INTRODUCTION
Lung cancer has been the most common cause of cancer-related death worldwide [1] and appears to remain a major health problem in the near future. Non-small cell lung cancer (NSCLC) constitutes 82% of the total lung cancer cases [2] with a 5-year survival rate of approximately 15% in developed countries [3]. Further understanding of the NSCLC’s molecular mechanism has led to the development of target therapies, which have improved overall survival, and the median OS has reached 3–5 years [4–11]. The Notch signaling pathway is one of many signaling pathways that have been shown to be important in NSCLC progression. The Notch protein has been shown to promote faster tissue growth and to up-regulate the self-renewal of the undifferentiated state of stem cells [12–20]. Notch activation has been shown to be stimulated by the differential O-linked glycosylation of its extracellular domain (NECD) [21], whereby O-linked glucose is added to a subset of epidermal growth factor-like (EGF) repeats in the NECD domain by protein O-glucosyltransferase 1 (POGLUT1) [22–24]. The chromosome 3 open reading frame 21 (C3orf21) gene encodes the Xyloside Xylosyltransferase 1 (XXYLT1) enzyme, which is involved in prolonging the O-linked xylose-glucose by activating the transfer of the second xylose into the notch extracellular protein EGF repetitive region and changing the Notch extracellular domain [25]. Our previous study showed that in the rs2131877 locus of the C3orf21 gene, the frequency of the T/T homozygous genotype was higher in the peripheral blood of a healthy population than in adenocarcinoma patients (40.5% vs 29.7%) [26]. Thus, we undertook this study to determine if there is a link between the changes in the C3orf21 gene mRNA expression and NSCLC development and its potential as a therapeutic target or a diagnostic molecular marker. We attempted to detect the mutation status of the rs2131877 locus in the C30rf21 gene by analyzing lung and adenocarcinoma tissues, respectively. We also attempted to identify the role of the C30rf21 gene in NSCLC using the high or low expressing lung cancer cell line MSTO-211H to detect its effects on cell proliferation, apoptosis and migration ability.
RESULTS
Correlation between patients’ clinical characteristics and C3orf21 rs2131877 genotype distribution
The 15 patients (7 males and 8 females) with lung adenocarcinoma were assessed for the genotype distribution of the C3orf21 rs2131877 locus. The analysis of 30 specimens (15 normal lung tissues and 15 tumor tissues) revealed that the frequencies of the rs2131877 T/C+C/C genotype were 20.0% and 33.3% in normal lung and adenocarcinoma tissues, respectively (Table 1).
Table 1: Clinical characteristics of patients
Patient |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
12 |
13 |
14 |
15 |
Sex |
M |
M |
F |
M |
M |
M |
F |
M |
F |
F |
F |
F |
F |
F |
M |
Age (years) |
52 |
55 |
54 |
63 |
58 |
74 |
63 |
58 |
40 |
65 |
67 |
41 |
70 |
54 |
68 |
Smoking |
yes |
yes |
No |
yes |
yes |
yes |
No |
No |
No |
No |
No |
No |
No |
No |
yes |
Lung |
TT |
TT |
TT |
CC |
TT |
TC |
CC |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
Cancer |
TC |
TC |
CC |
CC |
TC |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
TT |
Abbreviations: M, male; F, female; Lung, lung tissue genotype; Cancer, cancer tissue genotype.
The clinical characterization indicated that the mean ages of the patients with the rs2131877 T/C+C/C (mutation) and rs2131877 T/T (wild type) genotypes were 56.40 and 60.00 years, respectively (Table 2). Further comparison between these genotypes indicated that 4 male and 1 female patient displayed the rs2131877 T/C+C/C genotype, while 3 male and 7 female patients had the rs2131877 T/T genotype; this difference was not statistically significant (p = 0.067) (Table 2). Additionally, the patients who smoke had a significantly increased rs2131877 T/C+C/C genotype compared to the rs2131877 T/T genotype (p = 0.025) (Table 2). Similarly, the values of CEA, CA125, CA199 and SCC-Ag tumor markers were also higher in tumors with the rs2131877 T/C+C/C genotype; however, the differences were not significant (Table 2). Notably, the values of the CYF211 and NSE tumor markers (4.18 ± 0.42 ng/ml and 25.46 ± 11.99 ng/ml, respectively) were significantly higher in the tumor patients with the rs2131877 T/C+C/C genotype (p = 0.014 and p = 0.031, respectively) (Table 2).
