INTRODUCTION
Breast cancer is now recognized as a group of disease with significant heterogeneity, and traditional clinico-pathologic factors were no longer enough to meet the needs of prognosis prediction and treatment decision. In 2000, Perou et al characterized variation in gene expression patterns in a set of 65 breast tumor specimens using complementary DNA microarrays, and classified breast cancer into five intrinsic subtypes [1]. In 2012, The Cancer Genome Atlas Network (TCGA) produced a comprehensive catalogue of likely genomic drivers of the most common breast cancer subtypes [2]. Up to now, the management of breast cancer has entered into the era of molecular subtype with gene expression profiling.
Over the past decade, several multigene assays, based on findings of previous gene expression profiling, were developed and applied into routine practice of estrogen receptor(ER)-positive breast cancer. Among them, 21-gene recurrence score (RS, Oncotype DX) assay had gained most sufficient analytical as well as clinical validity [3–12]. 21-gene RS is a quantitative reverse transcriptase polymerase chain reaction (RT-PCR)–based test measuring 21 genes in formalin-fixed paraffin-embedded breast tumors. Retrospective studies from the NSABP B-14 showed that the RS predicted the likelihood of distant recurrence or breast cancer death in patients treated with endocrine therapy alone [3]. The ability of RS to predict benefit from adjuvant chemotherapy in node-negative or 1~3 node-positive patients was confirmed by retrospective analysis of the NSABP B-20 and SWOG-8814 trials [4, 5]. Furthermore, the latest prospective validation of RS in TAILORx (Trial Assigning IndividuaLized Options for Treatment) and West German Study Group PlanB trial revealed that among patients with hormone receptor (HR)-positive, human epidermal growth factor receptor2 (HER2)-negative, node-negative breast cancer and not treated with adjuvant chemotherapy, those with tumors that had a low-risk RS had very low rates of recurrence [11, 12].
Based on these findings, RS has changed patient treatment recommendation in 20% to 70% of cases and has resulted in a 13% to 34% reduction in adjuvant chemotherapy [13]. RS is now the only genomic test for breast cancer recommended by National Comprehensive Cancer Network (NCCN) which has the ability to predict response to adjuvant treatment [14]. However, due to the paucity of data in China, clinical application of RS in Chinese breast cancer patients remained unclear. The current study aimed to evaluate the distribution patterns of 21-gene RS in Chinese ER-positive, HER2-negative early breast cancer patients.
RESULTS
Distribution of RS
The baseline clinico-pathological features of the overall population are shown in Table 1. The median age at initial diagnosis was 56 years old (range: 27-93). Mastectomy was performed in 534(59.8%) patients. Seven hundred and ninety-five patients (88.7%) were diagnosed to have invasive ductal carcinoma (Table 2), and grade 1, 2, 3 tumors were documented in 17.0%, 57.9% and 25.1% of the patients. T1 and N0 tumors comprised 68.7% and 86.7% of all patients. Twenty-four patients (2.4%) had micrometastatic disease in their lymph nodes, and this part of patients was integrated into N0 subgroup for statistical analysis in the following part of the study.
Table 1: Characteristics of 980 eligible patients
Characteristics |
N |
% |
Valid % |
|---|---|---|---|
Age |
|
|
|
≤55 |
442 |
45.1 |
49.3 |
>55 |
454 |
46.3 |
50.7 |
Unknown |
84 |
8.6 |
|
Surgery |
|
|
|
Mastectomy |
534 |
54.5 |
59.8 |
BCS |
359 |
36.6 |
40.2 |
Unknown |
87 |
8.9 |
|
Pathology |
|
|
|
IDC |
795 |
81.1 |
88.7 |
Others |
101 |
10.3 |
11.3 |
Unknown |
84 |
8.6 |
|
Grade |
|
|
|
I |
135 |
13.8 |
17.0 |
II |
460 |
46.9 |
57.9 |
III |
199 |
20.3 |
25.1 |
Unknown |
186 |
19.0 |
|
T stage |
|
|
|
T1 |
608 |
62.0 |
68.7 |
T2-3 |
277 |
28.3 |
31.3 |
Unknown |
95 |
9.7 |
|
N stage |
|
|
|
N0 |
850 |
86.7 |
86.7 |
N1mi |
24 |
2.4 |
2.4 |
N1 |
106 |
10.8 |
10.8 |
ER |
|
|
|
Positive |
980 |
100 |
100 |
Negative |
0 |
0 |
0 |
PR |
|
|
|
Positive |
750 |
76.5 |
83.8 |
Negative |
145 |
14.8 |
16.2 |
Unknown |
85 |
8.7 |
|
Ki67 |
|
|
|
<14 |
430 |
43.9 |
49.7 |
≥14 |
436 |
44.5 |
50.3 |
Unknown |
114 |
11.6 |
|
HER2 |
|
|
|
Positive |
0 |
0 |
0 |
Negative |
980 |
100 |
100 |
Subtype |
|
|
|
Luminal A-like |
288 |
29.4 |
33.3 |
Luminal B-like |
578 |
59.0 |
66.7 |
Unknown |
114 |
11.6 |
|
RS category |
|
|
|
Low risk |
256 |
26.1 |
26.1 |
Intermediate risk |
483 |
49.3 |
49.3 |
High risk |
241 |
24.6 |
24.6 |
Abbreviations: BCS, breast conserving surgery; IDC, invasive ductal carcinoma; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth receptor 2; RS, recurrence score.
