INTRODUCTION
Nucleotide excision repair (NER) is a critical and versatile system that monitors and repairs a broad spectrum of DNA damage. NER is composed of global genome nucleotide excision repair (GGR) and transcription-coupled nucleotide excision repair (TCR) [1]. TCR specifically repairs RNA polymerase II (RNAPII) blocking DNA lesions and ensures the transcribed strand of the active gene to be repaired preferentially than the other sites of the genome [2]. ERCC6 (Excision repair cross-complementing group6, alternatively known as CSB) and ERCC8 (Excision repair cross-complementing group8, alternatively known as CSA) gene, are two indispensable core genes for the initiation of TCR pathway [3]. They were first described in the Cockayne syndrome (CS), a human autosomal recessive disease. ERCC6 and ERCC8 proteins, with direct interaction, jointly participate in DNA repair, transcriptional regulation, maintenance of the chromosome stability and chromatin remodeling [4–6].
Sequence variations and differential expression of the genes accounting for major components of human DNA repair system may be implicated in the carcinogenesis and development of cancer [7, 8]. In our previous study, we systematically analyzed the association of 43 SNPs of ten key NER pathway genes including ERCC6, ERCC8 with survival of gastric cancer (GC) patients [9], and found that ERCC6 SNP could predict GC prognosis. Moreover, ERCC6 rs1917799 and ERCC8 rs158572 polymorphisms were associated with increased GC risk separately [10, 11]. However, it is unclear whether there are interactions among ERCC6 and ERCC8 SNPs, by which it may enhance the risk warning for GC or its precancerous diseases because SNP-SNP interactions could be more valuable than a single SNP in cancer prediction [12–14]. In addition, it is also elusive whether the SNP-SNP combination is functional to influence ERCC6 and ERCC8 expression and whether the protein expression individually or jointly is associated with the development of GC, by which it may provide a genetic clue for phenotypic variations of gastric diseases and gastric carcinogenesis.
In this case-control study based on Chinese populations, we evaluated the two- and three-dimensional interactions between SNPs of ERCC6 and ERCC8 in GC and its precancerous diseases. We also performed protein level analysis to compare ERCC6 and ERCC8 expression (alone or in combination) in chronic superficial gastritis (CSG), chronic atrophic gastritis (CAG) and GC, and to correlate SNPs jointly with gene expression. We hoped to find potential combinations of biomarkers that could provide experimental evidence for the early diagnosis of GC.
RESULTS
Interaction analyses of ERCC6 and ERCC8 SNPs in different gastric diseases
Interactions between ERCC6 and ERCC8 SNPs in the risk of GC and CAG
We firstly focused on the pairwise interaction effects for SNPs of ERCC6 and ERCC8. In the two-way interaction analyses, significant interaction was observed. The results indicated that ERCC6 rs1917799 and ERCC8 rs158572 polymorphisms had interaction effects for CAG and GC (Pinteraction=0.013 and 0.021, separately). In addition, ERCC6 rs1917799 showed a significant interaction with ERCC8 rs158916 in relation to GC risk (Pinteraction=0.042) (Table 1). These results demonstrated that combination of the SNPs could have a synergistic effect on gastric carcinogenesis.
Table 1: Two-way interactions between ERCC6 and ERCC8 polymorphisms in the risk of GC and CAG
Gene |
Genotypes |
Number of Participants |
ERCC6 rs1917799 |
|||
|---|---|---|---|---|---|---|
TT |
GT/GG |
TT/GT |
GG |
|||
CAG vs CSG (n=700 vs.749) |
||||||
ERCC8 rs158572 |
AA |
No. of controls/cases |
229/228 |
401/382 |
524/510 |
106/100 |
OR (95% CI) |
1(Ref) |
0.92(0.72-1.19) |
1(Ref) |
0.96(0.70-1.33) |
||
GA/GG |
No. of controls/cases |
31/37 |
88/53 |
97/73 |
22/17 |
|
OR (95% CI) |
1.42(0.81-2.5) |
0.54(0.36-0.83) |
0.78(0.54-1.12) |
0.79(0.39-1.60) |
||
Pinteraction=0.013 |
Pinteraction=0.899 |
|||||
interaction index=0.41 |
interaction index=1.06 |
|||||
AA/GA |
No. of controls/cases |
259/263 |
486/431 |
618/578 |
127/116 |
|
OR (95% CI) |
1(Ref) |
0.82(0.65-1.04) |
1(Ref) |
1.71(0.36-8.21) |
||
GG |
No. of controls/cases |
1/2 |
3/4 |
3/5 |
1/1 |
|
OR (95% CI) |
2.29(0.18-29.2) |
1.07(0.20-5.67) |
0.97(0.72-1.31) |
1.00(0.05-20.4) |
||
Pinteraction=0.719 |
Pinteraction=0.772 |
|||||
interaction index=0.57 |
interaction index=0.60 |
|||||
ERCC8 rs158916 |
TT |
No. of controls/cases |
202/203 |
373/348 |
476/457 |
99/94 |
OR (95% CI) |
1(Ref) |
0.87(0.69-1.14) |
1(Ref) |
1.02(0.73-1.43) |
||
CT/CC |
No. of controls/cases |
58/62 |
116/87 |
145/126 |
29/23 |
|
OR (95% CI) |
1.00(0.64-1.56) |
1.07(0.20-5.67) |
0.87(0.65-1.17) |
0.68(0.37-1.25) |
||
Pinteraction=0.306 |
Pinteraction=0.475 |
|||||
interaction index=0.75 |
interaction index=0.77 |
|||||
TT/CT |
No. of controls/cases |
254/261 |
479/425 |
607/571 |
126/115 |
|
OR (95% CI) |
1(Ref) |
0.81(0.64-1.05) |
1(Ref) |
0.97(0.72-1.31) |
||
CC |
No. of controls/cases |
6/4 |
10/10 |
14/12 |
2/2 |
|
OR (95% CI) |
0.44(0.11-1.81) |
0.73(0.28-1.91) |
0.74(0.32-1.74) |
0.54(0.07-4.23) |
||
Pinteraction=0.406 |
Pinteraction=0.794 |
|||||
interaction index=2.06 |
