Introduction
Worldwide, colorectal cancer (CRC) is one of the most frequent malignant tumors and the fourth leading cause of cancer-related deaths in human cancers. Based on global cancer statistics, approximately 1,360,600 new cases of CRC were clinically diagnosed, and CRC caused an estimated 693,900 deaths, in 2012 [1]. Although the therapeutic opportunities have markedly improved in recent years for CRC patients, patients with liver, lung or lymph node metastases (advanced stage) still have a poor prognosis, with a 5-year survival rate of ~10% [2, 3].
Increasing evidence suggests that a small subpopulation of cancer cells, termed cancer stem cells (CSCs), are associated with self-renewal and uncontrolled proliferation, differentiation, and tumorigenicity [4, 5]. Some studies have shown that CSCs are responsible for the carcinogenesis, progression, metastasis and prognosis of human cancers [6-8]. Some CSC expression markers have become prognostic biomarkers in CRC (i.e., CD133, ALDH1 and Lgr5) [9-11]. CD166 is an important CSC marker; it is located on human chromosome 3q13.11, and it is also called activated leukocyte cell adhesion molecule (ALCAM) [12]. CD166 is involved in many biological functions, including CD166-CD6/CD166 cell-cell interactions, T-cell stimulation and proliferation, angiogenesis, and hematopoiesis [13-15]. CD166 expression is closely correlated with various types of human cancers, including CRC [16-18].
There are some inconsistent and controversial conclusions about CD166 expression in CRC. For example, a significant correlation between CD166 expression and overall survival (OS) of patients was reported based on multivariate analysis by Weichert et al [19]. However, Ribeiro et al reported that CD166 expression was not related to OS of patients with CRC using multivariate analysis [20]. Thus, we evaluated the prognostic and predictive role of CD166 expression in CRC patients with multivariate analysis. Moreover, we also evaluated the associations of CD166 expression between CRC and colonic adenomas and between CRC and normal colonic mucosa. Finally, we analyzed the correlation of CD166 expression with clinicopathological characteristics in this study.
Results
Characteristics of relevant studies
Initially, 391 publications were retrieved by the mentioned search strategy. According to the inclusion criteria, 15 eligible studies [18-32] were identified in the final meta-analysis (Figure 1), including 2,810 patients with CRC, 187 patients with colonic adenoma, and 335 controls with normal colonic mucosa. Of these studies, five studies analyzed the relationship of CD166 expression between CRC and normal colonic mucosa. Four studies analyzed the correlation of CD166 expression between CRC and colonic adenomas. Ten studies assessed the relationship between CD166 expression and the clinicopathological features in CRC. Five studies with the original multivariate analysis data analyzed the prognostic and predictive roles of CD166 expression. The general characteristics of the included studies are summarized in Table 1 and Supplementary Table 1.
Table 1: Basic characteristics of 15 eligible publications in this study.
First author |
Country |
Ethnicity |
Age |
Stage |
Staining |
Cut off scores (IHC) |
Cancer |
Adenoma |
Normal tissue |
Clinicopathological features |
MA-HR |
MA-OR |
||
N (E+ %) |
N (E+ %) |
N (E+ %) |
OS |
DFS |
PFS |
Predictor (survival) |
||||||||
Weichert 2004 [19] |
Germany |
Caucasians |
65 |
1-4 |
M |
0% |