Table 2: Differences between C3orf21 gene mutation and wildtype genotypes in tumor tissues
Tumor Mutation patients (genotype: CC + CT) |
Tumor Wildtype patients (genotype: TT) |
P value |
|||
|---|---|---|---|---|---|
Ages |
56.40 ± 4.28 |
60.00 ± 11.76 |
0.525 |
||
Gender |
Male |
Female |
Male |
Female |
0.067 |
4 |
1 |
3 |
7 |
||
Smoking |
Yes |
No |
Yes |
No |
0.025 |
4 |
1 |
2 |
8 |
||
CEA (ng/ml) |
33.29 ± 60.65 |
8.91 ± 7.89 |
0.217 |
||
CA125 (U/ml) |
59.06 ± 73.81 |
22.34 ± 20.50 |
0.155 |
||
CA199 (U/ml) |
105.34 ± 204.17 |
24.79 ± 16.74 |
0.220 |
||
SCC-Ag (ng/ml) |
1.64 ± 1.74 |
0.86 ± 0.45 |
0.192 |
||
CYF211 (ng/ml) |
4.18 ± 0.42 |
1.81 ± 1.81 |
0.014 |
||
NSE (ng/ml) |
25.46 ± 11.99 |
14.52 ± 5.86 |
0.031 |
||
We also performed a retrospective analysis of samples used in our previous study [26]. This analysis indicated that the mean age of the patients with the rs2131877 T/C+C/C (mutation) and rs2131877 T/T (wild type) genotypes were not significantly different (Tables 3, 4). Moreover, at the gender level, there were no significant differences between these genotypes (Tables 3, 4). Additionally, the CEA, CA125, CA199, SCC-Ag and CYF211 tumor markers did not show significant differences between patients with the rs2131877 T/C+C/C and rs2131877 T/T genotypes (Table 3). However, the values of the NSE tumor marker (15.46 ± 8.06) were significantly higher in the patients with the rs2131877 T/C+C/C genotype (p = 0.040) (Table 4).
Table 3: Differences between C3orf21 gene mutant and wildtype genotypes in patient’s blood (n = 103)
Patients with mutant genotype (genotype: CC + CT) |
Patients with wildtype genotype (genotype: TT) |
P value |
|||
|---|---|---|---|---|---|
Ages |
57.01 ± 9.42 |
56.45 ± 6.90 |
0.765 |
||
Gender |
Male |
Female |
Male |
Female |
0.699 |
47 |
25 |
19 |
12 |
||
CEA (ng/ml) |
108.91 ± 374.01 |
101.17 ± 350.05 |
0.922 |
||
CA125 (U/ml) |
197.41 ± 424.99 |
102.80 ± 303.15 |
0.265 |
||
CA199 (U/ml) |
233.45 ± 716.61 |
440.13 ± 2149.34 |
0.467 |
||
Table 4: Differences between C3orf21 gene mutant and wildtype genotypes in patient’s blood (n = 99)
Patients with mutant genotype (genotype: CC + CT) |
Patients with wildtype genotype (genotype: TT) |
P value |
|||
|---|---|---|---|---|---|
Ages |
57.16 ± 7.68 |
57.04±5.67 |
0.952 |
||
Gender |
Male |
Female |
Male |
Female |
0.835 |
45 |
24 |
17 |
10 |
||
SCC-Ag (ng/ml) |
1.69 ± 3.97 |
1.15 ± 0.91 |
0.488 |
||
CYF211 (ng/ml) |
9.84 ± 16.35 |
11.26 ± 19.11 |
0.718 |
||
NSE (ng/ml) |
15.46 ± 8.06 |
12.03 ± 4.43 |
0.040 |
||
Assessment of C3orf21 gene manipulation effects on cell proliferation, apoptosis and migration of a human lung cancer cell line
To understand the effects of C3orf21 gene manipulation in NSCLC, we either ablated its expression with siRNA or overexpressed it in MSTO-211H, a human lung cancer cell line. To overexpress C3orf21, we successfully cloned C3orf21 plasmid, and its length was consistent with the expected size (Figure 1, panel 1A). All 3 siRNAs appear to significantly ablate the expression of the C3orf21 gene at the mRNA level, as determined using fluorescence quantitative PCR (Figure 1, panel 1B). Similarly, the overexpression of the C3orf21 gene using a pcDNA3.1-based vector in these cells also significantly elevated its expression compared to control vector after 48 hrs of transfection (Figure 1, panel 1B, final two bars). Additionally, the ablation and overexpression effects of the C3orf21 gene were also validated at the protein level using western blotting (panel 1C) and quantified after normalization with the GAPDH protein, as shown in panel 1D. Thus, overall, these results demonstrate that C3orf21 gene manipulations were efficient in MSTO-211H cells.