Table 2: Pathology of 980 eligible patients
Pathologic type |
N |
% |
Valid % |
|---|---|---|---|
Invasive ductal carcinoma |
795 |
81.1 |
88.7 |
Mucinous carcinoma |
50 |
5.1 |
5.6 |
Invasive lobular carcinoma |
33 |
3.4 |
3.7 |
Invasive papillary carcinoma |
7 |
0.7 |
0.8 |
Invasive micropapillary carcinoma |
5 |
0.5 |
0.6 |
Neuroendocrine carcinoma |
3 |
0.3 |
0.3 |
Tubular carcinoma |
2 |
0.2 |
0.2 |
Medullary carcinoma |
1 |
0.1 |
0.1 |
Unknown |
84 |
8.6 |
|
Total |
980 |
100 |
100 |
Of the 980 patient, median RS was 23 (range: 0~90), and RS followed a skewed distribution (Kolmogorov and Shapiro test, p<0.001, Figure 1). There were 256 patients (26.1%) in the low-risk group (RS<18), 483 patients (49.3%) in the intermediate-risk group (RS 18 to 30), and 241 patients (24.6%) in the high-risk group (RS≥31).
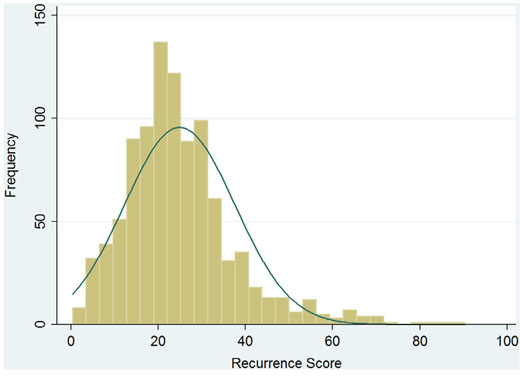
Figure 1: Recurrence score distribution of 980 eligible patients. No captions.
The distribution of RS varied significantly according to different tumor grade, T stage, PR status, Ki67 index and molecular subtypes (all p<0.05, Table 3). High grade, large tumor size, negative PR, high Ki67 index and luminal B subtype were more likely to have high-risk RSs (Figure 2). In grade 1 tumors, the proportion of low, intermediate, or high-risk patients were 40.0%, 50.4%, and 9.6%, whereas it was 13.1%, 39.7% and 47.2% in patients with grade 3 tumors (p<0.001). Likewise, T1, PR-positive and low-Ki67 tumors were more likely to be categorized as low or intermediate risk and less likely to be categorized as high risk compared with T2-3, PR-negative and high-Ki67 tumors (all p<0.05).