111 (30.6) |
NA |
NA |
NA |
Yes |
NA |
NA |
NA |
Zhuang 2007 [32] |
China |
Asians |
NA |
1-4 |
M/C |
10% |
66 (68.2) |
NA |
66 (0.0) |
Yes |
NA |
NA |
NA |
NA |
Horst 2009 [27] |
Germany |
Caucasians |
67 |
NA |
M |
0% |
110 (63.6) |
NA |
NA |
Yes |
NA |
NA |
NA |
NA |
Lugli 2010 [26] |
Switzerland |
Caucasians |
NA |
1-4 |
M |
65% |
1274 (60.8) |
NA |
NA |
Yes |
NA |
NA |
NA |
NA |
Chen 2011 [28] |
China |
Asians |
NA |
1-4 |
M/C |
5% |
69 (49.3) |
40 (25.0) |
69 (0.0) |
Yes |
NA |
NA |
NA |
NA |
Piscuoglio 2012 [25] |
Switzerland |
Caucasians |
63 |
NA |
M |
0 |
151 (42.4) |
87 (32.2) |
120 (28.3) |
NA |
NA |
NA |
NA |
NA |
Tachezy 2012 [24] |
Germany |
Caucasians |
65 |
NA |
M |
Spots |
300 (76.3) |
NA |
NA |
Yes |
Yes |
NA |
NA |
NA |
Hansen 2013 [23] |
USA |
Caucasians |
NA |
2 |
M |
NA |
105 (NA) |
NA |
NA |
NA |
NA |
NA |
NA |
Yes |
Shafaei 2013 [22] |
Iran |
Caucasians |
59 |
NA |
M |
50% |
121 (34.7) |
NA |
NA |
Yes |
NA |
NA |
NA |
NA |
Zhang 2013 [29] |
China |
Asians |
65 |
1-4 |
M |
10% |
57 (42.1) |
NA |
NA |
Yes |
NA |
NA |
NA |
NA |
Zhou 2013 [30] |
China |
Asians |
55 |
1-4 |
M/C |
10% |
120 (55.0) |
20 (20.0) |
40 (0.0) |
Yes |
NA |
NA |
NA |
NA |
Sim 2014 [18] |
Korea |
Caucasians |
62 |
3-4 |
M |
NA |
112 (NA) |
NA |
NA |
NA |
NA |
Yes |
NA |
NA |
Zhu 2015 [31] |
China |
Asians |
63 |
1-4 |
M/C |
0% |
102 (60.8) |
40 (15.0) |
40 (0.0) |
Yes |
NA |
NA |
NA |
NA |
Manhas 2016 [21] |
India |
Caucasians |
NA |
1-4 |
M/C |
0% |
54 (70.4) |
NA |
NA |
Yes |
NA |
NA |
NA |
NA |
Ribeiro 2016 [20] |
Brazil |
Caucasians |
65 |
2-4 |
M/C |
NA |
58 (43.1) |
NA |
NA |
NA |
Yes |
NA |
Yes |
NA |
IHC: immunohistochemistry; NA: not applicable; M: membrane; C: cytoplasm; N: number of total samples; E+: expression positive status; MA: multivariate analysis; OS: overall survival; DFS: disease-free survival; PFS: progression-free survival; OR: odds ratio; HR: hazard ratio.
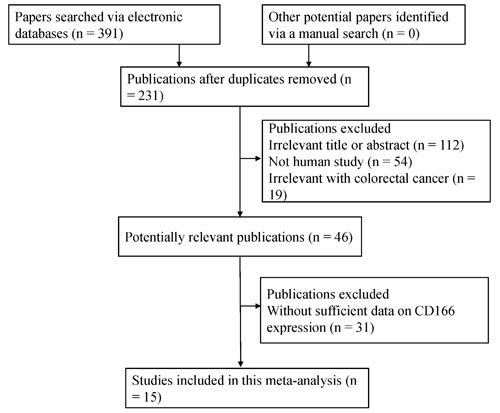
Figure 1: Flow diagram of the selection procedure for this study.
CD166 expression in CRC vs. control groups
The pooled data included five studies with 508 CRC patients and 335 patients with normal colonic mucosa and four studies with 442 CRC patients and 187 adenoma patients (Figure 2). Our results obtained using the random-effects model showed that CD166 expression in CRC had a significantly higher OR than its expression in colonic adenomas or normal colonic mucosa (OR = 3.48, 95% CI = 1.55-7.79, P = 0.002 and OR = 55.13, 95% CI = 2.04-1486.86, P = 0.017, respectively).
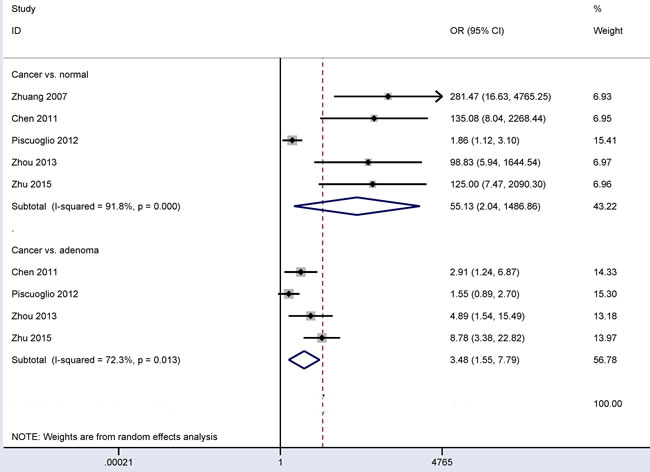
Figure 2: Forest plot of the relationship of CD166 expression between CRC and control groups, cancer vs. colonic adenoma: OR = 3.48, 95% CI = 1.55-7.79, P = 0.002; cancer vs. normal colonic mucosa: OR = 55.13, 95% CI = 2.04-1486.86, P = 0.017.
Associations between CD166 expression and gender and CD166 expression and vascular invasion in CRC
No evidence of heterogeneity was measured in relation to gender or vascular invasion (all I2 = 0.0%) (Figure 3), so the fixed-effects model was applied. The overall OR from three studies, including 411 patients with vascular invasion and 1,075 patients without vascular invasion, showed that CD166 expression was negatively correlated with vascular invasion (OR = 0.75, 95% CI = 0.60-0.95, P = 0.017).
The overall OR from nine studies, including 582 male and 417 female patients with CRC, demonstrated that CD166 expression was not correlated with gender (OR = 0.89, 95% CI = 0.68-1.17, P = 0.414).
