INTRODUCTION
Prostate cancer (PCa) is the second most common non-skin malignancy in developed countries like USA. More than 8,900,000 cases are detected and more than 260,000 deaths recorded worldwide every year [1]. The high death rate from PCa may be due to its natural history in which early stage cancer eventually progress into hormone refractory or castration resistant stage [2]. Histopathological analysis of clinical samples and animal studies have shown that infectious agents, dietary carcinogens, race, age and other factors can cause damage to the epithelial cells of prostate and elicit inflammatory responses predisposing to PCa [3]. Well-established pathophysiological evidence indicates that these inflammatory responses can lead to mutations [4, 5]. These mutations, together with inflammatory mediators and enhanced cell proliferation, are some of the causative factors for initiation and development of PCa [1]. These factors cause the formation of preneoplastic lesions including proliferative inflammatory atrophy (PIA) and prostate intraepithelial neoplasia (PIN) which can progress to malignancy and eventually lead to metastases. Progression of PCa is dependent on circulating levels of androgen until androgen independent, castration-resistant, cancer develops.
In view of the high mortality rate of PCa, a range of treatments have been developed for treating PCa. In most cases, localized PCa can be successfully treated by surgery or radiotherapy. But this is typically unsuccessful and complex when the cancer has become advanced and metastatic. Although conventional androgen deprivation therapy (ADT) is used to initially treat advanced PCa, unfortunately this approach invariably leads to resistance and its clinical use is only temporarily effective [6]. Failure of ADT leads to the development of so-called castration resistant prostate cancer (CRPC) [7–9]. Treatment options available for patients with CRPC are limited and more complicated than those for hormone-dependent PCa. Most chemotherapeutic drugs have inherent limitations such as development of resistance and side effects such as urinary dysfunction, bowel dysfunction and heart related complications. Therefore, there is an urgent need for the development of novel therapeutic approaches as well as chemopreventive strategies that can delay or reduce the progression of PCa with minimal side effects [10, 11]. This may be achieved through the identification and development of chemopreventive compounds from non-toxic natural sources such as dietary constituents.
Role of dietary constituents in chemoprevention of PCa
Epidemiological studies have indicated that diet and nutrition are important determinants of PCa risk with significant variations in incidences between different geographic regions [12, 13]. Western nations in America and Europe have higher PCa incidence rates, when compared to many Asian countries [14]. The lower rate of PCa in Asian countries may be due to dietary patterns with low fat intake and diets rich in plant-based agents. Hence, the addition of vegetables and fruits in the daily diet to prevent PCa has received considerable attention for cancer chemoprevention [15].
Chemoprevention is defined as prevention of cancer development or inhibition of cancer progression through pharmacological intervention with drugs, naturally occurring compounds, or dietary supplements [15]. According to Ansari et al. [16, 17] and Montironi et al. [18], chemoprevention of cancer may be defined as inhibition or delay of the onset of cancer growth by blocking neoplastic transformation as well as reversing the progression of transformed cells to the malignant stage. Epidemiological studies have indicated that populations consuming vegetables rich in micronutrients (anti-oxidants, vitamins, and trace minerals) have lower cancer incidence and cancer mortality [19]. Further research studies suggested that micronutrients present in vegetables and fruits may have potent anticancer properties [20]. Hence, there is a considerable interest in ascertaining whether these micronutrients, including vitamins such as VK, offer protection against PCa [19, 20]. A number of macro- and micro-nutrients have been proposed as chemopreventive agents [21]. Dietary or natural compounds may exert their chemopreventive effects by a variety of mechanisms, for example, by scavenging oxygen free radicals or inhibiting polyamine metabolism, thereby preventing carcinogenesis [22, 23]. Natural compounds are also known to exhibit anticancer effects through regulation of signal transduction pathways, induction of apoptosis, and activation of anti-metastasis genes [24]. Some well-known dietary agents that have been studied as potential chemopreventives include micronutrients such as selenium, vitamins C, D, E, and K, lycopene, green tea, silymarin, pomegranate and resveratrol. These agents may exert their putative anticancer activity through various mechanisms as shown in Table 1 [14].
Table 1: Chemopreventive mechanism of dietary agents in prostate cancer
Dietary agents |
Source of the dietary agent |
Mechanism |
|---|---|---|
Lycopene |
Tomato |
Influence expression of gap junction proteins and growth factor signaling [153] |
Vitamin A |
Fruits and vegetables |
Apoptosis, cell cycle arrest [154] |
Vitamin C |
Fruits |
Cellular chromosomal damage [37] |
Vitamin D |
Residential sunlight and milk with Vitamin D |
Apoptosis, cell cycle arrest [42] |
Vitamin E |
Fruits and vegetables |
Inhibition of protein kinase C, induction of NADPH detoxification enzyme and reduction of arachidonic acid metabolism [155] |
VK2 |
Fruits, vegetables, meat |
|
Selenium |
Fruits and vegetables |
Cell cycle arrest and apoptosis and reduction of angiogenesis [156] |
Selenium + Vitamin E |
Fruits and vegetables |
Apoptosis, cell cycle arrest [157] |
Soy flavonoid (genistein) |
Soy seeds |
Apoptosis and cell cycle arrest [158] |
Tea (catechins) |
Tea leaves |
Cell cycle arrest, inhibition of angiogenesis, inhibition of protein kinase C and apoptosis [159] |
Resveratrol |
Peanuts, pistachios, grapes, blueberries, cranberries |
Cell cycle arrest and apoptosis [160] |
Silymarin |
Medicinal plant |
Cell cycle arrest, inhibition of mitogenic cell survival signaling [161] |
Proanthocyanidins and procynadins |
Grape seed extract |
Inhibition of protein tyrosine kinase, matrix metalloproteinases and Rel/NF-kB family members [162] |
Apigenin |
Fruits and vegetables |
Cell cycle deregulation and apoptosis [163] |
Indoles and their derivatives |
Fruits and vegetables |
Modulations in cell cycle regulatory proteins and inhibition of cell survival pathways (PI3K/Akt) and NF-κB transcription factor [164] |
Isothiocyanates |
Cruciferous vegetables |
Cell cycle inhibition, induction of phase II enzymes, inhibition of extracellular signal-regulated kinases, suppression of NF-κB [165] |
Phenolic acids Curcumin |
Component of turmeric |
Tyrosine kinase and protein kinase C inhibition, down-regulation of AR gene expression, inhibition of PI3K/Akt and NF-κB [166] |
Vitamins and PCa chemoprevention
As indicated above, there is a considerable interest in micro-nutrients and other dietary agents as potential chemopreventive agents against malignancies including PCa [21]. The antioxidant and non-antioxidant activities of various dietary agents in PCa prevention have been examined in several studies [25, 26]. Most studies have addressed dietary sources that are rich in phytochemicals such as carotenoids, vitamins, flavonoids, selenium, dietary fiber, glucosinolate, indoles and phenols [27]. These phytochemical constituents have complimentary or overlapping mode of actions including antioxidant activity, enzyme detoxification, inhibition of nitrosamines formation, alteration of hormone metabolism and have the ability to modulate the carcinogenic cellular events [28].
It has been hypothesized that the lycopene present in tomato is associated with prevention of PCa. For instance, Hwang and Bowen [29] suggested that a diet rich in tomatoes and tomato products containing lycopene is associated with reduction of PCa risk. Some case-control studies and a meta-analysis also reveal that tomato products may play a role in the prevention of PCa [30, 31]. One preclinical study of PCa revealed that the consumption of tomato powder (but not pure lycopene) prevented prostate carcinogenesis in a rat model, suggesting that tomato powder contain some anticancer compounds other than the lycopene that inhibits the carcinogenic process [32]. On the other hand, other animal studies and a recent phase II intervention study with a lycopene-rich tomato product did not yield results indicative of preventive efficacy against PCa [33].
A major aspect in PCa chemoprevention through dietary constituents has focused on micronutrients, especially vitamins such as vitamin A, C, D and E, which have been extensively studied for their effects on PCa.
Vitamin A is essential for cell differentiation, visual functioning, physiological growth and is known to be able to modulate the cancer cell growth [34]. The potential chemopreventive mechanisms of vitamin A and its analogues have been shown in laboratory studies to specifically act on the tumor progression stage through the inhibition of cell proliferation, induction of apoptosis, cell cycle arrest and also a combination of these mechanisms [35]. However, toxicity has prevented clinical translation of the use of vitamin A and other retinoids to prevent PCa.
Vitamin C is a potent antioxidant which scavenges reactive oxygen species (ROS) and free radicals that cause DNA damage [36]. One potential chemopreventive mechanism of vitamin C has been shown to be the inhibition of neoplastic transformation by reducing cellular chromosomal damage [37]. Vitamin C not only acts as antioxidant by scavenging free radicals, but also reinstates the activity of α-tocopherol following its lipid peroxidation chain breaking effect and inhibits the growth of PCa cells in vitro [26]. In addition, Taper et al. [38] demonstrated that vitamin C inhibited the growth of both androgen-dependent and -independent human PCa cells in nude mice. Apart from the antioxidant mechanism of vitamin C, the combination of vitamin C with amino acids and other micronutrients are also effective in targeting the signal transduction pathways to inhibit the cell proliferation and cancer progression in laboratory studies, as has been shown for ovarian cancer [39].
Vitamin D (calcitrol) is synthesized in the skin following exposure of 7-dehydrocholesterol to ultraviolet light and is derived from dietary sources. Exposure to residential sunlight is associated with decreased risk of PCa that may be linked with calcitrol synthesis [40]. In addition, some epidemiological studies have suggested that increased risk of PCa is associated with a decreased production of vitamin D [41]. The biologically active form of vitamin D inhibits PCa cell proliferation in vitro through various mechanisms including induction of apoptosis, cell cycle arrest, and activation of growth factor signaling [42]. The combination of vitamin D with other dietary constituents such as genistein (component of soy) has also been shown to inhibit the growth of benign primary human prostate epithelial cells and PCa cells [43]. Statistical analysis of PCa mortality rates in 71 countries showed that exposure to increased sunlight and consumption of oilseeds and soybeans was inversely correlated with the rate of PCa [44].
