INTRODUCTION
Frontotemporal lobar degeneration (FTLD) is a common form of dementia that is characterized by focal atrophy of frontal and/or anterior temporal brain lobes [1]. The distinct clinical subtypes of FTLD include behavior variant frontotemporal dementia (bvFTD), semantic dementia (SD) and progressive non-fluent aphasia (PNFA) [2, 3]. Several genetic variants are associated with FTLD [4–6]. In the Italian population, C276T polymorphism of neuronal nitric oxide synthase (nNOS) gene is linked to increased susceptibility to sporadic FTLD [5]. Conversely, A2518G polymorphism in monocyte chemotactic protein 1 (MCP-1) gene is a protective factor of sporadic FTLD [6].
Human Apolipoprotein E (APOE) gene that is located on chromosome 19 is involved in lipid homeostasis and is implicated in cardiovascular disease [7, 8]. Altered structure and function of ApoE protein is associated with neurodegenerative disorders such as Alzheimer’s disease (AD) [8]. APOE gene has three common alleles (ε2, ε3 and ε4) and six related genotypes (ε3ε3, ε3ε2, ε2ε2, ε3ε4, ε4ε4, and ε2ε4) and distinct pathological roles have been attributed to all 3 alleles of APOE, namely, ε2, ε3, and ε4 [8]. The conclusions of various studies that have investigated the role of APOE polymorphism in FTLD have been inconsistent and contradictory. For instance, APOE ε4 was associated with increased FTLD risk in the Dutch population [9]. However, a negative association was reported between APOE polymorphism and FTLD risk in German patients [10]. In addition, genome wide association studies (GWAS) data of FTLD did not confirm a positive association with the APOE gene [11, 12].
So far, only two meta-analyses have reported on the relationship between APOE polymorphism and susceptibility to FTLD [13, 14]. Since many new studies have published on since 2013, we conducted an updated meta-analysis to reassess this association by systematically retrieving, screening and enrolling the available case-control studies to determine the association between APOE polymorphism and FTLD risk.
RESULTS
Selection criteria for eligible studies in the meta-analysis
Figure 1 shows the flow diagram of methodology used to search databases and select relevant studies based on “Preferred Reporting Items for Systematic Reviews and Meta-Analyses” (PRISMA). A total of 488 records were initially identified by searching four online databases, namely PubMed (n = 74), Cochrane Central Register of Controlled Trials (CENTRAL, n = 0), Excerpta Medica Database (EMBASE, n = 290) and Web of Science (WOS, n = 124). We removed 112 duplicate records after identifying them on Endnote. Further, 284 records that included case reports, posters, book articles, reviews, meeting abstracts (n = 53), non-FTLD, non-ApoE, non-clinical, non-mutation data (n = 223), and meta-analysis (n = 8) were also excluded. The remaining 92 full-text articles were then assessed for eligibility that resulted in excluding 41 articles for lack of relevant or control data. Finally, 51 case-control studies with 5123 cases and 20566 controls were included in our meta-analysis [5, 6, 9, 10, 13, 15–60]. The NOS assessment showed that three studies had a NOS score of 5 [39, 46, 47] and another three studies had a NOS score of 6 [26, 28, 32] indicating the medium-quality. The other 45 studies [5, 6, 9, 10, 13, 15–25, 27, 29–31, 33–38, 40–45, 48–60] were of high-quality with NOS scores > 6. Supplementary Table 1 shows the characteristics of eligible studies.
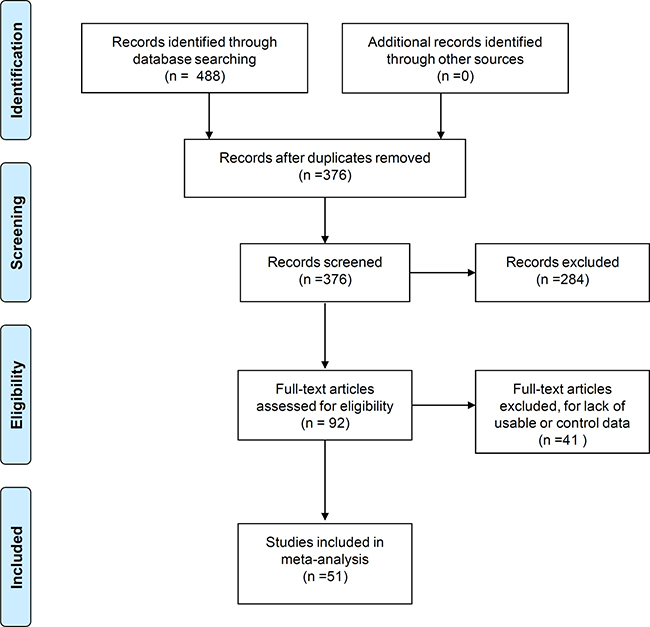
Figure 1: Flow diagram of database search and study selection.
APOE polymorphism and FTLD risk meta-analysis
The pooled values of OR and 95% confidence interval (CI) were analyzed by Mantel-Haenszel statistics to identify associations between APOE ε2, ε3, ε4 alleles and FTLD risks. As shown in Table 1, increased FTLD risk was observed in ε4 vs. ε3 allele model (P < 0.001, OR = 1.66, 95% CI = 1.35–2.03), ε4 vs. ε2 allele model (P = 0.008, OR = 1.52, 95% CI = 1.12–2.06), ε4 vs. ε2+ε3+ε4 allele model (P < 0.001, OR = 1.52, 95% CI = 1.31–1.76), ε4 vs. ε2+ε3+ε4 carrier model (P < 0.001, OR = 1.50, 95% CI = 1.32–1.70). Similarly, increased risk was observed for the genetic models of ε4ε4 vs. ε3ε3 (P < 0.001, OR = 3.23, 95% CI = 2.27–4.60), ε3ε4 vs. ε3ε3 (P < 0.001, OR = 1.62, 95% CI = 1.25–2.10), ε3ε4+ε4ε4 vs. ε3ε3 (P < 0.001, OR = 1.70, 95% CI = 1.33–2.19), and ε4ε4 vs. ε3ε3+ε3ε4 (P < 0.001, OR = 2.82, 95% CI = 1.99–3.98) as shown in Table 1. These data demonstrated that the APOE ε4 allele increased FTLD susceptibility in a dose-dependent manner.