Table 3: Distribution of RS according to clinico-pathologic factors
Characteristics |
Total N |
RS category, N (%) |
P value |
||
|---|---|---|---|---|---|
Low risk |
Intermediate risk |
High risk |
|||
Age |
896 |
|
|
|
0.325 |
≤55 |
|
106(24.0) |
221(50.0) |
115(26.0) |
|
>55 |
|
129(28.4) |
213(46.9) |
112(24.7) |
|
Surgery |
893 |
|
|
|
0.167 |
Mastectomy |
|
152(28.5) |
254(47.6) |
128(24.0) |
|
BCS |
|
83(23.1) |
177(49.3) |
99(27.6) |
|
Pathology |
896 |
|
|
|
0.321 |
IDC |
|
204(25.7) |
384(48.3) |
207(26.0) |
|
Others |
|
31(30.7) |
50(49.5) |
20(19.8) |
|
Grade |
794 |
|
|
|
<0.001 |
I |
|
54(40.0) |
68(50.4) |
13(9.6) |
|
II |
|
125(27.2) |
238(51.7) |
97(21.1) |
|
III |
|
26(13.1) |
79(39.7) |
94(47.2) |
|
T stage |
885 |
|
|
|
0.017 |
T1 |
|
172(28.3) |
299(49.2) |
137(22.5) |
|
T2-3 |
|
62(22.4) |
129(46.6) |
86(31.0) |
|
N stage |
980 |
|
|
|
0.686 |
N0 |
|
232(26.5) |
429(49.1) |
213(24.4) |
|
N1 |
|
24(22.6) |
54(50.9) |
28(26.4) |
|
PR |
895 |
|
|
|
<0.001 |
Positive |
|
221(29.5) |
372(49.6) |
157(20.9) |
|
Negative |
|
14(9.7) |
61(42.1) |
70(48.3) |
|
Ki67 |
866 |
|
|
|
<0.001 |
<14 |
|
137(31.9) |
226(52.6) |
67(15.6) |
|
≥14 |
|
88(20.2) |
200(45.9) |
148(33.9) |
|
Subtype |
866 |
|
|
|
<0.001 |
Luminal A-like |
|
117(40.6) |
143(49.7) |
28(9.7) |
|
Luminal B-like |
|
108(18.7) |
283(49.0) |
187(32.4) |
|
Abbreviations: BCS, breast conserving surgery; IDC, invasive ductal carcinoma; PR, progesterone receptor; RS, recurrence score.
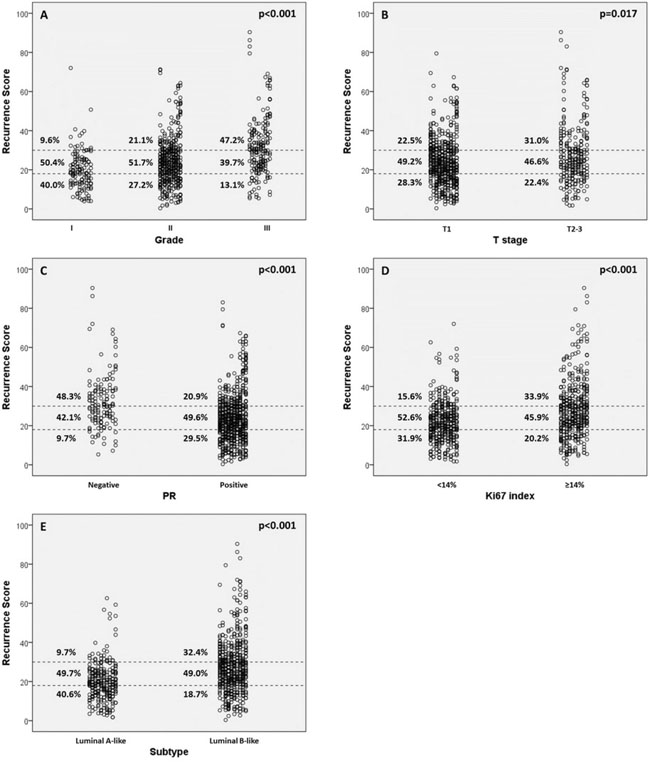
Figure 2: Recurrence score (RS) categories vs. clinic-pathologic factors. (A) RS vs grade; (B) RS vs clinical t stage; (C) RS vs progesterone receptor (PR); (D) RS vs Ki-67 index; (E) RS vs molecular subtype.
Among node-negative patients, all these factors remained significantly associated with RS (all p<0.05), whereas in 1~3 node-positive patients, T stage no longer had a significant correlation with RS.
Multivariate analysis
Logistic regression analysis demonstrated that grade, PR, and Ki67 were independent variables associated with RS (Table 4). Grade 2 and 3 were associated with significantly higher odds of intermediate(Grade 2 vs Grade 1, OR: 1.576, 95%CI: 1.006~2.470, p=0.047; Grade 3 vs Grade 1, OR: 2.607, 95%CI: 1.363~4.988, p=0.004) and high-risk(Grade 2 vs Grade 1, OR: 2.709, 95%CI: 1.309~5.606, p=0.007; Grade 3 vs Grade 1, OR: 10.035, 95%CI: 4.222~23.854, p<0.001) RSs. Patients with PR-negative tumors were more likely to have intermediate (OR: 4.397, 95%CI: 1.952~9.901, p<0.001) and high-risk (OR: 12.039, 95%CI: 5.207~27.836, p<0.001) RSs compared with those with PR-positive tumors. In addition, proportion of high-risk RS was also significantly higher among patients with high Ki67 index (OR: 1.719, 95%CI: 1.051~2.812, p=0.031). Tumor size had no significant impact on RS categories.