Vitamin E is a group of naturally occurring compounds: the tocopherols, tocotrienols and their derivatives. Of all the tocopherols, α-tocopherol is the predominant form of vitamin E found in plasma and tissues. Epidemiological studies have shown that consumption of a diet rich in vitamin E is inversely associated with the rate of PCa incidence [45, 46]. However, some epidemiological studies did not support an anticancer role of vitamin E in PCa [47, 48]. Due to their ability to trap reactive oxygen and nitrogen species (RONS), tocopherols are important biological antioxidants, and their cancer preventive activities have been extensively studied [49, 50]. Besides their anti-oxidant activity, vitamin E and its derivatives exert their anticancer effects through altered transforming growth factors-β and androgen receptor/prostate specific antigen (AR/PSA) signaling pathways and by regulating the cell cycle arrests at synthesis phase in PCa cell lines [51]. The mechanisms through which vitamin E inhibits cell proliferation include inhibition of protein kinase C activity, enzyme detoxification, induction of apoptosis, regulation of Fas levels in the membrane and cytoplasm and inhibition of matrix metallo-proteinases [52]. Male transgenic TRAMP (transgenic adenocarcinoma of the mouse prostate) mice fed with vitamin E succinate, selenium and lycopene supplemented diet had a significant reduction in PCa incidence [53]. Despite these promising studies indicating anti-PCa activity of vitamin E, α-tocopherol supplementation did not reduce, but slightly increased PCa risk in a large randomized clinical trial, SELECT (selenium and vitamin E cancer prevention trial) [54, 55].
Selenium is an essential component of several antioxidant enzymes such as glutathione peroxidase and oral supplementation with a baker’s yeast grown on a selenium-rich medium reduced PCa risk a small randomized trial. Furthermore, it causes cell cycle arrest and apoptosis and inhibits angiogenesis [56]. However, in the large randomized clinical trial, SELECT, it did not prevent PCa and in subgroups increased risk slightly [55, 57].
Vitamin K (VK) as an anticancer agent
VK is an essential micro nutrient, primarily associated with action in the coagulation cascade, and it also regulates bone metabolism through a mechanism involving gamma carboxylation of bone matrix proteins [58]. In addition to this activity, VK also had anticancer activity against human cancers such as hepatic, leukemia, lung, colonic, oral, breast and bladder cancers in vitro [59, 60]. Naturally occurring VK in the human diet is VK1 (phylloquinone) found in plants, particularly in green leafy vegetables where it is involved in photosynthesis. Mammalian intestinal bacteria convert VK1 into VK2 (menaquinone), the physiologically active form of VK. There are three synthetic forms of VK, VK3 (menadione), VK4, and VK5. VK3 is used as source of VK in pet food and as dietary supplement in some countries. VK1 and VK2 are non-toxic even at high doses, but large doses of VK3 are toxic capable of causing hemolytic anemia, liver toxicity, and allergic reactions.
Major anticancer efficacy of VK1 and VK2 is mediated by non-oxidative mechanisms, probably via transcription factors, but VK3 at higher concentrations works via reducing oxidative stress and arylation. Addition of catalase to the culture medium inhibits the in vitro effects of VK3 but not VK2 which suggested that the VK are a class of growth inhibitors that have a novel mechanism possibly involving carboxylation. [61, 62]. Pharmacological studies demonstrated that massive doses of VK2, up to more than 2.5 grams given per day, were safe and caused no enhancement of the toxicity of chemotherapy [63]. VK is a family of naturally occurring essential fat soluble compounds derived from 2-methyl-1,4-naphthoquinone that are structurally characterized by a common quinone naphthalene ring carrying two carbonyl moieties [58]. Interestingly, quinones are the functional unit in several cancer chemotherapeutic drugs such as doxorubicin, daunomycin, mitomycin C, and mitoxanthrone and the basic structure of VK is also a quinone (Naphthoquinone). This in part explains why VK has gained interest for the prevention and treatment of cancer [64].
Tamori et al. [65] reported that VK2 prevents hepatocarcinogenesis in patients with hepatic cirrhosis. In a meta-analysis of four randomized controlled studies of the preventive effect of VK analogues on the incidence of HCC recurrence after partial hepatectomy or local ablative therapy, indicated that VK and its derivatives prolonged disease-free survival [66, 67]. A recent meta-analysis conducted by Zhong et al. [68] reported that an analogue of VK2 may prevent the formation of secondary tumors in residual liver tissue following partial hepatectomy and increase the overall survival rate by inhibiting or activating certain signaling pathways (intrinsic apoptotic pathway and inhibition of nuclear factor kappa B activation) in HCC patients.
The antitumor activity of VK was first reported almost six decades ago [69]. The intravenous administration of the synthetic derivative of VK, menadione (VK3) increased the survival of inoperable bronchial carcinoma patients [58]. VK3 also synergistically inhibit cancer cell growth specifically in combination with vitamin C by activating oxidative stress and depleting cellular thiols [70]. Similarly, phylloquinones (VK1) and menaquinone (VK2) exert anti-proliferative activity by targeting transcription factors of proto-oncogenes such as c-myc, c-jun and c-fos, which result in cell cycle arrest and apoptosis [71]. Both phylloquinone (VK1) and menaquinone (VK2) have anticancer effects in various cancer cells including of the breast, stomach and liver [72, 73]. Menadione (VK3) was also shown to exhibit potent cytotoxicity against several cancers including oral, prostate, renal and breast cancer [74–77].
Epidemiological studies of VK and PCa
Epidemiological studies suggest that there is inverse association between dietary intake of VK (especially menaquinone) and overall cancer incidence [78]. This inverse association was seen in male patients with cancers of the prostate, colorectum, and lung, most profoundly for prostate and lung cancer [78]. A first confirmatory epidemiological study was reported by Nimptsch et al. [79] in which risk of advanced stage PCa was increased in participants with poor VK status as estimated by serum under-carboxylated osteocalcin. Nimptsch et al. [64] also conducted a cohort study in which they evaluated the association between dietary intake of VK (phylloquinone and menaquinone) and the risk of PCa. Their statistical analysis and experimental data indicated an inverse association of VK (menaquinone) and PCa risk. By contrast, such association was not observed for phylloquinone intake. In two other studies, short-term (6–12 weeks) and longtime treatment of VK were compared in their effect on urological cancers; the long-term VK treatment reduced risk of PCa by 40% more than the short-term VK treatment [80, 81]. By contrast, a small case–control study conducted by Blumentals et al. [17] suggested that there is no association between VK treatment and bladder cancer risk. Pottegard et al. [82] also reported no apparent association between intake of VK and total cancer risk in a population-based case control study. However, we recently reported that the administration of VK2 significantly inhibited proliferation of both androgen-dependent and androgen-independent PCa cells through molecular mechanisms involving induction of apoptosis and reduction of the angiogenic potential of the PCa cells [83]. Although the exact mode of action for the anticancer activity of VK2 is still unclear, some our above-mentioned study and other experimental studies suggest that VK2 has anticancer activity through the mechanisms such as induction of apoptosis, production of reactive oxygen species (ROS) and cell cycle arrest [84–86]. The anti-proliferative activity of VK2 has been most extensively studied in hepatocellular cancer in which ROS production, apoptosis and cell cycle arrest at S phase are implicated for its tumor suppressive role [82, 83]. Hepatoma-derived growth factor (HDGF) is a heparin- binding growth factor protein which was shown to be partially responsible for the anti-proliferative property of VK2 in hepatocellular cell lines [87]. Recently, we have reported that HDGF also plays an important role in the regulation of cell growth and apoptosis as well as invasion of human PCa cells, and hence it may serve as a therapeutic target for VK2 in PCa through the mechanism of the AKT and NF-kB pathways (Figure 1) [88]. Menadione (VK3), along with vitamin C (ascorbic acid), has been shown to reduce the rate of PCa growth in both in vitro and in vivo models [89]. The synergistic antitumor effects of VK3 and ascorbic acid appear to act through caspase-mediated apoptosis in PCa [74]. Recently, Gilloteaux et al. [90] reported that the combination of VK3 and ascorbic acid induces oxidative stress in DU-145 PCa cells. Scanning and transmission electron microscopy studies showed that the ROS induced by this combination damages nucleus, mitochondria, endomembranes, lysosomes, finally resulting in cell death [90].
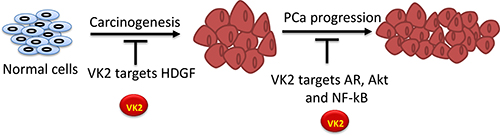
Figure 1: Proposed molecular mechanism of VK2 in prostate cancer. VK2 targets HDGF protein during the development of carcinogenesis and targets androgen receptors (AR), Akt and NF-kB during the progression of PCa.
Interestingly, pronounced anticancer effects of VK3 have also been observed when combined with other compounds such as plumbagin and juglone. Plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone) is naturally occurring napthoquinone sharing structural similarity with VK3 which is found in natural naphthoquinones such as roots, leaves, bark, and wood of Juglansregia (walnut). Zhou et al. [91] reported that plumbagin promotes the apoptosis and autophagy in PC-3 and DU145 PCa cells. They showed that plumbagin reduced the mitochondrial membrane potential followed by the release of cytochrome c in a dose-dependent manner which eventually led to apoptosis by the activation of the caspase cascade (caspase 9 and 3). They also reported that plumbagin induces autophagy by the inhibition of phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt)/ mammalian target of rapamycin (mTOR) and p38 mitogen-activated protein kinase (MAPK) pathways and activation of 5′-AMP-dependent kinase (AMPK) in PC-3 and DU145 cells. Plumbagin also shows anticancer effects on BRCA 1/2 defective castrate resistant prostate cancer cells as well as prostate cancer stem-like cells by the activation of apoptosis [92]. Recently, Hafeez et al. [93] reported that the dietary plumbagin inhibits the growth of both primary and castration-resistant prostate cancer (CRPC) in Pten-knockout mice through the inhibition of PKC, Stat3, AKT and EMT markers (vimentin and slug), which are linked to the induction and progression of PCa. Collectively these studies suggest that VK and its derivatives hold promise both as a chemopreventive and as a therapeutic agent for PCa.
Mechanisms underlying the anticancer effects of VK
All the three major analogues of VK (K1, K2 and K3) show anticancer activity such as induction of apoptosis and differentiation and cell cycle inhibition. Various anticancer mechanisms of VK are reviewed below.
Oxidative stress-mediated anticancer effects of VK
Oxidative stress-mediated anticancer activity is believed to be the primary mode of action of VK. Oxidative stress is induced via redox-cycling of the Quinone to generate ROS such as hydroxyl, superoxide radical, and hydrogen peroxide [58]. If both redox-cycling and ROS surpass the anti-oxidative capacity of the cell, cell death results. ROS are chemically reactive molecules containing oxygen molecules produced by eukaryotic cells during normal oxidative metabolism through various mechanisms. An imbalance between the intracellular production of ROS and their defense mechanism leads to the oxidative stress. Augmented levels of oxidative stress induce lipid and protein oxidative modifications and DNA damage leading to apoptotic cell death or carcinogenic cell transformation [94]. Therefore, oxidative stress is involved in most of the pathological conditions and diseases including cancer, which is characterized by increased cell proliferation, accumulating mutations or other DNA damage, and genomic instability [95, 96]. It is well documented that cancer cells contain higher level of ROS than normal cells. Hence, cancer cells are more susceptible to oxidative stress-induced cell death and this could be exploited for the development of therapeutic approaches. Some chemotherapeutic compounds (such as cisplatin, Buthionine sulphoximine, and Imexon) specifically increase ROS production or inhibit ROS elimination by targeting scavenging systems, which induces cell death by the accumulation of ROS in cancer cells [97, 98].
