INTRODUCTION
Biliary tract cancer (BTC) is a heterogeneous group of diseases that includes intrahepatic and extrahepatic cholangiocarcinoma (CCA) and gallbladder cancer. BTC is a rare malignancy and approximately 10,000 new cases are diagnosed annually in the United States and Europe [1]. Surgical resection is the only curative treatment modality for localized disease; however, most patients have a very poor prognosis, as the 5-year overall survival (OS) rate of advanced BTC is approximately 10% [2]. Although the combination of gemcitabine and cisplatin has been established as the standard first-line chemotherapy based on the success of the ABC-02 trial, its success is limited, with median survival of less than 1 year (11.7 months). This highlights the urgent need to develop novel treatments for BTC [3]. Although several agents targeting the EGFR and VEGFR pathways were evaluated in randomized trials, no agent has resulted in a survival benefit in patients with advanced BTC. However, most previous studies were conducted in an unselected patient population with a limited understanding of the biological characteristics of BTC. To enhance survival outcomes, novel agents targeting molecularly defined subgroups of patients may be the key for success, considering the very heterogeneous clinical features of BTC.
Recently, our understanding of the genetic landscape of BTC has improved with the wide application of next generation sequencing (NGS) [4, 5]. Studies indicate that significant proportions of BTC have potentially targetable genetic alterations; however, they also show that there is no single predominant target due to the large heterogeneity within the genetic landscape of BTC. These studies have suggested that potential therapeutic targets differ according to the tumor locations [5]. In intrahepatic CCA (iCCA), fibroblast growth factor receptor 2 (FGFR2) fusion, IDH1/2, BAP1, and ARID1A alterations have been considered as candidates for targeted therapy, and several trials are evaluating the therapeutic implications [6].
The FGFR pathway is involved in cell development, differentiation, survival, migration, and angiogenesis, and may also affect tumorigenesis [7]. In humans, there are 4 FGFRs, which are typical tyrosine kinase receptors (FGFR1-4) and 18 fibroblast growth factors (FGFs), which are ligands for FGFRs. FGF19 is involved in bile acid synthesis and gall bladder filling, and binds to FGFR4. Klotho-beta (KLB) is a transmembrane protein that acts as a cofactor for increased activation of FGFR4 [8]. There is growing evidence that the FGFR4 pathway may contribute to the development of hepatocellular carcinoma (HCC) [9, 10], and selective FGFR4 inhibitors have shown remarkable anti-tumor activity in HCC xenografts harboring FGF19-overexpression. Based on these results, a clinical trial of selective FGFR4 inhibitors is ongoing for patients with FGF19-amplified or overexpressed HCC [11]. Because iCCA shares many risk factors with HCC, we performed gene expression profiling analysis of iCCA to define the potential relevance of FGFR4 pathway as a therapeutic target.
RESULTS
Clinical outcomes
Clinicopathological characteristics of 46 patients with iCCA are presented in Table 1. Underlying liver cirrhosis was present in 3 patients (7%). Recurrence was observed in 30 patients (65%), and 32 patients (70%) had died by the time of analysis. The median follow-up duration was 25.2 months (range, 3–120 months), the median relapse-free survival time was 11.1 months (95% confidence interval [CI], 7.7–14.5 months), and the median OS was 27.9 months (95% CI, 13.1–42.7 months).
Table 1: Clinicopathological characteristics
Characteristics |
N (%), total n = 46 |
|---|---|
Age, median (range), years |
50 (30–78) |
Sex |
|
Male |
34 (74%) |
Female |
12 (26%) |
T stage |
0.69 |
T1–2 |
25 (55%) |
T3–4 |
21 (45%) |
N stage |
|
N0 or Nx |
21 (67%) |
N1 |
15 (33%) |
Resection margin status |
|
R0 resection |
37 (80%) |
R1 resection |
9 (20%) |
Differentiation |
|
Well or moderately differentiated |
38 (82%) |
Poorly differentiated |
8 (17%) |
Underlying viral hepatitis infection or liver cirrhosis |
5 (11%) |
Adjuvant treatment |
11 (24%) |
Expression of FGFR4-related genes
The expression levels of 4 FGFR4-related genes (FGF19, FGF21, FGFR4, and KLB) were dichotomized according to their median levels for further evaluation (Figure 1). The expression levels of these genes were not associated with clinical characteristics such as age, T stage, N stage, resection margin status, tumor differentiation and adjuvant treatment (p > 0.05); however, chronic hepatitis virus infection was associated with high FGFR4 expression (p = 0.049). In the correlative analysis of the expression of each of the 4 genes, there were significant relationships between the expression of FGF21 and FGFR4 (r = 0.33, p = 0.025), and FGF21 and KLB (r = 0.47, p = 0.001).