Table 4: Multivariate analysis of independent variables associated with RS
Characteristics |
Intermediate vs low risk |
High vs low risk |
||||
|---|---|---|---|---|---|---|
OR |
95%CI |
P value |
OR |
95%CI |
P value |
|
All patients(N=980) |
||||||
Grade |
|
|
|
|
|
|
I |
1 |
|
|
1 |
|
|
II |
1.576 |
1.006~2.470 |
0.047 |
2.709 |
1.309~5.606 |
0.007 |
III |
2.607 |
1.363~4.988 |
0.004 |
10.035 |
4.222~23.854 |
<0.001 |
T stage |
|
|
NS |
|
|
NS |
PR |
|
|
<0.001 |
|
|
<0.001 |
Positive |
1 |
|
|
1 |
|
|
Negative |
4.397 |
1.952~9.901 |
|
12.039 |
5.207~27.836 |
|
Ki67 |
|
|
NS |
|
|
0.031 |
<14 |
|
|
|
1 |
|
|
≥14 |
|
|
|
1.719 |
1.051~2.812 |
|
Node-negative patients(N=874) |
||||||
Grade |
|
|
|
|
|
|
I |
1 |
|
|
1 |
|
|
II |
1.662 |
1.041~2.655 |
0.033 |
2.724 |
1.303~5.694 |
0.008 |
III |
2.463 |
1.241~4.889 |
0.010 |
9.730 |
3.994~23.701 |
<0.001 |
PR |
|
|
<0.001 |
|
|
<0.001 |
Positive |
1 |
|
|
1 |
|
|
Negative |
4.451 |
1.969~10.063 |
|
10.760 |
4.604~25.149 |
|
Ki67 |
|
|
NS |
|
|
NS |
Node-positive patients(N=106) |
||||||
Grade |
|
|
NS |
|
|
NS |
PR |
|
|
NS |
|
|
NS |
Ki67 |
|
|
NS |
|
|
NS |
Abbreviations: PR, progesterone receptor; OR, odds ratio; CI, confidence interval; NS, not significant.
As for node-negative patients, both grade and PR status remained to be independent factors associated with RS. Conversely, no factors was identified to be correlated with RS in node-positive patients (Table 4).
Relationship between IHC and RT-PCR results
IHC and RT-PCR of ER were concordant in 98.8% of all cases (98.7% and 99.1% in node-negative and node-positive patients, Table 5, Figure 3A). The correlation between IHC and RT-PCR of ER as a continuous variable was weak but statistically significant (all cases, r=0.362; node-negative cases, r=0.356; node-positive cases, r=0.418; all p<0.001). The Spearman’s rank correlation between RS and ER IHC was -0.224, -0.213 and -0.328 in overall, node-negative and node-positive patients (all p<0.001).
Table 5: Relationship between IHC and RT-PCR results
|
All patients |
Node-negative patients |
Node-positive patients |
||||||
|---|---|---|---|---|---|---|---|---|---|
ER |
PR |
Ki67 |
ER |
PR |
Ki67 |
ER |
PR |
Ki67 |
|
Concordance between IHC and RT-PCR (%) |
98.8 |
88.3 |
/ |
98.7 |
87.5 |
/ |
99.1 |
94.3 |
/ |
Spearman’s correlation between IHC and RT-PCR |
0.362 |
0.676 |
0.446 |
0.356 |
0.683 |
0.449 |
0.418 |
0.624 |
0.462 |
Spearman’s correlation between IHC and RS |
-0.224 |
-0.434 |
0.303 |
-0.213 |
-0.439 |
0.290 |
-0.328 |
-0.397 |
0.402 |
Abbreviations: IHC, immunohistochemistry; RT-PCR, reverse transcriptase-polymerase chain reaction; RS, recurrence score; ER, estrogen receptor; PR, progesterone receptor.