Table 1: Meta-analysis for the association between APOE polymorphism and FTLD risks
Comparison |
Study number |
Sample size (case/control) |
Association Test |
Heterogeneity |
Model |
||
|---|---|---|---|---|---|---|---|
OR (95% CI) |
P |
I2 |
P |
||||
ε4 vs. ε3 allele |
34 |
2072/13661 |
1.66 (1.35–2.03) |
< 0.001 |
68.7% |
< 0.001 |
Random |
ε4 vs. ε2 allele |
34 |
2072/13661 |
1.52 (1.12–2.06) |
0.008 |
60.8% |
< 0.001 |
Random |
ε4 vs. ε2+ε3+ε4 allele |
40 |
2417/15059 |
1.52 (1.31–1.76) |
< 0.001 |
51.3% |
< 0.001 |
Random |
ε4 vs. ε2+ε3+ε4 carrier |
47 |
3511/18046 |
1.50 (1.32–1.70) |
< 0.001 |
40.9% |
0.002 |
Random |
ε4ε4 vs. ε3ε3 |
30 |
1650/11634 |
3.23 (2.27–4.60) |
< 0.001 |
0.0% |
0.922 |
Fixed |
ε3ε4 vs. ε3ε3 |
32 |
1696/11700 |
1.62 (1.25–2.10) |
< 0.001 |
67.3% |
< 0.001 |
Random |
ε3ε4+ε4ε4 vs. ε3ε3 |
32 |
1696/11700 |
1.70 (1.33–2.19) |
< 0.001 |
67.6% |
< 0.001 |
Random |
ε4ε4 vs. ε3ε3+ε3ε4 |
30 |
1650/11634 |
2.82 (1.99–3.98) |
< 0.001 |
0.0% |
0.962 |
Fixed |
ε2 vs. ε3 allele |
34 |
2072/13661 |
1.09 (0.87–1,37) |
0.462 |
51.5% |
< 0.001 |
Random |
ε2 vs. ε2+ε3+ε4 allele |
34 |
2072/13661 |
1.01 (0.82–1.24) |
0.953 |
43.1% |
0.005 |
Random |
ε2 vs. ε2+ε3+ε4 carrier |
32 |
1936/13591 |
0.93 (0.74–1.17) |
0.545 |
42.3% |
0.007 |
Random |
ε2ε2 vs. ε3ε3 |
22 |
944/9708 |
1.74 (1.03–2.96) |
0.039 |
0.0% |
0.774 |
Fixed |
ε3ε2 vs. ε3ε3 |
32 |
1346/10740 |
0.87 (0.73–1.04) |
0.132 |
24.2% |
0.110 |
Fixed |
ε3ε2+ε2ε2 vs. ε3ε3 |
32 |
1346/10740 |
0.95 (0.72–1.23) |
0.678 |
41.6% |
0.008 |
Random |
ε2ε2 vs. ε3ε3+ε3ε2 |
22 |
944/9708 |
1.84 (1.08–3.12) |
0.024 |
0.0% |
0.842 |
Fixed |
P < 0.05 of association test is shown in bold.
In contrast, APOE ε2 allele was not associated with FTLD risk. Our analyses for APOE ε2 showed significant difference only in the models of ε2ε2 vs. ε3ε3 (P = 0.039, OR = 1.74, 95% CI = 1.03–2.96) and ε2ε2 vs. ε3ε3+ε3ε2 (P = 0.024, OR = 1.84, 95% CI = 1.08–3.12), but not others (all P > 0.05). The forest plots for the allele models of ε4 vs. ε3 and ε2 vs. ε3 are shown in Figures 2 and 3, respectively.
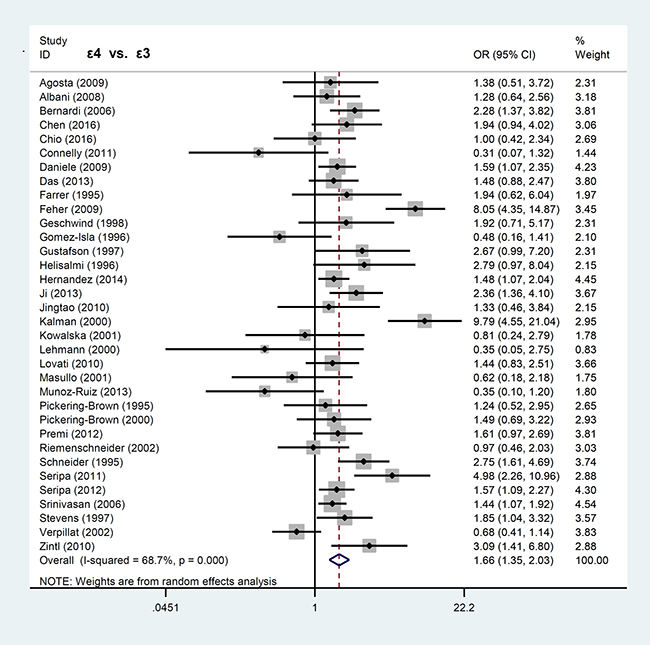
Figure 2: Forest plot of meta-analysis of the ε4 vs. ε3 allele model.
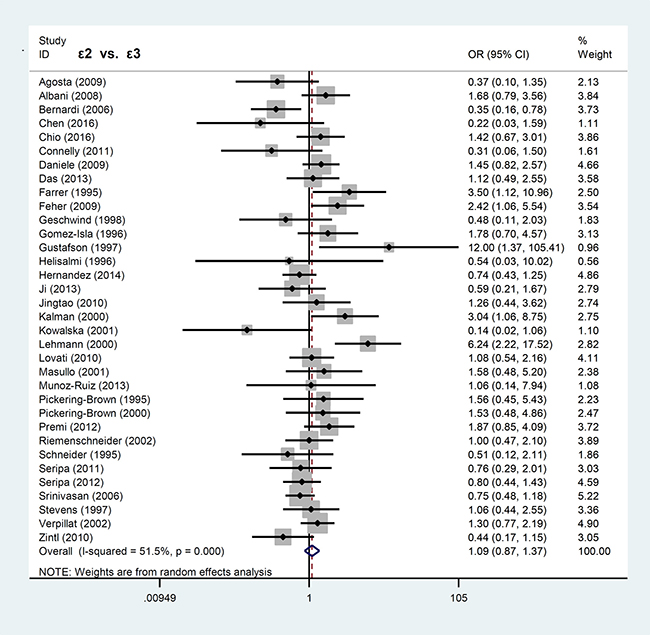
Figure 3: Forest plot of meta-analysis of the ε2 vs. ε3 allele model.