VK is a redox cycling compound that undergoes reduction producing semiquinone (one electron) followed by hydroquinone (two electrons) in cellular systems. Reduction of (one electron) VK is catalyzed by NADPH: cytochrome p450 reductase. In the presence of oxygen (O2), semiquinone is oxidized back to VK, which leads to reduction of O2 in the form of ROS. On the other hand, NQO1 uses NADH or NADPH as electron donors and catalyzes reduction of (2 electrons) VK (menaquinone) to hydroquinone in which reaction there is no generation of semiquinone and ROS production. The cyclic conversion of VK to semiquinone and back to quinone can result in the generation of ROS with adverse effects to the target cells. Oxidative stress increases DNA damage due to hydroxyl radicals produced by VK2 which leads to leads to cytotoxicity-mediated cell death in cancer cells (Figure 2) [99, 100].
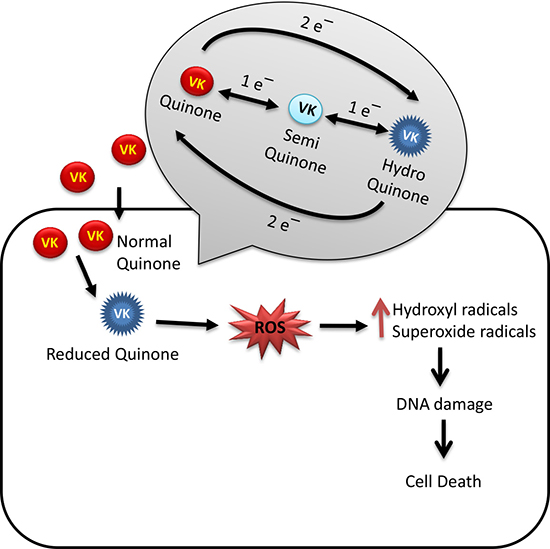
Figure 2: Oxidative stress mediated anticancer mechanism of VK. VK undergoes redox cycling to form hydroquinone and generate reactive oxygen species (ROS). ROS mediated oxidative stress causes DNA damage and finally cytotoxic mediated cell death in cancer cells.
Apoptosis inducing potential of VK and targeting of apoptotic pathways
Apoptosis is characterized by specific morphological changes such as nuclear condensation and fragmentation, membrane blabbing and formation of apoptotic bodies [101]. VK has been shown to induce apoptosis through functional activation of Fas/FasL signaling in Jurkat cells [102]. Several external stimuli such as T-cell receptor (TCR) ligation, UV and gamma irradiation and drugs are able to induce apoptosis by upregulating FasL through the activation of transcription factors like nuclear factor of activating T-cells and NF-kB [102, 103]. Ligation of the TCR induces the expression of FasL which is dependent on c-myc expression. c-Myc may function as transcription factors to induce apoptosis when deprived growth factors and this c-myc-induced cell death may be mediated by cell surface interaction of Fas with its ligand (FasL) [104, 105].
Apoptosis is defined as a synchronized mode of cell death, in which an intracellular events act to eliminate the unwanted or dangerous cells [106]. Apoptosis has a wide range of biological significance playing key role in physiological processes like homoeostasis, differentiation, regulation and immune functioning [107, 108]. Homeostasis is maintained by a strict balance between apoptosis and cell proliferation. Disruption between this balance implicated in tumor development, neurodegeneration and autoimmune diseases [106, 109]. It is well established that many tumor promoters inhibits apoptosis, leads to development of chemo resistant tumors [110]. Activation of apoptosis in chemo resistant tumors is one of the challenging strategies in cancer treatment [111, 112]. Since alterations in apoptosis program can lead to inappropriate changes in apoptotic proteins that are altered to blunt the effects of drug treatment. Recently, VK has been extensively studied and found to exhibit a wide range of cytotoxicity against various cancer types both in vitro and in vivo through the induction of various apoptotic pathways as discussed below and summarized in Table 2.
Table 2: Distinctive mechanisms of different forms of VK that triggers programmed cell death by apoptosis
Different forms of Vitamin K |
Cancer/Cancer Cell |
Target molecule |
|---|---|---|
VK |
Pancreatic cancer |
Caspase dependent apoptosis via the MAP kinase pathway [127] |
VK2 |
Leukemia |
G0/G1 arrest along with Apoptosis [116] |
VK3 |
Leukemia |
Fas/FasL [102] |
VK3 + Vitamin C |
Leukemia |
ROS, NF-kB [113] |
VK3 + Vitamin D-Fraction |
Renal cell carcinoma |
G0/G1 arrest along with Apoptosis [75] |
The aforementioned synergistic effect of VK3 with vitamin C also involves induction of apoptotic cell death in leukemia cells by sequential molecular events involving the activation of NF-kB, ROS production, p53/c-Jun transcription factor, mitochondrial depolarization and caspase-3 activation pathway [87]. Other studies also indicated that the combination of VK and vitamin C induces apoptosis in leukemia cells by oxidative stress [113–115]. Miyazawa et al. [116] reported that VK2 causes G0/G1 arrest along with apoptosis induction especially in leukemia cells that are resistant to VK1 inducing apoptosis.
VK targeting of cancer cells by mitochondrial mediated apoptosis
It is well established that mitochondria are an important component of the apoptosis execution pathway mediated by downregulation of Bcl-2 or Bcl-xl and mitochondrial membrane associated genes Bax and/or Bak [117, 118]. The anti-apoptotic Bcl-2 proteins regulates the mitochondrial membrane potential (MMP) and subsequent release of pro-apoptotic proteins such as cytochrome-c and apoptosis inducing factors (AIF) [117]. Cytoplasmic cytochrome-c interacts with apoptotic protease activating factor-1 (Apaf-1), leads to the activation of caspase family proteins which in turn leads to apoptosis through degradation of cellular proteins [119]. VK2 induces pro-apoptosis effects by regulating the MMP, in which mechanism VK2 produces superoxide within the mitochondrial membrane, followed by the release cytochrome c, activation of procaspase 3, and finally apoptotic cell death as shown in in TYK-nu ovarian cancer cells [120].
Yokoyama et al. [119] reported that VK2 induces apoptosis through mitochondrial caspase 3 mediated pathway in human myeloma cells and HL60 cells. In addition, Karasawa et al. [118] demonstrated the role of VK2 induced apoptosis through the activation of apoptotic regulators Bax and Bak in HL60 human promyelocytic leukemia cells. In this study, VK2-induced apoptosis was abrogated by the knockdown of Bak gene. Interestingly, VK2 directly binds to Bak, which is suppressed by anti-apoptotic Bcl-2 protein (Bcl-2 and Bcl-xL). Thus, the Bak gene is necessary for VK2-induced apoptosis and is therefore the molecular target of VK2. Furthermore, VK2 disrupted the mitochondrial membrane potential followed by the release of cytochrome C from mitochondria in a dose-dependent manner. Korsmeyer et al. [121] also reported that the loss of mitochondrial membrane potential and release of cytochrome C were associated with Bak and Bax in HL60 cells. Karasawa et al. [118] examined these pro-apoptotic proteins in human cervical carcinoma (HeLa) cells by immunofluorescent microscopy and found that VK2 induces activation and oligomerization of Bak and Bax. These results support the notion that VK2 induces mitochondrion-mediated apoptosis through the activation of Bak and Bax.
Fas/FasL mediated apoptosis
Several apoptotic inducers such as certain chemotherapeutic drugs, UV and gamma irradiation, and TCR ligation are thought to induce apoptosis through the inactivation of transcription factors such as NF-kB [122]. Moreover, in T-lymphocytes, oxidative stress mediated apoptosis has been linked to upregulation of FasL through the activation of members of the stress-mediated protein kinase family [123]. c-Myc may function as a transcription factor to drive apoptosis when cells are deprived of growth or survival factors [104]. When amounts of survival factors are low, Fas interacts with its ligand (FasL) to induce c-myc mediated apoptotic cell death [124]. Caricchio et al. [102] found evidence that there is a direct association between c-myc and the Fas/FasL system in the effect of VK, as VK3 treatment induced c-myc and also increased both FasL and Fas. Another important property of VK3 is that it induces oxidative stress through the production of ROS. Oxidative stress is also involved in Fas-mediated apoptosis, in which reactive oxygen intermediates induce FasL mRNA expression and the antioxidant glutathione rapidly decreases during apoptosis induced by crosslinking of Fas receptors [102, 125]. Laux and Nel [126] reported that the JNK (Jun amino-terminal kinases) cascade and FasL expression are involved in VK3 (menadione) mediated apoptosis (Figure 3).
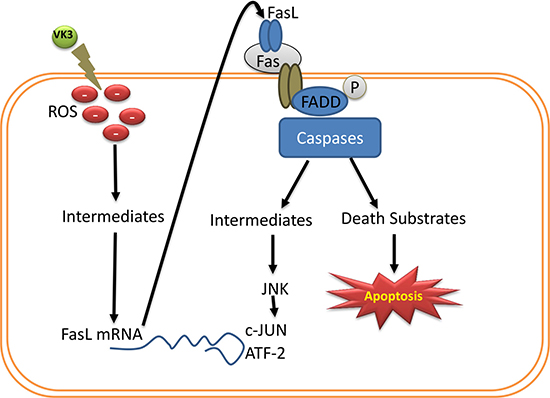
Figure 3: Regulation and expression of FasL during VK3 induced ROS. VK induced ROS involved in Fas mediated apoptosis, intermediates of ROS regulate the expression of FasL mRNA, which in turn induces the caspase mediated apoptosis through phosphorylation of Fas associated protein with death domain (FADD).