interaction index=0.74 |
|||||
GC vs CSG (n= 467vs.749) |
||||||
ERCC8 rs158572 |
AA |
No. of controls/cases |
229/119 |
401/268 |
524/315 |
106/72 |
OR (95% CI) |
1(Ref) |
1.27(0.94-1.71) |
1(Ref) |
1.20(0.83-1.73) |
||
GA/GG |
No. of controls/cases |
31/28 |
88/52 |
97/65 |
22/15 |
|
OR (95% CI) |
2.20(1.19-4.1) |
1.16(0.74-1.83) |
1.28(0.87-1.88) |
1.11(0.53-2.36) |
||
Pinteraction=0.021 |
Pinteraction=0.482 |
|||||
interaction index=0.41 |
interaction index=0.73 |
|||||
AA/GA |
No. of controls/cases |
259/145 |
486/315 |
618/373 |
127/87 |
|
OR (95% CI) |
1(Ref) |
1.11(0.84-1.45) |
NA |
NA |
||
GG |
No. of controls/cases |
1/2 |
3/5 |
3/7 |
1/0 |
|
OR (95% CI) |
1.50(0.13-17.8) |
3.45(0.69-17.22) |
NA |
NA |
||
Pinteraction=0.625 |
NA |
|||||
interaction index=2.09 |
NA |
|||||
ERCC8 rs158916 |
TT |
No. of controls/cases |
202/104 |
373/261 |
476/299 |
99/66 |
OR (95% CI) |
1(Ref) |
1.30(0.95-1.78) |
1(Ref) |
1.12(0.76-1.64) |
||
CT/CC |
No. of controls/cases |
58/43 |
116/59 |
145/174 |
29/21 |
|
OR (95% CI) |
1.27(0.77-2.10) |
0.86(0.56-1.32) |
0.82(0.59-1.15) |
1.00(0.53-1.90) |
||
Pinteraction=0.042 |
Pinteraction=0.816 |
|||||
interaction index=0.52 |
interaction index=1.10 |
|||||
TT/CT |
No. of controls/cases |
254/142 |
479/313 |
607/371 |
126/84 |
|
OR (95% CI) |
1(Ref) |
1.12(0.85-1.47) |
1(Ref) |
1.11(0.79-1.56) |
||
CC |
No. of controls/cases |
6/5 |
10/7 |
14/16 |
2/3 |
|
OR (95% CI) |
1.10(0.29-4.11) |
1.11(0.37-3.30) |
0.86(0.34-2.17) |
2.76(0.35-21.8) |
||
Pinteraction=0.905 |
Pinteraction=0.363 |
|||||
interaction index=0.90 |
interaction index=2.89 |
|||||
CSG: chronic superficial gastritis; CAG: chronic atrophic gastritis; GC: gastric cancer.
NA not available.
Indeed, such an interaction effect between polymorphisms of two or more genes may be indicative of epistasis [15], which refers to an interaction between a pair of loci, in which the phenotypic effect of one locus depends on the genotype at the second locus. As such, the genetic effects of the polymorphisms on disease risks would have been missed had they not been tested jointly. We therefore examined the epistatic effects between pairs of interacting factors. For ERCC6 rs1917799 and ERCC8 rs158572, GT/GG genotypes of rs1917799 and GA/GG genotypes of rs158572 each conferred a reduced risk of CAG (OR = 0.35 and 0.59, respectively), but only if they were both present; rs158572 GA/GG genotypes were associated with an increased risk of GC, but only in the presence of TT genotypes of rs1917799. Regarding ERCC8 rs158916, the CT/CC genotype was associated with a reduced risk of CAG (OR = 0.66), but only in the presence of the ERCC6 rs1917799 GT/GG genotype (Table 2).
Table 2: Epistatic effect of pair-wise interacting factors on the risks of GC and CAG
Interacted pair-wise SNPs |
Comparison |
Subset |
CAG vs. CSG |
GC vs. CSG |
||
|---|---|---|---|---|---|---|
P |
OR(95%CI) |
P |
OR(95%CI) |
|||
ERCC6 rs1917799 interacted with ERCC8 rs158572 |
ERCC6 rs1917799 GT/GG vs. TT |
ERCC8 rs158572 AA |
0.526 |
0.92(0.72-1.09) |
0.111 |
1.27(0.95-1.72) |
ERCC8 rs158572 GA/GG |
0.003 |
0.35(0.18-0.69) |
0.068 |
0.52(0.26-1.05) |
||
ERCC8 rs158572 GA/GG vs. AA |
ERCC6 rs1917799 TT |
0.240 |
1.40(0.80-2.45) |
0.014 |
2.20(1.18-4.11) |
|
ERCC6 rs1917799 GT/GG |
0.011 |
0.59(0.39-0.88) |
0.694 |
0.92(0.60-1.40) |
||
ERCC6 rs1917799 interacted with ERCC8 rs158916 |
ERCC6 rs1917799 GT/GG vs. TT |
ERCC8 rs158916TT |
0.310 |
0.87(0.67-1.14) |
0.098 |
1.30(0.95-1.78) |
ERCC8 rs158916 CT/CC |
0.093 |
0.65(0.40-1.07) |
0.153 |
0.67(0.38-1.16) |
||
ERCC8 rs158916 CT/CC vs. TT |
ERCC6 rs1917799 TT |
0.973 |
1.01(0.65-1.57) |
0.334 |
1.28(0.77-2.13) |
|
ERCC6 rs1917799 GT/GG |
0.088 |
0.74(0.53-1.05) |
0.035 |
0.66(0.45-0.97) |
||
CSG: chronic superficial gastritis; CAG: chronic atrophic gastritis; GC: gastric cancer.
Moreover, interactions involving multiple polymorphisms were also explored among the three SNPs of ERCC6 and ERCC8 involved in pairwise interactions. However, ERCC6 rs1917799-ERCC8 rs158572-ERCC8 rs158916 had no obvious interaction in relation to CAG or GC risk (both Pinteraction>0.05) (Table 3). All these evidences suggest that the SNP-SNP interaction is mainly in an epistatic pairwise pattern.
Table 3: The three dimensions interactions of the ERCC6 rs1917799-ERCC8 rs158572-ERCC8 rs158916 with the risk of GC and CAG
SNP genotypes |
CAG vs. CSG |
GC vs. CSG |
||||
|---|---|---|---|---|---|---|
P |
OR(95%CI) |
P |
OR(95%CI) |
|||
ERCC6 rs1917799-ERCC8 rs158572-ERCC8 rs158916 |
||||||
TT |
AA |
TT |
1(ref) |
1(ref) |
||
TT |
AA |
CT/CC |
0.375 |
1.24(0.77-1.97) |
0.188 |
1.44(0.84-2.49) |
TT |
GA/GG |
TT |
0.031 |
1.97(1.07-3.63) |
0.006 |
2.75(1.33-5.55) |
TT |
GA/GG |
CT/CC |
0.088 |
0.23(0.04-1.25) |
0.362 |
1.74(0.53-5.72) |
GT/GG |
AA |
TT |
0.718 |
1.06(0.79-1.41) |
0.013 |
1.56(1.10-2.20) |
GT/GG |
AA |
CT/CC |
0.153 |
0.75(0.51-1.11) |
0.962 |
1.01(0.64-1.59) |
GT/GG |
GA/GG |
TT |
0.032 |
0.61(0.39-0.96) |
0.252 |
1.33(0.81-2.20) |
GT/GG |
GA/GG |
CT/CC |
0.074 |
0.27(0.07-1.14) |
0.818 |
0.86(0.24-3.10) |
Pinteraction=0.119 |
Pinteraction=0.429 |
|||||
interaction index=6.61 |
interaction index=2.23 |
|||||
CSG: chronic superficial gastritis; CAG: chronic atrophic gastritis; GC: gastric cancer.