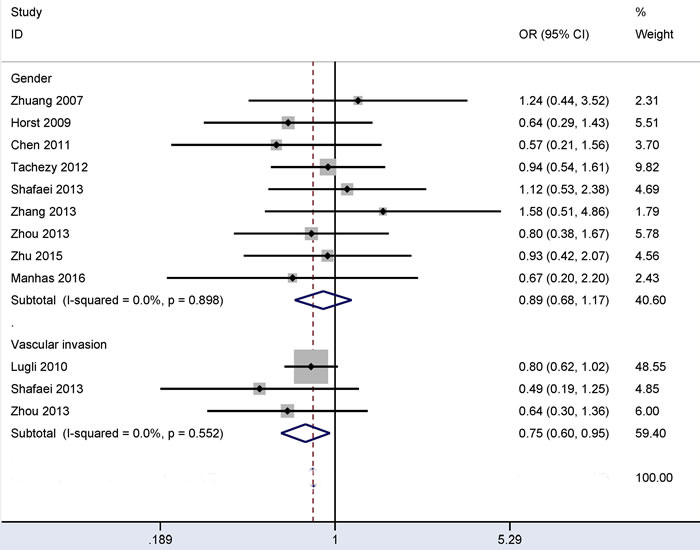
Figure 3: Forest plot of the relationship of CD166 expression with vascular invasion and gender status in colorectal cancer, vascular invasion (yes vs. no): OR = 0.75, 95% CI = 0.60-0.95, P = 0.017; gender (male vs. female): OR = 0.89, 95% CI = 0.68-1.17, P = 0.414.
Associations between CD166 expression and distant metastasis and between CD166 expression and lymph node status in CRC
Substantial heterogeneity was detected in relation to distant metastasis and lymph node status (all I2 > 50%), so the random-effects model was used. The results from four studies showed that CD166 expression was not linked to distant metastasis (OR = 1.60, 95% CI = 0.83-3.10, P = 0.16) (Figure 4). The results from nine studies showed that CD166 expression was not linked to lymph node status (OR = 1.35, 95% CI = 0.87-2.11, P = 0.183) (Figure 4). These data included the comparison of 221 CRC patients with metastasis and 664 CRC patients without metastasis and the comparison of 1,042 CRC patients with lymph node positive status and 1,100 CRC patients with lymph node negative status.
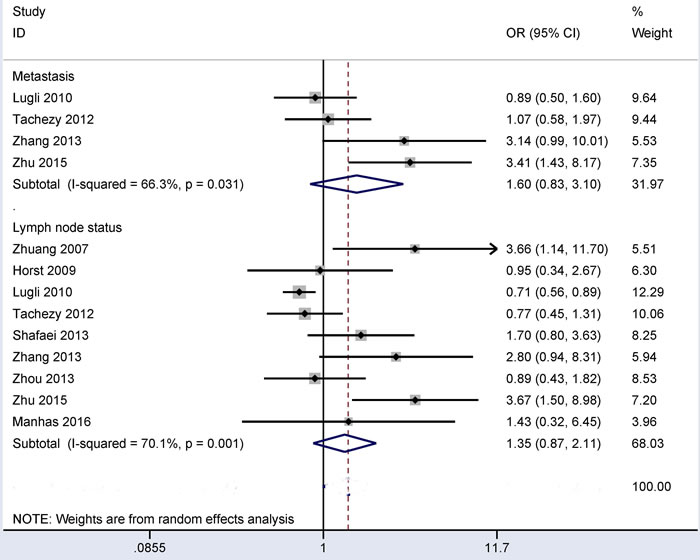
Figure 4: Forest plot of the correlation of CD166 expression with lymph node status and distant metastasis in colorectal cancer, distant metastasis (yes vs. no): OR = 1.60, 95% CI = 0.83-3.10, P = 0.16; lymph node status (yes vs. no): OR = 1.35, 95% CI = 0.87-2.11, P = 0.183.
Associations between CD166 expression and tumor location and between CD166 expression and T classification in CRC
In the comparison of 931 patients with left-sided CRC and 538 patients with right-sided CRC (Figure 5), the results from four studies demonstrated that no significant correlation was observed between CD166 expression and tumor location under the random-effects model (OR = 0.59, 95% CI = 0.28-1.21, P = 0.15). In the comparison of 1,577 patients with T3-4 classification and 427 patients with T1-2 classification (Figure 5), the results from seven studies demonstrated that no significant correlation was observed between CD166 expression and T classification under the random-effects model (OR = 1.55, 95% CI = 0.82-2.94, P = 0.181).
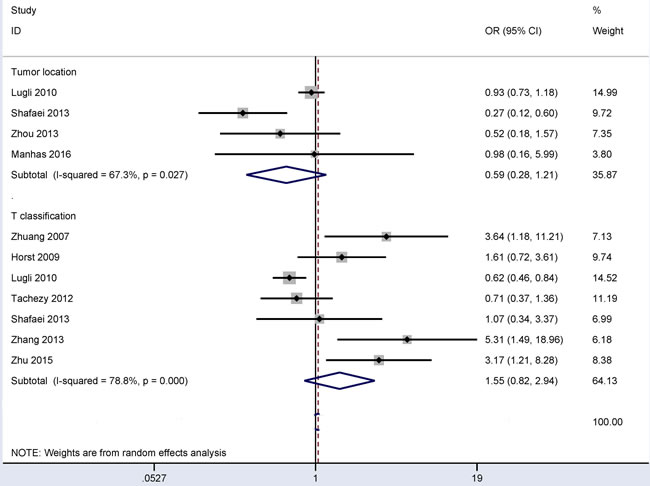
Figure 5: Forest plot of the association of CD166 expression with tumor location and T classification in colorectal cancer, tumor location (left vs. right): OR = 0.59, 95% CI = 0.28-1.21, P = 0.15; T classification (pT3-4 vs. pT1-2): OR = 1.55, 95% CI = 0.82-2.94, P = 0.181.