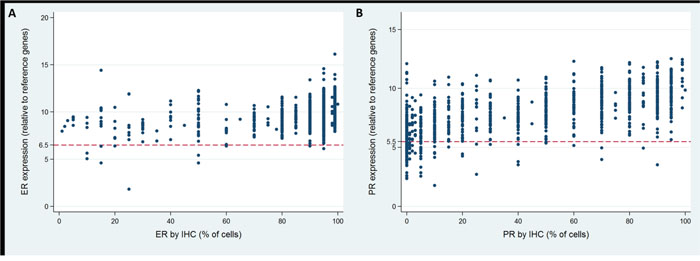
Figure 3: Concordance of estrogen receptor (ER) and progesterone receptor (PR) status between RT-PCR and IHC. (A) ER; (B) PR.
PR status between IHC and RT-PCR was concordant in 88.3% of all cases (87.5% and 94.3% in node-negative and node-positive patients, Table 5, Figure 3B). Comparing the continuous IHC and RT-PCR results of PR, a moderate relationship was observed (all cases, r=0.676; node-negative cases, r=0.683; node-positive cases, r=0.624; all p<0.001). Using Spearman’s rank correlation, the relationship between RS and PR IHC results was calculated to be -0.434, -0.439 and -0.397 in overall, node-negative and node-positive patients (all p<0.001).
Regarding Ki67 index, there was a weak to moderate correlation between IHC and RT-PCR (all cases, r=0.446; node-negative cases, r=0.449; node-positive cases, r=0.462; all p<0.001, Table 5). The Spearman’s rank correlation between RS and Ki67 IHC was 0.303, 0.290 and 0.402 in overall, node-negative and node-positive patients respectively (all p<0.001).
DISCUSSION
The current study assessed the patterns of distribution of 21-gene RS in Chinese ER-positive, HER2-negative, node-negative or 1~3 node-positive breast cancer patients. To the best our knowledge, this study had a sample size larger than any others reported before regarding RS in Chinese population. In our study, RS was demonstrated to be significantly associated with grade, tumor size, PR status, Ki67 index and molecular subtype. In grade 3, T 2~3, PR-negative, high Ki67 and luminal-B tumors, the proportion to have a high-risk RS ranged from 31.0% to 48.3%, significantly higher than their counterparts. Similar to our findings, retrospective study of NSABP B-20 trial revealed that 42%, 30% and 51% patients with grade 3, T 2~3, PR-negative tumors were categorized as high-risk [4]. In PlanB trial, RS (low risk: RS≤11, intermediate risk: RS 12~25, high risk: RS≥26) was also found to be significantly associated with grade, PR status and Ki67 index [12]. Besides, the distribution of RS differed little across subgroups with different lymph node status. Multivariate analysis in our study indicated that poorly-differentiated, PR-negative or high-proliferating tumors had much higher probabilities to have increased RSs (grade 2/3 and negative-PR predictive for high and intermediate-risk RS, Ki67≥14% predictive for high-risk RS, Table 4). These facts indicated that routine clinical and pathologic parameters might be of help in predicting RS.
Discordance between clinic-pathologic factors and RS was also noted in our study. Some patients with favorable clinic-pathologic factors were found to display high-risk RS (e.g. 9.6% of patients with grade 1 tumors were high-risk). On the other hand, there were cases of tumors with low-risk RS that had unfavorable clinic-pathologic factors (e.g. 13.1% of patients with grade 3 tumors were low-risk). These results suggested that RS might provide not only integration of cancer-related genes and quantification of risk assessment, but also additional prognostic information beyond routine biologic parameters of individual breast cancers in Chinese patients.
Of note, the proportions of patients categorized as having a low, intermediate, or high risk were 26.1%, 49.3% and 24.6% in our study, different from that of the NSABP B-14 trial (low risk: 51%, intermediate risk: 22%, high risk: 27%). Similar findings were also reported in the latest studies of RS. In TAILORx trial, the first prospective validation study of RS, the proportion of patients having a low-risk RS was lower than previously reported results of the NSABP B-14 and B-20 trials, despite the different definition of risk categories [3, 4, 11]. Moreover, the distribution of scores observed by the commercial laboratory (Genomic Health) also differed from the initial report [11]. We noticed that the disparities of distribution mainly existed in low-risk and intermediate groups, whereas the proportion of high-risk RS in our study was similar to previous retrospective reports. As mentioned above, the proportions of high-risk RS in subgroups of unfavorable biologic markers (grade 3, T2~3, PR negative) in our study were quite comparable to that of the NSABP B-20 trial. We postulate that those discordance may be due to clinicians selecting patients for the enrolling into those clinical trials in whom there was therapeutic equipoise regarding the treatment benefit of experimental arms (e.g. extended tamoxifen group in NSABP B-14 trial and chemotherapy group in TAILORx trial). Conversely, we consecutively recruited eligible patients in our center in the past few years, which may be more close to actual distribution of risk groups in clinical practice. Furthermore, the distribution of RS might differ between Eastern and Western populations, and this inconsistent RS distribution needs further validation.