Subgroup analysis of APOE polymorphism and FTLD risk
Next, we performed a series of subgroup analyses based on ethnicity (Caucasian and Asian), country (Italy, China, USA and UK), source of control (PB and HB), clinical subtypes (bvFTD, SD, PNFA, FTLD MND-, FTLD MND+), HWE (P value of HWE > 0.05 and < 0.05), and NOS (score > 6 and < = 6). We observed that Caucasian, Italian, PB, P value of HWE > 0.05, and NOS score > 6 subgroups for APOE ε4 demonstrated increased FTLD risk in the following models: ε4 vs. ε3 (Table 2, all P < 0.01, OR > 1); ε4 vs. ε2 (Table 2, all P < 0.05, OR > 1); ε4 vs. ε2+ε3+ε4 allele (Table 2, all P < 0.001, OR > 1); ε4ε4 vs. ε3ε3 (Table 3, all P < 0.001, OR > 1); ε3ε4 vs. ε3ε3 (Table 3, all P < 0.01, OR > 1); ε3ε4+ε4ε4 vs. ε3ε3 (Table 4, all P < 0.01, OR > 1); and ε4ε4 vs. ε3ε3+ε3ε4 (Table 4, all P < 0.01, OR > 1). These data demonstrated that both ε4ε4 and ε3ε4 genotypes of APOE conferred increased susceptibility to FTLD in the Caucasian population, especially people of Italian origin.
Table 2: Subgroup analysis of association between APOE ε4 and FTLD risks for ε4 vs. ε3, ε4 vs. ε2, and ε4 vs. ε2+ε3+ε4 allele models
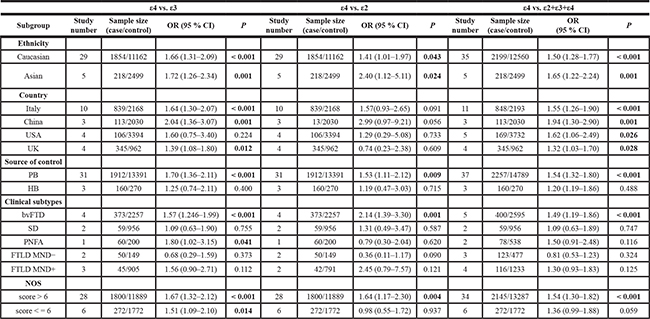
PB: population-based; HB: hospital-based; bvFTD: behavior variant frontotemporal dementia; SD: semantic dementia; PNFA: progressive non-fluent aphasia; FTLD: Frontotemporal lobar degeneration; MND: motor neuron disease; NOS: Newcastle-Ottawa scale; P < 0.05 is shown in bold.
Table 3: Subgroup analysis of association between APOE ε3/ε4 genotype frequency and FTLD risks for ε4ε4 vs. ε3ε3 and ε3ε4 vs. ε3ε3 models
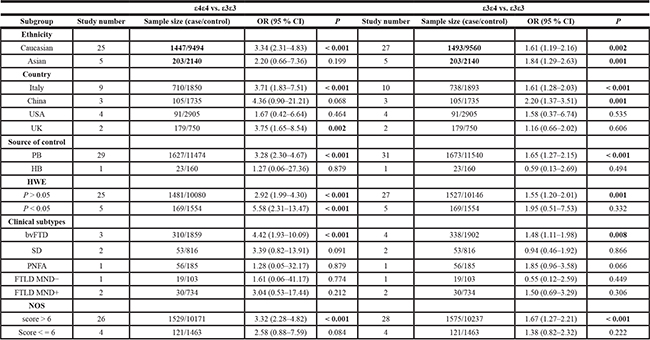
PB: population-based; HB: hospital-based; HWE: Hardy-Weinberg Equilibrium; bvFTD: behavior variant frontotemporal dementia; SD: semantic dementia; PNFA: progressive non-fluent aphasia; FTLD: Frontotemporal lobar degeneration; MND: motor neuron disease; NOS: Newcastle-Ottawa scale; P < 0.05 is shown in bold.
Table 4: Subgroup analysis of association between APOE ε3/ε4 genotype frequency and FTLD risks for ε3ε4+ε4ε4 vs. ε3ε3 and ε4ε4 vs. ε3ε3+ε3ε4 models
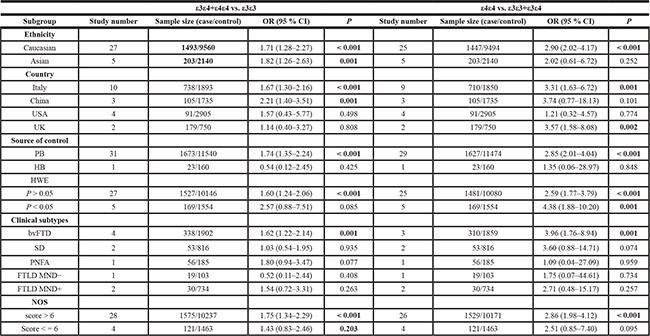
PB: population-based; HB: hospital-based; HWE: Hardy-Weinberg Equilibrium; bvFTD: behavior variant frontotemporal dementia; SD: semantic dementia; PNFA: progressive non-fluent aphasia; FTLD: Frontotemporal lobar degeneration; MND: motor neuron disease; NOS: Newcastle-Ottawa scale; P < 0.05 is shown in bold.
Moreover, our analysis for APOE ε4 in Asian populations, especially Chinese individuals demonstrated enhanced FTLD risk for the allele (Table 2, ε4 vs. ε3, P = 0.001, OR = 2.04; ε4 vs. ε2+ε3+ε4, P = 0.001, OR = 1.94), heterozygote (Table 3, ε3ε4 vs. ε3ε3, P = 0.001, OR = 2.20), dominant (Table 4, ε3ε4+ε4ε4 vs. ε3ε3, P = 0.001, OR = 2.21) and carrier (Supplementary Table 2, ε4 vs. ε2+ε3+ε4 carrier, P = 0.003, OR = 1.92) models, but were not significant for homozygote (Table 3, ε4ε4 vs. ε3ε3, P = 0.068) and recessive (Table 4, ε4ε4 vs. ε3ε3+ε3ε4, P = 0.101) models. These indicated that in the Asian population, including the Chinese individuals, the ε3ε4 genotype was linked to increased FTLD risk. The forest plots of subgroup analysis based on ethnicity for APOE ε4 under all genetic models were shown in Supplementary Figures 1–8.