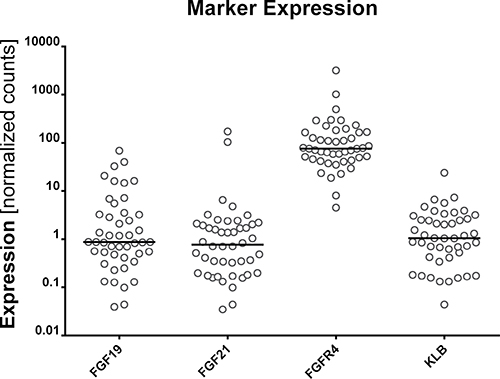
Figure 1: Expression of FGF19, FGF21, FGFR4, and KLB.
Correlative analysis of survival outcomes
Correlative analysis between gene expression and OS was performed for 44 patients after exclusion of 2 patients who had died from postoperative complications (Table 2). In univariate analysis, OS was significantly associated with FGFR4-related gene expression, such as FGF19 (> median vs. < median; unadjusted hazard ratio [HR] 0.48, p = 0.047; Figure 2A), FGF21 (0.47, p = 0.041; Figure 2B), FGFR4 (0.35, p = 0.004; Figure 2C), and KLB (0.44, p = 0.029; Figure 2D). In analyses of the expression of other genes, CYP8B1 (0.43, p = 0.024), ADH1B (0.47, p = 0.045), CDKN1B (0.35, p = 0.005), NCOR1 (0.45, p = 0.033), NF1 (0.36, p = 0.006), SUPV3L1 (0.44, p = 0.026), TIAL1 (0.45, p = 0.034), NROB2 (0.28, p = 0.001), MAP2K3 (0.38, p = 0.009), MYC (0.47, p = 0.040), CYP2E1 (0.25, p < 0.001), and LGR5 (0.45, p = 0.031) were significantly associated with OS.
Table 2: Univariate analysis for overall survival
Variables |
Hazard ratio (95% CI) |
p-value |
|---|---|---|
FGF19 |
0.48 (0.23–1.00) |
0.047 |
FGF21 |
0.47 (0.22–0.99) |
0.041 |
FGFR4 |
0.35 (0.17–0.74) |
0.004 |
KLB |
0.44 (0.20–0.94) |
0.029 |
CYP8B1 |
0.43 (0.20–0.92) |
0.024 |
ADH1B |
0.47 (0.22–1.00) |
0.045 |
CDKN1B |
0.35 (0.16–0.74) |
0.005 |
NCOR1 |
0.45 (0.22–0.96) |
0.033 |
NF1 |
0.36 (0.17–0.76) |
0.006 |
SUPV3L1 |
0.44 (0.21–0.92) |
0.026 |
TIAL1 |
0.45 (0.21–0.96) |
0.034 |
NROB2 |
0.28 (0.13–0.60) |
0.001 |
MAP2K3 |
0.38 (0.18–0.81) |
0.009 |
MYC |
0.47 (0.22–0.98) |
0.040 |
CYP2EA |
0.25 (0.11–0.56) |
< 0.001 |
LG5R |
0.45 (0.22–0.95) |
0.031 |
Abbreviations: CI = confidence interval.