Effect modification of H. pylori infection on the SNP–SNP interactions
Helicobacter pylori (H. pylori) infection is the most important environmental factor which may interact with hereditary factors to increase host susceptibility. We next investigated three-way interaction of ERCC6 and ERCC8 SNPs with H. pylori infection. There was no three-way combination had obvious interaction with CAG or GC risk (all Pinteraction>0.05) (Supplementary Table 2).
Individual and joint expression of ERCC6 and ERCC8 in different gastric diseases
Individual expression of ERCC6 and ERCC8 in different gastric tissues
We detected ERCC6 and ERCC8 in situ expression in CSG, precancerous CAG and GC by immunohistochemistry separately. ERCC6 and ERCC8 immunostaining were predominantly observed in the cytoplasm (Figure 1). Our results showed that expression of ERCC6 in CSG was higher than that in CAG and GC (P<0.001), but there was no obvious difference between CAG and GC (P=0.412). The expression of ERCC8 in CSG was higher than that in CAG and GC (P<0.001), but was lower in CAG than in GC (P=0.001) (Table 4) (Figure 2). GC cases were further classified into CAG group/non-CAG group by histopathological examination of adjacent non-cancerous tissues, and intestinal/diffuse group by Lauren’s Classification. In the stratified analysis, we found that positivity of ERCC6 and ERCC8 were higher in CSG than in different type of GC (all P<0.001). ERCC6 positivity was lower in diffuse GC than that in CAG (P=0.006), and ERCC8 positivity was lower in CAG compared with CAG-GC (P<0.001), intestinal-GC (P<0.001) and diffuse-GC (P=0.024). ERCC6 and ERCC8 expressions were significantly higher in intestinal GC than in diffuse GC (both P<0.05) (Table 4).
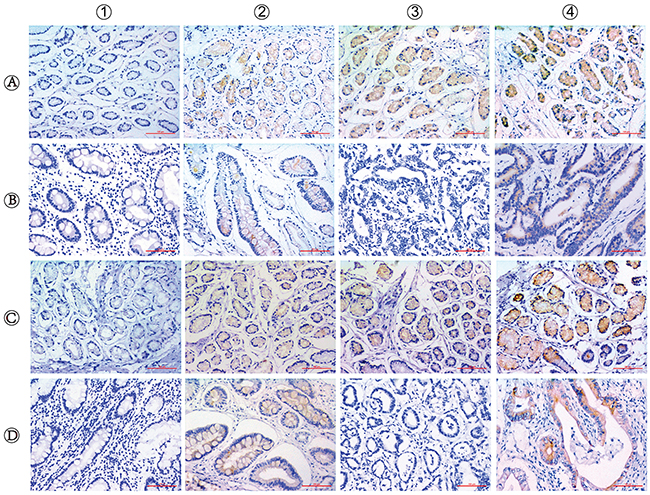
Figure 1: Immunohistochemical staining for ERCC6 and ERCC8 expression in CSG, CAG and GC. The staining of ERCC6 and ERCC8 are mainly located in the cytomembrane and cytoplasm. (A1-4) CSG with negative (−), weakly positive (+), moderately positive (++) and strongly positive (+++) ERCC6 expression; (B1-2) CAG with negative and positive ERCC6 expression; (B3-4) GC with negative and positive ERCC6 expression; (C1-4) CSG with negative (−), weakly positive (+), moderately positive (++) and strongly positive (+++) ERCC8 expression; (D1-2) CAG with negative and positive ERCC8 expression; (D3-4) GC with negative and positive ERCC8 expression. (Magnification, ×200; bar = 100μm).
Table 4: Individual and joint expression of ERCC6 and ERCC8 in different gastric diseases
Group |
case |
(–) |
(+) |
(+ +) |
(+ + +) |
Positive (%) |
Negative (%) |
P |
||
|---|---|---|---|---|---|---|---|---|---|---|
ERCC6 expression |
||||||||||
CSG |
109 |
12 |
52 |
31 |
14 |
97(89.0) |
12(11.0) |
ref. |
||
CAG |
109 |
59 |
40 |
10 |
0 |
50(45.9) |
59(54.1) |
<0.001 |
ref. |
|
GC |
109 |
65 |
40 |
4 |
0 |
44(40.4) |
65(59.6) |
<0.001 |
0.412 |
|
CAG-GC |
68 |
39 |
27 |
2 |
0 |
29(42.6) |
39(57.4) |
<0.001 |
0.756 |
ref. |
non CAG-GC |
41 |
26 |
13 |
2 |
0 |
15(36.6) |
26(63.4) |
<0.001 |
0.306 |
0.532 |
intestinal-GC |
24 |
10 |
14 |
0 |
0 |
14(58.3) |
10(41.7) |
<0.001 |
0.269 |
ref. |
diffuse-GC |
83 |
61 |
18 |
4 |
0 |
22(26.5) |
61(73.5) |
<0.001 |
0.006 |
0.004 |
ERCC8 expression |
||||||||||
CSG |
109 |
10 |
45 |
37 |
17 |
99(90.8) |
10(9.2) |
ref. |
||
CAG |
109 |
86 |
20 |
0 |
0 |
20(18.3) |
89(81.7) |
<0.001 |
ref. |
|
GC |
109 |
67 |
40 |
1 |
1 |
42(38.5) |
67(61.5) |
<0.001 |
0.001 |
|
CAG-GC |
68 |
38 |
29 |
1 |
0 |
30(44.1) |
38(55.9) |
<0.001 |
<0.001 |
ref. |
non CAG-GC |
41 |
29 |
11 |
0 |
1 |
12(29.3) |
29(70.7) |
<0.001 |
0.146 |
0.123 |
intestinal-GC |
24 |
9 |
15 |
0 |
0 |
15(62.5) |
9(37.5) |
<0.001 |
<0.001 |
ref. |
diffuse-GC |
83 |
56 |
25 |
1 |
1 |
27(32.5) |
56(67.5) |
<0.001 |
0.024 |
0.008 |
ERCC6-ERCC8 expression |
||||||||||
CSG |
109 |
91(83.5)* |
18(16.5)** |
ref. |
||||||
CAG |
109 |
14(12.8)* |
95(87.2)** |
<0.001 |
ref. |
|||||
GC |
109 |
21(19.3)* |
88(80.7)** |
<0.001 |
0.197 |
|||||
CAG-GC |
68 |
15(22.1)* |
53(77.9)** |
<0.001 |
0.107 |
ref. |
||||
non CAG-GC |
41 |
6(14.6)* |
35(85.4)** |
<0.001 |
0.774 |
0.341 |
||||
intestinal-GC |
24 |
8(33.3)* |
16(66.7)** |
<0.001 |
0.014 |
ref. |
||||
diffuse-GC |
83 |
10(12.0)* |
73(88.0)** |
<0.001 |
0.869 |
0.014 |
||||
CSG: chronic superficial gastritis; CAG: chronic atrophic gastritis; GC: gastric cancer.* double positive for ERCC6 and ERCC8 expression; ** single or double negative for ERCC6 and ERCC8 expression.
