INTRODUCTION
Endometrial cancer is the most frequently tumor of the female reproductive system in developed countries with an approximately 60,050 cases and 10,470 deaths for the year 2016 in the United States [1]. The surgical management of endometrial cancer is still controversial, especially in the early stage. The study shows that a complete lymphadenectomy may have no therapeutic benefit in patients with early-stage endometrial cancer [2]. Comprehensive lymphadenectomy not only increases operative time and blood loss but also is associated with surgical complications, such as blood vessel and nerve damage, lymphoedema, and lymphocyst formation [3]. The rate of long-term lymphedema directly attributed to lymphadenectomy was recently reported to be 23% [4]. Furthermore, lymphadenectomy imposes significant morbidity for the patients [3].
Lymph node status is a major prognostic factor and a criterion for adjuvant therapy in endometrial cancer. The concept of the sentinel lymph node (SLN), the node(s) most likely to harbor the first metastasis from the primary tumor, was first introduced in 1960 following observations associated with parotid gland carcinomas [5]. As the sentinel lymph node is relatively the first in a chain of lymph nodes, theoretically the sentinel node will be the first to encounter the effects of the metastatic form of the disease. If the sentinel node is negative, then it can be safely assumed that the remainder of the lymphatic basin is also unaffected by metastasis. The advantage of the distinctive benefits of SLN mapping is the opportunity to avoid “over-staging”, leading to a relatively lower morbidity than in the case of the performance of a full lymphadenectomy and the potential for improved diagnostic accuracy [6]. As a surgical technique, the SLN mapping has been implemented in the standard of treatment for patients with melanoma and breast cancer [7].
If the SLN concept is valid in endometrial cancer, most patients, especially women affected with endometrial cancer in the early stage could avoid the risks associated with the lymphadenectomy. However, the diagnostic efficacy of SLN mapping in endometrial cancer is still controversial. Therefore, we performed a meta-analysis to evaluate different sentinel lymph node mapping techniques and their corresponding detection rates and sensitivity in endometrial cancer.
RESULTS
Overall, 2,205 studies were retrieved through the electronic databases searching. Among these, 859 (39.0%) studies were removed as duplicates. After title and abstract evaluation, 233 (10.6%) remained for full review. Of these, 189 (8.6%) full-text articles were additionally excluded for the following reasons: 8 (0.36%) had low sample size(fewer than 7 patients); 56 (2.5%) not concerned with endometrial cancer; 62 (2.8%) not mainly focused on SLN mapping; 12 (0.54%) without histological or immunohistological results; 6 (0.27%) reported only the numbers of metastasized lymph nodes but not the number of patients; 6 (0.27%) from the same research team using the same database; 10 (0.45%) were animal studies; 3 (0.1%) were case reports; 7 (0.32%) were letters; 19 (0.86%) were reviews. Thus, 44 (2.0%) studies [11–54] were deemed as eligible by the authors (involving 2,236 patients) after conducting a comprehensive and through literature search. Figure 1 shows the process involved in the assessment of the studies and in accordance to the process highlighted in the figure the studies were identified, those of them that fulfilled the conditions detailed earlier were included and those that didn’t were excluded.
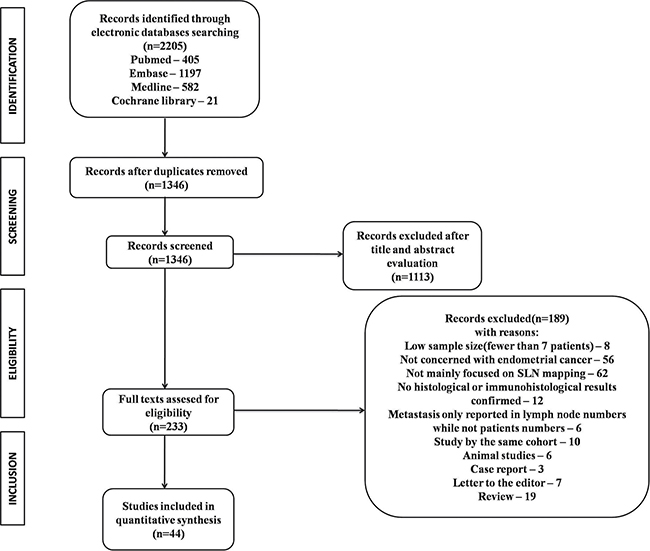
Figure 1: Flow diagram of studies identified, included, and excluded.
Sensitivity
The pooled sensitivity for all studies deemed eligible (n = 44) by the authors was 91% (95% CI: 87–95%), the Cochran Q value was 49.90 (p = 0.22 and I2 = 13.82%). The forest plot of sensitivity pooling is shown in Figure 2.