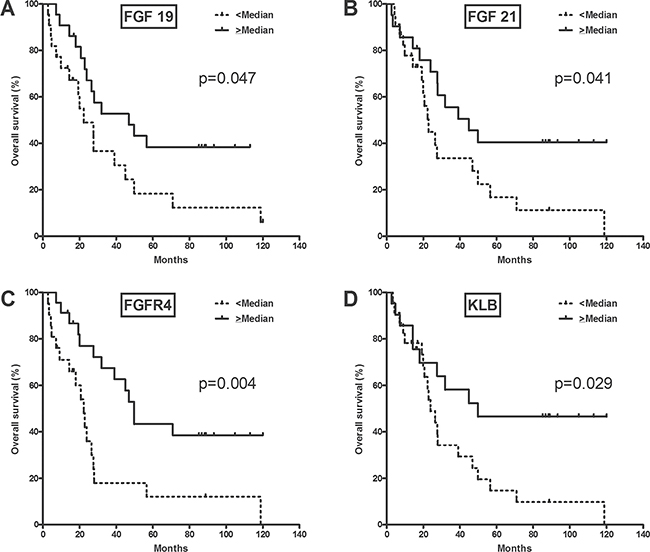
Figure 2: Overall survival according to FGFR4-related gene expression.
Multivariate analysis was performed to validate the relevance of FGFR4-related gene expression (FGF19, FGF21, FGFR4 and KLB) as a prognostic factor (Table 3). In the analyses including potential prognostic factors of iCCA, high expression of FGF19, FGF21, and FGFR4 was associated with better OS (FGF19, adjusted HR = 0.38 [0.18–0.81], p = 0.012; FGF21, adjusted HR = 0.41 [0.19–0.89], p = 0.024; FGFR4, adjusted HR = 0.32 [0.14–0.72], p = 0.006). The expression of KLB showed a marginal association with OS (adjusted HR = 0.47 [0.20–1.01], p = 0.77).
Table 3: Multivariate analysis for overall survival according to the expression of FGFR4-related genes
Variables |
Hazard ratio (95% CI) |
p-value |
|---|---|---|
FGF19 |
||
FGF19 (high vs. low) |
0.38 (0.18–0.81) |
0.012 |
Sex (male vs. female) |
1.36 (0.52–3.55) |
0.53 |
Age (≥ 60 years vs. < 60 years) |
0.76 (0.34–1.69) |
0.50 |
N stage (N1 vs. N0 or Nx) |
1.44 (0.68–3.01) |
0.35 |
Differentiation (poor vs. well or moderate) |
0.24 (0.07–0.86) |
0.029 |
FGF21 |
||
FGF21 (high vs. low) |
0.41 (0.19–0.89) |
0.024 |
Sex (male vs. female) |
1.16 (0.44–3.03) |
0.76 |
Age (≥ 60 years vs. < 60 years) |
0.73 (0.33–1.60) |
0.43 |
N stage (N1 vs. N0 or Nx) |
1.84 (0.85–3.99) |
0.13 |
Differentiation (poor vs. well or moderate) |
0.30 (0.08–1.08) |
0.065 |
FGFR4 |
||
FGFR4 (high vs. low) |
0.32 (0.14–0.72) |
0.006 |
Sex (male vs. female) |
1.12 (0.41–3.01) |
0.83 |
Age (≥ 60 years vs. < 60 years) |
0.95 (0.44–2.16) |
0.95 |
N stage (N1 vs. N0 or Nx) |
1.90 (0.87–4.16) |
0.11 |
Differentiation (poor vs. well or moderate) |
0.30 (0.08–1.10) |
0.069 |
KLB |
||
KLB (high vs. low) |
0.47 (0.20–1.01) |
0.077 |
Sex (male vs. female) |
1.11 (0.42–2.94) |
0.84 |
Age (≥ 60 years vs. < 60 years) |
0.81 (0.37–1.80) |
0.61 |
N stage (N1 vs. N0 or Nx) |
1.83 (0.83–4.05) |
0.14 |
Differentiation (poor vs. well or moderate) |
0.40 (0.11–1.46) |
0.17 |
Abbreviations: CI = confidence interval.