In addition, stratified analysis of clinical subtypes (bvFTD, SD, PNFA, FTLD with or without motor neuron disease) showed that all genetic models were associated with increased bvFTD risk (Tables 2–4, Supplementary Table 2, all P < 0.01, OR > 1). This suggested that APOE ε4 was a risk factor for bvFTD.
In regard to APOE ε2, no significant differences were observed in the subgroup analyses for almost all genetic models (Supplementary Tables 2–5, P > 0.05). These findings further confirmed the negative genetic association between APOE ε2 and FTLD risks.
Heterogeneity, publication bias and sensitivity analysis
We assessed heterogeneity between studies by performing the Q statistic and I2 tests. As shown in Table 1, there was no heterogeneity among different studies for the following models: ε4ε4 vs. ε3ε3, ε4ε4 vs. ε3ε3+ε3ε4, ε2ε2 vs. ε3ε3, ε3ε2 vs. ε3ε3, and ε2ε2 vs. ε3ε3+ε3ε2 (all P value of heterogeneity > 0.1, I2< 25 %). Hence, we used the fixed-effect model for their analysis. The random-effect model was applied for others.
In addition, Begg’s test and Egger’s test analyses suggested absence of publication bias (Supplementary Table 6, all P value > 0.1). Begg’s funnel plot of publication bias for ε4 vs. ε3 and ε2 vs. ε3 allele models are shown in Figure 4. Furthermore, sensitivity analysis was performed to evaluate the reliability of data and strengthen the validity of genetic relationship. We observed that similar pooled ORs were obtained when individual studies were omitted one by one, thereby indicating that the original statistical data were genuine and robust (Figure 5).
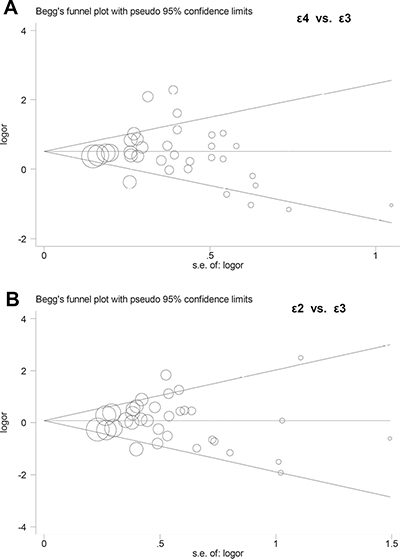
Figure 4: Begg’s funnel plots of publication bias. (A) ε4 vs. ε3 allele model; (B) ε2 vs. ε3 allele model.
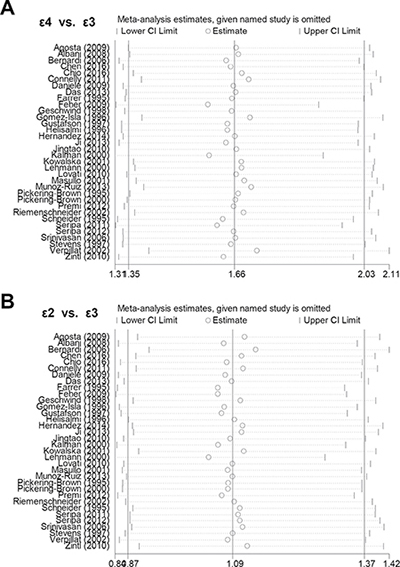
Figure 5: Sensitivity analyses. (A) ε4 vs. ε3 allele model; (B) ε2 vs. ε3 allele model.
DISCUSSION
In 2002, Verpillat et al. [13] carried out a meta-analysis of 11 studies, and reported that APOE ε2 was associated with an increased risk of FTLD in the Caucasian population. However, in 2013, another meta-analysis based on 28 studies by Rubino et al. [14] in 2013 showed that FTLD susceptibility was associated with APOE ε4, but not ε2. These contradictory conclusions may have been a result of small and different sample sizes.
Recently, mutations in valosin-containing protein (VCP), progranulin (GRN), and the microtubule-associated protein tau (MAPT) genes were reported by us in 38 Chinese FTLD cases [61]. Further, our analysis of 62 Chinese FTLD patients and 381 sex- and age-matched elderly controls demonstrated significant association between FTLD susceptibility and APOE ε4, but not ε2 [36]. However, both conclusions were limited by small sample sizes. Therefore, to comprehensively assess the factors that are associated with FTLD, we enrolled 51 case-control studies and conducted an updated meta-analysis that also included subtype analyses of factors such as country, ethnicity, source of controls and clinical subtypes. Our data demonstrated a strong positive association between APOE ε4 and FTLD risks in the allele, homozygote, heterozygote, dominant recessive and carrier models. However, no statistically correlation was observed between APOE ε2 and FTLD risks, thereby confirming our previous finding [36] and partly agreeing with the results reported by Verpillat et al. [13].
FTLD and Alzheimer’s disease (AD) are main contributors to dementia [62]. The molecular mechanisms underlying the role of APOE ε4 in the pathogenesis of FTLD and AD are unclear. APOE ε4 reduced the clearance of beta-amyloid (Aβ) that resulted in enhanced Aβ deposition within the neurons in the AD mouse model [63, 64]. APOE ε4 was also associated with Aβ deposition in the brain of a FTLD case [65]. Hence, the link between APOE ε4 and Aβ deposition merits further investigation. In addition, APOE ε4 enhanced phosphorylation of tau protein in brains of transgenic mice [66]. Since FTLD-tau is a neuropathological subtype of FTLD [4, 67], abnormal Tau phosphorylation may be partly involved in the pathogenesis of FTLD by APOE ε4.