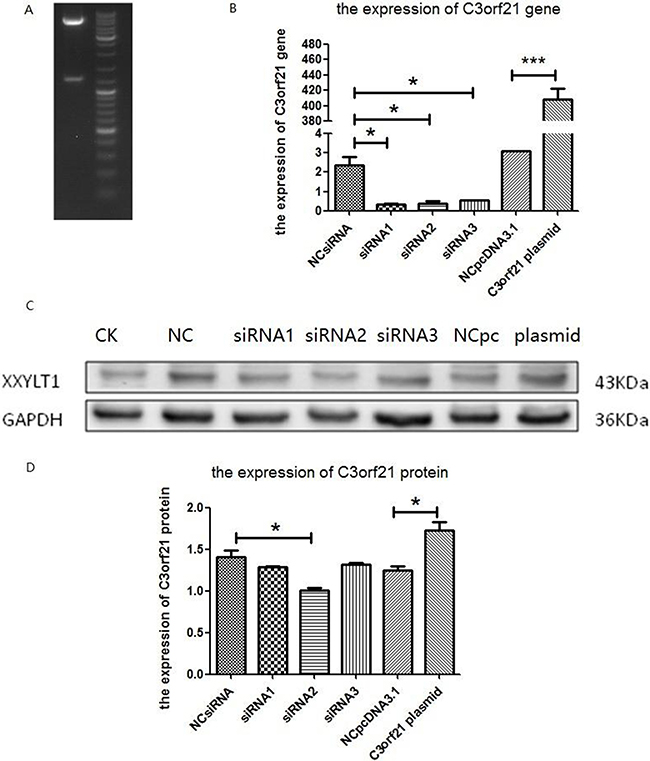
Figure 1: Confirmation of C3orf21 gene expression manipulation. (Panel A) C3orf21 plasmid digested with HindIII and BamH1 restriction enzymes. (Panel B) the fluorescence quantitative PCR-based expression of the C3orf21 gene in MSTO-211H cells after its ablation with three siRNAs or overexpression. NC siRNA indicated that MSTO-211H cells were transfected with control siRNA; siRNA1, siRNA2 and siRNA3 indicated that MSTO-211H cells were transfected with three C3orf21 gene-specific siRNAs; NCpcDNA3.1 represented cells transfected with the control vector; and C3orf21 plasmid represented MSTO-211H cells overexpressing C3orf21 gene cDNA. * indicates a P value of < 0.05, while *** represents a P value of < 0.001. (Panel C) indicates the C3orf21 gene expression by western blotting. (Panel D) represents the normalized quantification of C3orf21 protein expression in different cell types. * indicates a P value of < 0.05.
Next, we assessed the effect of C3orf21 gene manipulations on MSTO-211H cell proliferation by measuring CFSE staining through flow cytometry. Our data revealed that siRNA ablation of the C3orf21 gene substantially promoted cell proliferation, while elevated C3orf21 gene expression did not show any significant reduction, as observed in Figure 2.
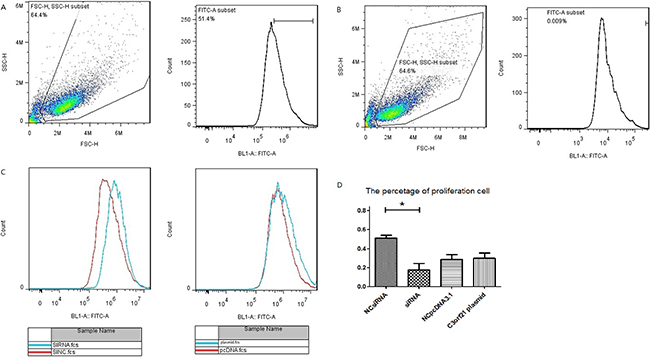
Figure 2: C3orf21 gene ablation promotes cell proliferation. (Panel A and B) represent the strategy to assess cell proliferation by including MSTO-211H cell (A) and negative control (B). (Panel C) shows that C3orf21 gene ablation leads to faster MSTO-211H cell proliferation than normal cells, p < 0.05, while its overexpression has a very minimal effect. (Panel D) represents the quantification of the percent cell proliferation under different conditions. * indicates a P value of < 0.05.
We also analyzed the effect of C3orf21 gene manipulation on cell apoptosis using Annexin V and PI staining analysis via flow cytometry. Our results indicated that C3orf21 gene overexpression in MSTO-211H cells slightly accelerated the trend of lung cancer cell apoptosis; however, this accelerating effect was not significant compared to control vector (Figure 3, panel 3B). However, the siRNA-based ablation of C3orf21 gene expression significantly (p < 0.05) inhibited cell apoptosis observed in Figure 3 (panel 3B, first two bars).
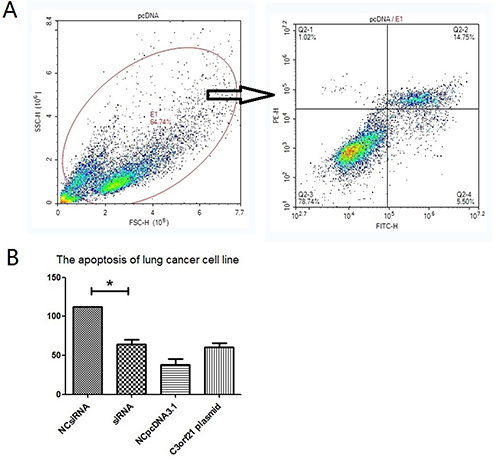
Figure 3: C3orf21 gene ablation inhibits cell apoptosis. (Panel A) represents the strategy to assess percent cell apoptosis by flow cytometry. (Panel B) shows the comparison of percent cell apoptosis after C3orf21 manipulation. * indicates a P value of < 0.05.