Validation using the the cancer genome atlas (TCGA) dataset
The TCGA iCCA dataset includes a total of 36 patients. Overexpression of FGF19, KLB, FGF21, and FGFR4 was noted in 6 (17%), 4 (11%), 2 (6%), and 2 (6%) patients, respectively (Figure 3A). Patients who had mRNA overexpression of at least one of FGF19, FGF21, FGFR4 and KLB showed significantly better disease-free survival compared to those without any overexpression in all these genes (p = 0.0137, Figure 3B).
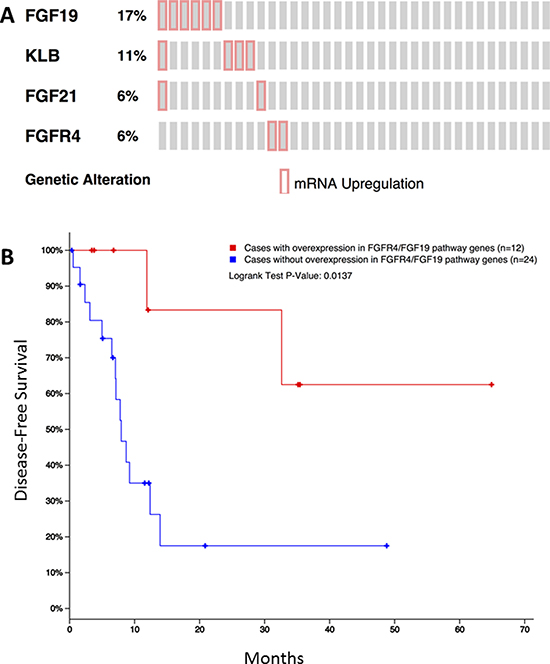
Figure 3: Overexpression of FGF19, KLB, FGF21, and FGFR4 in the public TCGA dataset for iCCA (A) and its impact on disease-free survival (B).
DISCUSSION
Our results show that gene aberrations in the FGFR4 pathway may be a distinct molecular phenotype of CCA, and the prognosis of patients with iCCA may be stratified according to mRNA expression of FGFR4-related genes.
Primary activating FGFR aberrations are observed in a variety of cancers, and have been recognized as novel targets for cancer therapy. A previous study based on an NGS assay of 4,853 tumors showed that FGFR aberrations were found in 7.1% of cancers, with the majority being gene amplification (66%), followed by mutation (26%), and rearrangement (8%) [12]. In this study, FGFR4 was the least affected among the FGFRs, as the frequency of FGFR4 aberrations was 0.5% across the whole study population. Gene amplification was the most common type of FGFR4 aberration (78%).
Previous genomic sequencing studies have revealed that FGFR gene aberrations are observed in 11–50% of iCCA [5, 13–15]; in contrast, these aberrations are rarely detected in extrahepatic CCA or gallbladder cancer. Although FGFR1-3 gene rearrangement is well known as the genetic aberration in iCCA, the role of the FGFR4 signaling pathway has not been well elucidated in iCCA. A recent Japanese study showed that FGF19 gene amplification is detected in 3% of the overall BTC patient population (260 cases, including 145 iCCA), and it was more frequently observed in iCCA compared with extrahepatic CCA and gallbladder cancer [5]. Another Western study which included 377 BTC patients also showed consistent results as the gene aberrations in FGF19/FGFR4 were observed in 13 patients (3.4%), which consisted of 14% of all detected FGFR pathway gene alterations (n = 95) [16].