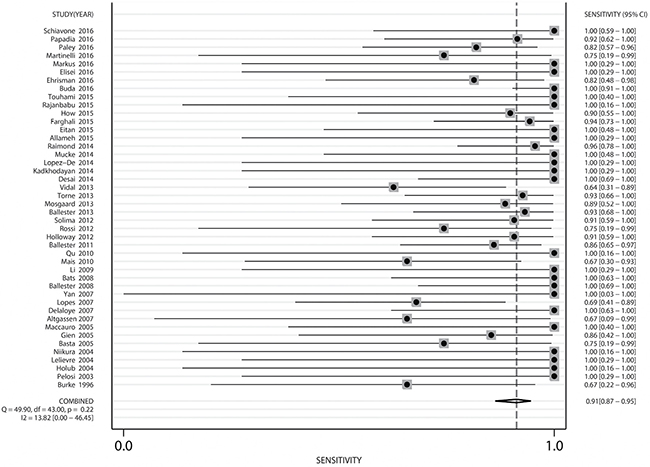
Figure 2: Forest plot of pooled sensitivity and 95% CI in SLN mapping in endometrial cancer.
The funnel plot of the sensitivity pooling and the funnel plot of the sensitivity pooling by using trim and fill method are shown in Figure 3. The egger’s regression intercept was found out to be 2.34 (p = 0.031).
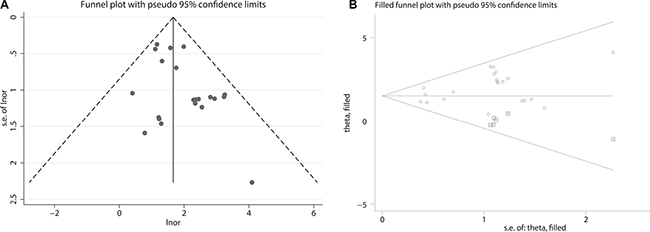
Figure 3: (A) Funnel plot of pooled sensitivity. (B) Funnel plot of pooled sensitivity by using trim and fill method.
Overall SLN detection rate
Figure 4 shows the forest plot associated with the overall SLN detection rate. The pooled detection rate was 83 % (95% CI: 80–86%), with heterogeneity I2 = 78.9% (p = 0.000).
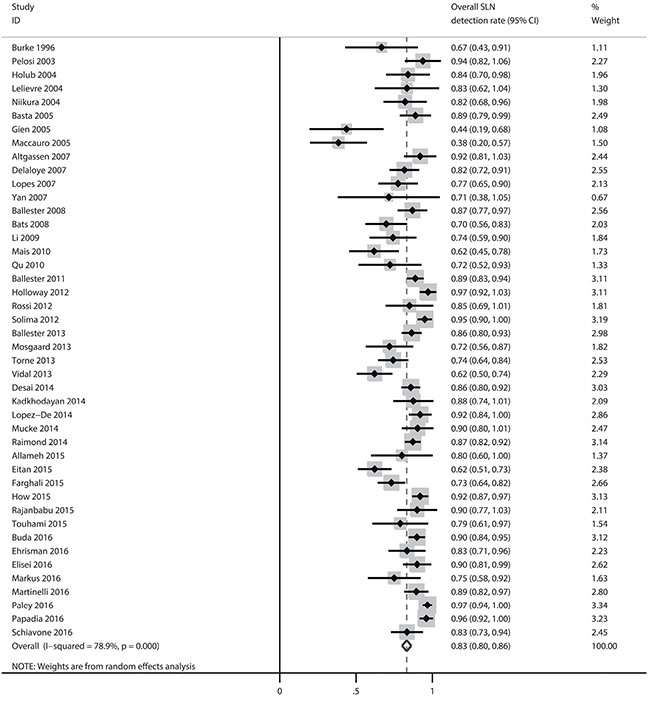
Figure 4: Forest plot of pooled overall detection rate and 95% CI in SLN mapping in endometrial cancer.
The funnel plot of the overall SLN detection rate pooling and the funnel plot of the overall SLN detection rate pooling are shown in Figure 5. The egger’s regression intercept was −7.06 (p = 0.000).
Figure 5: (A) Funnel plot of pooled overall SLN detection rate. (B) Funnel plot of pooled overall SLN detection rate by using trim and fill method.
Bilateral SLN detection rate
Figure 6 shows the forest plot of the bilateral SLN detection rate. The bilateral SLN detection rate was observed to be 56% (95% CI: 48–63%), with heterogeneity I2 = 91.4% (p = 0.000).
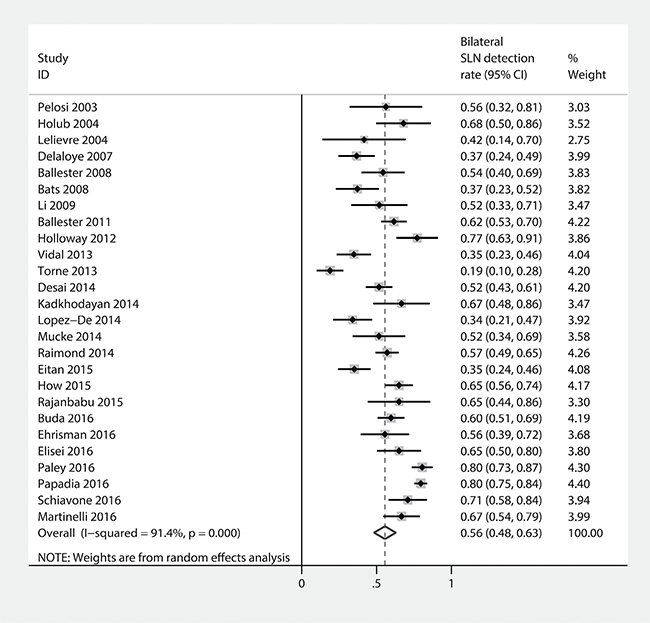
Figure 6: Forest plot of pooled bilateral SLN detection rate and 95%CI in SLN mapping in endometrial cancer.