We also tested the effect of C3orf21 gene manipulation on the cell migration ability of MSTO-211H cells using RTCA detection technology, which measured cell migration every 15 minutes over a period of 72 hrs. Our results indicated that C3orf21 ablation enhanced cell migration (blue curve), while its overexpression inhibited migration (light blue curve) compared to control cells (red curve), as observed in Figure 4.
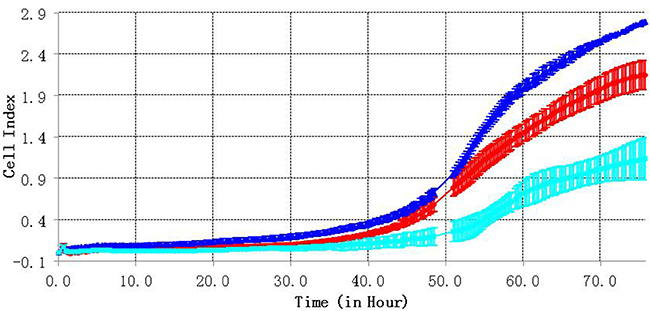
Figure 4: C3orf21 gene ablation accelerates cell migration. The red curve represents the migration of the MSTO-211H control group cells, the blue line represents cells from the C3orf21 ablation group, and the light blue line indicates cells from the C3orf21 overexpression group.
DISCUSSION
Based on our analysis, lung adenocarcinoma tissues displayed a lower frequency of the T/T homozygous genotype (wild-type) in the C3orf21 gene rs 2131877 locus compared with normal lung tissue (66.7% vs 80%). Additionally, the values for tumor markers, including CYF211 and NSE, were higher in patients with the rs 21319877 T/C+C/C genotype in tumor tissue, and NSE was higher in patients with the rs 2131877 T/C+C/C genotype in blood. These observations indicate specific differences between the clinical characteristics of lung adenocarcinoma patients with different genotypes. The high expression levels of the NOTCH1 and NOTCH3 genes have been observed to be significantly associated with poor prognosis in lung adenocarcinoma [27, 28]. Additionally, DLL4 (Delta-like 1, one of five Notch ligands) was observed to be positively associated with poor OS in NSCLC patients [29]. More specifically, patients exhibiting high Notch 1, Notch 3, JAG1 (Jagged, one of five Notch ligands) or DLL4 protein levels had a significantly worse OS rate [30]. In this context, our results also suggested that the C30rf21 rs 2131877 T/C+C/C genotype may act as a marker for higher malignant adenocarcinoma. Moreover, we also observed that patients who smoke had a significantly increased rate of the C30rf21 rs 2131877 T/C+C/C genotype. However, smoking was not substantially associated with adenocarcinoma; this indicated that the C30rf21 rs 2131877 T/C+C/C genotype patients may have higher nicotine addiction. The number of patients was small in our study; thus, the results were difficult to conclude decisively. A large study is required to validate our observation.
Furthermore, the Notch signaling pathway has been demonstrated to up-regulate cell proliferation and control differentiation and apoptotic processes [31]. The reduced activity of UDP-xylose:-xyloside 1,3-xylosyltransferase (XXYLT1), which is encoded by the C3orf21 gene [29], would lead to enhanced Notch signaling [32]. Notch signaling participates in TIC (tumor-initiating cells) fate in NSCLC [33, 34], and Notch deregulation has been reported to be associated with the development of lung cancer [35]. However, Notch signaling inhibitors exhibited potent antitumor activity in the lungs [36]. Our study revealed that the ablation of the C3orf21 gene (XXYLT1) promoted lung cancer MSTO-211H cell proliferation, inhibited apoptosis and accelerated cell migration. However, its overexpression did not significantly alter cell proliferation or apoptosis, and this could be attributed to the fact that C3orf21 (XXYLT1) over-expression in Notch signaling was dependent on the specific tissue context, microenvironment, and crosstalk with other signaling pathways. The Notch 1 protein has also been known to suppress tumor proliferation under normoxia in lung cancer; however, it exhibited the opposing role of tumor promotion under hypoxia conditions [37]. Additional research is required to elucidate this aspect.