The mitochondrion is a major target for VK# (menadione)-induced cytotoxicity. Menadione induced ROS and thereby possible alteration of the oxidation-reduction state of cystenyl groups in the mitochondrial membrane. This, in turn, leads to the cross linking of sulphydryl (SH) groups in the permeability transition (PT) pore, leading to an increased/enlarged pore. PT pore opening initiates sequential events including decreasing mitochondrial membrane potential (Ψ) and disruption of inner membrane integrity. Ultimately it may result in the release of apoptotic proteins such as cyto-c, Apaf-1 which trigger caspase-9 activation. ROS intermediates induce expression of FasL protein which translocates from cytoplasm to plasma membrane to interact with Fas, which then recruits FADD (Fas-Associated protein with Death Domain) to bind to its death domain. This FasL-Fas activates the caspase cascade that subsequently leads to apoptosis and JNK-mediated cell death. Similar findings were observed in leukemia cells, in which VK2 induces apoptosis through the activation of caspase-3. It has been well established data that VK induces apoptosis in various cancer cell lines such as from renal cell carcinoma, leukemia, pancreatic cancer, glioma, and prostate cancer [75, 83, 116, 127, 128].
VK2 induced autophagy
Similar to apoptosis, autophagy is an evolutionary conserved membrane mediated process that leads to the degradation of proteins and organelles present in cytosol by lysosomes [129]. Hence, autophagy is an important mechanism of living cells to remove damaged or long-lived cytosolic proteins. Autophagy-defective cells undergo susceptibility to metabolic stress and genomic damage, which finally may lead to tumorigenesis. Loss of the important autophagic gene Beclin1 has been found in 75% of human breast, ovarian, and prostate cancers, suggesting that autophagy plays a critical role in preventing tumor cell growth. Paradoxically, autophagy has a dual role in cancer cells, functioning as both tumor suppressor by the removal of damaged proteins and organelles and promotion of tumor cell growth under adverse conditions due to stress tolerance [130]. In autophagy, cytosolic proteins and other organelles are engulfed into double membrane vesicles called autophagosomes which subsequently fuse with lysosomes to form autolysosomes to be degraded by lysosomal hydrolases [119]. Canonically autophagy is induced by nutrient deprivation and starvation and hence it is sensitive to levels of growth factors and hormones. Some cancer chemotherapeutic drugs also induce autophagy, such as a plant-derived alkaloid (voacamine), a Chinese herbal remedy (diterpenoidoritonin), and natural quinonoidplumbagin. Some autophagy inducing nutritional dietary constituents and their anticancer mechanisms are listed in Table 3 [131, 132]. Rapamycin is the currently available standard and potent drug to induce autophagy as therapy of various cancers [133]. Some trace elements and vitamins including vitamins C, D, and E have been shown to stimulate the autophagy in non-small cell lung cancer cells (vitamin C), head and neck squamous cell carcinoma cells (vitamin D3), and pancreatic cancer cells (vitamin E) [131]. VK2 also can induce autophagy as indicated by accumulation of autophagic vacuoles in cholangiocellular carcinoma cells treated with VK2 [134]. Other experimental studies indicated that VK2 inhibits the tumor cell growth by inducing both apoptosis and autophagy in leukemia and colon cancer cells [119].
Table 3: Induction of autophagy through nutritional and dietary constituents
Dietary Constituent |
Cancer/Cancer Cell |
Target molecule |
|---|---|---|
Curcumin |
Brain cancer |
Akt/mTOR/S6 kinase, ERK1/2 [167] |
Genistein |
Ovarian cancer |
Akt [168] |
Resveratrol |
Ovarian cancer |
|
Sulforaphane |
Prostate cancer |
Mitochondria, mitophagy [171] |
Vitamin C |
Glial cells, |
|
Vitamin D3 |
Head and Neck cancer |
p19INK4D [174] |
VK2 |
Liver cancer |
Not known |
Ascorbate |
Prostate cancer/PC-3 cells |
ROS production [175] |
Piperlongumine |
PCa/PC-3 cells |
ROS production and Akt/mTOR [176] |
Curcumin |
PCa/22Rv1 |
Prodeath [177] |
Gossypol |
PCa xenograft models |
Prodeath [178] |
VK targeting of cancer cells by cell cycle arrest
Cell cycle checkpoint regulatory proteins (Cdk1, Cdk2 and Cdk4) are major effectors of regulation of the cell cycle. These check point regulators are coordinated by the interaction of various cyclins with their cyclin dependent kinases (CDKs) to form active complexes. Cell cycle check point failure often causes mutations and genetic instability which, in turn, can lead to the development of cancer [135]. Hence the identification of novel compounds that are capable of selective inhibition of these kinases (CDKs) are attractive strategies in cancer chemotherapy. Khan et al. [136] reported that pseudolaric acid B induces apoptosis through inhibition of CDKs activity, thereby causing cell cycle arrest, in glioblastoma cells.
One of the important activities of VK3 is inducing cell cycle arrest by inhibiting the activity of CDK-1 check point as has been shown in cervical cancer cells [137]. VK3 causes hyper-phosphorylation on tyrosine residues on the epidermal growth factor receptor (EGFR) and cyclin dependent kinase-1 (CDK-1). Phosphorylation of CDK-1 itself is associated with reduced activity of both CDK-1 and protein tyrosine phosphates [138]. VK3 induces CDK-1 hyper-phosphorylation and also inhibit the cell division cycle 25A (CDC-25A) phosphatase activity, which is a dual specificity phosphatase in cell division and cell cycle, and finally leads to cycle arrest and cell death [137]. This is probably due to ability of VK3 to bind the catalytic domain of cdc25phophatase, thus interfering with the activity of CDC-25 phosphatase and delaying the cellular entry into the S and G2/M phase of cell cycle [137]. Cell division cycle 25 (CDC-25) is a protein tyrosine phosphate critical for cell division, especially at entry of S phase to G2/M phase. Miyazawa et al. [116] reported that VK2 stimulates one of the key CDK-1 inhibitors, p21, in a p53-independent manner. Generally, expression of p21 is upregulated by the p53 tumor suppressor gene, but p21 expression can also be regulated independently of p53 [116]. p21 inhibits cell cycle progression through the inhibition of CDK-2 activity, which is important for the phosphorylation of Rb and activation of E2F dependent gene expression. p21 is shown to uniquely function as a central inhibitor of cdk2 that is activated or induced in response to a variety of cellular signals to promote the tumor suppressor activities as shown in Figure 4.
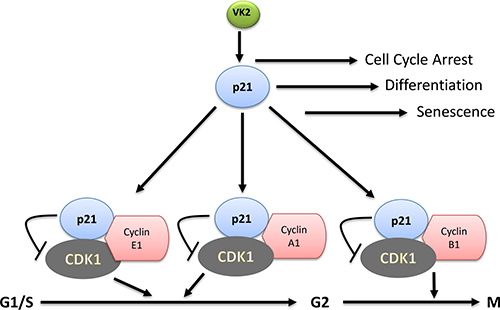
Figure 4: VK2 induced cell cycle regulation in cancer cells. VK2 regulate the cell cycle arrest, cell differentiation and cellular senescence through cdk-1 inhibitor, p21. Subsequently, p21 inhibits the activity of cdk1 by binding with cyclin A1 and E1 and arrest the cell cycle at G1/S-G2 phase. Similarly, p21 inhibits activity of cdk1 by binding with cyclin B1 and arrest the growth at G2/M phase.
Analogues of VK
Some analogues of VK have potent anticancer activity. For example, Nishikawa et al. [139] demonstrated that synthetic quinoid analogues of VK have growth inhibitory effects on Hep3B hepatoma cells through both apoptotic and necrotic cell death and that these effects may be mediated by interaction at position 3 of their quinoid nuclei with cellular thiols. Tamura et al. [140] reported that the 2-(2-mercaptoethanol)-3-methyl-1,4- naphthoquinone analogue of VK had potent anticancer activity through the cell cycle arrest at both G1 and G2/M in hepatoma cells. The same analogue of VK was also shown to have more potent pro-apoptotic activity in primary rat liver cells via the mitogen-activated protein kinase pathway [141]. Kuo et al. [142] reported that plumbagin, an anologue of VK3, has anticancer effects by inhibiting cell proliferation by inducing cell cycle arrest at G2/M phase and autophagic cell death in breast cancer cells. Plumbagin may exert anticancer effects through various mechanisms including disruption of the microtubule network, production of ROS and cell cycle arrest [143]. Lee et al. [144] also reported that Plumbagin had potent antiproliferative activity through the intracellular production of ROS in breast cancer cells. In addition, Yan et al. [145] reported that Plumbagin can trigger DNA damage and also induce cell cycle arrest at S-phase in MG-63 human osteosarcoma cells. Furthermore, plumbagin has been shown to exhibit radiosensitizing effects both in vitro as well as in mouse models [146, 147].
Role of VK as an adjuvant in cancer treatment
Cancer therapy modalities after surgery are either chemo-therapy or radiotherapy, sometimes in combination. Factors that can increase the effectiveness of cancer chemo- or radiotherapy are used as cancer adjuvant therapy. The combination of vitamins C and VK3 has been proposed as a non-toxic mixture of drugs active as an adjuvant cancer therapy by increasing chemo- or radiotherapy effects through alteration of deoxyribonuclease activity [148]. Gilloteaux et al. [149] reported that the co-administration of the non-toxic adjuvant Apatone (Vitamin C + VK3) to radiation and/or chemotherapy treatments to kill bladder and other cancer cells in vitro without any risk or side effects for patients. The combination of VK3 and ascorbate exhibited synergistic anticancer effect, associated with extracellular production of H2O2 that promoted cell death through DNA damage, lysosomal-mitochondrial perturbation and release of cytochrome c [150]. Several reports have indicated that combination of VK2 with certain cytotoxic drugs (sorafenib) exert synergistic anticancer effects [151, 152]. Sorafenib is an oral multikinase inhibitor and function as a single agent chemotherapeutic drug against hepatocellular carcinoma. Zhang et al. [151] investigated that the combination of both sorafenib and VK2 treatment strategy might be effective therapy against hepatocellular carcinoma than the treatment with sorafenib alone. Taken together, these studies support the potential of VK as an adjunct treatment for various cancers.
CONCLUSIONS
In this review, we have summarized the recent progress of VK in various cancers especially PCa. Collective data from different studies indicate that VK is a potential anticancer compound. In particular, the following observations make VK a unique therapeutic agent for treatment of various cancers: (a) It exhibits a broad-spectrum of toxicity toward a wide range of human cancer cells of different origins; (b) It induces apoptosis by interfering with multiple mechanisms that are considered central to cancer development and progression; (c) It can inhibit multiple signaling pathways which are frequently deregulated in human cancers and associated with drug resistance.
Considering aforementioned outcomes, it can be speculated that VK and its derivatives and analogs may become potential compounds for future development of anticancer therapy. However, extensive preclinical and clinical trials are yet required to elucidate the full spectrum of anticancer effects of VK, either alone or in synergistic combination with other standard drugs, to validate its usefulness as a potent anticancer agent.