Sub-group analysis
The sentinel lymph node detection rate and sensitivity were related to the mapping method employed (blue dye alone, radiotracer dye with blue dye, ICG), the surgical approach followed (laparotomy, laparoscopy, robotic assistance) and the site of dye injection (cervical injection, uterine injection) (Table 1). Use of ICG showed high pooled overall detection rate: 93% (95% CI: 89–96%) and high pooled bilateral detection rate: 78% (95% CI: 72–84%). The Robot-assisted surgery also had high pooled overall detection rate: 86% (95% CI: 79–93%) and pooled bilateral detection rate: 62% (95%CI: 43–80%). The sensitivity and overall detection rate were high in patients in whom cervical injection was the site of dye injection 93% (95% CI: 87–96%) and 86% (95% CI: 83–89%), respectively and both blue dye and radiotracer dye were used for mapping 92% (95% CI: 84–96%) and 86% (95% CI: 82–90%), respectively. Laparoscopic surgery showed higher pooled sensitivities 96 % (95% CI: 88–99%).
DISCUSSION
To our knowledge, this is the newest meta-analysis focused on the diagnostic efficacy of sentinel lymph node mapping in endometrial cancer. In our meta-analysis of 44 studies comprising 2,236 cases, sentinel node mapping yielded a pooled detection rate of 83%. The pooled sensitivity implies that 91% of occult lymph node metastases could be diagnosed by SLN mapping, which seems to be equivalent to those achieved in patients with breast cancer (sensitivity 91%) [55]. However, bilateral nodes were detected in only 59% (26 studies) of the patients. The Endometrium as a midline organ and it exhibits two different pathways of lymphatic drainage: right and left [28]. The bilateral detection method has been used for other midline organs such as the penis with fairly promising results [56]. In the present study,the rate of bilateral SLN detection was between 89% (in the study conducted by Martinelli et al.) [51] and 19% (in the study conducted by Torné et al.) [34], and the pooled bilateral detection rate was 56% (95% CI: 48–64%). Therefore, achieving high bilateral SLN detection rates of endometrial cancer is mandatory to implement the SLN mapping as a routine component of clinical practice.
It should be mentioned that in the previous meta-analyses focusing on the diagnostic efficacy of SLN mapping in endometrial cancer, Kang et al. [57] reported a pooled detection rate of 78% (95% CI: 73–84%) (n = 1,101), while Ansari et al. [58] reported a pooled detection rate and sensitivity of 77.8% (95% CI: 74–82%) and 89% (95% CI: 83–93%) respectively (n = 2,071). Compared to these previous meta-analyses, our research found higher detection rate of 83% (95% CI: 80–86%) (n = 2,236) and sensitivity of 91% (95% CI: 87–95%). Since previous two meta-analyses were published several years ago, our meta-analysis included the newest results of the recent studies ,which makes it more valid.
In addition, the studies included in our systematic review were heterogeneous and therefore gave rise to the need for conducting a sub-group analysis to explore the reasons for the observed heterogeneity. Laparoscopic surgery and robot-assisted surgery, were associated with high detection rates and sensitivities when compared with an open surgery based approach. The pilot study conducted by Mais et al. [26] showed that the high detection rates obtainable through laparoscopy were not reproducible through laparotomy. The different detection rates observed through laparoscopy or through laparotomy might depend on the different time intervals elapsing between the injection of the vital dye into the cervix and the surgical SLN assessment in the pelvic basin. In fact, this time interval was always found to be shorter in the case of laparoscopy than for laparotomy. The current study also showed that the pooled sensitivity and detection rates in endometrial cancer patients are high for SLN mapping when the mapping was carried out using ICG. When the mapping was carried out using solely the blue dye, the pooled detection rate was observed to be rather low. Moreover, there are some disadvantages of the use of the blue dye. Allergic reactions to the blue dye were observed in 0.14–3% of the patients, including urticaria, skin rashes, erythema, blue hives, cardiovascular collapse, and anaphylactic shock. Other side effects include temporary skin tattooing, blue discoloration of the operative field following peritumoral injection, blue-colored urine for up to 24 hrs. following administration, and a factitious drop in intraoperative oxygen saturation measured by pulse oximetry. Furthermore, the teratogenicity and the long-term toxicity associated with the blue dye are unknown and could have serious effects on pregnancy [59, 60]. Indocyanine green (ICG) represents a feasible alternative to the more traditional methods of sentinel lymph node (SLN) mapping, and the interest associated with this promising tracer is growing. A recent meta-analysis conducted by Ruscito et al. [61] demonstrated that ICG SLN mapping seems to be equivalent to the combination of blue dyes and Tc-99m in terms of overall and bilateral detection rates in uterine malignancies. The good toxicity profile and ease of use of ICG, which does not require the injection to be administered in a controlled environment is also very desirable. Another issue in SLN mapping pertains to the identification of the optimal injection site for the radiocolloid /dye in patients with endometrial cancer. A comprehensive sub-group analysis showed that the use of cervical injection as the dye injection approach was found to cause an increase of 10% and 5% in the detection rate and sensitivity respectively, as compared to the use of uterine injection as the dye injection approach. It seems to be a lack of sufficient consensus about the best site of injection. The cervical injection approach is a method which is easy to carry out, reproducible, and is very suitable for standardization.
