoo NK. Analysis of argentinated peptide complexes using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry: Peptide = oxytocin, arg(8) -vasopressin, bradykinin, bombesin, somatostatin, neurotensin. Rapid Commun Mass Spectrom. 2016; 30:1313–22.
268. Tyndall JD, Pfeiffer B, Abbenante G, Fairlie DP. Over one hundred peptide-activated G protein-coupled receptors recognize ligands with turn structure. Chem Rev. 2005; 105:793–826.
269. Moreno P, Mantey SA, Nuche-Berenguer B, Reitman ML, González N, Coy DH, Jensen RT. Comparative pharmacology of bombesin receptor subtype-3, nonpeptide agonist MK-5046, a universal peptide agonist, and peptide antagonist Bantag-1 for human bombesin receptors. J Pharmacol Exp Ther. 2013; 347:100–16.
270. Martín-Gago P, Gomez-Caminals M, Ramón R, Verdaguer X, Martin-Malpartida P, Aragón E, Fernández-Carneado J, Ponsati B, López-Ruiz P, Cortes MA, Colás B, Macias MJ, Riera A. Fine-tuning the π-π aromatic interactions in peptides: somatostatin analogues containing mesityl alanine. Angew Chem Int Ed Engl. 2012; 51:1820–25.