There are several limitations in this meta-analysis that need to be highlighted. Firstly, out of 51 case-control studies included in our pooled analysis, 19 studies [5, 6, 17, 18, 20, 21, 27, 35, 43, 44, 46, 47, 49, 50, 54, 55, 57–59] contained only allele or carrier data and did not provide information regarding the specific genotype frequencies of ε3ε4 and ε3ε2 that could have weakened the statistical output. Secondly, genetic heterogeneity existed between studies for majority of comparisons because of hospital based controls, lack of the pathology or autopsy confirmed FTLD diagnoses, clinical complexity, and pathological heterogeneity. Although poor quality studies were excluded based on NOS analysis, six medium quality articles [26, 28, 32, 39, 46, 47] were still included in the analysis. Hence, more high quality studies with large sample sizes are required to avoid false positives. Thirdly, our meta-analysis included only five articles based on Asian populations [22, 26, 36, 37, 39] compared to 46 articles based on Caucasian populations [5, 6, 9, 10, 13, 15–21, 23–25, 27–35, 38, 40–60]. Among these were 15 articles based on Italian populations [5, 6, 15–17, 19, 20, 23, 25, 41, 42, 48, 52, 53, 59]. In addition, only full-text articles in Chinese or English were collected for this meta-analysis. All these factors might lead to selection bias. Fourthly, bvFTD, the most frequent clinical subtype of FTLD is a clinical syndrome characterized by progressive changes of personality, abnormalities of social behavior and cognitive function, and lack of emotional response [4, 68]. Our subgroup analysis of bvFTD contained seven articles [6, 15, 21, 22, 53, 54, 56] that showed significant association with APOE ε4 . It is probable that APOE ε4 may serve as a disease modifier of bvFTD. However, this result needs to be verified since our analysis was based on a small sample size. Similarly, only four articles for PNFA [6, 21, 48, 54] and five articles for SD [6, 21, 22, 48, 56] were available and therefore the role of APOE polymorphisms in PNFA and SD could not be determined conclusively. This was true of the subgroup analysis of FTLD with or without MND. Finally, in view of the unclear etiology of FTLD, more factors, including age at onset, male/female, pathological criteria, clinical presentation, living habits, the combination of APOE and other related genes (e.g. VCP, GRN, MAPT) should be considered in future meta-analysis. Also, pathogenesis of APOE ε4 in the memory function, behavioral symptoms and brain morphological changes in FTLD-spectrum disease should be investigated.
In conclusion, this meta-analysis demonstrated that APOE ε4 was a genetic risk factor for FTLD patients in Caucasian and Asian populations, thereby corroborating the role of APOE genetic variants in FTLD. Also, our study demonstrated that APOE ε2 was not a susceptibility factor for FTLD.
MATERIALS AND METHODS
Database search and study selection
We searched four databases, including PubMed, CENTRAL, EMBASE and WOS until February 27th, 2017 with specific search terms listed in Supplementary Table 7 and identified 488 records. After removing the duplicates by endnote software (Thomson Reuters), the remaining 376 records were screened according to our inclusion/exclusion criteria. We excluded the records of case reports, posters, books, reviews, meeting abstracts, meta-analysis, and the articles with non-FTLD, non-ApoE, non-clinical, non-mutation data. The remaining 92 full-text articles were then assessed to identify 51 eligible case-control studies while removing articles that lacked control or other usable data for this meta-analysis. The PRISMA was used in this study [69]. The PRISMA 2009 checklist is shown in Supplementary Table 8.
Quality assessment of eligible studies and data extraction
Three authors independently assessed the methodological quality of the selected case-control studies using the Newcastle-Ottawa Scale (NOS) (http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp) and extracted the relevant data. Studies with a NOS score > 6 were considered high quality, whereas studies with NOS score < 5 were considered poor and removed from the included studies. Whenever there was a disagreement, it was resolved by discussion among the three authors. The following information was collected from all the selected studies and summarized: first author, year of publication, country, ethnicity, genotype distributions (ε3ε3, ε3ε2, ε2ε2, ε3ε4, ε4ε4, and ε2ε4) in case group and control group, clinical subtypes of case, source of control, and genotyping assay. The first or the corresponding author was contacted by email whenever relevant data was not available.
Statistical analyses
Stata/SE 12.0 software (StataCorp, USA) was used for Mantel-Haenszel statistic, Q statistic and I2 tests from P values, pooled ORs, and 95% CIs. P<0.05 was considered statistically significant. Six genetic models, namely allele (ε4 vs. ε3; ε2 vs. ε3; ε4 vs. ε2; ε4 vs. ε2+ε3+ε4, ε2 vs. ε2+ε3+ε4), homozygote (ε4ε4 vs. ε3ε3, ε2ε2 vs. ε3ε3), heterozygote (ε3ε4 vs. ε3ε3, ε3ε2 vs. ε3ε3), dominant (ε3ε4+ε4ε4 vs. ε3ε3, ε3ε2+ε2ε2 vs. ε3ε3), recessive (ε4ε4 vs. ε3ε3+ε3ε4, ε2ε2 vs. ε3ε3+ε3ε2) or carrier (ε4 vs. ε2+ε3+ε4 carrier; ε2 vs. ε2+ε3+ε4 carrier) were used and Hardy-Weinberg Equilibrium (HWE) was calculated by chi-squared test. P values of Q statistic >0.1 or I2 values ≤ 25% indicated heterogeneity between studies and the fixed-effect model was used for analysis. If not, the random-effect model was used. Subgroup analyses were performed based on ethnicity, country, source of control, clinical subtypes, HWE, and NOS score. Furthermore, Begg’s funnel plot (Begg’s test) and Egger’s publication bias plot (Egger’s test) was used to evaluate the potential publication bias. The P value of Begg’s test and Egger’s test > 0.05 was regarded as the absence of publication bias. Sensitivity analysis was also performed to evaluate the stability of statistical results.
ACKNOWLEDGMENTS AND FUNDING
This study was supported by National Natural Science Foundation of China (Grant Number: 81571057 and 81300947), Tianjin Science and Technology Support Programs (Grant Numbers: 13ZCZDSY01600), Tianjin Natural Science Foundation (Grant Number: 13JCYBJC21300), Key Research Project of Tianjin Public Health (Grant Number: 14KG117), the Science and Technology Project of Tianjin Municipal Health Bureau (Grant number: 2014KR10).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Pan XD, Chen XC. Clinic, neuropathology and molecular genetics of frontotemporal dementia: a mini-review. Transl Neurodegener. 2013; 2:8.
2. Tsai RM, Boxer AL. Therapy and clinical trials in frontotemporal dementia: past, present, and future. J Neurochem. 2016; 138:211–221.
3. Oeckl P, Steinacker P, Feneberg E, Otto M. Neurochemical biomarkers in the diagnosis of frontotemporal lobar degeneration: an update. J Neurochem. 2016; 138:184–192.
4. Li YQ, Tan MS, Yu JT, Tan L. Frontotemporal Lobar Degeneration: Mechanisms and Therapeutic Strategies. Mol Neurobiol. 2016; 53:6091–6105.
5. Venturelli E, Villa C, Scarpini E, Fenoglio C, Guidi I, Lovati C, Marcone A, Cortini F, Scalabrini D, Clerici F, Bresolin N, Mariani C, Cappa S, et al. Neuronal nitric oxide synthase C276T polymorphism increases the risk for frontotemporal lobar degeneration. Eur J Neurol. 2008; 15:77–81.