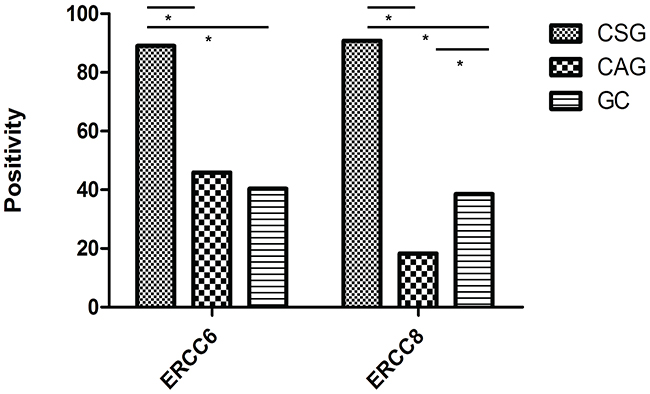
Figure 2: Positivity for ERCC6 and ERCC8 expression in CSG, CAG and GC. (*, P<0.05).
Joint expression of ERCC6 and ERCC8 in different gastric tissues
For the joint expression of ERCC6 and ERCC8, the results showed that ERCC6-ERCC8 double positive expression in CSG was significantly higher than in CAG and GC (both P<0.001), while no obvious difference was observed between CAG and GC (P=0.197). For subgroup analysis, ERCC6-ERCC8 expression was higher in CSG compared with different type of GC (all P<0.001), higher in intestinal-GC than in CAG (P=0.014) and diffuse-GC (P=0.014) (Table 4).
Effects of ERCC6 and ERCC8 SNPs on the protein expression
Effects of a single SNP on ERCC6 and ERCC8 expression
To examine whether the gene expression are correlated with ERCC6 and ERCC8 SNPs, 109 CSG subjects with both SNP genotype information and relatively high protein expression were used for analysis, which can rule out the interference of disease’s factor. In separate analysis of a single SNP, no correlation between the three SNPs and corresponding gene expression was observed (all P>0.05) (Supplementary Table 1).
Joint effects of SNP combination on ERCC6 and ERCC8 expression
As for joint analysis of two SNPs, in the CSG group, the ERCC6 rs1917799–ERCC8 rs158572 pair had significant effect on ERCC6 and ERCC6-ERCC8 expression (P=0.021 and 0.011, respectively). The lowest protein expression was observed in TT–GA/GG combined genotype, and GT/GG–GA/GG genotype had the highest protein expression (Table 5).
Table 5: Joint effects of SNP combination on ERCC6 and ERCC8 expression
SNP genotypes |
Total |
(–) |
(+) |
(+ +) |
(+ + +) |
ERCC6 positive n(%) |
(–) |
(+) |
(+ +) |
(+ + +) |
ERCC8 positive n(%) |
ERCC6-ERCC8 positive* n(%) |
||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
ERCC6rs1917799-ERCC8 rs158572 |
||||||||||||||
TT |
AA |
46 |
6 |
23 |
10 |
7 |
40(87.0) |
4 |
19 |
14 |
9 |
42(91.3) |
38(82.6) |
|
TT |
GA/GG |
7 |
3 |
1 |
3 |
0 |
4(57.1) |
2 |
3 |
2 |
0 |
5(74.1) |
3(42.9) |
|
GT/GG |
AA |
44 |
3 |
24 |
13 |
4 |
41(93.2) |
4 |
19 |
14 |
7 |
40(90.9) |
38(86.4) |
|
GT/GG |
GA/GG |
12 |
0 |
4 |
5 |
3 |
12(100.0) |
0 |
4 |
7 |
1 |
12(100.0) |
12(100.0) |
|
P=0.021 |
P=0.223 |
P=0.011 |
||||||||||||
ERCC6rs1917799-ERCC8 rs158916 |
||||||||||||||
TT |
TT |
41 |
5 |
22 |
9 |
5 |
36(87.8) |
4 |
20 |
10 |
7 |
37(92.0) |
33(80.5) |
|
TT |
CT/CC |
0 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
GT/GG |
TT |
53 |
7 |
22 |
17 |
7 |
46(86.8) |
5 |
18 |
25 |
5 |
48(90.6) |
44(83.0) |
|
GT/GG |
CT/CC |
15 |
0 |
8 |
5 |
2 |
15(100.0) |
1 |
7 |
2 |
5 |
14(93.3) |
14(93.3) |
|
P=0.337 |
P=0.935 |
P=0.514 |
||||||||||||
* double positive for ERCC6 and ERCC8 expression
DISCUSSION
As important members of the NER repair pathway, ERCC6 and ERCC8 are the only two necessary factors for TCR recognition. The novelty of the present study is that the results of genetic model dependent-analyses at protein levels provide a new prospective for a link between interaction of ERCC6-ERCC8 SNPs, interaction of ERCC6-ERCC8 gene expression and phenotypic variations of gastric diseases. To the best of our knowledge, this is the first study to investigate the relationship between ERCC6-ERCC8 SNP interactions, joint protein expression and GC/CAG risk. To some extent, it may help us to gain an in-depth understanding towards the association between ERCC6 and ERCC8 functional genotypes, coding protein and gastric carcinogenesis.