MATERIALS AND METHODS
Patient selection and C3orf21 rs2131877 genotype identification
Fifteen patients (seven male and eight female) who were newly diagnosed with lung adenocarcinoma and underwent surgical treatment between March 2008 and November 2009 at Zhejiang Cancer hospital were recruited for this study. All subjects provided informed consent, and the study was approved by the ethics committee of Zhejiang Cancer Hospital. A total of 15 normal lung tissues and 15 cancer tissues were obtained from these patients during surgery and were stored at −80°C for further analysis. The droplet Digital PCR (dd-PCR) method was used to evaluate the genotype of C3orf21 in normal lung tissue and adenocarcinoma specimens. The DNA was isolated from these lung and tumor tissue homogenates, respectively, using the FastPrep-24 sample processing system (MP Biomedical, Santa Ana, California, USA), and the genotyping was performed with the PCR sequencing method using an S100-PCR Amplification Device (Bio-Rad, Samoa, USA). The assay designer’s software version 3.0 (Sequenom) was used to design primers for the polymerase chain reaction and single base extension. The primers for the C3orf21 rs2131877 genotype were:
1st, 3′-CTGGCCTTCAGGACCTAC-5′
2nd, 3′-CTGCCTGTAACCTCAAAGA5′
Finally, the purified PCR products were sequenced using an ABI 3100 DNA sequencer (USA). Other tumor markers, such as carcinoembryonic antigen (CEA), carbohydrate antigen 125 (CA125), cell keratin fragments 211 (CYF211), carbohydrate antigen 199 (CA199), squamous cell antigen (SCC-Ag), and neuron enolization enzymes (NSE), were detected in the peripheral blood before surgery with a Chemiluminescent Microparticle Immunoassay in the Abbott I400 AxSYM system (Abbott, Chicago, USA). All patients were followed up until June 2016. We also performed a retrospective analysis of two hundred NSCLC patients including 145 patients with adenocarcinoma and 55 patients with squamous cell carcinoma from our previous study [26]. All of these patients were non-smokers. Among these, one hundred and three adenocarcinoma patients displayed tumor markers (CEA, CA125, CA199) (Table 3); however, only ninety-nine patients had records for the values of all markers (CEA, CA125, CA199, SCC-Ag, CYF211 and NSE) (Table 4).
Cell culture and C3orf21 overexpression and ablation
The MSTO-211H human lung cancer cell line, which was procured from the cell bank of the Chinese Academy of Sciences Library/Stem Cells, was cultured in a humidified incubator with 5% CO2 at 37°C in six-well plates (1 × 106 cells/ml) containing RPMI-1640 media (Gibco, Gaithersburg, MD, USA) supplemented with 10% fetal bovine serum (Gibco, Gaithersburg, MD, USA). To perform C3orf21 gene ablation, the following three siRNAs sequences were designed by the bioinformatics software Primer Premier 5.0 (Premier Biosoft International, Palo Alto CA, USA):
siRNA1 sense: 5′-GGAACUGCAACACUCCCAU TT-3′ and antisense: 5′-AUGGGAGUGUUGCA GUUCCTT-3′; siRNA2 sense: 5′-GCAUCAGAU CAUGCCCAAATT-3′ and antisense: 5′-UUUGGGCAUG AUCUGAUGCTT-3′; siRNA3 sense: 5′-GCACGAGGUG CUUAACCUUTT-3′ and antisense: 5′-AAGGUUAAG CACCUCGUGCTT-3′; Similarly, to overexpress the C3orf21 (XXYLT1) gene, it was cloned into pcDNA3.1 plasmid using HindIII/BamHI sites with T4 DNA ligase (Figure 1, panel A). The ligation product was further transformed into a competent Escherichia coli strain, and positive clones were selected by ampicillin selection and then sequenced using ABI3730 sequencing analysis (Sangon, Shanghai, China). The siRNAs were transfected into an MSTO-211H cell line with RNAiMAX (Lipofectamine) transfection agent, while the plasmid overexpressing C3orf21 cDNA was transfected using Lipo3000 reagent. The C3orf21 gene ablation or overexpression was verified at the mRNA and protein levels using quantitative PCR and western blotting, respectively.
RNA isolation, reverse transcription, and PCR
The RNA was isolated from MSTO-211H cells either ablated for or overexpressing the C3orf21 gene using an RNAiso Plus (TaKaRa, Dalian, China) kit. To generate cDNA, the isolated RNA (1 μg) was reverse transcribed using an RT transcription kit (TaKaRa, Dalian, China) with oligo (dT18) primer. Next, the quantitative PCR was conducted using a real time qPCR (Bio-Rad, USA) machine with the following specific primers synthesized by Sangon Biotech (Shanghai, China): 5′-CTGGCCTTCAGGACCTAC-3′ and 5′-CTGCCTGTAACCTCAAAGA-3′. The quantitative PCR reaction mix consisted of 20 μl total volumes including 10 μl SYBR® Premix Ex Taq (TaRaKa), 0.8 μl of each primer (10 μM), 0.2 μl cDNA, and 6.4 μl of ddH2O. The following PCR amplification conditions were used: denaturation at 95°C for 30 seconds, then 40 cycles of 95°C for 5 seconds and 60°C for 20 seconds, and a final cycle of 72°C for 1 minute. For all PCR analyses experiments, the GAPDH expression served as a loading control.
Western blotting
Cells were collected and lysed on ice for 30 min in RIPA lysis buffer (cell-land, Hangzhou, China). Protein fractions were collected by centrifugation at 15,000 g at 4°C for 10 min, were subjected to 10% SDS-PAGE and transferred to PVDF membranes (Bio-Rad, USA). After blocking with 5% BSA, the membranes were incubated with specific antibodies overnight at 4°C. The following day, a secondary antibody was added, and the proteins were visualized using an enhanced chemiluminescence detection system as recommended by the manufacturer Bio-Rad Laboratories, Inc.