In our study, when mRNA expression of FGFR4-related genes was dichotomized according to the median expression level, patients with high expression showed better prognosis after surgical resection than those with low expression. This finding was consistent even after adjustment for clinical prognostic factors such as lymph node metastasis, age, and sex. Our results could suggest that the FGFR4 signaling pathway might be the distinct molecular subgroup that could stratify iCCA patients based on their prognosis; however, the reasons for the association between this genetic aberration and a better prognosis is unexplained in this study. Despite this, our results from the analysis using the public TCGA dataset also suggest that mRNA overexpression of FGFR4-related genes may have prognostic implications by indicating better survival outcomes. Our findings are also consistent with the recent study which showed that the BTC patients with FGFR pathway genetic aberrations were significantly associated with improved OS [15]. However, conflicting data exist in terms of the prognostic implication of FGFR4 overexpression in BTC patients. Previous study evaluated the prognostic impact of FGFR4 overexpression in 83 iCCA patients using immunohistochemistry and this study showed that overexpression of FGFR4 was significantly associated with poor prognosis [17]. In addition, FGF19 overexpression correlated with poor survival outcomes in patients with hepatocellular carcinoma [18]. At this point, however, we could not conclude on the reason for these conflicting results on the prognostic implication of FGFR4: these might be related with different methodology (immunohistochemistry vs mRNA expression using NanoString platform) or cancer types (HCC vs iCCA). Further in vitro/in vivo studies and biomarker validation analysis in large patient population are needed to define the biological differences in iCCA according to the expression of FGFR4-related genes.
Until recently, the development of targeted agents against iCCA has been mostly focused on FGFR1-3 gene rearrangement or IDH1-2 mutations. Our study revealed that FGFR4 may also be a valuable target for the management of patients with iCCA. FGFR inhibitors have been widely investigated in a variety of cancer types using non-selective or selective FGFR-tyrosine kinase inhibitors (TKIs), monoclonal antibodies, and FGF-ligand traps [19]. Therefore, it is necessary to explore the clinical relevance of these FGFR inhibitors in patients with iCCA harboring FGFR4-related gene aberrations to determine whether they effectively inhibit the FGFR4 signaling pathway in vitro or in vivo. For successful clinical investigation and future development of FGFR4-targeted drugs, however, the FGFR4 pathway-aberrant molecular phenotype should be defined. It is still unclear which markers (DNA, mRNA, or protein) and methodologies are optimal to define the patient population for which FGFR inhibitors are effective [20]. Further studies are needed for this subject.
Several caveats exist in our study. Although we performed extensive mRNA expression analysis of the FGFR pathway, integrative bioinformatics analysis to define the molecular signature grouping could not be performed due to the small number of patients. All patients included in this study had undergone surgical resection. Considering that the clinical behavior of tumors harboring specific genetic aberrations may be different between localized resectable and metastatic disease, our data on prognosis may have some limitations for indicating the impact of high FGFR4 expression in patients with metastatic iCCA. Considering multiple hypothesis generating compaisons were inevitably done because of the small sample size, there is a possibility that the strength of correlation seen in this study might be overemphasized.
In conclusion, mRNA expression of FGFR4-related genes may define a distinctive molecular phenotype of iCCA. Further validation in the preclinical and clinical settings is needed to determine prevalence and prognostic impact.
MATERIALS AND METHODS
Patients
We obtained formalin-fixed paraffin-embedded (FFPE) tumor tissues from 46 patients with histologically confirmed iCCA who underwent curative surgery in Asan Medical Center, Seoul, Korea between July 2005 and March 2011. Tissue samples were obtained from primary tumor (n = 45, 98%) and recurrent tumor (n = 1, 2%). All procedures were conducted according to the guidelines from the Declaration of Helsinki. Medical records of all included patients were retrospectively reviewed for clinical characteristics and outcomes. The Institutional Review Board at Asan Medical Center approved the protocol.
NanoString analysis
Total RNA was extracted from 5 to 10 sections of 4-μm thick FFPE sections. Non-tumor elements were removed using manual microdissection guided by H&E-stained slides before transferring to the extraction tube. Total RNA was then extracted using the High Pure RNA paraffin kit (Roche), according to the manufacturer’s protocol. A Qubit 3.0 Fluorometer (Thermo Fisher) was used to quantify concentrations of extracted RNA. RNA fragment size and concentration was assessed by an Agilent BioAnalyzer 2100 using the RNA 6000 Pico kit according to manufacturer’s protocol. Fragments above 50 bp were integrated to calculate an adjusted RNA concentration. 100 ng of fragment length adjusted RNA was used in NanoString hybridizations.