ACKNOWLEDGMENTS AND FUNDING
This study is partly supported by funding received from NIH, United States (R21 CA184646-01A1).
CONFLICTS OF INTEREST
Authors declare that they do not have conflicts of interest.
REFERENCES
1. Joshi G, Singh PK, Negi A, Rana A, Singh S, Kumar R. Growth factors mediated cell signalling in prostate cancer progression: Implications in discovery of anti-prostate cancer agents. Chem Biol Interact. 2015; 240:120–133.
2. Dean M, Lou H. Genetics and genomics of prostate cancer. Asian J Androl. 2013; 15:309–313.
3. Nelson WG, Sfanos KS, DeMarzo AM, Yegnasubramanian S. Prostate Inflammation and Prostate Cancer. In: Klein AE, Jones SJ, eds. Management of Prostate Cancer. (Totowa, NJ: Humana Press). 2013; 103–115.
4. Karan D, Lin MF, Johansson SL, Batra SK. Current status of the molecular genetics of human prostatic adenocarcinomas. Int J cancer. 2003; 103:285–293.
5. Shukla S, MacLennan GT, Marengo SR, Seftel AD, Resnick MI, Gupta S. Genetic Abnormalities in Prostate Cancer. Current Genomics. 2004; 5:67–83.
6. Amato R, Stepankiw M, Gonzales P. A phase II trial of androgen deprivation therapy (ADT) plus chemotherapy as initial treatment for local failures or advanced prostate cancer. Cancer Chemother Pharmacol. 2013; 71:1629–1634.
7. Shafi AA, Yen AE, Weigel NL. Androgen receptors in hormone-dependent and castration-resistant prostate cancer. Pharmacol Ther. 2013; 140:223–238.
8. Yuan X, Cai C, Chen S, Chen S, Yu Z, Balk SP. Androgen receptor functions in castration-resistant prostate cancer and mechanisms of resistance to new agents targeting the androgen axis. Oncogene. 2014; 33:2815–2825.
9. Bahl A, Oudard S, Tombal B, Ozguroglu M, Hansen S, Kocak I, Gravis G, Devin J, Shen L, de Bono JS, SartorAO. Impact of cabazitaxel on 2-year survival and palliation of tumour-related pain in men with metastatic castration-resistant prostate cancer treated in the TROPIC trial. Ann Oncol. 2013; 24:2402–2408.
10. Acar O, Esen T, Lack NA. New therapeutics to treat castrate-resistant prostate cancer. Scientific World Journal. 2013; 2013:379641.
11. Kelloff GJ, Lieberman R, Steele VE, Boone CW, Lubet RA, Kopelovitch L, Malone WA, Crowell JA, Sigman CC. Chemoprevention of prostate cancer: concepts and strategies. Eur Urol. 1999; 35:342–350.
12. Penson DF, Albertsen PC. Lessons learnt about early prostate cancer from large scale databases: population-based pearls of wisdom. Surg Oncol. 2002; 11:3–11.
13. Freedland SJ, Krupski TL, Moul JW. Update on outcomes research databases in prostate cancer 2006. Curr Opin Urol. 2006; 16:168–172.
14. Shukla S, Gupta S. Dietary agents in the chemoprevention of prostate cancer. Nutr Cancer. 2005; 53:18–32.
15. Sporn MB, Suh N. Chemoprevention: an essential approach to controlling cancer. Nat Rev Cancer. 2002; 2:537–543.
16. Ansari MS, Gupta NP, Hemal AK. Chemoprevention of carcinoma prostate. A review. Int Urol Nephrol. 2002; 34:207–214.
17. Blumentals WA, Foulis PR, Schwartz SW, Mason TJ. Does warfarin therapy influence the risk of bladder cancer? Thromb Haemost. 2004; 91:801–805.
18. Montironi R, Barbisan F, Mazzucchelli R. Target populations, pathological biomarkers and chemopreventive agents in prostate cancer prevention. Arch Ital Urol Androl. 2003; 75:127–134.
19. Riboli E, Norat T. Epidemiologic evidence of the protective effect of fruit and vegetables on cancer risk. Am J Clin Nutr. 2003; 78:559s–569s.
20. Surh YJ. Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer. 2003; 3:768–780.
21. Donaldson MS. Nutrition and cancer: a review of the evidence for an anti-cancer diet. Nutr J. 2004; 3:19.
22. Gonzalez CA, Riboli E. Diet and cancer prevention: Contributions from the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Eur J Cancer (Oxford, England : 1990). 2010; 46:2555–2562.
23. Dasari S, Kathera C, Janardhan A, Praveen Kumar A, Viswanath B. Surfacing role of probiotics in cancer prophylaxis and therapy: A systematic review. Clin Nutr (Edinburgh, Scotland). 2016 Nov 24. [Epub ahead of print].
24. Kakizoe T. Chemoprevention of cancer—focusing on clinical trials. Jpn J Clin Oncol. 2003; 33:421–442.
25. Cohen LA. Nutrition and prostate cancer: a review. Ann N Y Acad Sci. 2002; 963:148–155.
26. Willis MS, Wians FH. The role of nutrition in preventing prostate cancer: a review of the proposed mechanism of action of various dietary substances. Clin Chim Acta. 2003; 330:57–83.
27. Greenwald P, Milner JA, Anderson DE, McDonald SS. Micronutrients in cancer chemoprevention. Cancer Metastasis Rev. 2002; 21:217–230.
28. Hamilton Z, Parsons JK. Prostate Cancer Prevention: Concepts and Clinical Trials. Curr Urol Rep. 2016; 17:35.
29. Hwang ES, Bowen PE. Can the consumption of tomatoes or lycopene reduce cancer risk? Integr Cancer Ther. 2002; 1:121–132; discussion 132.
30. Jian L, Du CJ, Lee AH, Binns CW. Do dietary lycopene and other carotenoids protect against prostate cancer? Int J Cancer. 2005; 113:1010–1014.
31. Etminan M, Takkouche B, Caamano-Isorna F. The role of tomato products and lycopene in the prevention of prostate cancer: a meta-analysis of observational studies. Cancer Epidemiol Biomarkers Prev. 2004; 13:340–345.
32. Boileau TW, Liao Z, Kim S, Lemeshow S, Erdman JW Jr, Clinton SK. Prostate carcinogenesis in N-methyl-N-nitrosourea (NMU)-testosterone-treated rats fed tomato powder, lycopene, or energy-restricted diets. J Natl Cancer Inst. 2003; 95:1578–1586.
33. Gann PH, Deaton RJ, Rueter EE, van Breemen RB, Nonn L, Macias V, Han M, Ananthanarayanan V. A Phase II Randomized Trial of Lycopene-Rich Tomato Extract Among Men with High-Grade Prostatic Intraepithelial Neoplasia. Nutr Cancer. 2015; 67:1104–1112.
34. Blomhoff R, Green MH, Norum KR. Vitamin A: physiological and biochemical processing. Annu Rev Nutr. 1992; 12:37–57.
35. Igawa M, Tanabe T, Chodak GW, Rukstalis DB. N-(4-hydroxyphenyl) retinamide induces cell cycle specific growth inhibition in PC3 cells. The Prostate. 1994; 24:299–305.
36. Hu ML, Shih MK. Ascorbic acid inhibits lipid peroxidation but enhances DNA damage in rat liver nuclei incubated with iron ions. Free Radic Res. 1997; 26:585–592.
37. Menon M, Maramag C, Malhotra RK, Seethalakshmi L. Effect of vitamin C on androgen independent prostate cancer cells (PC3 and Mat-Ly-Lu) in vitro: involvement of reactive oxygen species-effect on cell number, viability and DNA synthesis. Cancer Biochem Biophys. 1998; 16:17–30.
38. Taper HS, Jamison JM, Gilloteaux J, Gwin CA, Gordon T, Summers JL. In vivo reactivation of DNases in implanted human prostate tumors after administration of a vitamin C/K combination. J Histochem Cytochem. 2001; 49:109–120.
39. Niedzwiecki A, Roomi MW, Kalinovsky T, Rath M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients. 2016; 8.
40. John EM, Dreon DM, Koo J, Schwartz GG. Residential sunlight exposure is associated with a decreased risk of prostate cancer. J Steroid Biochem Mol Biol. 2004; 89–90:549–552.
41. Stewart LV, Weigel NL. Vitamin D and prostate cancer. Exp Biol Med (Maywood). 2004; 229:277–284.
42. Polek TC, Stewart LV, Ryu EJ, Cohen MB, Allegretto EA, Weigel NL. p53 Is required for 1,25-dihydroxyvitamin D3-induced G0 arrest but is not required for G1 accumulation or apoptosis of LNCaP prostate cancer cells. Endocrinology. 2003; 144:50–60.
43. Rao A, Woodruff RD, Wade WN, Kute TE, Cramer SD. Genistein and vitamin D synergistically inhibit human prostatic epithelial cell growth. J Nutr. 2002; 132:3191–3194.
44. Lucia MS, Torkko KC. Inflammation as a target for prostate cancer chemoprevention: pathological and laboratory rationale. J Urol. 2004; 171:S30–34; discussion S35.
45. Brawley OW, Barnes S, Parnes H. The future of prostate cancer prevention. Ann N Y Acad Sci. 2001; 952:145–152.
46. Bosland MC, Ozten N, Eskra JN, Mahmoud AM. A Perspective on Prostate Carcinogenesis and Chemoprevention. Curr Pharmacol Rep. 2015; 1:258–265.
47. Rodriguez C, Jacobs EJ, Mondul AM, Calle EE, McCullough ML, Thun MJ. Vitamin E supplements and risk of prostate cancer in U.S. men. Cancer Epidemiol Biomarkers Prev. 2004; 13:378–382.
48. Kirsh VA, Hayes RB, Mayne ST, Chatterjee N, Subar AF, Dixon LB, Albanes D, Andriole GL, Urban DA, Peters U. Supplemental and dietary vitamin E, beta-carotene, and vitamin C intakes and prostate cancer risk. J Natl Cancer Inst. 2006; 98:245–254.
49. Barve A, Khor TO, Nair S, Reuhl K, Suh N, Reddy B, Newmark H, Kong AN. Gamma-tocopherol-enriched mixed tocopherol diet inhibits prostate carcinogenesis in TRAMP mice. Int J Cancer. 2009; 124:1693–1699.
50. Chen JX, Li G, Wang H, Liu A, Lee MJ, Reuhl K, Suh N, Bosland MC, Yang CS. Dietary tocopherols inhibit PhIP-induced prostate carcinogenesis in CYP1A-humanized mice. Cancer lett. 2016; 371:71–78.