Cell proliferation analysis
The 1 × 106 cells (MSTO-211H) per ml in a logarithmic growth phase were incubated with 10 μm CFSE staining solution at 37°C for 30 min. These cells were later washed twice with PBS and then cultured in a humidified incubator with 5% CO2 at 37°C in six-well plates. After 72 hr, the cells were collected and analyzed for cell proliferation using flow cytometry detection technology.
Apoptosis analysis
Following incubation for 24 hrs, MSTO-211H cells were incubated with 5 μl of FITC-conjugated Annexin V dye and 10 μl of PI staining reagent in the dark for 5 min at room temperature. These cells were then immediately analyzed using flow cytometry.
Assessing cell migration using real time cell analysis (RTCA)
To assess the cell migration of MSTO-211H cells, we performed a real time cell analysis. First, 165 μl serum containing culture medium was added to the lower chamber of the cell invasion and migration (CIM) plate, while 30 ul of serum-free medium was added to the upper chamber, and the plate was incubated at 37°C and 5% CO2 for 1 hr to balance the baseline. Next, 100 μl of the logarithmic phase cells (concentration 4 × 105 per ml) in serum-free medium were added into the CIM upper chamber, and the cell migration was detected every 15 min. The real-time cell migration data were collected for 72 hrs.
Statistical analysis
The flow cytometry data were analyzed using FlowJo 7.6 software. The results were analyzed using the statistical software GraphPad Prism 5.0 and presented as the means ± standard error for continuous variables. An analysis of variance and the following independent t tests were used for all comparisons. Correlations between two variables were estimated using Spearman’s rank sum test, and a P value of < 0.05 was considered statistically significant.
CONCLUSIONS
In summary, we demonstrated that the C3orf21 gene T/C+C/C genotype at the rs 2131877 locus may act as a susceptibility marker for lung adenocarcinomas with a higher degree of malignance. Importantly, C3orf21 mRNA expression appears to be associated with lung cancer risk, and its ablation promoted lung cancer MSTO-211H cell proliferation, inhibited apoptosis and accelerated cell migration.
Authors’ contributions
Litao Yang, Ying Wang and Yongjun Zhang designed this study and drafted the manuscript. Meiyu Fang and Dehou Deng carried out the collection of patients’ clinical data and performed the statistical analysis. Litao Yang carried out the collection of normal lung and tumor tissue.
ACKNOWLEDGMENTS AND FUNDING
This study was supported by grants from Zhejiang Provincial Science Department Public Technology Application Research Fund (#2016C33056) and Zhejiang Provincial Medical and Health Scientific and Technical Fund (#2017171306).
CONFLICTS OF INTEREST
None.
REFERENCES
1. Siegel R, Naishadh D, Jemal A. Cancer statistics. CA Cancer J Clin. 2013; 63:11–30. doi: 10.3322/caac.21166.
2. Pao W, Chmielecki J. Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer. 2010; 10:760–74. doi: 10.1038/nrc2947.
3. Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc. 2008; 83: 584–94. doi: 10.4065/83.5.584.
4. Jackman DM, Miller VA, Cioffredi LA, Yeap BY, Jnne PA, Riely GJ, Ruiz MG, Giaccone G, Sequist LV, Johnson BE. Impact of epidermal growth factor receptor and KRAS mutations on clinical outcomes in previously untreated non-small cell lung cancer patients: results of an online tumor registry of clinical trials. Clin Cancer Res. 2009; 15: 5267–73. doi: 10.1158/1078-0432.CCR-09-0888.
5. Yang JC, Wu YL, Schuler M, Sebastian M, Popat S, Yamamoto N, Zhou C, Hu CP, O’Byrne K, Feng J, Lu S, Huang Y, Geater SL, et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol. 2015; 16:141–51. doi: 10.1016/S1470-2045(14)71173-8.
6. Jänne PA, Yang JC, Kim DW, Planchard D, Ohe Y, Ramalingam SS, Ahn MJ, Kim SW, Su WC, Horn L, Haggstrom D, Felip E, Kim JH, et al. AZD9291 in EGFR inhibitor-resistant non-small-cell lung cancer. N Engl J Med. 2015; 372:1689–99. doi: 10.1056/NEJMoa1411817.
7. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014; 371:2167–77. doi: 10.1056/NEJMoa1408440.
8. Shaw AT, Kim DW, Mehra R, Tan DS, Felip E, Chow LQ, Camidge DR, Vansteenkiste J, Sharma S, De Pas T, Riely GJ, Solomon BJ, Wolf J, et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med. 2014; 370:1189–97. doi: 10.1056/NEJMoa1311107.