Evidence is accumulating that ERCC6 and ERCC8 polymorphisms have a close relationship with the susceptibility, progression and prognosis of various malignancies [16–20]. In our previous pilot study, three potential functional SNPs, ERCC6 rs1917799, ERCC8 rs158572 and rs158916 were detected and two of them were supposed to be associated with GC risk [10, 11]. In the current study, we further analyzed the interaction effect of the SNPs, and found that ERCC6 rs1917799 and ERCC8 rs158572 polymorphisms were associated with CAG and GC. ERCC6 rs1917799 also showed a significant interaction with ERCC8 rs158916 in relation to GC risk. It is worth noting that such an interaction effect between polymorphisms of two or more genes may be indicative of epistasis [15], which has been involved in susceptibility to various malignancies [21, 22]. In this study, the strongest epistatic interaction was the pairwise ERCC6 rs1917799-ERCC8 rs158572 combination. Both the rs1917799 GT/GG genotype and the rs158572 GA/GG genotype were associated with a reduced risk of CAG, but only in combination. Another two pairs of factors with epistatic effects were identified: rs158572 GA/GG genotypes were associated with an increased risk of GC, but only in the presence of TT genotypes of rs1917799; and rs158916 CT/CC genotype was associated with a reduced risk of CAG, but only in the presence of the rs1917799 GT/GG genotype. We also performed interaction analysis involving multiple polymorphisms of ERCC6 and ERCC8 and the effect modification of H. pylori infection. However, no significant interaction was observed for the three SNPs or SNP-SNP-H. pylori combination. These results demonstrated that the three polymorphisms contribute to gastric carcinogenesis mainly in an epistatic pairwise pattern. In a word, these evidences suggest that the combination of ERCC6 and ERCC8 SNPs could have a synergistic effect on gastric carcinogenesis.
Furthermore, individual and joint expression of ERCC6 and ERCC8 were also evaluated by immunohistochemistry in different gastric diseases, which have never been reported before. Our results showed the expressions of ERCC6, ERCC8 and ERCC6-ERCC8 combination have similarities, that higher positivity was observed in CSG compared with CAG and GC. Specifically, ERCC6 positivity decreased as CSG→CAG→GC development, whereas lowest positivity of ERCC8 was observed in CAG group. For ERCC6-ERCC8 joint expression, both CAG and GC group had very low positivity. Therefore, we supposed that ERCC6-ERCC8 joint expression was more suitable for discriminating normal and diseased mucosa, and ERCC8 was a better marker to distinguish CAG from other gastric diseases. Although ERCC6 and ERCC8 might play important roles in the initiation of GC and could serve as biomarkers for this disease, the underlying mechanisms remain unexplored. Physiologically, the expression of DNA repair genes reflects the cellular ability to meet repair demand once cells are stimulated by carcinogens. ERCC6 and ERCC8 defects specifically disrupt the coupling of transcription and repair, resulting in a loss of normal rapid repair [23]. Recently, it was reported that ERCC6 and ERCC8 deficient mice were more susceptible to both UV- and chemically-induced skin cancer [24, 25]. Javeri et al. [26] found that ERCC6 knockdown in human keratinocytes decreased DNA repair capacity for UV-induced cyclobutane dimers as well as 8-oxo-deoxyguanine, providing mechanistic evidence of a role for ERCC6 in skin carcinogenesis. In addition, different histological type of gastric cancer is thought to differ in pathogenesis, based on dissimilar variants and expression of various genes. A clear difference in genomic instability and DNA mismatch repair between intestinal-GC and diffuse-GC was found [27]. We observed that ERCC6 and ERCC8 positive rates were higher in intestinal-GC than diffuse-GC, from a side confirmed that intestinal subtype is more associated with genomic instability and DNA repair. In brief, we hypothesized that downregulation of ERCC6 and ERCC8 may result in lower DNA repair capacity, thus elevating cancer susceptibility by allowing unrepaired DNA damage to remain, ultimately leading to carcinogenesis.
Genetically determined factors may lead to inter-individual variation in protein expression and function. Population-based epidemiologic studies have shown that ERCC6 and ERCC8 polymorphisms have a significant impact on the risk of some human malignancies, including gastric cancer [10, 11, 16, 28–30]. In vitro evidence indicated that ERCC6 polymorphisms may alter protein function [16, 31], and reduced ERCC6 protein levels were associated with increased cancer risk [32, 33]. However, it is unclear whether the interactions of ERCC6-ERCC8 SNPs influence protein expression in gastric mucosa, which could help to reveal hidden heritability for gastric carcinogenesis. Therefore, we further investigated the functional relevance of ERCC6 and ERCC8 SNPs on gene expression in a fixed group (CSG) at protein level. We found that single SNP had no correlation with corresponding gene expression. As for joint analysis of two SNPs, the ERCC6 rs1917799–ERCC8 rs158572 pair had significant effect on ERCC6 and ERCC6-ERCC8 expression. The lowest protein expression was observed in TT–GA/GG combined genotype, and GT/GG–GA/GG genotype had the highest protein expression. Interestingly, the TT–GA/GG combination was found to have a correlation with increased GC risk, and GT/GG–GA/GG genotype can decrease CAG risk. ERCC6_rs1917799 polymorphism is located in 5’ regulatory region of ERCC6 gene and predicted having promoter activity. ERCC8_ rs158572 is located in region of intron in ERCC8 gene and predicted having enhancer activity (FASTSNP, http://fastsnp.ibms.sinica.edu.tw/pages/input_CandidateGeneSearch.jsp). It is reported that distal regulatory enhancer elements in the genome can interact with proximal promoter regions to regulate the target gene’s expression, and variants that change such interactions will cause the target gene to be dysregulated [34, 35]. We speculated that GT/GG-GA/GG genotype combination may activate such enhancer-promoter interaction to upregulate the gene expression, whereas there is no such effect for TT-GA/GG genotype. The regulation of gene expression by SNP combination may partially explain why individuals with specific genotypes had decreased risk of CAG or increased risk of GC.
In summary, ERCC6 rs1917799, ERCC8 rs158572 and rs158916 demonstrated pairwise epistatic interactions to affect CAG and GC risk. Expression of ERCC6, ERCC8 and ERCC6-ERCC8 combination have similarities that higher positivity was observed in CSG than CAG and GC. The ERCC6 rs1917799–ERCC8 rs158572 pair had significant influence on ERCC6 and ERCC6-ERCC8 expression. The regulation of SNP combination on gene expression may provide the genetic evidence for phenotypic variations of gastric diseases. Nevertheless, more thorough functional analysis will verify the interactions of genetic variation and gene expression between ERCC6 and ERCC8 in gastric carcinogenesis.
MATERIALS AND METHODS
Study population
The design of this study was approved by the Human Ethics Committee of the First Affiliated Hospital of China Medical University (Shenyang, China) before the outset of the research. Written informed consent was obtained from each study participant.