51. Greenwald P, Clifford CK, Milner JA. Diet and cancer prevention. Eur J Cancer. 2001; 37:948–965.
52. Sarkar FH, Li Y. Cell signaling pathways altered by natural chemopreventive agents. Mutat Res. 2004; 555:53–64.
53. Kumar NB, Hopkins K, Allen K, Riccardi D, Besterman-Dahan K, Moyers S. Use of complementary/integrative nutritional therapies during cancer treatment: implications in clinical practice. Cancer Control. 2002; 9:236–243.
54. Klein EA, Thompson IM Jr, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, Karp DD, Lieber MM, Walther PJ, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). Jama. 2011; 306:1549–1556.
55. Lippman SM, Klein EA, Goodman PJ, Lucia MS, Thompson IM, Ford LG, Parnes HL, Minasian LM, Gaziano JM, Hartline JA, Parsons JK, Bearden JD 3rd, Crawford ED, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). Jama. 2009; 301:39–51.
56. Lu J, Zhang J, Jiang C, Deng Y, Ozten N, Bosland MC. Cancer chemoprevention research with selenium in the post-SELECT era: Promises and challenges. Nutr Cancer. 2016; 68:1–17.
57. Kristal AR, Darke AK, Morris JS, Tangen CM, Goodman PJ, Thompson IM, Meyskens FL Jr, Goodman GE, Minasian LM, Parnes HL, Lippman SM, Klein EA. Baseline selenium status and effects of selenium and vitamin e supplementation on prostate cancer risk. J Natl Cancer Inst. 2014; 106:djt456.
58. Lamson DW, Plaza SM. The anticancer effects of vitamin K. Altern Med Rev. 2003; 8:303–318.
59. Kitano T, Yoda H, Tabata K, Miura M, Toriyama M, Motohashi S, Suzuki T. Vitamin K3 analogs induce selective tumor cytotoxicity in neuroblastoma. Biol Pharm Bull. 2012; 35:617–623.
60. Ishibashi M, Arai M, Tanaka S, Onda K, Hirano T. Antiproliferative and apoptosis-inducing effects of lipophilic vitamins on human melanoma A375 cells in vitro. Biol Pharm Bull. 2012; 35:10–17.
61. Wang Z, Wang M, Finn F, Carr BI. The growth inhibitory effects of vitamins K and their actions on gene expression. Hepatology. 1995; 22:876–882.
62. Sata N, Klonowski-Stumpe H, Han B, Haussinger D, Niederau C. Menadione induces both necrosis and apoptosis in rat pancreatic acinar AR4-2J cells. Free Radic Biol Med. 1997; 23:844–850.
63. Margolin KA, Akman SA, Leong LA, Morgan RJ, Somlo G, Raschko JW, Ahn C, Doroshow JH. Phase I study of mitomycin C and menadione in advanced solid tumors. Cancer Chemother Pharmacol. 1995; 36:293–298.
64. Nimptsch K, Rohrmann S, Linseisen J. Dietary intake of vitamin K and risk of prostate cancer in the Heidelberg cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Heidelberg). Am J Clin Nutr. 2008; 87:985–992.
65. Tamori A, Habu D, Shiomi S, Kubo S, Nishiguchi S. Potential role of vitamin K(2) as a chemopreventive agent against hepatocellular carcinoma. Hepatol Res. 2007; 37:S303–307.
66. Chu KJ, Lai EC, Yao XP, Zhang HW, Lau WY, Fu XH, Lu CD, Shi J, Cheng SQ. Vitamin analogues in chemoprevention of hepatocellular carcinoma after resection or ablation—a systematic review and meta-analysis. Asian J Surg. 2010; 33:120–126.
67. Riaz IB, Riaz H, Riaz T, Rahman S, Amir M, Badshah MB, Kazi AN. Role of vitamin K2 in preventing the recurrence of hepatocellular carcinoma after curative treatment: a meta-analysis of randomized controlled trials. BMC Gastroenterol. 2012; 12:170.
68. Zhong JH, Mo XS, Xiang BD, Yuan WP, Jiang JF, Xie GS, Li LQ. Postoperative use of the chemopreventive vitamin K2 analog in patients with hepatocellular carcinoma. PloS one. 2013; 8:e58082.
69. Mitchell JS, Simon-Reuss I. Combination of some effects of x-radiation and a synthetic vitamin K substitute. Nature. 1947; 159:98.
70. Verrax J, Cadrobbi J, Delvaux M, Jamison JM, Gilloteaux J, Summers JL, Taper HS, Buc Calderon P. The association of vitamins C and K3 kills cancer cells mainly by autoschizis, a novel form of cell death. Basis for their potential use as coadjuvants in anticancer therapy. Eur J Med Chem. 2003; 38:451–457.
71. Tokita H, Tsuchida A, Miyazawa K, Ohyashiki K, Katayanagi S, Sudo H, Enomoto M, Takagi Y, Aoki T. Vitamin K2-induced antitumor effects via cell-cycle arrest and apoptosis in gastric cancer cell lines. Int J Mol Med. 2006; 17:235–243.
72. Otsuka M, Kato N, Shao RX, Hoshida Y, Ijichi H, Koike Y, Taniguchi H, Moriyama M, Shiratori Y, Kawabe T, Omata M. Vitamin K2 inhibits the growth and invasiveness of hepatocellular carcinoma cells via protein kinase A activation. Hepatology. 2004; 40:243–251.
73. Okayasu H, Ishihara M, Satoh K, Sakagami H. Cytotoxic activity of vitamins K1, K2 and K3 against human oral tumor cell lines. Anticancer Res. 2001; 21:2387–2392.
74. Tomasetti M, Nocchi L, Neuzil J, Goodwin J, Nguyen M, Dong L, Manzella N, Staffolani S, Milanese C, Garrone B, Alleva R, Borghi B, Santarelli L, et al. Alpha-tocopheryl succinate inhibits autophagic survival of prostate cancer cells induced by vitamin K3 and ascorbate to trigger cell death. PloS one. 2012; 7:e52263.
75. Degen M, Alexander B, Choudhury M, Eshghi M, Konno S. Alternative therapeutic approach to renal-cell carcinoma: induction of apoptosis with combination of vitamin K3 and D-fraction. J Endourol. 2013; 27:1499–1503.
76. Yang CR, Liao WS, Wu YH, Murugan K, Chen C, Chao JI. CR108, a novel vitamin K3 derivative induces apoptosis and breast tumor inhibition by reactive oxygen species and mitochondrial dysfunction. Toxicol Appl Pharmacol. 2013; 273:611–622.
77. Suresh S, Raghu D, Karunagaran D. Menadione (Vitamin K3) induces apoptosis of human oral cancer cells and reduces their metastatic potential by modulating the expression of epithelial to mesenchymal transition markers and inhibiting migration. Asian Pac J Cancer Prev. 2013; 14:5461–5465.
78. Nimptsch K, Rohrmann S, Kaaks R, Linseisen J. Dietary vitamin K intake in relation to cancer incidence and mortality: results from the Heidelberg cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Heidelberg). Am J Clin Nutr. 2010; 91:1348–1358.
79. Nimptsch K, Rohrmann S, Nieters A, Linseisen J. Serum undercarboxylated osteocalcin as biomarker of vitamin K intake and risk of prostate cancer: a nested case-control study in the Heidelberg cohort of the European prospective investigation into cancer and nutrition. Cancer Epidemiol Biomarkers Prev. 2009; 18:49–56.
80. Schulman S, Lindmarker P. Incidence of cancer after prophylaxis with warfarin against recurrent venous thromboembolism. Duration of Anticoagulation Trial. N Engl J Med. 2000; 342:1953–1958.
81. Taliani MR, Agnelli G, Prandoni P, Becattini C, Moia M, Bazzan M, Ageno W, Tomasi C, Guazzaloca G, Ambrosio GB, Bertoldi A, Salvi R, Poggio R, et al. Incidence of cancer after a first episode of idiopathic venous thromboembolism treated with 3 months or 1 year of oral anticoagulation. J Thromb Haemost. 2003; 1:1730–1733.
82. Pottegard A, Friis S, Hallas J. Cancer risk in long-term users of vitamin K antagonists: a population-based case-control study. Int J Cancer. 2013; 132:2606–2612.
83. Samykutty A, Shetty AV, Dakshinamoorthy G, Kalyanasundaram R, Zheng G, Chen A, Bosland MC, Kajdacsy-Balla A, Gnanasekar M. Vitamin k2, a naturally occurring menaquinone, exerts therapeutic effects on both hormone-dependent and hormone-independent prostate cancer cells. Evid Based Complement Alternat Med. 2013; 2013:287358.
84. Yokoyama T, Miyazawa K, Yoshida T, Ohyashiki K. Combination of vitamin K2 plus imatinib mesylate enhances induction of apoptosis in small cell lung cancer cell lines. Int J Oncol. 2005; 26:33–40.
85. Sibayama-Imazu T, Fujisawa Y, Masuda Y, Aiuchi T, Nakajo S, Itabe H, Nakaya K. Induction of apoptosis in PA-1 ovarian cancer cells by vitamin K2 is associated with an increase in the level of TR3/Nur77 and its accumulation in mitochondria and nuclei. J Cancer Res Clin Oncol. 2008; 134:803–812.
86. Yao Y, Li L, Zhang H, Jia R, Liu B, Zhao X, Zhang L, Qian G, Fan X, Ge S. Enhanced therapeutic efficacy of vitamin K2 by silencing BCL-2 expression in SMMC-7721 hepatocellular carcinoma cells. Oncology lett. 2012; 4:163–167.
87. Yamamoto T, Nakamura H, Liu W, Cao K, Yoshikawa S, Enomoto H, Iwata Y, Koh N, Saito M, Imanishi H, Shimomura S, Iijima H, Hada T, et al. Involvement of hepatoma-derived growth factor in the growth inhibition of hepatocellular carcinoma cells by vitamin K(2). J Gastroenterol. 2009; 44:228–235.
88. Shetty A, Dasari S, Banerjee S, Gheewala T, Zheng G, Chen A, Kajdacsy-Balla A, Bosland MC, Munirathinam G. Hepatoma-derived growth factor: A survival-related protein in prostate oncogenesis and a potential target for vitamin K2. Urol Oncol. 2016; 34:483.e481–483.e488.
89. Jamison JM, Gilloteaux J, Taper HS, Summers JL. Evaluation of the in vitro and in vivo antitumor activities of vitamin C and K-3 combinations against human prostate cancer. J Nutr. 2001; 131:158s–160s.
90. Gilloteaux J, Jamison JM, Neal D, Summers JL. Synergistic antitumor cytotoxic actions of ascorbate and menadione on human prostate (DU145) cancer cells in vitro: nucleus and other injuries preceding cell death by autoschizis. Ultrastruct Pathol. 2014; 38:116–140.