9. Ou SH, Ahn JS, De Petris L, Govindan R, Yang JC, Hughes B, Lena H, Moro-Sibilot D, Bearz A, Ramirez SV, Mekhail T, Spira A, Bordogna W, et al. Alectinib in crizotinib-refractory ALK-rearranged non-small-cell lung cancer: a phase II global study. J Clin Oncol. 2016; 34:661–8. doi: 10.1200/JCO.2015.63.9443.
10. Shaw AT, Gandhi L, Gadgeel S, Riely GJ, Cetnar J, West H, Camidge DR, Socinski MA, Chiappori A, Mekhail T, Chao BH, Borghaei H, Gold KA, et al. Alectinib in ALK-positive, crizotinib-resistant, non-small-cell lung cancer: a single-group, multicentre, phase 2 trial. Lancet Oncol. 2016; 17:234–42. doi: 10.1016/S1470- 2045(15)00488-X.
11. Shaw AT, Friboulet L, Leshchiner I, Gainor JF, Bergqvist S, Brooun A, Burke BJ, Deng YL, Liu W, Dardaei L, Frias RL, Schultz KR, Logan J, et al. Resensitization to crizotinib by the lorlatinib ALK resistance mutation L1198F. N Engl J Med. 2016; 374:54–61. doi: 10.1056/NEJMoa1508887.
12. Axelson H. Notch signaling and cancer:emerging complexit. Sem in Cancer Biol. 2004; 14:3l7–9. doi: 10.1016/j.semcancer.2004.04.010.
13. Hubmann R, Schwarzmeier JD, Shehata M, Hilgarth M, Duechler M, Dettke M, Berger R. Notch2 is involved in the overexpression of CD23 in B-cell chronic lymphocytic leukemia. Blood. 2002; 99:3742–7. doi: 10.1182/blood.V99.10.3742
14. Purow BW, Haque RM, Noel MW, Su Q, Burdick MJ, Lee J, Sundaresan T, Pastorino S, Park JK, Mikolaenko I, Maric D, Eberhart CG, Fine HA. Expression of Notchl and its ligands, Delta-like-1 and Jagged-1, is critical for glioma cell survival and proliferation. Cancer Res. 2005; 65:2353–63. doi: 10.1158/0008-5472.CAN-04-1890.
15. Collins BJ, Kleeberger W, Ball DW. Notch in lung development and lung cancer. Semin Cancer Biol. 2004; 14:357–64. doi: 10.1016/j.semcancer.2004.04.015.
16. Doucas H, Mann CD, Sutton CD, Garcea G, Neal CP, Berry DP, Manson MM. Expression of nuclear Notch3 in pancreatic adenocarcinoma is associated with adverse clinical features, and correlates with the expression of STAT3 and phosphorylated Akt. J Surg Oncol. 2008; 97:63–8. doi: 10.1002/jso.20894.
17. Bellavia D, Campese AF, Checquolo S, Balestri A, Biondi A, Cazzaniga G, Lendahl U, Fehling HJ, Hayday AC, Frati L, von Boehmer H, Gulino A, Screpanti I. Combined expression of pT alpha and Notch3 in T cell leukemia identifies the requirement of preTCR for leukemogenesis. Proc Natl Acad Sci USA. 2002; 99:3788–93. doi: 10.1073/pnas.062050599
18. Liu ZJ, Xiao M, Balint K, Smalley KS, Brafford P, Qiu R, Pinnix CC, Li X, Herlyn M. Notch1 signaling promotes primary melanoma progression by activating mitogen-activated protein kinase/phosphatidylinositol 3-kinase-Akt pathways and upregulating N-cadherin expression. Cancer Res. 2006; 66:4182–90. doi: 10.1158/0008-5472.CAN- 05-3589.
19. McLaughlin KA, Rones MS, Mercola M. Notch regulates cell fate in developing pronephros. Dev Biol. 2000; 227:567–80. doi: 10.1006/dbio.2000.9913.
20. Guilmeau S, Flandez M, Bancroft L, Sellers RS, Tear B, Stanley P, Augenlicht LH. Intestinal deletion of pofut1 in the mouse inactivates notch signaling and causes enterocolitis. Gastroenterology. 2008; 135:849–60. doi: 10.1053/j.gastro.2008.05.050.
21. Takeuchi H, Haltiwanger RS. Significance of glycosylation in Notch signaling. Biochem. Biophys. Res Commun. 2014; 453:235–42. doi: 10.1016/j.bbrc.2014.05.115.
22. Acar M, Jafar-Nejad H, Takeuchi H, Rajan A, Ibrani D, Rana NA, Pan H, Haltiwanger RS, Bellen HJ. Rumi is a CAP10 domain glycosyltransferase that modifies Notch and is required for Notch signaling. Cell. 2008; 132:247–58. doi: 10.1016/j.cell.2007.12.016
23. Fernandez-Valdivia R, Takeuchi H, Samarghandi A, Lopez M, Leonardi J, Haltiwanger RS, Jafar-Nejad H. Regulation of mammalian Notch signaling and embryonic development by the protein O-glucosyltransferase Rumi. Development. 2011; 138:1925–34. doi: 10.1242/dev.060020.