For the genetic association study, 1916 subjects were recruited from a population-based, combined serologic/endoscopic screening program for GC in the Zhuanghe area of Liaoning Province between 1997 and 2011. The screening population selection and recruitment process were reported previously [36]. All the enrolled subjects were diagnosed based on the gastroscopic and histopathological examinations by two independent pathologists. Patients with a history of other malignant tumors were excluded from our study. Histopathological findings were assessed according to the visual analog scale of the updated Sydney System for gastritis [37] and the World Health Organization (WHO) criteria for GC [38]. A 5-ml fasting venous blood sample was obtained for DNA isolation. The segregated blood clots were immediately frozen and stored until analysis. All the enrolled subjects were histologically classified into three groups: GC, CAG, and CSG (healthy control subjects with relative normal mucosa or only mild superficial gastritis). The demographic and geographic characteristics of study participants are shown in Table 6.
For the protein expression study, tissue samples were obtained from 109 GC patients from the anorectal department of the First Affiliated Hospital of China Medical University, who underwent surgical treatment, without neoadjuvant chemoradiotherapy or other therapy between 2012 and 2015. Additionally, 109 individuals with CSG and 109 patients with CAG were randomly matched from the above mentioned genetic association study. There was no statistical difference among the groups in terms of age and gender composition (Table 6).
Table 6: Baseline characteristics of the subjects for the genetic association and the protein expression study
Variable |
N |
Subjects |
P |
||
|---|---|---|---|---|---|
GC |
CAG |
CSG |
|||
The genetic association study |
|||||
Total n |
1916 |
n=467 |
n=700 |
n=749 |
|
Age |
<0.001 |
||||
<60 |
1292 |
263 |
487 |
542 |
|
≥60 |
624 |
204 |
213 |
207 |
|
Gender |
<0.001 |
||||
Male |
1109 |
321 |
401 |
387 |
|
Female |
807 |
146 |
299 |
362 |
|
H. pylori-IgG |
<0.001 |
||||
Positive |
830 |
167 |
420 |
243 |
|
Negative |
1086 |
582 |
280 |
224 |
|
The protein expression study |
|||||
Total n |
327 |
109 |
109 |
109 |
|
Age (years) |
0.364 |
||||
<60 |
186 |
59 |
59 |
68 |
|
≥60 |
141 |
50 |
50 |
41 |
|
Gender |
0.84 |
||||
Male |
212 |
73 |
70 |
69 |
|
Female |
115 |
36 |
39 |
40 |
|
CSG: chronic superficial gastritis; CAG: chronic atrophic gastritis; GC: gastric cancer.
SNP genotyping
Whole blood from individuals was collected, and blood clots were allowed to form by incubating clot-activating tubes at room temperature for 1 h. Each clot was transferred to a 2-ml centrifuge tube and stored at −80 °C until DNA extraction. Genomic DNA of the blood samples from included subjects was extracted from venous blood as previously described using routine phenol–chloroform method [39]. For genotyping, the DNA concentration was adjusted to 50 ng/μl. SNP genotyping was performed using Matrix assisted laser desorption/ionization time of flight mass spectrometry, as previously described [40]. Briefly, all samples were placed randomly into 384-well plates and blinded for disease status. The genotyping assay was performed by CapitalBio (Beijing, China) using the Sequenom MassARRAY platform (Sequenom, San Diego, CA, USA). To evaluate the quality of the genotyping, 5% samples were repeatedly genotyped and the consistency rate was higher than 99%.
In situ protein expression by immunohistochemistry
Paraffin-embedded tissues were sectioned into 4-μm-thick sections, and mounted onto positive-charged glass slides. Briefly, slides were deparaffinized in xylene, rehydrated in a graded alcohol series, washed in tap water and boiled at 95°C for 30 min in citric acid buffer (pH 6.0) for antigen retrieval. Endogenous peroxidase activity was blocked by washing with 3% hydrogen peroxide solution for 10 min, and the sections were then washed with phosphate-buffered saline (PBS, pH 7.4). Tissue collagen was blocked to avoid nonspecific binding by adding 10% normal goat serum at 37°C for 30 min. The sections were incubated with primary antibodies against ERCC6 (TA313375, 1:300, Origene, Rockville, MD, USA) and ERCC8 (AV31542, 1:500, Sigma, Saint Louis, MO, USA) at 37°C for 1 h. After rinsing three times with PBS, the sections were incubated with biotinylated secondary goat anti-rabbit antibody (Maixin, Fuzhou, Fujian, China), and then with streptavidin-biotin peroxidase for 10 min each. After secondary antibody staining, diaminobenzidine (DAB) was used as the chromogen for 1 min. Finally, slides were rinsed with water, counterstained with hematoxylin blued in water, dehydrated, cleared in xylene, and mounted.
Standards for immunohistochemistry evaluation
ERCC6 and ERCC8 staining were mainly located in the cell membrane and cytoplasm. Staining results were evaluated independently by two pathologists blinded to the clinicopathological characteristics of patients. ERCC6 and ERCC8 expression were scored using a semi-quantitative method that took into account both the staining intensity and the percentage of cells at that intensity [41]. For each sample, staining intensity was scored according to intensity (0, no staining; 1, mild staining; 2, moderate staining; 3, strong staining) and range (0, ≤5%; 1, 5%–25%; 2, 25%–50%; 3, 50%–75%; 4, ≥75%) of the staining. Finally, the staining intensity and the staining range were multiplied to generate an immunoreactivity score (IS) for each sample, which was divided as: no staining, 0; mild staining, 1–4; moderate staining, 5–8; severe staining, 9–12. A score of 0 meant negative expression, while all others indicated ranges of positive expression.
Detection of the serum H. pylori IgG titer
We tested the serum H. pylori immunoglobulin G (IgG) antibody titer by enzyme-linked immunosorbent assay (Helicobacter pylori IgG kit; Biohit, Helsinki, Finland) according to a previously described method [42]. Patients with a serum titer > 34 IU were diagnosed as H. pylori positive.
Statistical analysis
This study defined the heterozygote plus rare homozygote compared with the wild-type genotype as the dominant model, the mutant genotype compared with the wild-type genotype plus the heterozygote as the recessive model [14]. The distribution of demographic characteristics and the genotypes in case and control groups was tested using chi-square test. Multivariate logistic regression analysis adjusted by age and sex was used to assess the OR and 95% confidence interval for the GC and CAG risks. The log likelihood ratio test was used for the interaction analysis between SNPs. Chi-square test was used to analyze differential expression of ERCC6 and ERCC8 between different gastric diseases and the correlation between SNP and gene expression. All statistical tests were performed using SPSS 18.0 (SPSS Inc., Chicago, IL, USA). P values less than 0.05 were considered statistically significant.
Abbreviations
ERCC6: Excision repair cross-complementing group 6; ERCC8: Excision repair cross-complementing group 8; CSG: Chronic superficial gastritis; CAG: Chronic atrophic gastriitis; GC: Gastric Cancer; SNP: Single nucleotide polymorphism; NER: Nucleotide excision repair; TCR: Transcription-coupled nucleotide excision repair; CS: Cockayne syndrome.