91. Zhou ZW, Li XX, He ZX, Pan ST, Yang Y, Zhang X, Chow K, Yang T, Qiu JX, Zhou Q, Tan J, Wang D, Zhou SF. Induction of apoptosis and autophagy via sirtuin1- and PI3K/Akt/mTOR-mediated pathways by plumbagin in human prostate cancer cells. Drug Des Devel Ther. 2015; 9:1511–1554.
92. Reshma RS, Sreelatha KH, Somasundaram V, Satheesh Kumar S, Nadhan R, Nair RS, Srinivas P. Plumbagin, a naphthaquinone derivative induces apoptosis in BRCA 1/2 defective castrate resistant prostate cancer cells as well as prostate cancer stem-like cells. Pharmacol Res. 2016; 105:134–145.
93. Hafeez BB, Fischer JW, Singh A, Zhong W, Mustafa A, Meske L, Sheikhani MO, Verma AK. Plumbagin Inhibits Prostate Carcinogenesis in Intact and Castrated PTEN Knockout Mice via Targeting PKCepsilon, Stat3, and Epithelial-to-Mesenchymal Transition Markers. Cancer Prev Res (Phila). 2015; 8:375–386.
94. Valko M, Izakovic M, Mazur M, Rhodes CJ, Telser J. Role of oxygen radicals in DNA damage and cancer incidence. Mol Cell Biochem. 2004; 266:37–56.
95. Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB. Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med. 2010; 49:1603–1616.
96. Visconti R, Grieco D. New insights on oxidative stress in cancer. Curr Opin Drug Discov Devel. 2009; 12:240–245.
97. Schumacker PT. Reactive oxygen species in cancer cells: live by the sword, die by the sword. Cancer Cell. 2006; 10:175–176.
98. Fang J, Seki T, Maeda H. Therapeutic strategies by modulating oxygen stress in cancer and inflammation. Adv Drug Deliv Rev. 2009; 61:290–302.
99. Nutter LM, Ngo EO, Fisher GR, Gutierrez PL. DNA strand scission and free radical production in menadione-treated cells. Correlation with cytotoxicity and role of NADPH quinone acceptor oxidoreductase. J Biol Chem. 1992; 267:2474–2479.
100. Ross D, Thor H, Threadgill MD, Sandy MS, Smith MT, Moldeus P, Orrenius S. The role of oxidative processes in the cytotoxicity of substituted 1,4-naphthoquinones in isolated hepatocytes. Arch Biochem Biophys. 1986; 248:460–466.
101. Hetts SW. To die or not to die: an overview of apoptosis and its role in disease. Jama. 1998; 279:300–307.
102. Caricchio R, Kovalenko D, Kaufmann WK, Cohen PL. Apoptosis provoked by the oxidative stress inducer menadione (Vitamin K(3)) is mediated by the Fas/Fas ligand system. Clin Immunol. 1999; 93:65–74.
103. Gottlieb RA, Nordberg J, Skowronski E, Babior BM. Apoptosis induced in Jurkat cells by several agents is preceded by intracellular acidification. Proc Natl Acad Sci USA. 1996; 93:654–658.
104. Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M, Waters CM, Penn LZ, Hancock DC. Induction of apoptosis in fibroblasts by c-myc protein. Cell. 1992; 69:119–128.
105. Hug H, Strand S, Grambihler A, Galle J, Hack V, Stremmel W, Krammer PH, Galle PR. Reactive oxygen intermediates are involved in the induction of CD95 ligand mRNA expression by cytostatic drugs in hepatoma cells. J Biol Chem. 1997; 272:28191–28193.
106. Fuchs Y, Steller H. Programmed cell death in animal development and disease. Cell. 2011; 147:742–758.
107. Wu H, Che X, Zheng Q, Wu A, Pan K, Shao A, Wu Q, Zhang J, Hong Y. Caspases: a molecular switch node in the crosstalk between autophagy and apoptosis. Int J Biol Sci. 2014; 10:1072–1083.
108. Cotter TG. Apoptosis and cancer: the genesis of a research field. Nat Rev Cancer. 2009; 9:501–507.
109. Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007; 35:495–516.
110. Toshiya Kuno TT, Akira Hara, Takuji Tanaka. Cancer chemoprevention through the induction of apoptosis by natural compounds. Biophys Chem. 2012; 3:156–173.
111. Ghobrial IM, Witzig TE, Adjei AA. Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin. 2005; 55:178–194.
112. Roth W, Reed JC. Apoptosis and cancer: when BAX is TRAILing away. Nat Med. 2002; 8:216–218.
113. Sakagami H, Satoh K, Hakeda Y, Kumegawa M. Apoptosis-inducing activity of vitamin C and vitamin K. Cell Mol Biol (Noisy-le-grand). 2000; 46:129–143.
114. Ogawa M, Nakai S, Deguchi A, Nonomura T, Masaki T, Uchida N, Yoshiji H, Kuriyama S. Vitamins K2, K3 and K5 exert antitumor effects on established colorectal cancer in mice by inducing apoptotic death of tumor cells. Int J Oncol. 2007; 31:323–331.
115. Wang J, Yi J. Cancer cell killing via ROS: to increase or decrease, that is the question. Cancer Biol Ther. 2008; 7:1875–1884.
116. Miyazawa K, Yaguchi M, Funato K, Gotoh A, Kawanishi Y, Nishizawa Y, Yuo A, Ohyashiki K. Apoptosis/differentiation-inducing effects of vitamin K2 on HL-60 cells: dichotomous nature of vitamin K2 in leukemia cells. Leukemia. 2001; 15:1111–1117.
117. Henry-Mowatt J, Dive C, Martinou JC, James D. Role of mitochondrial membrane permeabilization in apoptosis and cancer. Oncogene. 2004; 23:2850–2860.
118. Karasawa S, Azuma M, Kasama T, Sakamoto S, Kabe Y, Imai T, Yamaguchi Y, Miyazawa K, Handa H. Vitamin K2 covalently binds to Bak and induces Bak-mediated apoptosis. Mol Pharmacol. 2013; 83:613–620.
119. Yokoyama T, Miyazawa K, Naito M, Toyotake J, Tauchi T, Itoh M, Yuo A, Hayashi Y, Georgescu MM, Kondo Y, Kondo S, Ohyashiki K. Vitamin K2 induces autophagy and apoptosis simultaneously in leukemia cells. Autophagy. 2008; 4:629–640.
120. Shibayama-Imazu T, Aiuchi T, Nakaya K. Vitamin K2-mediated apoptosis in cancer cells: role of mitochondrial transmembrane potential. Vitam Horm. 2008; 78:211–226.
121. Korsmeyer SJ, Wei MC, Saito M, Weiler S, Oh KJ, Schlesinger PH. Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that result in the release of cytochrome c. Cell Death Differ. 2000; 7:1166–1173.
122. Kasibhatla S, Genestier L, Green DR. Regulation of fas-ligand expression during activation-induced cell death in T lymphocytes via nuclear factor kappaB. J Biol Chem. 1999; 274:987–992.
123. Faris M, Latinis KM, Kempiak SJ, Koretzky GA, Nel A. Stress-induced Fas ligand expression in T cells is mediated through a MEK kinase 1-regulated response element in the Fas ligand promoter. Mol Cell Biol. 1998; 18:5414–5424.
124. Hueber AO, Zornig M, Lyon D, Suda T, Nagata S, Evan GI. Requirement for the CD95 receptor-ligand pathway in c-Myc-induced apoptosis. Science. 1997; 278:1305–1309.
125. van den Dobbelsteen DJ, Nobel CS, Schlegel J, Cotgreave IA, Orrenius S, Slater AF. Rapid and specific efflux of reduced glutathione during apoptosis induced by anti-Fas/APO-1 antibody. J Biol Chem. 1996; 271:15420–15427.
126. Laux I, Nel A. Evidence that oxidative stress-induced apoptosis by menadione involves Fas-dependent and Fas-independent pathways. Clin Immunol. 2001; 101:335–344.
127. Showalter SL, Wang Z, Costantino CL, Witkiewicz AK, Yeo CJ, Brody JR, Carr BI. Naturally occurring K vitamins inhibit pancreatic cancer cell survival through a caspase-dependent pathway. J Gastroenterol Hepatol. 2010; 25:738–744.
128. Du W, Zhou JR, Wang DL, Gong K, Zhang QJ. Vitamin K1 enhances sorafenib-induced growth inhibition and apoptosis of human malignant glioma cells by blocking the Raf/MEK/ERK pathway. World J Surg Oncol. 2012; 10:60.
129. Yoshimori T. Autophagy: a regulated bulk degradation process inside cells. Biochem Biophys Res Commun. 2004; 313:453–458.
130. Yang ZJ, Chee CE, Huang S, Sinicrope FA. The role of autophagy in cancer: therapeutic implications. Mol Cancer Ther. 2011; 10:1533–1541.
131. Singletary K, Milner J. Diet, autophagy, and cancer: a review. Cancer Epidemiol Biomarkers Prev. 2008; 17:1596–1610.
132. Naponelli V, Modernelli A, Bettuzzi S, Rizzi F. Roles of autophagy induced by natural compounds in prostate cancer. Biomed Res Int. 2015; 2015:121826.
133. Tsang CK, Qi H, Liu LF, Zheng XF. Targeting mammalian target of rapamycin (mTOR) for health and diseases. Drug Discov Today. 2007; 12:112–124.
134. Enomoto M, Tsuchida A, Miyazawa K, Yokoyama T, Kawakita H, Tokita H, Naito M, Itoh M, Ohyashiki K, Aoki T. Vitamin K2-induced cell growth inhibition via autophagy formation in cholangiocellular carcinoma cell lines. Int J Mol Med. 2007; 20:801–808.
135. Millimouno FM, Dong J, Yang L, Li J, Li X. Targeting apoptosis pathways in cancer and perspectives with natural compounds from mother nature. Cancer Prev Res (Phila). 2014; 7:1081–1107.
136. Khan M, Zheng B, Yi F, Rasul A, Gu Z, Li T, Gao H, Qazi JI, Yang H, Ma T. Pseudolaric Acid B induces caspase-dependent and caspase-independent apoptosis in u87 glioblastoma cells. Evid Based Complement Alternat Med. 2012; 2012:957568.
137. Wu FY, Sun TP. Vitamin K3 induces cell cycle arrest and cell death by inhibiting Cdc25 phosphatase. Eur J Cancer. 1999; 35:1388–1393.
