INTRODUCTION
Liver cancer is a malignancy of high incidence and mortality. It is estimated that nearly 750,000 new cases and more than 690,000 cancer deaths occur annually worldwide. Unfortunately, half of these cases were estimated to occur in China [1]. Among the liver cancers, hepatocellular carcinoma (HCC) is the major histological subtype. Various anticancer drugs such as tamoxifen, octreotide, interferon, and interleukin-2 are used in the treatment of HCC; however, these drugs are not specific for HCC and demonstrate low treatment efficacy [2]. Multidrug resistance (MDR) to anticancer drugs is the most common cause of failure in cancer chemotherapy [3].
The mechanisms of resistance to anticancer drugs consist of changes of pharmacokinetic or tumor micro-environment, cancer cell-specific factors which include increased drug efflux or decreased drug influx, drug inactivation, drug target modification, and apoptosis evasion [4]. Of these mechanisms, the increased drug efflux/reduced drug accumulation in the tumor cell appears to be a very common mechanism of MDR. Hydrophobic drugs are expelled from cells by activation of energy-dependent transport systems [5, 6]. The first of these transporters to be identified was P-glycoprotein (P-gp), the product of the human MDR1 gene [7]. P-gp is a member of the large adenosine triphosphate (ATP)-binding cassette (ABC) family of proteins, also known as ABCB1. P-gp/ABCB1 has a molecular weight of 170 kDa and comprises of two nucleotide-binding domains (NBD1 and NBD2) and two transmembrane-binding domains (TMD1 and TMD2) [8, 9]. P-gp/ABCB1 utilizes energy from the hydrolysis of ATP to efflux drugs from intracellular to extracellular matrix, leading to decreased intracellular drug concentration [10]. Overexpression of P-gp/ABCB1 can produce MDR in cancer cells [11].
Over the past few decades, efforts were made to look for new compounds as resistance reversal agents to overcome MDR in tumor cells. Verapamil was one of the first generation of these MDR reversal agents [12]. But the effective concentration of verapamil in reversing MDR in vitro was too high to be achieved in vivo [13]. The dose of verapamil required was much larger than the clinically safe dose, resulting in toxic reactions in almost all patients. Although second and third generations of reversal agents were explored, they were inhibited respectively by P450 3A4 enzymes and by anticancer drugs [14–16]. In order to obtain more effective and safer resistance reversal agents, some natural products and their derivatives have been considered for use in combination with anticancer drugs [17].
Epigallocatechin-3-gallate (EGCG) is one of the MDR reversal modulators (Figure 1A) [18]. It is the most abundant catechin in green tea polyphenols. A previous study revealed that EGCG could significantly inhibit the proliferation of human HCC cell BEL-7404/DOX in vitro and the tumor growth of the xenograft mouse model when it was administrated at lower doses with doxorubicin, compared to treatment with doxorubicin alone [19]. However, many phenolic hydroxyl groups are included in the structure of EGCG, which makes the compound unstable due to rapid oxidation, low lipid solubility, low bioavailability, and short duration of action. Therefore, its application became limited [20]. Y6 is an ethylation product of EGCG with strong stability (Figure 1B). The above limitations were overcome in Y6 because most of the phenolic hydroxyl groups were replaced by ethyl groups. In the present study, we evaluate the potential effect of Y6 as a reversal agent that specifically reverses ABC transporter-mediated MDR in vitro.
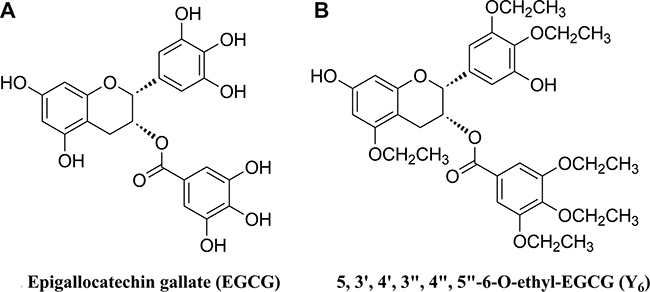
Figure 1: Chemical structures of two monomers of catechin.