The meta-analysis performed during the course of this study has the following limitations that must be taken into account. First, the results presented in the current study were based on unadjusted estimates; more accurate outcomes would result from the adjustments made to be considered for other confounders such as age, body mass index, cancer stage, and so on. Second, the studies included in this analysis were not sufficient enough, especially from the perspective of a subgroup analysis. Thus, a potential publication bias is very likely to be associated with the results provided in this study in spite of evidence obtained from the statistical tests performed. Finally, only English and Chinese reports have been included in the analysis and consequently this might lead to the study not considering the data from other relevant studies published in other languages, which may result in causing a potential language bias.
In conclusion, the present results confirmed that sentinel lymph node mapping is a feasible and reliable approach that performs well diagnostically for the assessment of lymph nodal involvement in endometrial cancer. We also found that SLN mapping using some new techniques, such as ICG and robot-assisted surgery demonstrated higher detection rates compared to other modalities. The use of cervical injection and the mode of dye injection for both the blue dye and the radiotracer of the mapping material can optimize the sensitivity and detection rate of the technique. Further clinical trials are required to investigate the relationship between lymphadenectomy guided by SLN mapping and prognosis of endometrial cancer in the future.
MATERIALS AND METHODS
Search strategy
A comprehensive literature search on the retrieved publications (the last search was done in November, 2016) was performed independently by two authors associated with this current study. The language of studies was limited to English and Chinese only. The primary sources for the literature search were the electronic databases: Pubmed, Embase, Medline and the Cochrane Library. The predefined keywords used for the search were “sentinel lymph node” and “endometrial cancer”. A search algorithm that selected and screened results based on a combination with the following search terms: “sentinel AND (endometri* OR uterus OR uterine OR corpus uteri ) AND (cancer OR neoplasm* OR carcinoma* OR malignanc* OR tumo*)” was used to perform the literature search detailed in this study.
Inclusion and exclusion criteria
For evaluating the diagnostic performance of sentinel lymph node mapping in endometrial cancers, the studies in accordance to the following inclusion criteria were included: (1) Studies with the enrollment of at least 7 women diagnosed with endometrial cancer; (2) The SLN mapping was the study’s primary focus; (3) Studies validated by pelvic with/without para-aortic lymph node dissection and pathological examination including H&E (hematoxylin-eosin) staining or immunohistochemistry (IHC) were taken as the reference standard; (4) SLN mapping as the diagnostic method; (5) Studies where the total number of enrolled patients as well as those with detected SLN were both reported; (6) Studies that reported the total number of patients with a positive lymph node diagnosis, as well as those with false negative results.
Review articles, letters, comments, conference proceedings, unpublished data and case-reports were excluded. To avoid overlapping patient data in duplicate publications, we included the more recent articles with the largest sample sizes.
Study quality assessment
The quality assessment of studies included in this article was undertaken by authors Lin and Zhang. The “QUADAS-2” (Quality Assessment of Diagnostic Accuracy Studies-2) tool, an official tool for assessing the quality of the diagnosis accuracy of a study (launched in 2011) was used to assist with the above-mentioned quality assessment [8]. The core “QUADAS-2” items used in our study are detailed outlined in the Appendix Table 1. “QUADAS-2” divided the assessment items into the risk of bias and the applicability, and has several items including: patient selection, index test, reference standard, flow and timing. The criteria could be scored as “yes”,”no”, or “not reported” in the publication.
Table 1: Results of sub-group analysis
Sub-group |
Detection rate (95% CI) |
Sensitivity (95% CI) |
Bilateral detection rate (95% CI) |
|
|---|---|---|---|---|
Surgical |
Laparoscopy |
82% (78–87%) |
96% (88–99%) |
58% (47–69%) |
approach |
Laparotomy |
77% (71–84%) |
89% (80–95%) |
47% (32–61%) |
|
Robotic assistance |
86% (79–93%) |
90% (77–96%) |
62% (43–80%) |
Mapping |
Blue dye |
76% (71–81%) |
90% (79–96%) |
44% (38–50%) |
method |
ICG |
93% (89–96%) |
87% (76–93%) |
78% (72–84%) |
|
Tc-99m+blue dye |
86% (82–90%) |
92% (84–96%) |
56% (41–71%) |
Injection |
Cervical injection |
86% (83–89%) |
93% (87–96%) |
60% (52–69%) |
site |
Uterine injection |
76% (68–83%) |
88% (78–93%) |
47% (11–84%) |
Abbreviations: ICG, indocyanine green; Tc-99m, technecium-99.
Data abstraction
Data from the included studies were extracted and summarized independently by the two authors mentioned earlier (Lin and Zhang). The extracted data primarily included (Supplementary Table 1): first author, publication year, country, sample size of the study, detection rate, tracer, injection site, pathology assessment and surgical approach. Wherever possible, the SLN detection rate was calculated in patients with bilateral sentinel node identification. The sensitivity associated with the sentinel lymph node procedure was defined as the total number of true positives in patients with a positive histopathology. A positive sentinel node was considered as true positive (TP) irrespective to the status of the other nodes, and a true negative (TN) was a negative sentinel node only if all other non-sentinel nodes were negative. A false negative (FN) was defined as positive non-SLN with negative SLN. The SLN detection rate can be defined as the percentage of patients in which at least one SLN was identified.