Associations between CD166 expression and tumor differentiation and between CD166 expression and clinical stage in CRC
The random-effects model was applied when there was obvious evidence of heterogeneity in relation to tumor differentiation or clinical stage (all I2 > 50%). When 383 poorly differentiated CRC patients were compared to 1,831 moderately or well differentiated CRC patients, the result from ten studies demonstrated that no significant relationship was found between CD166 expression and tumor differentiation (OR = 1.45, 95% CI = 0.80-2.63, P = 0.217) (Figure 6).
The result from six studies with 457 CRC patients showed that no obvious association was found between CD166 expression and clinical stage (OR = 1.73, 95% CI = 0.91-3.27, P = 0.092) (Figure 6).
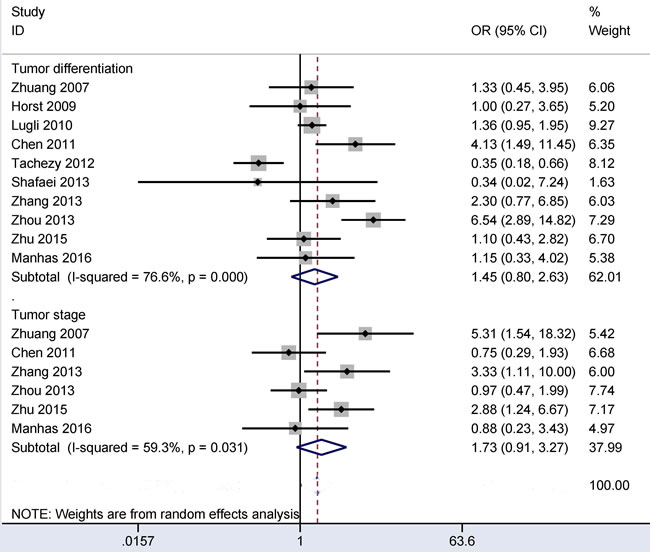
Figure 6: Forest plot of the relationship between CD166 expression and tumor differentiation and clinical stage in colorectal cancer, tumor differentiation (poor vs. moderate/well): OR = 1.45, 95% CI = 0.80-2.63, P = 0.217; clinical stage (stage 3-4 vs. stage 1-2): OR = 1.73, 95% CI = 0.91-3.27, P = 0.092.
Prognostic role of CD166 expression in CRC patients using multivariate regression analysis
As shown in Figure 7, the results from three studies involving 469 patients with CRC revealed that CD166 expression was not correlated with the overall survival (OS) of patients in multivariate regression analysis (HR = 1.20, 95% CI = 0.45-3.22, P = 0.72). One study involving 58 CRC patients reported that CD166 expression was not linked to progression free survival (PFS) in multivariate regression analysis (HR = 0.65, 95% CI = 0.32-1.32, P = 0.233). One study including 112 CRC patients reported that a significant association between CD166 expression and disease free survival (DFS) was found in patients with CRC (HR = 5.61, 95% CI = 1.82-17.36, P = 0.003) using multivariate regression analysis.
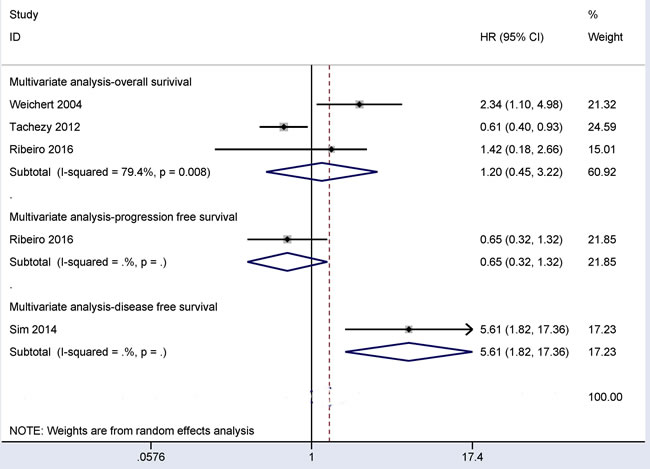
Figure 7: Forest plot showing the prognostic values of CD166 expression in overall survival (HR = 1.20, 95% CI = 0.45-3.22, P = 0.72), disease free survival (HR = 5.61, 95% CI = 1.82-17.36, P = 0.003), and progression free survival (HR = 0.65, 95% CI = 0.32-1.32, P = 0.233) for multivariate analysis.