Author contributions
Conceived and designed the experiments: YY. Performed the experiments: JJ-J YZ-L NN-D. Collected the blood/tissue samples: YH-G LP-S QX. Analyzed the data: JJ-J JW-L YZ-L. Contributed reagents/materials/analysis tools: YY. Wrote and revised the paper: JJ-J YY.
CONFLICTS OF INTEREST
All authors do not have a commercial or other association that might pose a conflicts of interest.
FUNDING
This work is supported by grants from the National Science-technology Support Plan Project (ref no. 2015BAI13B07)
REFERENCES
1. Marteijn JA, Lans H, Vermeulen W, Hoeijmakers JH. Understanding nucleotide excision repair and its roles in cancer and ageing. Nat Rev Mol Cell Biol. 2014; 15: 465-81. doi: 10.1038/nrm3822.
2. Fousteri M, Mullenders LH. Transcription-coupled nucleotide excision repair in mammalian cells: molecular mechanisms and biological effects. Cell Res. 2008; 18: 73-84. doi: 10.1038/cr.2008.6.
3. Fousteri M, Vermeulen W, van Zeeland AA, Mullenders LH. Cockayne syndrome A and B proteins differentially regulate recruitment of chromatin remodeling and repair factors to stalled RNA polymerase II in vivo. Mol Cell. 2006; 23: 471-82. doi: 10.1016/j.molcel.2006.06.029.
4. Groisman R, Kuraoka I, Chevallier O, Gaye N, Magnaldo T, Tanaka K, Kisselev AF, Harel-Bellan A, Nakatani Y. CSA-dependent degradation of CSB by the ubiquitin-proteasome pathway establishes a link between complementation factors of the Cockayne syndrome. Genes Dev. 2006; 20: 1429-34. doi: 10.1101/gad.378206.
5. Henning KA, Li L, Iyer N, McDaniel LD, Reagan MS, Legerski R, Schultz RA, Stefanini M, Lehmann AR, Mayne LV, Friedberg EC. The Cockayne syndrome group A gene encodes a WD repeat protein that interacts with CSB protein and a subunit of RNA polymerase II TFIIH. Cell. 1995; 82: 555-64.
6. Mullenders L. DNA damage mediated transcription arrest: Step back to go forward. DNA Repair (Amst). 2015; 36: 28-35. doi: 10.1016/j.dnarep.2015.09.005.
7. Liu J, He C, Xing C, Yuan Y. Nucleotide excision repair related gene polymorphisms and genetic susceptibility, chemotherapeutic sensitivity and prognosis of gastric cancer. Mutat Res. 2014; 765: 11-21. doi: 10.1016/j.mrfmmm.2014.04.008.
8. Friedberg EC. How nucleotide excision repair protects against cancer. Nat Rev Cancer. 2001; 1: 22-33. doi: 10.1038/35094000.
9. Liu J, Deng N, Xu Q, Sun L, Tu H, Wang Z, Xing C, Yuan Y. Polymorphisms of multiple genes involved in NER pathway predict prognosis of gastric cancer. Oncotarget. 2016; 7: 48130-42. doi: 10.18632/oncotarget.10173.
10. Jing JJ, Sun LP, Xu Q, Yuan Y. Effect of ERCC8 tagSNPs and their association with H. pylori infection, smoking, and alcohol consumption on gastric cancer and atrophic gastritis risk. Tumour Biol. 2015; 36: 9525-35. doi: 10.1007/s13277-015-3703-9.
11. Liu JW, He CY, Sun LP, Xu Q, Xing CZ, Yuan Y. The DNA repair gene ERCC6 rs1917799 polymorphism is associated with gastric cancer risk in Chinese. Asian Pac J Cancer Prev. 2013; 14: 6103-8.
12. Moore JH, Williams SM. New strategies for identifying gene-gene interactions in hypertension. Ann Med. 2002; 34: 88-95.
13. Cordell HJ. Detecting gene-gene interactions that underlie human diseases. Nat Rev Genet. 2009; 10: 392-404. doi: 10.1038/nrg2579.
14. He C, Xu Q, Tu H, Sun L, Gong Y, Liu J, Yuan Y. Polymorphic rs9471643 and rs6458238 upregulate PGC transcription and protein expression in overdominant or dominant models. Mol Carcinog. 2016; 55: 586-99. doi: 10.1002/mc.22305.
15. Carlborg O, Haley CS. Epistasis: too often neglected in complex trait studies? Nat Rev Genet. 2004; 5: 618-25. doi: 10.1038/nrg1407.
16. Lin Z, Zhang X, Tuo J, Guo Y, Green B, Chan CC, Tan W, Huang Y, Ling W, Kadlubar FF, Lin D, Ning B. A variant of the Cockayne syndrome B gene ERCC6 confers risk of lung cancer. Hum Mutat. 2008; 29: 113-22. doi: 10.1002/humu.20610.
17. Ma H, Hu Z, Wang H, Jin G, Wang Y, Sun W, Chen D, Tian T, Jin L, Wei Q, Lu D, Huang W, Shen H. ERCC6/CSB gene polymorphisms and lung cancer risk. Cancer Lett. 2009; 273: 172-6. doi: 10.1016/j.canlet.2008.08.002.
18. Savina NV, Nikitchenko NV, Kuzhir TD, Rolevich AI, Krasny SA, Goncharova RI. The Cellular Response to Oxidatively Induced DNA Damage and Polymorphism of Some DNA Repair Genes Associated with Clinicopathological Features of Bladder Cancer. Oxid Med Cell Longev. 2016; 2016: 5710403. doi: 10.1155/2016/5710403.
19. Gu J, Zhao H, Dinney CP, Zhu Y, Leibovici D, Bermejo CE, Grossman HB, Wu X. Nucleotide excision repair gene polymorphisms and recurrence after treatment for superficial bladder cancer. Clin Cancer Res. 2005; 11: 1408-15. doi: 10.1158/1078-0432.ccr-04-1101.
20. Fleming ND, Agadjanian H, Nassanian H, Miller CW, Orsulic S, Karlan BY, Walsh CS. Xeroderma pigmentosum complementation group C single-nucleotide polymorphisms in the nucleotide excision repair pathway correlate with prolonged progression-free survival in advanced ovarian cancer. Cancer. 2012; 118: 689-97. doi: 10.1002/cncr.26329.
21. He C, Tu H, Sun L, Xu Q, Gong Y, Jing J, Dong N, Yuan Y. SNP interactions of Helicobacter pylori-related host genes PGC, PTPN11, IL1B, and TLR4 in susceptibility to gastric carcinogenesis. Oncotarget. 2015; 6: 19017-26. doi: 10.18632/oncotarget.4231.