A NanoString panel was designed to assess 98 genes, including 10 FGF pathway genes (FGFR1-4, KLB, FGF3, 4, 19, 21, and 23), 19 response marker genes (AFP, ABCB11, ACACB, ACOX2, AQP8, CYP7A1, CYP7B1, CYP8B1, CYP17A1, CYP27A1, EGR1, HMGCR, IGFBP2, LEPR, NR0B2, NRIP1, SCD, SQLE, and FRS2), 31 genes relevant to the oncogenic signaling of HCC and CCA (AKT1, ARID1A, BAD, BAX, BCL2L1, BIRC5, BRD7, CASP9, CDK1, CDKN1A, CDKN1B, CTNNB1, DAXX, GLUL, HGF, LGR5, MIR21, NCOR1, NF1, TGFB1, TSC1, TSC2, ARID2, CDKN3, AURKB, CCNB1, CYP2E1, ADH1B, HGFAC, APOF, and FCN2), 18 copy number variation matched genes (AXIN1, BICC1, CCND1, CDKN2A, ELL, ERBB2, MAP2K3, MET, MYC, ORAOV1, PTEN, RB1, RECQL4, SAV1, TERT, TP53, TRIM45, and VEGFA), and 20 control genes (ACTB, BRAP, CNOT2, COX15, CTCF, EIF2B1, FAM149B1, FAM175B, FBXO18, GAPDH, NRF1, SDHAF2, SF1, SUPT7L, SUPV3L1, TIAL1, VTI1B, WDR33, YY1 and ZNF143). The details and brief introduction of the analyzed genes are presented in Supplementary Table 1. Hybridization of probes and total RNA was performed according to the manufacturer’s protocol. A NanoString nCounter Digital Analyzer (NanoString Technologies, Seattle, Washington, USA) was used to count the digital barcodes that indicate the number of transcripts. The raw expression data were normalized using nSolver Analysis software. A normalization factor was calculated by obtaining the geometric mean of the positive controls used for each sample and applying it to the raw counts of the nCounter output data to eliminate any variability that was unrelated to the samples. The resulting data were normalized again using the geometric mean of the housekeeping genes. Normalized data were log2 transformed for further analyses.
Statistical analysis
Gene expression levels were dichotomized according to the median level (< median vs. > median). OS was defined as the time from surgery to death from any cause. We used Kaplan-Meier curves to estimate OS, and compared groups using the log-rank test. Student t-test or Fisher’s exact test were used to correlate gene expression levels with patient characteristics. Multivariate analyses were conducted using Cox proportional hazards regression model. All results were considered statistically significant if a p value of < 0.05 was obtained. SPSS version 21.0 (IBM Co., Armonk, NY) was used for all statistical analyses. Because small sample size may result in overfitting problem in the multivariate model, all potentially related genes and clinical factors in the univariate analyses could not be included in the multivariate analysis. Therefore, we included each FGFR4-related gene (FGF19, FGF21, FGFR4 and KLB) and key clinical prognostic factors (sex, age, lymph node metastasis, and differentiation of tumor) in the multivariate analyses to adjust the impact of potential confounding factors.
Analysis using the iCCA TCGA dataset
To further evaluate the potential implication of the FGFR4 pathway in iCCA, we analyzed the TCGA gene expression data in the public cBioPortal. Normalized mRNA expression data for iCCA in TCGA was obtained from the portal (http://www.cbioportal.org). The frequency of overexpression (z-score > 2.0) in FGFR4 pathway-related genes, including FGFR4, FGF19, FGF21, and KLB, was estimated. Survival outcomes were compared according to the overexpression of FGFR4, FGF19, FGF21, and KLB. All detailed information from the iCCA dataset was available in the public cBioPortal (Cholangiocarcinoma, TCGA, provisional).
Abbreviations
BTC = Biliary tract cancer, iCCA = cholangiocarcinoma, EGFR = epithelial growth factor receptor, VEGFR = vascular endothelial growth factor receptor, FGFR = fibroblast growth factor receptor, HCC = hepatocellular carcinoma, OS = overall survival, CI = confidence interval, KLB = Klotho-beta, TCGA = The Cancer Genome Atlas, TKI = tyrosine kinase inhibitor
Authors’ contributions
Dr. Kyu-pyo Kim had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Changhoon Yoo, Kyu-pyo Kim, Seung-Mo Hong, Klaus Hoeflich, Oleg Schmidt-Kittler, Stephen Miller, Eun Kyung Choi.