Predictive role of CD166 expression in CRC patients using multivariate logistic regression analysis
Only one study recorded that CD166 expression may be a predictive marker of clinical outcomes in stage II CRC patients with multivariate logistic regression analysis (OS: OR = 9.97, 95% CI = 1.17-84.90, P = 0.035; disease-specific survival: OR = 29.02, 95% CI = 2.17-389.08, P = 0.011) [23].
Publication bias
Egger’s test was applied to detect a possible publication bias in relation to gender, tumor differentiation, clinical stage, T classification or lymph node status (Figure 8). There was obvious evidence of a publication bias in relation to T classification and lymph node status (P < 0.05). No evidence of a publication bias was detected in relation to gender, tumor differentiation or clinical stage (P > 0.1).
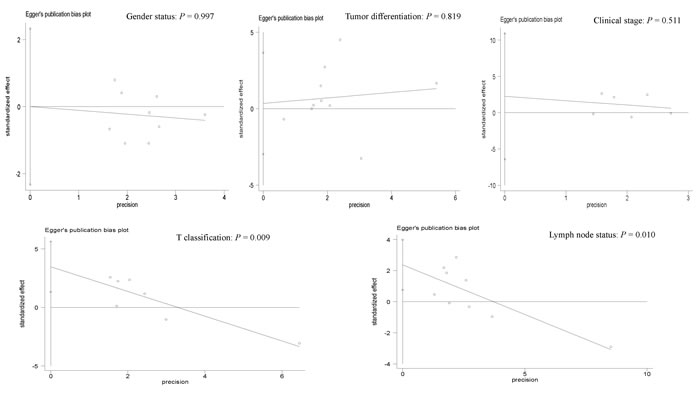
Figure 8: Forest plot of the possible publication bias in relation to gender status (P = 0.997), tumor differentiation (P = 0.819), clinical stage (P = 0.511), T classification (P = 0.009) and lymph node status (P = 0.010) using Egger’s test.
Discussion
There are a variety of cell surface markers used to identify and enrich CSCs from different human cancers, such as CD44, CD24, CD29, CD133 and epithelial cell adhesion molecule (EpCAM) [33, 34]. As a CSC marker, CD166 expression may be involved in the progression, metastasis and prognosis of several malignant tumors [16, 35, 36]. Immunohistochemical staining of CD166 was found to be frequently expressed in CRC [18, 20, 21], with frequency ranging from 30.6% [19] to 76.3% [24]. CRC generally derives from normal colonic mucosa that turns into adenomas; then these adenomas progress to malignancy through genetic and environment factors [37, 38]. Our findings indicated that CD166 expression was notably higher in CRC than in colonic adenomas or normal colonic mucosa, suggesting that CD166 expression may be related to the carcinogenesis of CRC. We found that the number of cancer and control groups were not well matched, which may have led to the heterogeneous results.
Next, the associations of CD166 expression with clinicopathological characteristics were controversial in CRC. Horst et al reported that no significant relationship was found between CD166 expression and clinicopathological features, including gender, lymph node status, T category, and tumor differentiation [27]. Tachezy et al found CD166 expression was significantly associated with tumor grade, but not linked to gender, lymph node status and T category [24]. CD166 expression was found to be associated with T category and lymph node status in CRC by Lugli et al [26]. Our study in a large series of patients with CRC suggested that CD166 expression was not associated with these clinicopathological features, including gender, tumor location, distant metastasis, lymph node status, clinical stage, T classification or tumor differentiation. Interestingly, CD166 expression was correlated with vascular invasion status. Lugli et al reported that CD166 expression showed a trend towards a strong negative association with vascular invasion (P = 0.076) in a large population (1,245 CRC patients) [26]. Two other studies with small populations (less than 130 CRC patients per study) reported no correlation between CD166 expression and vascular invasion status (Figure 3). The current meta-analysis involving 1,486 patients with CRC showed that CD166 expression was significantly lower in patients with vascular invasion than in patients without vascular invasion (OR = 0.75, P = 0.017). This finding suggested that CD166 expression may significantly decrease the risk of CRC patients with vascular invasion and that CD166 may serve as a potential drug therapy target for patients with vascular invasion.
Finally, the results on the prognostic role of CD166 expression were inconsistent for OS using multivariate regression analysis [19, 20, 24]. The current study involving 469 CRC patients revealed that CD166 expression was not correlated with OS of CRC patients in multivariate regression analysis (HR = 1.20, 95% CI = 0.45-3.22, P = 0.72). In addition, Ribeiro et al reported that no correlation was observed between CD166 expression and PFS using multivariate regression analysis (HR = 0.65, 95% CI = 0.32-1.32, P = 0.233) [20]. The correlation between CD166 expression and DFS was observed using multivariate regression analysis by Sim et al (HR = 5.61, P = 0.003) [18]. In the future, additional studies with large sample sizes will be needed to validate the prognostic value of CD166 expression in OS, PFS and DFS using multivariate regression analysis.