Statistical analysis
The statistical analysis tool, Stata 12.0 software (StataCorp, College Station, Texas, USA) was employed to perform the aggregate data meta-analyses and evaluate the heterogeneity of the included studies. The random effects model was applied for calculating the overall detection rate, the bilateral detection rate, and the sensitivity from the data provided in the source articles. The results were depicted graphically as forest plots. The pooled data was presented with 95% confidence intervals (95% CI). The potential heterogeneity of the sensitivity and the detection rate was analyzed with the Cochrane Q test (the p-values less than 0.05 were considered as statistically significant). I2 index was used to quantify the heterogeneity, analyzing how much of the variance associated with the included studies was real and wasn’t due to sampling errors. Funnel plots, Egger’s regression intercepts [9], and the Duval and Tweedie’s [10] “trim and fill” method were used for the evaluation of publication bias. A subgroup analysis was performed for expolring the heterogeneity in three variables: surgical approach, mapping method, injection site.
ACKNOWLEDGMENTS AND FUNDING
The study is supported by the National Natural Science Foundation of China, Grant No. 81371881 and The Natural Science Foundation of Zhejiang Province, China Grant No. LY12H16012.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
The work has not been published previously (except in the form of an abstract or as part of a published lecture or academic thesis or as an electronic preprint).
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66: 7–30. doi: 10.3322/caac.21332.
2. May K, Bryant A, Dickinson HO, Kehoe S, Morrison J. Lymphadenectomy for the management of endometrial cancer. Cochrane Database Syst Rev. 2010; 65:1399–1400. doi: 10.1002/14651858.CD007585.pub2.
3. Dowdy SC, Borah BJ, Bakkum-Gamez JN, Weaver AL, McGree ME, Haas LR, Keeney GL, Mariani A, Podratz KC. Prospective assessment of survival, morbidity, and cost associated with lymphadenectomy in low-risk endometrial cancer. Gynecol Oncol. 2012; 127:5–10. doi: 10.1016/j.ygyno.2012.06.035.
4. Yost KJ, Cheville AL, Al-Hilli MM, Mariani A, Barrette BA, McGree ME, Weaver AL, Dowdy SC. Lymphedema after surgery for endometrial cancer: prevalence, risk factors, and quality of life. Obstet Gynecol. 2014; 124:307–15. doi: 10.1097/AOG.0000000000000372.
5. Gould EA, Winship T, Philbin PH, Kerr HH. Observations on a “sentinel node” in cancer of the parotid. Cancer. 1960; 13:77–8. doi: 10.1002/1097–0142(196001/02)13:1<77::AID-CNCR2820130114>3.0.CO;2-D.
6. Emerson J, Robison K. Evaluation of sentinel lymph nodes in vulvar, endometrial and cervical cancers. World J Obstet Gynecol. 2016; 5:78–86. doi: 10.5317/WJOG.v5.i1.78.
7. Cabanas RM. An approach for the treatment of penile carcinoma. Cancer. 1977; 39:456–466. doi: 10.1002/1097-0142(197702)39:2<456::AID-CNCR2820390214>3.0.CO;2-I.
8. Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM. QUADAS-2 Group. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011; 155:529–36. doi: 10.7326/0003-4819-155-8-201110180-00009.
9. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34. doi: 10.1136/bmj.315.7109.629.
10. Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000; 56:455–63. doi : 10.1111/j.0006-341X.2000.00455.x.
11. Burke TW, Levenback C, Tornos C, Morris M, Wharton JT, Gershenson DM. Intraabdominal lymphatic mapping to direct selective pelvic and paraaortic lymphadenectomy in women with high-risk endometrial cancer: results of a pilot study. Gynecol Oncol. 1996; 62:169–73. doi: 10.1006/gyno.1996.0211.
12. Pelosi E, Arena V, Baudino B, Bellò M, Giusti M, Gargiulo T, Palladin D, Bisi G. Preoperative lymphatic mapping and intraoperative sentinel lymph node detection in early stage endometrial cancer. Nucl Med Commun. 2003; 24:971–5. doi: 10.1097/00006231-200309000-00005.
13. Holub Z, Jabor A, Lukac J, Kliment L. Laparoscopic detection of sentinel lymph nodes using blue dye in women with cervical and endometrial cancer. Med Sci Monit. 2004; 10: CR587–91.
14. Lelievre L, Camatte S, Le Frere-belda MA, Kerrou K, Froissart M, Taurelle R, Vilde F, Lecuru F. Sentinel lymph node biopsy in cervix and corpus uteri cancers. Int J Gynecol Cancer. 2004; 14:271–8. doi: 10.1111/j.1048-891X.2004.014210.x.
15. Niikura H, Okamura C, Utsunomiya H, Yoshinaga K, Akahira J, Ito K, Yaegashi N. Sentinel lymph node detection in patients with endometrial cancer. Gynecol Oncol. 2004; 92:669–74. doi: 10.1016/j.ygyno.2003.10.039.