6. Galimberti D, Venturelli E, Villa C, Fenoglio C, Clerici F, Marcone A, Benussi L, Cortini F, Scalabrini D, Perini L, Restelli I, Binetti G, Cappa S, et al. MCP-1 A-2518G polymorphism: effect on susceptibility for frontotemporal lobar degeneration and on cerebrospinal fluid MCP-1 levels. J Alzheimers Dis. 2009; 17:125–133.
7. Lin-Lee YC, Kao FT, Cheung P, Chan L. Apolipoprotein E gene mapping and expression: localization of the structural gene to human chromosome 19 and expression of ApoE mRNA in lipoprotein- and non-lipoprotein-producing tissues. Biochemistry. 1985; 24:3751–3756.
8. Mahley RW. Apolipoprotein E: from cardiovascular disease to neurodegenerative disorders. J Mol Med (Berl). 2016; 94:739–746.
9. Stevens M, van Duijn CM, de Knijff P, van Broeckhoven C, Heutink P, Oostra BA, Niermeijer MF, van Swieten JC. Apolipoprotein E gene and sporadic frontal lobe dementia. Neurology. 1997; 48:1526–1529.
10. Riemenschneider M, Diehl J, Müller U, Förstl H, Kurz A. Apolipoprotein E polymorphism in German patients with frontotemporal degeneration. J Neurol Neurosurg Psychiatry. 2002; 72:639–641.
11. Ferrari R, Hernandez DG, Nalls MA, Rohrer JD, Ramasamy A, Kwok JB, Dobson-Stone C, Brooks WS, Schofield PR, Halliday GM, Hodges JR, Piguet O, Bartley L, et al. Frontotemporal dementia and its subtypes: a genome-wide association study. Lancet Neurol. 2014; 13:686–699.
12. Girard SL, Rouleau GA. Genome-wide association study in FTD: Divide to conquer. Lancet Neurol. 2014; 13:643–644.
13. Verpillat P, Camuzat A, Hannequin D, Thomas-Anterion C, Puel M, Belliard S, Dubois B, Didic M, Lacomblez L, Moreaud O, Golfier V, Campion D, Brice A, et al. Apolipoprotein E gene in frontotemporal dementia: an association study and meta-analysis. Eur J Hum Genet. 2002; 10:399–405.
14. Rubino E, Vacca A, Govone F, De Martino P, Pinessi L, Rainero I. Apolipoprotein E polymorphisms in frontotemporal lobar degeneration: a meta-analysis. Alzheimers Dement. 2013; 9:706–713.
15. Agosta F, Vossel KA, Miller BL, Migliaccio R, Bonasera SJ, Filippi M, Boxer AL, Karydas A, Possin KL, Gorno-Tempini ML. Apolipoprotein E epsilon 4 is associated with disease-specific effects on brain atrophy in Alzheimer’s disease and frontotemporal dementia. Proc Natl Acad Sci USA. 2009; 106:2018–2022.
16. Albani D, Prato F, Fenoglio C, Batelli S, Dusi S, De Mauro S, Polito L, Lovati C, Galimberti D, Mariani C, Scarpini E, Forloni G. Association study to evaluate the serotonin transporter and apolipoprotein E genes in frontotemporal lobar degeneration in Italy. J Hum Genet. 2008; 53: 1029–1033.
17. Bagnoli S, Piaceri I, Tedde A, Bessi V, Bracco L, Sorbi S, Nacmias B. TOMM40 polymorphisms in Italian Alzheimer’s disease and frontotemporal dementia patients. Neurol Sci. 2013; 34:995–998.
18. Balasa M, Sanchez-Valle R, Antonell A, Bosch B, Olives J, Rami L, Castellvi M, Molinuevo JL, Llado A. Usefulness of biomarkers in the diagnosis and prognosis of early-onset cognitive impairment. J Alzheimers Dis. 2014; 40:919–927.
19. Bernardi L, Maletta RG, Tomaino C, Smirne N, Di Natale M, Perri M, Longo T, Colao R, Curcio SAM, Puccio G, Mirabelli M, Kawarai T, Rogaeva E, et al. The effects of APOE and tau gene variability on risk of frontotemporal dementia. Neurobiol Aging. 2006; 27:702–709.
20. Boccardi M, Laakso MP, Bresciani L, Galluzzi S, Geroldi C, Beltramello A, Soininen H, Frisoni GB. The MRI pattern of frontal and temporal brain atrophy in fronto-temporal dementia. Neurobiol Aging. 2003; 24:95–103.
21. Borroni B, Yancopoulou D, Tsutsui M, Padovani A, Sawcer SJ, Hodges JR, Spillantini MG. Association between tau H2 haplotype and age at onset in frontotemporal dementia. Arch Neurol. 2005; 62:1419–1422.
22. Chen KL, Sun YM, Zhou Y, Zhao QH, Ding D, Guo QH. Associations between APOE polymorphisms and seven diseases with cognitive impairment including Alzheimer’s disease, frontotemporal dementia, and dementia with Lewy bodies in southeast China. Psychiatr Genet. 2016; 26:124–131.
23. Chio A, Brunetti M, Barberis M, Iazzolino B, Montuschi A, Ilardi A, Cammarosano S, Canosa A, Moglia C, Calvo A. The Role of APOE in the Occurrence of Frontotemporal Dementia in Amyotrophic Lateral Sclerosis. JAMA Neurol. 2016; 73:425–430.
24. Connelly SJ, Mukaetova-Ladinska EB, Abdul-All Z, Alves da Silva J, Brayne C, Honer WG, Mann DM. Synaptic changes in frontotemporal lobar degeneration: correlation with MAPT haplotype and APOE genotype. Neuropathol Appl Neurobiol. 2011; 37:366–380.
25. Daniele A, Matera MG, Seripa D, Acciarri A, Bizzarro A, Pilotto A, Masullo C. APOE epsilon 2/epsilon 4 genotype a risk factor for primary progressive aphasia in women. Arch Neurol. 2009; 66:910–912.
26. Das G, Sadhukhan T, Sadhukhan D, Biswas A, Pal S, Ghosh A, Das SK, Ray K, Ray J. Genetic study on frontotemporal lobar degeneration in India. Parkinsonism Relat Disord. 2013; 19:487–489.