There has been growing interest in molecular biomarker testing by RT-PCR with conflicting results [15–20]. In our study, ER displayed a very high concordance of 98.8% between IHC and RT-PCR, in accordance with the literature (93% to 100%). Spearman’s rank correlation showed that ER status by IHC had a positively weak relationship with ER by RT-PCR (r=0.362) and a negatively weak relationship with RS (r=-0.224). Previous studies reported that concordant cases had higher ER expression while discordant cases exhibited lower level of ER expression, which might partly account for the false-negative results by RT-PCR [15]. According to the present study, rarely ER by RT-PCR incorrectly determine ER status. Nonetheless, it’s so far contraindicated to deny hormone therapies according to RT-PCR assay that would otherwise be indicated.
In the case of PR, a slightly lower concordance rate (88.3%) was detected than for ER. Similar concordance rates (87.7% to 94.2%) were reported in previous studies [15–20]. Meanwhile, spearman’s rank correlation revealed that PR by IHC had a positively moderate relationship with PR by RT-PCR(r=0.676) and a negatively moderate relationship with RS(r=-0.434), higher than that of ER. This is comparable with some reports [15, 17–19], and lower than some others [16, 20]. Possible explanations for the lower concordance of PR status than ER was the change in the clones used to generate the ER and PR antibodies used for IHC and the positive staining criteria within the period of our study at our institution.
Limitations of this study could be the retrospective design and lack of follow-up. The follow-up visits is ongoing and it’s immature to draw any conclusions regarding the prognostic significance of RS at this moment. Nonetheless, our study delineated the distribution patterns of RS across different biologic subgroups in Chinese ER-positive, HER2-negative early breast cancer patients for the first time, and explored the relationship of between molecular biomarker by IHC and 21-gene RT-PCR assay. Our findings appear to be plausible and warrant confirmation in further studies with long-term follow-up.
In Conclusion, 21-gene RS correlated significantly with tumor grade, T stage, PR status, Ki67 index and subtypes in Chinese ER-positive, HER2-negative early breast cancer patients. Grade, PR status and Ki67 index could independently predict RS. ER, PR status and Ki67 index between RT-PCR and IHC test had remarkable concordance.
PATIENTS AND METHODS
Study population
Nine hundred and eighty consecutive invasive breast cancer patients who underwent radical surgery between January 2009 and May 2016 in Shanghai Ruijin Hospital were retrospectively reviewed. Women with axillary node-negative or 1~3 nodes-positive breast cancer were eligible for inclusion if they had ER positive and HER2 negative tumors. Exclusion criteria were: T1a or T4 tumor, metastatic breast cancer, and previous neoadjuvant treatment. Following data were also collected: age at initial diagnosis, surgery type, pathology, histologic grade, progesterone receptor (PR) status and Ki67 index. Patients were subdivided into luminal A-like and luminal B-like subtypes according to 2013 St. Gallen Expert Consensus [21]. This retrospective study has been approved by the Ethical Committees of Shanghai Ruijin Hospital. The results of this study do not affect the treatment decision of any patient enrolled. All the clinical and pathological data was collected only after the written informed consent form was obtained from the patient.
Analytic methods of recurrence score
The RS was determined from formalin-fixed, paraffin-embedded tissue as previously described [3]. In brief, hematoxylin and eosin-stained slides were reviewed to ensure sufficient invasive breast cancer (pathologist XC Fei), and then RNA was extracted from two 10μm unstained sections. Total RNA content was measured, and the absence of DNA contamination was verified. Gene-specific reverse transcription was performed followed by standardized quantitative RT-PCR reactions in 96 well plates using Applied Biosystems (Foster City, CA) 7500 Real-Time PCR System. Expression of each gene was measured in triplicate, and normalized relative to a set of five reference genes. Reference-normalized expression measurements range from 0 to 15, and a 1-unit increase reflected approximately a two-fold increase in RNA. A tumor is considered ER-negative if expression units <6.5, ER-positive ≥6.5, PR-negative <5.5, PR-positive ≥5.5 [15]. The RS, ranging from 0 to 100, was derived from the reference-normalized expression measurements for the 16 cancer-related genes. Patients were then categorized into low-risk (RS<18), intermediate-risk (RS 18~30), and high-risk (RS≥31) groups [3].