Limitations and prospects
Our study had several potential limitations. First, although the PubMed, EMBASE, EBSCO, CNKI, and Wanfang literature databases were systematically searched to minimize any potential publication bias as completely as possible, potential publication bias was observed in relation to T classification and lymph node status. The possible reasons for publication bias are stated as follows: 1) articles with positive conclusions are more easily published than articles with negative conclusions, which are absent; and 2) publications with other styles, such as unpublished papers and conference abstracts, were excluded based on insufficient information. Second, the main population consisted of Caucasian and Asian populations, while the inclusion of other ethnic populations, such as Africans, was limited. Third, the cut-off values of CD166 expression from the eligible studies were different; in the future, CD166 expression should be defined as positive or negative based on a standard. Fourth, the prognostic role of CD166 expression using multivariate regression analysis in OS, PFS and DFS was analyzed in a small population with CRC; additional large-scale studies are necessary to further validate the clinical outcomes of CD166 expression on CRC patients based on larger sample sizes. Finally, only one study in stage II CRC patients reported that CD166 expression may become a predictive biomarker of OS and disease-specific survival using multivariate logistic regression analysis [23], which suggests that additional studies with large populations are needed to confirm the predictive role of CD166 expression in CRC patients with the detailed tumor staging.
Conclusions
Our meta-analysis suggested that CD166 expression is associated with the carcinogenesis of CRC and a decreased risk of patients with vascular invasion. CD166 expression may be correlated with a poor prognosis in DFS, and CD166 may become a predictive biomarker of survival for stage II CRC patients. CD166 expression is not correlated with gender, tumor location, distant metastasis, lymph node status, clinical stage, T classification, tumor differentiation, or the prognosis of CRC patients in OS and PFS using multivariate regression analysis. Further large-scale studies with larger sample sizes should be conducted to validate the prognostic and predictive values of CD166 expression in patients with CRC.
MATERIALS AND Methods
Search strategy
The PubMed, EMBASE, EBSCO, CNKI and Wanfang literature databases were searched to obtain the relevant literature published before November 07, 2016. The following key words and terms were used during the search: (CD166 OR ALCAM OR activated leukocyte cell adhesion molecule) AND (colorectal cancer OR colorectal tumor OR colorectal carcinoma OR colorectal neoplasm OR CRC). The references of eligible publications were also scanned to find additional potential studies.
Selection criteria
The eligible studies in this meta-analysis had to meet the following inclusion criteria: 1) patients with CRC were diagnosed based on histopathological examination; 2) studies regarding CD166 expression were detected using an immunohistochemistry (IHC) method; 3) CD166 expression was defined as positive in the original publications; 4) studies provided sufficient data to estimate the correlation of CD166 expression between CRC and colonic adenomas, and normal colonic mucosa, and in relation to the clinical features of CRC; 5) studies provided the original information from multivariate regression analysis to assess the prognostic or predictive role of CD166 expression in patients with CRC. Only the complete study with the most information was included when authors published more than one paper using overlapping samples.
Data extraction
All available data were collected from all of studies, including the surname of the first author, year of publication, country, ethnicity, cut-off value (positivity), age, immunohistochemical staining patterns, number of tissue samples, frequency of expression, clinicopathological features, such as gender, tumor location, distant metastasis, lymph node status, vascular invasion, clinical stage, T classification and tumor differentiation, as well as OS, DFS, PFS and disease-specific survival with multivariate analysis. Control groups included colonic adenomas and normal colonic mucosa. Stages ≤ 2 were defined as early stage, and stages of 3-4 were defined as later stage.
Statistical analysis
Data analyses were conducted using Stata software 12.0 (STATA Corp., College Station, TX, USA). The overall odds ratios (ORs) and their corresponding 95% confidence intervals (95% CIs) were calculated to estimate the correlation of CD166 expression between CRC and control groups, and in relation to patient characteristics (such as gender, tumor location, distant metastasis, lymph node status, vascular invasion, clinical stage, T classification and tumor differentiation.) The combined hazard ratios (HRs) with their 95% CIs were also calculated to analyze the prognostic role of CD166 expression in patients using multivariate analysis. The Cochran’s Q statistic and I² test were applied to evaluate possible heterogeneity among studies [39]. A random-effects model was used to make the results more reliable for this meta-analysis when there was substantial heterogeneity (I2 > 50% or P < 0.1); otherwise, the fixed-effects model was performed [40, 41]. The potential publication bias was detected using Egger’s test in cases with more than five studies [42]. A P value less than 0.05 was considered statistically significant.
Authors’ contributions
FH and QS contributed to the conception and design of this research. SH and WY contributed to drafts of the article and provided final approval of the submitted version. SH, WY, SZ, HL, SL and WL contributed to the analyses and interpretation of the data and the completion of figures and tables. All authors read and approved the final manuscript.
Acknowledgments and Funding
This research was supported by grants from the Natural Science Foundation of China (81473624).
CONFLICTS OF INTEREST
All authors declare that they have no competing financial interests.
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87-108.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5-29.
3. Labianca R, Merelli B. Screening and diagnosis for colorectal cancer: present and future. Tumori. 2010; 96:889-901.
4. Yuan J, Zhang F, Niu R. Multiple regulation pathways and pivotal biological functions of STAT3 in cancer. Sci Rep. 2015; 5:17663.