27. Fabre SF, Forsell C, Viitanen M, Sjogren M, Wallin A, Blennow K, Blomberg M, Andersen C, Wahlund LO, Lannfelt L. Clinic-based cases with frontotemporal dementia show increased cerebrospinal fluid tau and high apolipoprotein E epsilon4 frequency, but no tau gene mutations. Exp Neurol. 2001; 168:413–418.
28. Farrer LA, Abraham CR, Volicer L, Foley EJ, Kowall NW, McKee AC, Wells JM. Allele epsilon 4 of apolipoprotein E shows a dose effect on age at onset of Pick disease. Exp Neurol. 1995; 136:162–170.
29. Feher A, Juhasz A, Rimanoczy A, Kalman J, Janka Z. Association between BDNF Val66Met polymorphism and Alzheimer disease, dementia with Lewy bodies, and Pick disease. Alzheimer Dis Assoc Disord. 2009; 23:224–228.
30. Geschwind D, Karrim J, Nelson SF, Miller B. The apolipoprotein E epsilon4 allele is not a significant risk factor for frontotemporal dementia. Ann Neurol. 1998; 44:134–138.
31. Gomez-Isla T, West HL, Rebeck GW, Harr SD, Growdon JH, Locascio JJ, Perls TT, Lipsitz LA, Hyman BT. Clinical and pathological correlates of apolipoprotein E epsilon 4 in Alzheimer’s disease. Ann Neurol. 1996; 39:62–70.
32. Gustafson L, Abrahamson M, Grubb A, Nilsson K, Fex G. Apolipoprotein-E genotyping in Alzheimer’s disease and frontotemporal dementia. Dement Geriatr Cogn Disord. 1997; 8:240–243.
33. Helisalmi S, Linnaranta K, Lehtovirta M, Mannermaa A, Heinonen O, Ryynänen M, Riekkinen P Sr, Soininen H. Apolipoprotein E polymorphism in patients with different neurodegenerative disorders. Neurosci Lett. 1996; 205:61–64.
34. Hernandez I, Mauleon A, Rosense-Roca M, Alegret M, Vinyes G, Espinosa A, Sotolongo-Grau O, Becker JT, Valero S, Tarraga L, Lopez OL, Ruiz A, Boada M. Identification of misdiagnosed fronto-temporal dementia using APOE genotype and phenotype-genotype correlation analyses. Curr Alzheimer Res. 2014; 11:182–191.
35. Ingelson M, Fabre SF, Lilius L, Andersen C, Viitanen M, Almkvist O, Wahlund LO, Lannfelt L. Increased risk for frontotemporal dementia through interaction between tau polymorphisms and apolipoprotein E ε4. Neuroreport. 2001; 12:905–909.
36. Ji Y, Liu M, Huo YR, Liu S, Shi Z, Liu S, Wisniewski T, Wang J. Apolipoprotein E ε4 Frequency Is Increased among Chinese Patients with Frontotemporal Dementia and Alzheimer’s Disease. Dement Geriatr Cogn Disord. 2013; 36:163–170.
37. Jingtao W. The association study of apolipoprotein E gene polymorphism with different types of dementia in a Chinese Han population. CAMS & PUMC. 2010; 28–29.
38. Kalman J, Juhasz A, Majtenyi K, Rimanoczy A, Jakab K, Gardian G, Rasko I, Janka Z. Apolipoprotein E polymorphism in Pick’s disease and in Huntington’s disease. Neurobiol Aging. 2000; 21:555–558.
39. Kowalska A, Asada T, Arima K, Kumakiri C, Kozubski W, Takahashi K, Tabira T. Genetic analysis in patients with familial and sporadic frontotemporal dementia: two tau mutations in only familial cases and no association with apolipoprotein epsilon4. Dement Geriatr Cogn Disord. 2001; 12:387–392.
40. Lehmann DJ, Smith AD, Combrinck M, Barnetson L, Joachim C. Apolipoprotein E epsilon2 may be a risk factor for sporadic frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2000; 69:404–405.
41. Lovati C, Galimberti D, Albani D, Bertora P, Venturelli E, Cislaghi G, Guidi I, Fenoglio C, Cortini F, Clerici F, Finazzi D, Forloni G, Scarpini E, et al. APOE epsilon2 and epsilon4 influence the susceptibility for Alzheimer’s disease but not other dementias. Int J Mol Epidemiol Genet. 2010; 1:193–200.
42. Masullo C, Daniele A, Fazio VM, Seripa D, Gravina C, Filippini V, Grossi D, Fragassi N, Nichelli P, Leone M, Gainotti G. The Apolipoprotein E genotype in patients affected by syndromes with focal cortical atrophy. Neurosci Lett. 2001; 303:87–90.
43. Minthon L, Hesse C, Sjogren M, Englund E, Gustafson L, Blennow K. The apolipoprotein E epsilon4 allele frequency is normal in fronto-temporal dementia, but correlates with age at onset of disease. Neurosci Lett. 1997; 226:65–67.
44. Morenas-Rodriguez E, Cervera-Carles L, Vilaplana E, Alcolea D, Carmona-Iragui M, Dols-Icardo O, Ribosa-Nogue R, Munoz-Llahuna L, Sala I, Belen Sanchez-Saudinos M, Blesa R, Clarimon J, Fortea J, et al. Progranulin Protein Levels in Cerebrospinal Fluid in Primary Neurodegenerative Dementias. J Alzheimers Dis. 2016; 50:539–546.
45. Munoz-Ruiz MA, Hartikainen P, Hall A, Mattila J, Koikkalainen J, Herukka SK, Julkunen V, Vanninen R, Liu Y, Lotjonen J, Soininen H. Disease state fingerprint in frontotemporal degeneration with reference to Alzheimer’s disease and mild cognitive impairment. J Alzheimers Dis. 2013; 35:727–739.
46. Pickering-Brown SM, Owen F, Isaacs A, Snowden J, Varma A, Neary D, Furlong R, Daniel SE, Cairns NJ, Mann DM. Apolipoprotein E epsilon4 allele has no effect on age at onset or duration of disease in cases of frontotemporal dementia with pick- or microvacuolar-type histology. Exp Neurol. 2000; 163:452–456.
47. Pickering-Brown SM, Siddons M, Mann DM, Owen F, Neary D, Snowden JS. Apolipoprotein E allelic frequencies in patients with lobar atrophy. Neurosci Lett. 1995; 188:205–207.