22. Xu Q, Wu YF, Li Y, He CY, Sun LP, Liu JW, Yuan Y. SNP-SNP interactions of three new pri-miRNAs with the target gene PGC and multidimensional analysis of H. pylori in the gastric cancer/atrophic gastritis risk in a Chinese population. Oncotarget. 2016; 7: 23700-14. doi: 10.18632/oncotarget.8057.
23. Venema J, Mullenders LH, Natarajan AT, van Zeeland AA, Mayne LV. The genetic defect in Cockayne syndrome is associated with a defect in repair of UV-induced DNA damage in transcriptionally active DNA. Proc Natl Acad Sci U S A. 1990; 87: 4707-11.
24. van der Horst GT, van Steeg H, Berg RJ, van Gool AJ, de Wit J, Weeda G, Morreau H, Beems RB, van Kreijl CF, de Gruijl FR, Bootsma D, Hoeijmakers JH. Defective transcription-coupled repair in Cockayne syndrome B mice is associated with skin cancer predisposition. Cell. 1997; 89: 425-35.
25. van der Horst GT, Meira L, Gorgels TG, de Wit J, Velasco-Miguel S, Richardson JA, Kamp Y, Vreeswijk MP, Smit B, Bootsma D, Hoeijmakers JH, Friedberg EC. UVB radiation-induced cancer predisposition in Cockayne syndrome group A (Csa) mutant mice. DNA Repair (Amst). 2002; 1: 143-57.
26. Javeri A, Lyons JG, Huang XX, Halliday GM. Downregulation of Cockayne syndrome B protein reduces human 8-oxoguanine DNA glycosylase-1 expression and repair of UV radiation-induced 8-oxo-7,8-dihydro-2’-deoxyguanine. Cancer Sci. 2011; 102: 1651-8. doi: 10.1111/j.1349-7006.2011.02005.x.
27. Vauhkonen M, Vauhkonen H, Sajantila A, Sipponen P. Differences in genomic instability between intestinal- and diffuse-type gastric cancer. Gastric Cancer. 2005; 8: 238-44. doi: 10.1007/s10120-005-0346-3.
28. Chen M, Kamat AM, Huang M, Grossman HB, Dinney CP, Lerner SP, Wu X, Gu J. High-order interactions among genetic polymorphisms in nucleotide excision repair pathway genes and smoking in modulating bladder cancer risk. Carcinogenesis. 2007; 28: 2160-5. doi: 10.1093/carcin/bgm167.
29. Wheless L, Kistner-Griffin E, Jorgensen TJ, Ruczinski I, Berthier-Schaad Y, Kessing B, Hoffman-Bolton J, Francis L, Shugart YY, Strickland PT, Kao WH, Alani RM, Smith MW, et al. A community-based study of nucleotide excision repair polymorphisms in relation to the risk of non-melanoma skin cancer. J Invest Dermatol. 2012; 132: 1354-62. doi: 10.1038/jid.2012.4.
30. Berndt SI, Platz EA, Fallin MD, Thuita LW, Hoffman SC, Helzlsouer KJ. Genetic variation in the nucleotide excision repair pathway and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev. 2006; 15: 2263-9. doi: 10.1158/1055-9965.epi-06-0449.
31. Mallery DL, Tanganelli B, Colella S, Steingrimsdottir H, van Gool AJ, Troelstra C, Stefanini M, Lehmann AR. Molecular analysis of mutations in the CSB (ERCC6) gene in patients with Cockayne syndrome. Am J Hum Genet. 1998; 62: 77-85. doi: 10.1086/301686.
32. Cheng L, Spitz MR, Hong WK, Wei Q. Reduced expression levels of nucleotide excision repair genes in lung cancer: a case-control analysis. Carcinogenesis. 2000; 21: 1527-30.
33. Cheng L, Sturgis EM, Eicher SA, Spitz MR, Wei Q. Expression of nucleotide excision repair genes and the risk for squamous cell carcinoma of the head and neck. Cancer. 2002; 94: 393-7. doi: 10.1002/cncr.10231.
34. Guo Y, Xu Q, Canzio D, Shou J, Li J, Gorkin DU, Jung I, Wu H, Zhai Y, Tang Y, Lu Y, Wu Y, Jia Z, et al. CRISPR Inversion of CTCF Sites Alters Genome Topology and Enhancer/Promoter Function. Cell. 2015; 162: 900-10. doi: 10.1016/j.cell.2015.07.038.
35. Zhang Y, Wong CH, Birnbaum RY, Li G, Favaro R, Ngan CY, Lim J, Tai E, Poh HM, Wong E, Mulawadi FH, Sung WK, Nicolis S, et al. Chromatin connectivity maps reveal dynamic promoter-enhancer long-range associations. Nature. 2013; 504: 306-10. doi: 10.1038/nature12716.
36. Tu H, Sun L, Dong X, Gong Y, Xu Q, Jing J, Yuan Y. Serum anti-Helicobacter pylori immunoglobulin G titer correlates with grade of histological gastritis, mucosal bacterial density, and levels of serum biomarkers. Scand J Gastroenterol. 2014; 49: 259-66. doi: 10.3109/00365521.2013.869352.
37. Stolte M, Meining A. The updated Sydney system: classification and grading of gastritis as the basis of diagnosis and treatment. Can J Gastroenterol. 2001; 15: 591-8.
38. Hamilton S, Aaltonen L. World Health Organization classification of tumours. Pathology and genetics of turnouts of the digestive system. Lyon: IARC Press; 2000.
39. Xu Q, Yuan Y, Sun LP, Gong YH, Xu Y, Yu XW, Dong NN, Lin GD, Smith PN, Li RW. Risk of gastric cancer is associated with the MUC1 568 A/G polymorphism. Int J Oncol. 2009; 35: 1313-20.
40. He CY, Sun LP, Xu Q, Liu JW, Jiang JY, Dong NN, Yuan Y. PGC TagSNP and its interaction with H. pylori and relation with gene expression in susceptibility to gastric carcinogenesis. PLoS One. 2014; 9: e115955. doi: 10.1371/journal.pone.0115955.
41. Detre S, Saclani Jotti G, Dowsett M. A “quickscore” method for immunohistochemical semiquantitation: validation for oestrogen receptor in breast carcinomas. J Clin Pathol. 1995; 48: 876-8.
42. Gong YH, Sun LP, Jin SG, Yuan Y. Comparative study of serology and histology based detection of Helicobacter pylori infections: a large population-based study of 7,241 subjects from China. Eur J Clin Microbiol Infect Dis. 2010; 29: 907-11. doi: 10.1007/s10096-010-0944-9.