16. Basta A, Pityński K, Basta P, Hubalewska-Hoła A, Opławski M, Przeszlakowski D. Sentinel node in gynaecological oncology. Rep Pract Oncol Radiother. 2005; 10:24–27. doi: 10.1016/S1507-136771114-7.
17. Gien LT, Kwon JS, Carey MS. Sentinel node mapping with isosulfan blue dye in endometrial cancer. J Obstet Gynaecol Can. 2005; 27:1107–12. doi: 10.1016/S1701-216330393-0.
18. Maccauro M, Lucignani G, Aliberti G, Villano C, Castellani MR, Solima E, Bombardieri E. Sentinel lymph node detection following the hysteroscopic peritumoural injection of 99mTc-labelled albumin nanocolloid in endometrial cancer. Eur J Nucl Med Mol Imaging. 2005; 32:569–74. doi: 10.1007/s00259-004-1709-4.
19. Altgassen C, Pagenstecher J, Hornung D, Diedrich K, Hornemann A. A new approach to label sentinel nodes in endometrial cancer. Gynecol Oncol. 2007; 105:457–61. doi: 10.1016/j.ygyno.2007.01.021.
20. Delaloye JF, Pampallona S, Chardonnens E, Fiche M, Lehr HA, De Grandi P, Delaloye AB. Intraoperative lymphatic mapping and sentinel node biopsy using hysteroscopy in patients with endometrial cancer. Gynecol Oncol. 2007; 106:89–93. doi: 10.1016/j.ygyno.2007.03.003.
21. Lopes LA, Nicolau SM, Baracat FF, Baracat EC, Gonçalves WJ, Santos HV, Lopes RG, Lippi UG. Sentinel lymph node in endometrial cancer. Int J Gynecol Cancer. 2007; 17:1113–7. doi: 10.1111/j.1525-1438.2007.00909.x.
22. Yan X, Li G, Chen L, Wang G, Shang H, Lin T, Han Y, Guan J. A pilot study on sentinel lymph nodes identification in patients with cervical cancer or endometrial cancer. Pract Obstet Gynecol. 2007; 23:737–39. doi: 10.3969/j.issn.1003-6946.2007.12.014.
23. Ballester M, Dubernard G, Rouzier R, Barranger E, Darai E. Use of the sentinel node procedure to stage endometrial cancer. Ann Surg Oncol. 2008; 15: 1523–9. doi: 10.1245/s10434-008-9841-1.
24. Bats AS, Clément D, Larousserie F, Le Frère-Belda MA, Pierquet-Ghazzar N, Hignette C, Lécuru F. Does sentinel node biopsy improve the management of endometrial cancer? Data from 43 patients. J Surg Oncol. 2008; 97:141–5. doi: 10.1002/jso.20857.
25. Li B, Wu L, Li X, Lu H, Bai P, Li S, Zhang W, Gao J. Sentinel lymph node identification in endometrial cancer. Clin Oncol Cancer Res. 2009; 6:124–8. doi: 10.1007/s11805-009-0124-9.
26. Mais V, Peiretti M, Gargiulo T, Parodo G, Cirronis MG, Melis GB. Intraoperative sentinel lymph node detection by vital dye through laparoscopy or laparotomy in early endometrial cancer. J Surg Oncol. 2010; 101: 408–12. doi: 10.1002/jso.21496.
27. Qu J, Qi Y, Li W. The preliminary research of sentinel lymph node enhancement identification detection in endometrial cancer. J Jining Med Univ. 2010; 33:398–400. doi: 10.3969/j.issn.1000-9760.2010.06.008.
28. Ballester M, Dubernard G, Lécuru F, Heitz D, Mathevet P, Marret H, Querleu D, Golfier F, Leblanc E, Rouzier R, Daraï E. Detection rate and diagnostic accuracy of sentinel-node biopsy in early stage endometrial cancer: a prospective multicentre study (SENTI-ENDO). Lancet Oncol. 2011; 12:469–76. doi: 10.1016/S1470-204570070-5.
29. Holloway RW, Bravo RA, Rakowski JA, James JA, Jeppson CN, Ingersoll SB, Ahmad S. Detection of sentinel lymph nodes in patients with endometrial cancer undergoing robotic-assisted staging: a comparison of colorimetric and fluorescence imaging. Gynecol Oncol. 2012; 126:25–9. doi: 10.1016/j.ygyno.2012.04.009.
30. Rossi EC, Ivanova A, Boggess JF. Robotically assisted fluorescence-guided lymph node mapping with ICG for gynecologic malignancies: a feasibility study. Gynecol Oncol. 2012; 124:78–82. doi: 10.1016/j.ygyno.2011.09.025.
31. Solima E, Martinelli F, Ditto A, Maccauro M, Carcangiu M, Mariani L, Kusamura S, Fontanelli R, Grijuela B, Raspagliesi F. Diagnostic accuracy of sentinel node in endometrial cancer by using hysteroscopic injection of radiolabeled tracer. Gynecol Oncol. 2012; 126:419–23. doi: 10.1016/j.ygyno.2012.05.025.