48. Premi E, Pilotto A, Alberici A, Papetti A, Archetti S, Seripa D, Daniele A, Masullo C, Garibotto V, Paghera B, Caobelli F, Padovani A, Borroni B. FOXP2, APOE, and PRNP: new modulators in primary progressive aphasia. J Alzheimers Dis. 2012; 28:941–950.
49. Rosso SM, Van Swieten JC, Roks G, Van Duijn CM, Heutink P, Cruts M, Van Broeckhoven C. Apolipoprotein E4 in the temporal variant of frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2002; 72:820.
50. Ruiz A, Dols-Icardo O, Bullido MJ, Pastor P, Rodriguez-Rodriguez E, Lopez de Munain A, de Pancorbo MM, Perez-Tur J, Alvarez V, Antonell A, Lopez-Arrieta J, Hernandez I, Tarraga L, et al. Assessing the role of the TREM2 p.R47H variant as a risk factor for Alzheimer’s disease and frontotemporal dementia. Neurobiol Aging. 2014; 35:444.e441–444.
51. Schneider JA, Gearing M, Robbins RS, de l’Aune W, Mirra SS. Apolipoprotein E genotype in diverse neurodegenerative disorders. Ann Neurol. 1995; 38:131–135.
52. Seripa D, Bizzarro A, Panza F, Acciarri A, Pellegrini F, Pilotto A, Masullo C. The APOE gene locus in frontotemporal dementia and primary progressive aphasia. Arch Neurol. 2011; 68:622–628.
53. Seripa D, Bizzarro A, Pilotto A, Palmieri O, Panza F, D’Onofrio G, Gravina C, Archetti S, Daniele A, Borroni B, Padovani A, Masullo C. TOMM40, APOE, and APOC1 in primary progressive aphasia and frontotemporal dementia. J Alzheimers Dis. 2012; 31:731–740.
54. Short RA, Graff-Radford NR, Adamson J, Baker M, Hutton M. Differences in tau and apolipoprotein E polymorphism frequencies in sporadic frontotemporal lobar degeneration syndromes. Arch Neurol. 2002; 59:611–615.
55. Sleegers K, Roks G, Theuns J, Aulchenko YS, Rademakers R, Cruts M, Van Gool WA, Van Broeckhoven C, Heutink P, Oostra BA, Van Swieten JC, Van Duijn CM. Familial clustering and genetic risk for dementia in a genetically isolated Dutch population. Brain. 2004; 127:1641–1649.
56. Srinivasan R, Davidson Y, Gibbons L, Payton A, Richardson AM, Varma A, Julien C, Stopford C, Thompson J, Horan MA, Pendleton N, Pickering-Brown SM, Neary D, et al. The apolipoprotein E epsilon4 allele selectively increases the risk of frontotemporal lobar degeneration in males. J Neurol Neurosurg Psychiatry. 2006; 77:154–158.
57. Steenland K, MacNeil J, Seals R, Levey A. Factors affecting survival of patients with neurodegenerative disease. Neuroepidemiology. 2010; 35:28–35.
58. van Blitterswijk M, Mullen B, Wojtas A, Heckman MG, Diehl NN, Baker MC, DeJesus-Hernandez M, Brown PH, Murray ME, Hsiung GY, Stewart H, Karydas AM, Finger E, et al. Genetic modifiers in carriers of repeat expansions in the C9ORF72 gene. Mol Neurodegener. 2014; 9:38.
59. Villa C, Ghezzi L, Fenoglio C, Clerici F, Marcone A, Benussi L, Ghidoni R, Gallone S, Serpente M, Cantoni C, Ridolfi E, Bonsi R, Cerami C, et al. Genetics and expression analysis of the specificity protein 4 gene (SP4) in patients with Alzheimer’s disease and frontotemporal lobar degeneration. J Alzheimers Dis. 2012; 31:537–542.
60. Zintl M, Petkov M, Schmitz G, Hajak G, Klunemann HH. [Frontotemporal dementia in association with a family history of dementia and ApoE polymorphism]. [Article in German]. Nervenarzt. 2010; 81:75–78.
61. Shi Z, Liu S, Xiang L, Wang Y, Liu M, Liu S, Han T, Zhou Y, Wang J, Cai L, Gao S, Ji Y. Frontotemporal dementia-related gene mutations in clinical dementia patients from a Chinese population. J Hum Genet. 2016.
62. Rogers BS, Lippa CF. A clinical approach to early-onset inheritable dementia. Am J Alzheimers Dis Other Demen. 2012; 27:154–161.
63. Holtzman DM, Bales KR, Tenkova T, Fagan AM, Parsadanian M, Sartorius LJ, Mackey B, Olney J, McKeel D, Wozniak D, Paul SM. Apolipoprotein E isoform-dependent amyloid deposition and neuritic degeneration in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci USA. 2000; 97:2892–2897.
64. Castellano JM, Kim J, Stewart FR, Jiang H, DeMattos RB, Patterson BW, Fagan AM, Morris JC, Mawuenyega KG, Cruchaga C, Goate AM, Bales KR, Paul SM, et al. Human apoE isoforms differentially regulate brain amyloid-beta peptide clearance. Sci Transl Med. 2011; 3:89ra57.
65. Mann DM, McDonagh AM, Pickering-Brown SM, Kowa H, Iwatsubo T. Amyloid beta protein deposition in patients with frontotemporal lobar degeneration: relationship to age and apolipoprotein E genotype. Neurosci Lett. 2001; 304:161–164.
66. Brecht WJ, Harris FM, Chang S, Tesseur I, Yu GQ, Xu Q, Dee Fish J, Wyss-Coray T, Buttini M, Mucke L, Mahley RW, Huang Y. Neuron-specific apolipoprotein e4 proteolysis is associated with increased tau phosphorylation in brains of transgenic mice. J Neurosci. 2004; 24:2527–2534.
67. Mackenzie IR, Neumann M, Bigio EH, Cairns NJ, Alafuzoff I, Kril J, Kovacs GG, Ghetti B, Halliday G, Holm IE, Ince PG, Kamphorst W, Revesz T, et al. Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathol. 2010; 119:1–4.
68. Sorbi S, Hort J, Erkinjuntti T, Fladby T, Gainotti G, Gurvit H, Nacmias B, Pasquier F, Popescu BO, Rektorova I, Religa D, Rusina R, Rossor M, et al. EFNS-ENS Guidelines on the diagnosis and management of disorders associated with dementia. Eur J Neurol. 2012; 19:1159–1179.
69. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6:e1000097.