138. Wu FYH JCC, Sun TP, Chang WT. Vitamin K3- induced cell cycle progression delay in human tumor cells correlates with decreased activity of p34cdc2 kinase. Proc Am Assoc Cancer Res. 1993; 34:291.
139. Nishikawa Y, Carr BI, Wang M, Kar S, Finn F, Dowd P, Zheng ZB, Kerns J, Naganathan S. Growth inhibition of hepatoma cells induced by vitamin K and its analogs. J Biol Chem. 1995; 270:28304–28310.
140. Tamura K, Southwick EC, Kerns J, Rosi K, Carr BI, Wilcox C, Lazo JS. Cdc25 inhibition and cell cycle arrest by a synthetic thioalkyl vitamin K analogue. Cancer Res. 2000; 60:1317–1325.
141. Wang Z, Nishikawa Y, Wang M, Carr BI. Induction of apoptosis via mitogen-activated protein kinase pathway by a K vitamin analog in rat hepatocytes. J Hepatol. 2002; 36:85–92.
142. Kuo PL, Hsu YL, Cho CY. Plumbagin induces G2-M arrest and autophagy by inhibiting the AKT/mammalian target of rapamycin pathway in breast cancer cells. Mol Cancer Ther. 2006; 5:3209–3221.
143. Tian L, Yin D, Ren Y, Gong C, Chen A, Guo FJ. Plumbagin induces apoptosis via the p53 pathway and generation of reactive oxygen species in human osteosarcoma cells. Mol Med Rep. 2012; 5:126–132.
144. Lee JH, Yeon JH, Kim H, Roh W, Chae J, Park HO, Kim DM. The natural anticancer agent plumbagin induces potent cytotoxicity in MCF-7 human breast cancer cells by inhibiting a PI-5 kinase for ROS generation. PloS one. 2012; 7:e45023.
145. Yan CH, Li F, Ma YC. Plumbagin shows anticancer activity in human osteosarcoma (MG-63) cells via the inhibition of S-Phase checkpoints and down-regulation of c-myc. Int J Clin Exp Med. 2015; 8:14432–14439.
146. Devi PU, Rao BS, Solomon FE. Effect of plumbagin on the radiation induced cytogenetic and cell cycle changes in mouse Ehrlich ascites carcinoma in vivo. Indian J Exp Biol. 1998; 36:891–895.
147. Ganasoundari A, Zare SM, Devi PU. Modification of bone marrow radiosensensitivity by medicinal plant extracts. Br J Radiol. 1997; 70:599–602.
148. Taper HS. Altered deoxyribonuclease activity in cancer cells and its role in non toxic adjuvant cancer therapy with mixed vitamins C and K3. Anticancer Res. 2008; 28:2727–2732.
149. Gilloteaux J, Jamison JM, Neal DR, Loukas M, Doberzstyn T, Summers JL. Cell damage and death by autoschizis in human bladder (RT4) carcinoma cells resulting from treatment with ascorbate and menadione. Ultrastruct Pathol. 2010; 34:140–160.
150. Tomasetti M, Strafella E, Staffolani S, Santarelli L, Neuzil J, Guerrieri R. alpha-Tocopheryl succinate promotes selective cell death induced by vitamin K3 in combination with ascorbate. Br J Cancer. 2010; 102:1224–1234.
151. Zhang Y, Zhang B, Zhang A, Zhao Y, Zhao J, Liu J, Gao J, Fang D, Rao Z. Synergistic growth inhibition by sorafenib and vitamin K2 in human hepatocellular carcinoma cells. Clinics (Sao Paulo). 2012; 67:1093–1099.
152. Jung DH, Hwang S, Song GW, Ryoo BY, Kim N, Tak E, Hong HN. An interim safety analysis of hepatocellular carcinoma patients administrating oral vitamin K with or without sorafenib. Korean J Hepatobiliary Pancreat Surg. 2015; 19:1–5.
153. Aust O, Ale-Agha N, Zhang L, Wollersen H, Sies H, Stahl W. Lycopene oxidation product enhances gap junctional communication. Food Chem Toxicol. 2003; 41:1399–1407.
154. Liang JY, Fontana JA, Rao JN, Ordonez JV, Dawson MI, Shroot B, Wilber JF, Feng P. Synthetic retinoid CD437 induces S-phase arrest and apoptosis in human prostate cancer cells LNCaP and PC-3. The Prostate. 1999; 38:228–236.
155. Fazzio A, Marilley D, Azzi A. The effect of alpha-tocopherol and beta-tocopherol on proliferation, protein kinase C activity and gene expression in different cell lines. Biochem Mol Biol Int. 1997; 41:93–101.
156. Sinha R, El-Bayoumy K. Apoptosis is a critical cellular event in cancer chemoprevention and chemotherapy by selenium compounds. Curr Cancer Drug Targets. 2004; 4:13–28.
157. Lippman SM, Goodman PJ, Klein EA, Parnes HL, Thompson IM Jr, Kristal AR, Santella RM, Probstfield JL, Moinpour CM, Albanes D, Taylor PR, Minasian LM, Hoque A, et al. Designing the Selenium and Vitamin E Cancer Prevention Trial (SELECT). J Natl Cancer Inst. 2005; 97:94–102.
158. Davis JN, Singh B, Bhuiyan M, Sarkar FH. Genistein-induced upregulation of p21WAF1, downregulation of cyclin B, and induction of apoptosis in prostate cancer cells. Nutr Cancer. 1998; 32:123–131.
159. Siddiqui IA, Adhami VM, Afaq F, Ahmad N, Mukhtar H. Modulation of phosphatidylinositol-3-kinase/protein kinase B- and mitogen-activated protein kinase-pathways by tea polyphenols in human prostate cancer cells. J Cell Biochem. 2004; 91:232–242.
160. Hsieh TC, Wu JM. Differential effects on growth, cell cycle arrest, and induction of apoptosis by resveratrol in human prostate cancer cell lines. Exp Cell Res. 1999; 249:109–115.
161. Singh RP, Agarwal R. Prostate cancer prevention by silibinin. Curr Cancer Drug Targets. 2004; 4:1–11.
162. Agarwal C, Singh RP, Dhanalakshmi S, Agarwal R. Anti-angiogenic efficacy of grape seed extract in endothelial cells. Oncol Rep. 2004; 11:681–685.
163. Shukla S, Gupta S. Molecular mechanisms for apigenin-induced cell-cycle arrest and apoptosis of hormone refractory human prostate carcinoma DU145 cells. Mol Carcinog. 2004; 39:114–126.
164. Howells LM, Gallacher-Horley B, Houghton CE, Manson MM, Hudson EA. Indole-3-carbinol inhibits protein kinase B/Akt and induces apoptosis in the human breast tumor cell line MDA MB468 but not in the nontumorigenic HBL100 line. Mol Cancer Ther. 2002; 1:1161–1172.
165. Xu C, Shen G, Chen C, Gelinas C, Kong AN. Suppression of NF-kappaB and NF-kappaB-regulated gene expression by sulforaphane and PEITC through IkappaBalpha, IKK pathway in human prostate cancer PC-3 cells. Oncogene. 2005; 24:4486–4495.
166. Kumar AP, Garcia GE, Ghosh R, Rajnarayanan RV, Alworth WL, Slaga TJ. 4-Hydroxy-3-methoxybenzoic acid methyl ester: a curcumin derivative targets Akt/NF kappa B cell survival signaling pathway: potential for prostate cancer management. Neoplasia. 2003; 5:255–266.
167. Aoki H, Takada Y, Kondo S, Sawaya R, Aggarwal BB, Kondo Y. Evidence that curcumin suppresses the growth of malignant gliomas in vitro and in vivo through induction of autophagy: role of Akt and extracellular signal-regulated kinase signaling pathways. Mol Pharmacol. 2007; 72:29–39.
168. Gossner G, Choi M, Tan L, Fogoros S, Griffith KA, Kuenker M, Liu JR. Genistein-induced apoptosis and autophagocytosis in ovarian cancer cells. Gynecol Oncol. 2007; 105:23–30.
169. Kueck A, Opipari AW Jr, Griffith KA, Tan L, Choi M, Huang J, Wahl H, Liu JR. Resveratrol inhibits glucose metabolism in human ovarian cancer cells. Gynecol Oncol. 2007; 107:450–457.
170. Ohshiro K, Rayala SK, Kondo S, Gaur A, Vadlamudi RK, El-Naggar AK, Kumar R. Identifying the estrogen receptor coactivator PELP1 in autophagosomes. Cancer Res. 2007; 67:8164–8171.
171. Herman-Antosiewicz A, Johnson DE, Singh SV. Sulforaphane causes autophagy to inhibit release of cytochrome C and apoptosis in human prostate cancer cells. Cancer Res. 2006; 66:5828–5835.
172. Martin A, Joseph JA, Cuervo AM. Stimulatory effect of vitamin C on autophagy in glial cells. J Neurochem. 2002; 82:538–549.
173. Ohtani S, Iwamaru A, Deng W, Ueda K, Wu G, Jayachandran G, Kondo S, Atkinson EN, Minna JD, Roth JA, Ji L. Tumor suppressor 101F6 and ascorbate synergistically and selectively inhibit non-small cell lung cancer growth by caspase-independent apoptosis and autophagy. Cancer Res. 2007; 67:6293–6303.
174. Tavera-Mendoza L, Wang TT, Lallemant B, Zhang R, Nagai Y, Bourdeau V, Ramirez-Calderon M, Desbarats J, Mader S, White JH. Convergence of vitamin D and retinoic acid signalling at a common hormone response element. EMBO reports. 2006; 7:180–185.
175. Chen P, Yu J, Chalmers B, Drisko J, Yang J, Li B, Chen Q. Pharmacological ascorbate induces cytotoxicity in prostate cancer cells through ATP depletion and induction of autophagy. Anticancer Drugs. 2012; 23:437–444.
176. Makhov P, Golovine K, Teper E, Kutikov A, Mehrazin R, Corcoran A, Tulin A, Uzzo RG, Kolenko VM. Piperlongumine promotes autophagy via inhibition of Akt/mTOR signalling and mediates cancer cell death. Br J Cancer. 2014; 110:899–907.
177. Teiten MH, Gaascht F, Cronauer M, Henry E, Dicato M, Diederich M. Anti-proliferative potential of curcumin in androgen-dependent prostate cancer cells occurs through modulation of the Wingless signaling pathway. Int J Oncol. 2011; 38:603–611.
178. Lian J, Ni Z, Dai X, Su C, Smith AR, Xu L, He F. Sorafenib sensitizes (-)-gossypol-induced growth suppression in androgen-independent prostate cancer cells via Mcl-1 inhibition and Bak activation. Mol Cancer Ther. 2012; 11:416–426.