32. Ballester M, Naoura I, Chéreau E, Seror J, Bats AS, Bricou A, Daraï E. Sentinel node biopsy upstages patients with presumed low- and intermediate-risk endometrial cancer: results of a multicenter study. Ann Surg Oncol. 2013; 20:407–12. doi: 10.1245/s10434-012-2683-x.
33. Mosgaard BJ, Skovlund VR, Hendel HW. Promising results using sentinel node biopsy as a substitute for radical lymphadenectomy in endometrial cancer staging. Dan Med J. 2013; 60: A4665.
34. Torné A, Pahisa J, Vidal-Sicart S, Martínez-Roman S, Paredes P, Puerto B, Albela S, Fusté P, Perisinotti A, Ordi J. Transvaginal ultrasound-guided myometrial injection of radiotracer (TUMIR): a new method for sentinel lymph node detection in endometrial cancer. Gynecol Oncol. 2013; 128:88–94. doi: 10.1016/j.ygyno.2012.10.008.
35. Vidal F, Leguevaque P, Motton S, Delotte J, Ferron G, Querleu D, Rafii A. Evaluation of the sentinel lymph node algorithm with blue dye labeling for early-stage endometrial cancer in a multicentric setting. Int J Gynecol Cancer. 2013; 23:1237–43. doi: 10.1097/IGC.0b013e31829b1b98.
36. Desai PH, Hughes P, Tobias DH, Tchabo N, Heller PB, Dise C, Slomovitz BM. Accuracy of robotic sentinel lymph node detection (RSLND) for patients with endometrial cancer (EC). Gynecol Oncol. 2014; 135:196–200. doi: 10.1016/j.ygyno.2014.08.032.
37. Kadkhodayan S, Shiravani Z, Hasanzadeh M, Sharifi N, Yousefi Z, Fattahi A, Sadeghi R. Lymphatic mapping and sentinel node biopsy in endometrial cancer—a feasibility study using cervical injection of radiotracer and blue dye. Nucl Med Rev Cent East Eur. 2014; 17: 55–8. doi: 10.5603/NMR.2014.0017.
38. López-De la Manzanara Cano C, Cordero García JM, Martín-Francisco C, Pascual-Ramírez J, Parra CP, Céspedes Casas C. Sentinel lymph node detection using 99mTc combined with methylene blue cervical injection for endometrial cancer surgical management: a prospective study. Int J Gynecol Cancer. 2014; 24:1048–53. doi: 10.1097/IGC.0000000000000158.
39. Mücke J, Klapdor R, Schneider M, Länger F, Gratz KF, Hillemanns P, Hertel H. Isthmocervical labelling and SPECT/CT for optimized sentinel detection in endometrial cancer: technique, experience and results. Gynecol Oncol. 2014; 134:287–92. doi: 10.1016/j.ygyno.2014.05.001.
40. Raimond E, Ballester M, Hudry D, Bendifallah S, Daraï E, Graesslin O, Coutant C. Impact of sentinel lymph node biopsy on the therapeutic management of early-stage endometrial cancer: Results of a retrospective multicenter study. Gynecol Oncol. 2014; 133:506–11. doi: 10.1016/j.ygyno.2014.03.019.
41. Allameh T, Hashemi V, Mohammadizadeh F, Behnamfar F. Sentinel lymph node mapping in early stage of endometrial and cervical cancers. J Res Med Sci. 2015; 20:169–73.
42. Eitan R, Sabah G, Krissi H, Raban O, Ben-Haroush A, Goldschmit C, Levavi H, Peled Y. Robotic blue-dye sentinel lymph node detection for endometrial cancer - Factors predicting successful mapping. Eur J Surg Oncol. 2015; 41:1659–63. doi: 10.1016/j.ejso.2015.09.006.
43. Farghali MM, Allam IS, Abdelazim IA, El-Kady OS, Rashed AR, Gareer WY, Sweed MS. Accuracy of Sentinel Node in Detecting Lymph Node Metastasis in Primary Endometrial Carcinoma. Asian Pac J Cancer Prev. 2015; 16:6691–6. doi: 10.7314/APJCP.2015.16.15.6691.
44. How J, Gotlieb WH, Press JZ, Abitbol J, Pelmus M, Ferenczy A, Probst S, Gotlieb R, Brin S, Lau S. Comparing indocyanine green, technetium, and blue dye for sentinel lymph node mapping in endometrial cancer. Gynecol Oncol. 2015; 137:436–42. doi: 10.1016/j.ygyno.2015.04.004.
45. Rajanbabu A, Murali V, Nataraj YS, Vijaykumar DK. Detection of Sentinel Lymph Nodes in Endometrial Cancer with Intracervical Indocyanine Green Injection and Robotically Assisted Near Infrared Imaging: A Feasibility Study in Indian Setting. Indian Journal of Gynecologic Oncology. 2015; 13:1–6. doi: 10.1007/s40944-015-0020-6.
46. Touhami O, Trinh XB, Gregoire J, Sebastianelli A, Renaud MC, Grondin K, Plante M. Is a More Comprehensive Surgery Necessary in Patients With Uterine Serous Carcinoma? Int J Gynecol Cancer. 2015; 25:1266–70. doi: 10.1097/IGC.0000000000000488.