RESULTS
Y6 improved the chemosensitivity of BEL-7404/DOX cells to doxorubicin treatment
HCC cell lines (parental BEL-7404 and doxorubicin-selected resistant BEL-7404/DOX cells) were used to examine the cytotoxicity of doxorubicin, EGCG, and Y6 on cell proliferation using the 3-(4, 5-dimethylthiazol-2-y1)-2, 5-diphenyltetrazolium bromide (MTT) assay. As shown in Figure 2, higher concentration of doxorubicin is required to inhibit the same ratio of BEL-7404/DOX cell proliferation than that required in BEL-7404 cells. The IC50 value (44.14 μM) for doxorubicin in the BEL-7404/DOX cells was 29.2 times higher than that (1.51 μM) in the parental BEL-7404 cells (Table 1). The IC50 values of Y6 and EGCG in this pair of cell groups were > 50 μM. The concentrations for combined use with doxorubicin were non-cytotoxic and below the IC10 values (Figure 2).
Table 1: Reversal effect of Y6, EGCG, and VER in BEL-7404 cells and BEL-7404/DOX cells
Compounds |
IC50 ± SD (μM)a (Resistance folds)b |
|
|---|---|---|
BEL-7404 |
BEL-7404/DOX |
|
DOX |
1.51 ± 0.14 (1.0) |
44.14 ± 3.97 (29.2) |
+VER 10 μM |
1.47 ± 0.13 (1.0) |
8.39 ± 1.43 (5.6)* |
+EGCG 10 μM |
1.49 ± 0.32 (1.0) |
8.04 ± 1.08 (5.3)* |
+Y6 10 μM |
1.29 ± 0.21 (0.9) |
5.74 ± 0.73 (3.8)*# |
+Y6 15 μM |
1.15 ± 0.31 (0.8) |
4.36 ± 0.15 (2.9)*# |
Cell survival was determined by MTT assay. a: Values represent means ± SDs of at least three independent experiments performed in triplicate. b: The fold reversal of MDR (values given in parentheses) was calculated by dividing the IC50 values of substrate in BEL-7404/DOX cells in the presence or absence of an inhibitor, or BEL-7404 cells with inhibitors, by the IC50 of BEL-7404cells without an inhibitor. *p < 0.05, significantly different from values obtained in the absence of an inhibitor; #p < 0.05, significantly different from values obtained in the presence of inhibitors VER or EGCG.
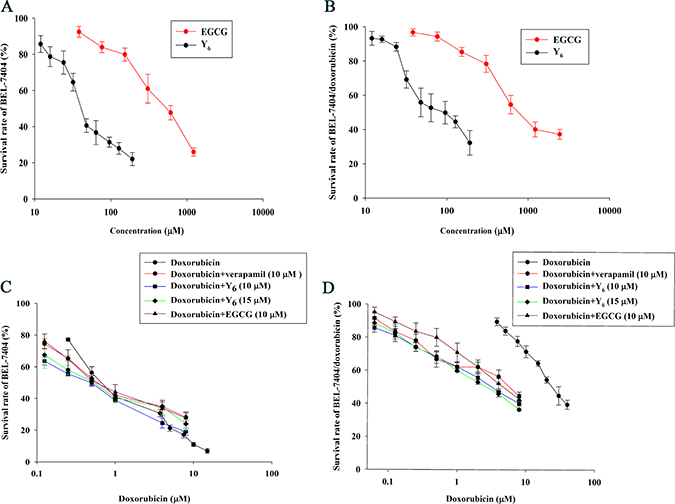
Figure 2: The Reversal effect of Y6, EGCG, and verapamil in parental and resistant cells. (A) The cytotoxicity of EGCG and Y6 in the parental cell line BEL-7404. (B) The cytotoxicity of EGCG and Y6 in the doxorubicin-resistance cell line BEL-7404/DOX. (C) (D) the concentration-response curves of hepatocellular carcinoma cell lines (C) BEL-7404 and (D) BEL-7404/DOX treated with doxorubicin alone and doxorubicin combined with verapamil, EGCG, or Y6. Cell survival rate was determined by MTT assay. Points with error bars represent the mean ± SD. Experiments were performed for three independent times.