Immunohistochemistry (IHC)
Positive staining for ER/PR was defined as nuclear staining in ≥1% of the tumor cells. Negative HER2 status was considered as 0 to 1+ by IHC or negative on FISH. Ki-67 index was characterized as the proportion of positively nuclear staining cells among at least 1000 tumor cells in the area counted. The following antibodies were used for the IHC test: ER: clone 1D5 (rabbit monoclonal, Gene), PR: clone PR636 (mouse monoclonal, Dako), HER2: 4B5 (rabbit monoclonal, Roche), Ki67: MIB-1 (mouse monoclonal, Dako).
Statistical analysis
The Chi-square test were applied to evaluate the distribution of RS risk category among patients with different clinico-pathologic factors. Fisher’s exact test was performed when necessary. Multivariate logistic regression analysis was used to estimate the odds ratio (OR), 95% confidence interval (CI), and the association between clinical and pathological variables and RS. Spearman’s rank correlation was used to compare the results of ER, PR and Ki67 on RT-PCR and IHC. All statistical tests were two sided and p<0.05 was considered significant. The software package STATA (version 14.0, College Station, TX, US) for Windows 10 was used for analysis.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
FUNDING
This work was supported by grants from the Joint Research Project of the Emerging Cutting-edge Technology of Shanghai Shen-kang Hospital Development Center (Grant Number: SHDC12014103), Medical Guidance Foundation of Shanghai Municipal Science and Technology Commission (15411966400) and Technology Innovation Act Plan of Shanghai Municipal Science and Technology Commission (Grant Number: 14411950200, 14411950201). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
REFERENCES
1. Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, et al. Molecular portraits of human breast tumours. Nature. 2000; 406:747-52. doi: 10.1038/35021093.
2. Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012; 490:61-70. doi: 10.1038/nature11412.
3. Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, Baehner FL, Walker MG, Watson D, Park T, Hiller W, Fisher ER, Wickerham DL, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004; 351:2817-26. doi: 10.1056/NEJMoa041588.
4. Paik S, Tang G, Shak S, Kim C, Baker J, Kim W, Cronin M, Baehner FL, Watson D, Bryant J, Costantino JP, Geyer CE Jr, Wickerham DL, et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol. 2006; 24:3726-34. doi: 10.1200/JCO.2005.04.7985.
5. Albain KS, Barlow WE, Shak S, Hortobagyi GN, Livingston RB, Yeh IT, Ravdin P, Bugarini R, Baehner FL, Davidson NE, Sledge GW, Winer EP, Hudis C, et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 2010; 11:55-65. doi: 10.1016/S1470-2045(09)70314-6.
6. Dowsett M, Cuzick J, Wale C, Forbes J, Mallon EA, Salter J, Quinn E, Dunbier A, Baum M, Buzdar A, Howell A, Bugarini R, Baehner FL, et al. Prediction of risk of distant recurrence using the 21-gene recurrence score in node-negative and node-positive postmenopausal patients with breast cancer treated with anastrozole or tamoxifen: a TransATAC study. J Clin Oncol. 2010; 28:1829-34. doi: 10.1200/JCO.2009.24.4798.
7. Goldstein LJ, Gray R, Badve S, Childs BH, Yoshizawa C, Rowley S, Shak S, Baehner FL, Ravdin PM, Davidson NE, Sledge GW Jr, Perez EA, Shulman LN, et al. Prognostic utility of the 21-gene assay in hormone receptor-positive operable breast cancer compared with classical clinicopathologic features. J Clin Oncol. 2008; 26:4063-71. doi: 10.1200/JCO.2007.
8. Mamounas EP, Tang G, Fisher B, Paik S, Shak S, Costantino JP, Watson D, Geyer CE Jr, Wickerham DL, Wolmark N. Association between the 21-gene recurrence score assay and risk of locoregional recurrence in node-negative, estrogen receptor-positive breast cancer: results from NSABP B-14 and NSABP B-20. J Clin Oncol. 2010; 28:1677-83. doi: 10.1200/JCO.2009.23.7610.