24. Takeuchi H, Fernández-Valdivia RC, Caswell DS, Nita-Lazar A, Rana NA, Garner TP, Weldeghiorghis TK, Macnaughtan MA, Jafar-Nejad H, Haltiwanger RS. Rumi functions as both a protein O-glucosyltransferase and a protein O-xylosyltransferase. Proc Nat Acad Sci USA. 2011; 108:16600–5. doi: 10.1073/pnas.1109696108.
25. Sethi MK, Buettner FF, Krylov VB, Takeuchi H, Nifantiev NE, Haltiwanger RS, Gerardy-Schahn R, Bakker H. Identification of glycosyltransferase 8 family members as xylosyltransferases acting on O-glucosylated notch epidermal growth factor repeats. J Biol Chem. 2010; 285:1582–6. doi: 10.1074/jbc.C109.065409.
26. Zhang Y, Gu C, Shi H, Zhang A, Kong X, Bao W, Deng D, Ren L, Gu D. Association between C3orf21, TP63 polymorphisms and environment and NSCLC in never-smoking Chinese population. Gene. 2012; 497:93–7. doi: 10.1016/j.gene.2012.01.053
27. Donnem T, Andersen S, Al-Shibli K, Al-Saad S, Busund LT, Bremnes RM. Prognostic impact of Notch ligands and receptors in nonsmall cell lung cancer: coexpression of Notch-1 and vascular endothelial growth factor-A predicts poor survival. Cancer. 2010; 116:5676–85. doi: 10.1002/cncr.25551.
28. Ye YZ, Zhang ZH, Fan XY, Xu XL, Chen ML, Chang BW, Zhang YB. Notch3 overexpression associates with poor prognosis in human non-small-cell lung cancer. Med Oncol. 2013; 30:595. doi: 10.1007/s12032-013-0595-7.
29. Yuan X, Wu H, Xu H, Han N, Chu Q, Yu S, Chen Y, Wu K. Meta-analysis reveals the correlation of Notch signaling with non-small cell lung cancer progression and prognosis. Sci Rep. 2015; 5:10338. doi: 10.1038/srep10338.
30. Jin MM, Ye YZ, Qian ZD, Zhang YB. Notch signaling molecules as prognostic biomarkers for non-small cell lung cancer. Oncol Lett. 2015; 10:3252–60. doi: 10.3892/ol.2015.3662.
31. Yuan X, Wu H, Han N, Xu H, Chu Q, Yu S, Chen Y, Wu K. Notch signaling and EMT in non-small cell lungcancer: biological significance and therapeutic application. J Hematol Oncol. 2014; 7:87. doi: 10.1186/s13045-014-0087-z.
32. Yu H, Takeuchi M, LeBarron J, Kantharia J, London E, Bakker H, Haltiwanger RS, Li H, Takeuchi H. Notch-modifying xylosyltransferase structures support an SNi-like retaining mechanism. Nat Chem Biol. 2015; 11:847–54. doi: 10.1038/nchembio.1927.
33. Sullivan JP, Spinola M, Dodge M, Raso MG, Behrens C, Gao B, Schuster K, Shao C, Larsen JE, Sullivan LA, Honorio S, Xie Y, Scaglioni PP, et al. Aldehyde dehydrogenase activity selects for lung adenocarcinoma stem cells dependent on notch signaling. Cancer Res. 2010; 70:9937–48. doi: 10.1158/0008-5472.CAN-10-0881.
34. Zheng Y, de la Cruz CC, Sayles LC, Alleyne-Chin C, Vaka D, Knaak TD, Bigos M, Xu Y, Hoang CD, Shrager JB, Fehling HJ, French D, Forrest W, et al. A rare population of CD24(+) ITGB4(+) Notch(hi) cells drives tumor propagation in NSCLC and requires Notch3 for self-renewal. Cancer Cell. 2013; 24:59–74. doi: 10.1016/j.ccr.2013.05.021.
35. Donnem T, Andersen S, Al-Shibli K, Al-Saad S, Busund LT, Bremnes RM. Prognostic impact of Notch ligands andreceptors in nonsmall cell lung cancer: coexpression ofNotch-1 and vascular endothelial growth factor-A predictspoor survival. Cancer. 2010; 116:5676–85. doi: 10.1002/cncr.25551.
36. Konishi J, Kawaguchi KS, Vo H, Haruki N, Gonzalez A, Carbone DP, Dang TP. Gamma-secretase inhibitor prevents Notch3 activation and reduces proliferation in human lung cancers. Cancer Res. 2007; 67:8051–7. doi: 10.1158/0008-5472.CAN-07- 1022.
37. Chen Y, De Marco MA, Graziani I, Gazdar AF, Strack PR, Miele L, Bocchetta M. Oxygen concentration determines the biological effects of NOTCH-1 signaling in adenocarcinoma of the lung. Cancer Res. 2007; 67:7954–9. doi: 10.1158/0008-5472.CAN- 07-1229