Acquisition of data: Changhoon Yoo, Jihoon Kang, Deokhoon Kim, Kyu-pyo Kim, Baek-Yeol Ryoo, Seung-Mo Hong, Jung Jin Hwang, Seong-Yun Jeong, Shin Hwang, Ki-Hun Kim, Young-Joo Lee, Klaus Hoeflich, Oleg Schmidt-Kittler, Stephen Miller, Eun Kyung Choi.
Analysis and interpretation of data: Changhoon Yoo, Jihoon Kang, Deokhoon Kim, Kyu-pyo Kim, Baek-Yeol Ryoo, Seung-Mo Hong, Jung Jin Hwang, Seong-Yun Jeong, Shin Hwang, Ki-Hun Kim, Young-Joo Lee, Klaus Hoeflich, Oleg Schmidt-Kittler, Stephen Miller, Eun Kyung Choi.
Drafting of the manuscript: Changhoon Yoo, Kyu-pyo Kim, Seung-Mo Hong, Klaus Hoeflich, Oleg Schmidt-Kittler, Stephen Miller.
Critical revision of the manuscript for important intellectual content: Changhoon Yoo, Jihoon Kang, Deokhoon Kim, Kyu-pyo Kim, Baek-Yeol Ryoo, Seung-Mo Hong, Jung Jin Hwang, Seong-Yun Jeong, Shin Hwang, Ki-Hun Kim, Young-Joo Lee, Klaus Hoeflich, Oleg Schmidt-Kittler, Stephen Miller, Eun Kyung Choi.
Statistical analysis: Changhoon Yoo, Deokhoon Kim, Kyu-pyo Kim.
ACKNOWLEDGMENTS AND FUNDING
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number : HI06C0868, HI15C0972).
CONFLICTS OF INTEREST
The authors have no conflicts of interest to disclose.
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7–30.
2. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th Edition of the AJCC Cancer Staging Manual and the Future of TNM. Ann Surg Oncol. 2010; 17:1471–1474.
3. Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, Madhusudan S, Iveson T, Hughes S, Pereira SP, Roughton M, Bridgewater J. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med. 2010; 362:1273–1281.
4. Lee H, Wang K, Johnson A, Jones DM, Ali SM, Elvin JA, Yelensky R, Lipson D, Miller VA, Stephens PJ, Javle M, Ross JS. Comprehensive genomic profiling of extrahepatic cholangiocarcinoma reveals a long tail of therapeutic targets. J Clin Pathol. 2016; 69:403–408.
5. Nakamura H, Arai Y, Totoki Y, Shirota T, Elzawahry A, Kato M, Hama N, Hosoda F, Urushidate T, Ohashi S, Hiroka N, Ojima H, Shimada K, et al. Genomic spectra of biliary tract cancer. Nat Genet. 2015; 47:1–11.
6. Kelley RK, Bardeesy N. Biliary Tract Cancers: Finding Better Ways to Lump and Split. J Clin Oncol. 2015; 33:2588–90.
7. Turner N, Grose R. Fibroblast growth factor signalling: from development to cancer. Nat Rev Cancer. 2010; 10:116–129.
8. Goetz R, Beenken A, Ibrahimi OA, Kalinina J, Olsen SK, Eliseenkova AV, Xu C, Neubert TA, Zhang F, Linhardt RJ, Yu X, White KE, Inagaki T, et al. Molecular Insights into the Klotho-Dependent, Endocrine Mode of Action of Fibroblast Growth Factor 19 Subfamily Members. Mol Cell Biol. 2007; 27:3417–3428.
9. Ahn SM, Jang SJ, Shim JH, Kim D, Hong SM, Sung CO, Baek D, Haq F, Ansari AA, Lee SY, Chun SM, Choi S, Choi HJ, et al. Genomic portrait of resectable hepatocellular carcinomas: implications of RB1 and FGF19 aberrations for patient stratification. Hepatology. 2014; 60:1972–82.