47. Buda A, Crivellaro C, Elisei F, Di Martino G, Guerra L, De Ponti E, Cuzzocrea M, Giuliani D, Sina F, Magni S, Landoni C, Milani R. Impact of Indocyanine Green for Sentinel Lymph Node Mapping in Early Stage Endometrial and Cervical Cancer:Comparison with Conventional Radiotracer (99m)Tc and/or Blue Dye. Ann Surg Oncol. 2016; 23:2183–91. doi: 10.1245/s10434-015-5022-1.
48. Ehrisman J, Secord AA, Berchuck A, Lee PS, Di Santo N, Lopez-Acevedo M, Broadwater G, Valea FA, Havrilesky LJ. Performance of sentinel lymph node biopsy in high-risk endometrial cancer. Gynecol Oncol Rep. 2016; 17:69–71. doi: 10.1016/j.gore.2016.04.002.
49. Elisei F, Crivellaro C, Giuliani D, Dolci C, De Ponti E, Montanelli L, La Manna M, Guerra L, Arosio M, Landoni C, Buda A. Sentinel-node mapping in endometrial cancer patients: comparing SPECT/CT, gamma-probe and dye. Ann Nucl Med. 2017; 31: 93–99. doi: 10.1007/s12149-016-1137-0.
50. Markus A, Ray AS, Bolla D, Müller J, Diener PA, Wendler T, Hornung R. Sentinel lymph node biopsy in endometrial and cervical cancers using freehand SPECT—first experiences. Gynecol Surg. 2016: 1–8. doi: 10.1007/s10397-016-0969-x.
51. Martinelli F, Ditto A, Bogani G, Signorelli M, Chiappa V, Lorusso D. Raspagliesi F.Laparoscopic sentinel node mapping in endometrial cancer following hysteroscopic injection of indocyanine green. J Minim Invasive Gynecol. 2016; 23: S99-S100. doi: 10.1016/j.jmig.2016.08.244.
52. Paley PJ, Veljovich DS, Press JZ, Isacson C, Pizer E, Shah C. A prospective investigation of fluorescence imaging to detect sentinel lymph nodes at robotic-assisted endometrial cancer staging. Am J Obstet Gynecol. 2016; 215:117.e1–7. doi: 10.1016/j.ajog.2015.12.046.
53. Papadia A, Imboden S, Siegenthaler F, Gasparri ML, Mohr S, Lanz S, Mueller MD. Laparoscopic Indocyanine Green Sentinel Lymph Node Mapping in Endometrial Cancer. Ann Surg Oncol. 2016; 23:2206–11. doi: 10.1245/s10434-016-5090-x.
54. Schiavone MB, Zivanovic O, Zhou Q, Leitao MM Jr, Levine DA, Soslow RA, Alektiar KM, Makker V, Iasonos A, Abu-Rustum NR. Survival of Patients with Uterine Carcinosarcoma Undergoing Sentinel Lymph Node Mapping. Ann Surg Oncol. 2016; 23:196–202. doi: 10.1245/s10434-015-4612-2.
55. Veronesi U, Paganelli G, Viale G, Luini A, Zurrida S, Galimberti V, Intra M, Veronesi P, Robertson C, Maisonneuve P, Renne G, De Cicco C, De Lucia F, et al. A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. N Engl J Med. 2003; 349:546–53. doi: 10.1056/NEJMoa012782.
56. Sadeghi R, Gholami H, Zakavi SR, Kakhki VR, Tabasi KT, Horenblas S. Accuracy of sentinel lymph node biopsy for inguinal lymph node staging of penile squamous cell carcinoma: systematic review and meta-analysis of the literature. J Urol. 2012; 187:25–31. doi: 10.1016/j.juro.2011.09.058.
57. Kang S, Yoo HJ, Hwang JH, Lim MC, Seo SS, Park SY. Sentinel lymph node biopsy in endometrial cancer: meta-analysis of 26 studies. Gynecol Oncol. 2011; 123:522–7. doi: 10.1016/j.ygyno.2011.08.034.
58. Ansari M, Rad MA, Hassanzadeh M, Gholami H, Yousefi Z, Dabbagh VR, Sadeghi R. Sentinel node biopsy in endometrial cancer: systematic review and meta-analysis of the literature. Eur J Gynaecol Oncol. 2013; 34: 387–401.
59. Argon AM, Duygun U, Acar E, Daglioz G, Yenjay L, Zekioglu O, Kapkac M. The use of periareolar intradermal Tc-99m tin colloid and peritumoral intraparenchymal isosulfan blue dye injections for determination of the sentinel lymph node. Clin Nucl Med. 2006; 31: 795–800. doi: 10.1097/01.rlu.0000246855.80027.b7.
60. Bagaria SP, Faries MB, Morton DL. Sentinel node biopsy in melanoma: Technical considerations of the procedure as performed at the john wayne cancer institute. J Surg Oncol. 2010; 101: 669–676. doi: 10.1002/jso.21581.
61. Ruscito I, Gasparri ML, Braicu EI, Bellati F, Raio L, Sehouli J, Mueller MD, Panici PB, Papadia A. Sentinel Node Mapping in Cervical and Endometrial Cancer:Indocyanine Green Versus Other Conventional Dyes-A Meta-Analysis. Ann Surg Oncol. 2016; 23:3749–56. doi: 10.1245/s10434-016-5236-x.