9. Tang G, Cuzick J, Costantino JP, Dowsett M, Forbes JF, Crager M, Mamounas EP, Shak S, Wolmark N. Risk of recurrence and chemotherapy benefit for patients with node-negative, estrogen receptor-positive breast cancer: recurrence score alone and integrated with pathologic and clinical factors. J Clin Oncol. 2011; 29:4365-72. doi: 10.1200/JCO.2011.35.3714.
10. King TA, Lyman JP, Gonen M, Voci A, De Brot M, Boafo C, Sing AP, Hwang ES, Alvarado MD, Liu MC, Boughey JC, McGuire KP, Van Poznak CH, et al. Prognostic Impact of 21-Gene Recurrence Score in Patients With Stage IV Breast Cancer: TBCRC 013. J Clin Oncol. 2016; 34:2359-65. doi: 10.1200/JCO.2015.63.1960.
11. Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, Geyer CE Jr, Dees EC, Perez EA, Olson JA Jr, Zujewski J, Lively T, Badve SS, et al. Prospective Validation of a 21-Gene Expression Assay in Breast Cancer. N Engl J Med. 2015; 373:2005-14. doi: 10.1056/NEJMoa1510764.
12. Gluz O, Nitz UA, Christgen M, Kates RE, Shak S, Clemens M, Kraemer S, Aktas B, Kuemmel S, Reimer T, Kusche M, Heyl V, Lorenz-Salehi F, et al. West German Study Group Phase III PlanB Trial: First Prospective Outcome Data for the 21-Gene Recurrence Score Assay and Concordance of Prognostic Markers by Central and Local Pathology Assessment. J Clin Oncol. 2016; 34:2341-9. doi: 10.1200/JCO.2015.63.5383.
13. Hornberger J, Alvarado MD, Rebecca C, Gutierrez HR, Yu TM, Gradishar WJ. Clinical validity/utility, change in practice patterns, and economic implications of risk stratifiers to predict outcomes for early-stage breast cancer: a systematic review. J Natl Cancer Inst. 2012; 104:1068-79. doi: 10.1093/jnci/djs261.
14. Gradishar WJ, Anderson BO, Balassanian R, Blair SL, Burstein HJ, Cyr A, Elias AD, Farrar WB, Forero A, Giordano SH, Goetz M, Goldstein LJ, Hudis CA, et al. NCCN clinical practice guidelines in oncology: Breast Cancer Version I.2016. http://nccn.org. Accessed February 24, 2016.
15. Park MM, Ebel JJ, Zhao W, Zynger DL. ER and PR immunohistochemistry and HER2 FISH versus oncotype DX: implications for breast cancer treatment. Breast J. 2014; 20:37-45. doi: 10.1111/tbj.12223.
16. Kraus JA, Dabbs DJ, Beriwal S, Bhargava R. Semi-quantitative immunohistochemical assay versus oncotype DX(®) qRT-PCR assay for estrogen and progesterone receptors: an independent quality assurance study. Mod Pathol. 2012; 25:869-76. doi: 10.1038/modpathol.2011.219.
17. Badve SS, Baehner FL, Gray RP, Childs BH, Maddala T, Liu ML, Rowley SC, Shak S, Perez EA, Shulman LJ, Martino S, Davidson NE, Sledge GW, et al. Estrogen- and progesterone-receptor status in ECOG 2197: comparison of immunohistochemistry by local and central laboratories and quantitative reverse transcription polymerase chain reaction by central laboratory. J Clin Oncol. 2008; 26:2473-81. doi: 10.1200/JCO.2007.13.6424.
18. Chaudhary LN, Jawa Z, Szabo A, Visotcky A, Chitambar CR. Relevance of progesterone receptor immunohistochemical staining to Oncotype DX recurrence score. Hematol Oncol Stem Cell Ther. 2016; 9:48-54. doi: 10.1016/j.hemonc.2015.12.001.
19. Khoury T, Yan L, Liu S, Bshara W. Oncotype DX RT-qPCR assay for ER and PR correlation with IHC: a study of 3 different clones. Appl Immunohistochem Mol Morphol. 2015; 23:178-87. doi: 10.1097/PAI.0000000000000078.
20. O'Connor SM, Beriwal S, Dabbs DJ, Bhargava R. Concordance between semiquantitative immunohistochemical assay and oncotype DX RT-PCR assay for estrogen and progesterone receptors. Appl Immunohistochem Mol Morphol. 2010; 18:268-72. doi: 10.1097/PAI.0b013e3181cddde9.
21. Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, Senn HJ; Panel members. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013; 24:2206-23. doi: 10.1093/annonc/mdt303.