5. Majumdar A, Curley SA, Wu X, Brown P, Hwang JP, Shetty K, Yao ZX, He AR, Li S, Katz L, Farci P, Mishra L. Hepatic stem cells and transforming growth factor beta in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2012; 9:530-538.
6. da Silva-Diz V, Simon-Extremera P, Bernat-Peguera A, de Sostoa J, Urpi M, Penin RM, Sidelnikova DP, Bermejo O, Vinals JM, Rodolosse A, Gonzalez-Suarez E, Moruno AG, Pujana MA, et al. Cancer Stem-like Cells Act via Distinct Signaling Pathways in Promoting Late Stages of Malignant Progression. Cancer Res. 2016; 76:1245-1259.
7. Xu R, Cai MY, Luo RZ, Tian X, Han JD, Chen MK. The Expression Status and Prognostic Value of Cancer Stem Cell Biomarker CD133 in Cutaneous Squamous Cell Carcinoma. JAMA Dermatol. 2016; 152:305-311.
8. Giordano C, Chemi F, Panza S, Barone I, Bonofiglio D, Lanzino M, Cordella A, Campana A, Hashim A, Rizza P, Leggio A, Gyorffy B, Simoes BM, et al. Leptin as a mediator of tumor-stromal interactions promotes breast cancer stem cell activity. Oncotarget. 2016; 7:1262–1275. doi: 10.18632/oncotarget.6014.
9. Zhao Y, Peng J, Zhang E, Jiang N, Li J, Zhang Q, Zhang X, Niu Y. CD133 expression may be useful as a prognostic indicator in colorectal cancer, a tool for optimizing therapy and supportive evidence for the cancer stem cell hypothesis: a meta-analysis. Oncotarget. 2016; 7:10023-10036. doi: 10.18632/oncotarget.7054.
10. Chen J, Xia Q, Jiang B, Chang W, Yuan W, Ma Z, Liu Z, Shu X. Prognostic Value of Cancer Stem Cell Marker ALDH1 Expression in Colorectal Cancer: A Systematic Review and Meta-Analysis. PLoS One. 2015; 10:e0145164.
11. Jiang Y, Li W, He X, Zhang H, Jiang F, Chen Z. Lgr5 expression is a valuable prognostic factor for colorectal cancer: evidence from a meta-analysis. BMC Cancer. 2015; 15:948.
12. Swart GW, Lunter PC, Kilsdonk JW, Kempen LC. Activated leukocyte cell adhesion molecule (ALCAM/CD166): signaling at the divide of melanoma cell clustering and cell migration? Cancer Metastasis Rev. 2005; 24:223-236.
13. Hansen AG, Swart GW, Zijlstra A. ALCAM: Basis Sequence: Mouse. AFCS Nat Mol Pages. 2011; 2011.
14. van Kempen LC, Nelissen JM, Degen WG, Torensma R, Weidle UH, Bloemers HP, Figdor CG, Swart GW. Molecular basis for the homophilic activated leukocyte cell adhesion molecule (ALCAM)-ALCAM interaction. J Biol Chem. 2001; 276:25783-25790.
15. Bowen MA, Aruffo A. Adhesion molecules, their receptors, and their regulation: analysis of CD6-activated leukocyte cell adhesion molecule (ALCAM/CD166) interactions. Transplant Proc. 1999; 31:795-796.
16. Donizy P, Zietek M, Halon A, Leskiewicz M, Kozyra C, Matkowski R. Prognostic significance of ALCAM (CD166/MEMD) expression in cutaneous melanoma patients. Diagn Pathol. 2015; 10:86.
17. Clauditz TS, von Rheinbaben K, Lebok P, Minner S, Tachezy M, Borgmann K, Knecht R, Sauter G, Wilczak W, Blessmann M, Munscher A. Activated leukocyte cell adhesion molecule (ALCAM/CD166) expression in head and neck squamous cell carcinoma (HNSSC). Pathol Res Pract. 2014; 210:649-655.
18. Sim SH, Kang MH, Kim YJ, Lee KW, Kim DW, Kang SB, Eom KY, Kim JS, Lee HS, Kim JH. P21 and CD166 as predictive markers of poor response and outcome after fluorouracil-based chemoradiotherapy for the patients with rectal cancer. BMC Cancer. 2014; 14:241.
19. Weichert W, Knosel T, Bellach J, Dietel M, Kristiansen G. ALCAM/CD166 is overexpressed in colorectal carcinoma and correlates with shortened patient survival. J Clin Pathol. 2004; 57:1160-1164.
20. Ribeiro KB, da Silva Zanetti J, Ribeiro-Silva A, Rapatoni L, de Oliveira HF, da Cunha Tirapelli DP, Garcia SB, Feres O, da Rocha JJ, Peria FM. KRAS mutation associated with CD44/CD166 immunoexpression as predictors of worse outcome in metastatic colon cancer. Cancer Biomark. 2016; 16:513-521.