10. Ho HK, Pok S, Streit S, Ruhe JE, Hart S, Lim KS, Loo HL, Aung MO, Lim SG, Ullrich A. Fibroblast growth factor receptor 4 regulates proliferation, anti-apoptosis and alpha-fetoprotein secretion during hepatocellular carcinoma progression and represents a potential target for therapeutic intervention. J Hepatol. 2009; 50:118–127.
11. Hagel M, Miduturu C, Sheets M, Rubin N, Weng W, Stransky N, Bifulco N, Kim JL, Hodous B, Brooijmans N, Shutes A, Winter C, Lengauer C, et al. First Selective Small Molecule Inhibitor of FGFR4 for the Treatment of Hepatocellular Carcinomas with an Activated FGFR4 Signaling Pathway. Cancer Discov. 2015; 5:424–437.
12. Helsten T, Elkin S, Arthur E, Tomson BN, Carter J, Kurzrock R. The FGFR Landscape in Cancer: Analysis of 4,853 Tumors by Next-Generation Sequencing. Clin Cancer Res. 2016; 22:259–267.
13. Ross JS, Wang K, Gay L, Al-Rohil R, Rand JV, Jones DM, Lee HJ, Sheehan CE, Otto GA, Palmer G, Yelensky R, Lipson D, Morosini D, et al. New Routes to Targeted Therapy of Intrahepatic Cholangiocarcinomas Revealed by Next-Generation Sequencing. Oncologist. 2014; 19:235–242.
14. Arai Y, Totoki Y, Hosoda F, Shirota T, Hama N, Nakamura H, Ojima H, Furuta K, Shimada K, Okusaka T, Kosuge T, Shibata T. Fibroblast growth factor receptor 2 tyrosine kinase fusions define a unique molecular subtype of cholangiocarcinoma. Hepatology. 2014; 59:1427–1434.
15. Javle M, Bekaii-Saab T, Jain A, Wang Y, Kelley RK, Wang K, Kang HC, Catenacci D, Ali S, Krishnan S, Ahn D, Bocobo AG, Zuo M, et al. Biliary cancer: Utility of next-generation sequencing for clinical management. Cancer. 2016; 122:3838–3847.
16. Jain A, Shroff RT, Kelley RK, Kaseb AO, Wang Y, Abdel-Wahab R, Ahn DH, Bocobo AG, Meric-Bernstam F, Isaac R, Bekaii-Saab TS, Javle M. FGFR pathway genetic aberrations in cholangiocarcinoma: Demographics and experience with targeted therapy. J Clin Oncol. 2016; 34:109.
17. Xu YF, Yang XQ, Lu XF, Guo S, Liu Y, Iqbal M, Ning SL, Yang H, Suo N, Chen YX. Fibroblast growth factor receptor 4 promotes progression and correlates to poor prognosis in cholangiocarcinoma. Biochem Biophys Res Commun. 2014; 446:54–60.
18. Miura S, Mitsuhashi N, Shimizu H, Kimura F, Yoshidome H, Otsuka M, Kato A, Shida T, Okamura D, Miyazaki M. Fibroblast growth factor 19 expression correlates with tumor progression and poorer prognosis of hepatocellular carcinoma. BMC Cancer. 2012; 12:56.
19. Dieci MV, Arnedos M, Andre F, Soria JC. Fibroblast Growth Factor Receptor Inhibitors as a Cancer Treatment: From a Biologic Rationale to Medical Perspectives. Cancer Discov. 2013; 3:264–279.
20. Seo S, Park SJ, Ryu MH, Park SR, Ryoo BY, Park YS, Na YS, Lee CW, Lee JK, Kang YK. Prognostic impact of fibroblast growth factor receptor 2 gene amplification in patients receiving fluoropyrimidine and platinum chemotherapy for metastatic and locally advanced unresectable gastric cancers. Oncotarget. 2017; 8:33844–54. doi: 10.18632/oncotarget.12953.