21. Manhas J, Bhattacharya A, Agrawal SK, Gupta B, Das P, Deo SV, Pal S, Sen S. Characterization of cancer stem cells from different grades of human colorectal cancer. Tumour Biol. 2016; 37:14069-14081.
22. Shafaei S, Sharbatdaran M, Kamrani G, Khafri S. The association between CD166 detection rate and clinicopathologic parameters of patients with colorectal cancer. Caspian J Intern Med. 2013; 4:768-772.
23. Hansen AG, Freeman TJ, Arnold SA, Starchenko A, Jones-Paris CR, Gilger MA, Washington MK, Fan KH, Shyr Y, Beauchamp RD, Zijlstra A. Elevated ALCAM shedding in colorectal cancer correlates with poor patient outcome. Cancer Res. 2013; 73:2955-2964.
24. Tachezy M, Zander H, Gebauer F, Marx A, Kaifi JT, Izbicki JR, Bockhorn M. Activated leukocyte cell adhesion molecule (CD166)—its prognostic power for colorectal cancer patients. J Surg Res. 2012; 177:e15-20.
25. Piscuoglio S, Lehmann FS, Zlobec I, Tornillo L, Dietmaier W, Hartmann A, Wunsch PH, Sessa F, Rummele P, Baumhoer D, Terracciano LM. Effect of EpCAM, CD44, CD133 and CD166 expression on patient survival in tumours of the ampulla of Vater. J Clin Pathol. 2012; 65:140-145.
26. Lugli A, Iezzi G, Hostettler I, Muraro MG, Mele V, Tornillo L, Carafa V, Spagnoli G, Terracciano L, Zlobec I. Prognostic impact of the expression of putative cancer stem cell markers CD133, CD166, CD44s, EpCAM, and ALDH1 in colorectal cancer. Br J Cancer. 2010; 103:382-390.
27. Horst D, Kriegl L, Engel J, Kirchner T, Jung A. Prognostic significance of the cancer stem cell markers CD133, CD44, and CD166 in colorectal cancer. Cancer Invest. 2009; 27:844-850.
28. Chen X, Mei GL, Wang D, Zhu JW. Clinical significance of cortactin and CD166 expression in tissue samples from patients with colorectal cancer. The Journal of practical medicine. 2011; 27:2973-2975.
29. Zhang YS, Wang K, Huang J. Expression of CD166 in colon cancer and its biological significance. Zhejiang medical journal. 2013; 35:1123-1126.
30. Zhou XY, Jin JQ, Qi XW. Expression and Clinical Significance of CD166 and TSPAN-1 in Colorectal Carcinoma. Journal of Aerospace Medicine. 2013; 24:1315-1318.
31. Zhu XT, Huang J, Li SG, Lu XF, Hu B. Expression and its clinical significance of cancer markers CD133 and CD166 in colorectal carcinoma. Chinese journal of health laboratory technology. 2015; 25:1692-1695.
32. Zhuang JL, Pan QX, Su ZJ, Xu RY, Wang CR. Expressions and significances of CD24 and ALCAM in colorectal carcinoma. International Journal of Pathology & Clinical Medicine. 2007; 27:382-387.
33. Zhang X, Hua R, Wang X, Huang M, Gan L, Wu Z, Zhang J, Wang H, Cheng Y, Li J, Guo W. Identification of stem-like cells and clinical significance of candidate stem cell markers in gastric cancer. Oncotarget. 2016; 7:9815-9831. doi: 10.18632/oncotarget.6890.
34. Feng D, Wang N, Hu J, Li W. Surface markers of hepatocellular cancer stem cells and their clinical potential. Neoplasma. 2014; 61:505-513.
35. Morise M, Hishida T, Takahashi A, Yoshida J, Ohe Y, Nagai K, Ishii G. Clinicopathological significance of cancer stem-like cell markers in high-grade neuroendocrine carcinoma of the lung. J Cancer Res Clin Oncol. 2015; 141:2121-2130.
36. Eom DW, Hong SM, Kim G, Bae YK, Jang KT, Yu E. Prognostic Significance of CD44v6, CD133, CD166, and ALDH1 Expression in Small Intestinal Adenocarcinoma. Appl Immunohistochem Mol Morphol. 2015; 23:682-688.
37. Amankwatia EB, Chakravarty P, Carey FA, Weidlich S, Steele RJ, Munro AJ, Wolf CR, Smith G. MicroRNA-224 is associated with colorectal cancer progression and response to 5-fluorouracil-based chemotherapy by KRAS-dependent and -independent mechanisms. Br J Cancer. 2015; 112:1480-1490.
38. Jass JR, Young J, Leggett BA. Evolution of colorectal cancer: change of pace and change of direction. J Gastroenterol Hepatol. 2002; 17:17-26.
39. Zintzaras E, Ioannidis JP. HEGESMA: genome search meta-analysis and heterogeneity testing. Bioinformatics. 2005; 21:3672-3673.
40. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557-560.
41. DerSimonian R. Meta-analysis in the design and monitoring of clinical trials. Stat Med. 1996; 15:1237-1248; discussion 1249-1252.
42. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629-634.