The non-toxic doses of verapamil, EGCG, and Y6 were individually combined with a series of doxorubicin concentrations, and were used to treat BEL-7404 cells and BEL-7404/DOX cells. Among them, verapamil, a known chemosensitizer, was used as the positive control inhibitor of P-gp in this experiment. The MTT assay results showed that the IC50 values for doxorubicin was significantly reduced from 44.14 to 8.39 μM by verapamil, to 8.04 μM by EGCG, and to 5.74 μM and 4.36 μM by Y6 (10 μM) and Y6 (15 μM) respectively in the BEL-7404/DOX cells compared with doxorubicin alone, representing drug resistance folds decreasing from 29.2 to 5.6, 5.3, 3.8, and 2.9 respectively (Table 1). Combination groups were compared with the doxorubicin alone group, and the differences were statistically significant (*p < 0.05). These results indicated that the capability of Y6 in reversing drug resistance was higher than that of EGCG combined with doxorubicin (#p < 0.05) (Table 1, Figure 2).
Y6 induced apoptosis in BEL-7404/DOX cells
The induction of cell apoptosis is a common mechanism for many anti-tumor drugs. To examine whether Y6 can induce cell apoptosis, we detected apoptotic cells in HCC BEL-7404/DOX cells treated with verapamil (10 μM), EGCG (10 μM), and Y6 (10 and 15 μM) in combination with doxorubicin (10 μM) and compared with doxorubicin (10 μM) alone. The cells that treated with verapamil were served as the positive controls of P-gp inhibitors. BEL-7404/DOX cells were incubated in anoxic condition for 48 h, then subjected to Annexin V-FITC labeling and Propidium iodide (PI) staining as described in the Materials and Methods section. We used flow cytometry analysis to determine the apoptotic rate of BEL-7404/DOX cells treated with the drug combinations or doxorubicin alone. The results showed that after treatment with any drug combination, the number of cells increased in late apoptotic stage (Table 2, Figure 3). Only 12.17% of cells showed apoptotic signals when treated with doxorubicin alone, but the percentage increased to 17.91% with verapamil (10 μM), to 19.52% with EGCG (10 μM), to 27.89% with Y6 (10 μM) and to 40.03% withY6 (15 μM). The differences were statistically significant when compared with the doxorubicin group (*p < 0.05). Furthermore, at the same concentration, Y6 had a higher effect than verapamil and EGCG on the induction of apoptosis in the late apoptotic stage, and the differences were statistically significant (#p < 0.05). In addition, the higher the concentrations of Y6, the more cell apoptosis in the BEL-7404/DOX cells.
Table 2: The late stage apoptosis rates in BEL-7404/DOX cells (n = 3)
Drugs (μM) |
Apoptosis rate (%) ± SD (μM)a |
|---|---|
Controlb |
5.67 ± 1.10 |
DOX 10 μMc |
12.17 ± 1.26 |
+VER 10 μMd |
17.91 ± 3.35* |
+EGCG 10 μMd |
19.52 ± 4.41* |
+Y6 10 μMd |
27.89 ± 2.53*# |
+Y6 15 μMd |
40.03 ± 3.21*# |
a: Values represent means ± SDs of at least three independent experiments performed in triplicate. b: Control group, no drugs were used; c: DOX group, 10 μM of DOX alone was used; d: 10 μM of DOX combined with 10 μM of VER, 10 μM of EGCG, 10 μM of Y6, or 15 μM of Y6 were used; (VER 10 μM+DOX 10 μM) group was used as the positive control; *p < 0.05 vs. the DOX alone group. #p < 0.05 vs. the (VER 10 μM +DOX 10 μM) group or (EGCG 10 μM +DOX 10 μM) group.
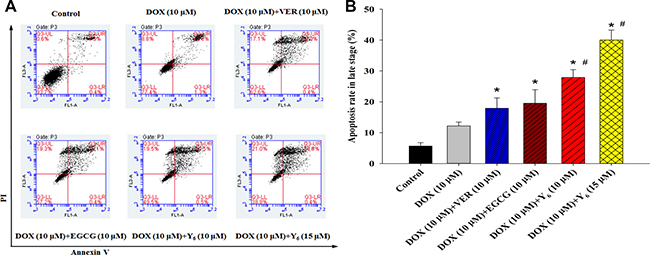
Figure 3: Apoptosis was measured in BEL-7404/DOX cell line. (A) Apoptotic cells as a result of Y6, EGCG, and verapamil treatment were quantified by the Annexin V/PI assay. Cells were stained using an Annexin V/PI staining kit and then detected by flow cytometry. (B) *p < 0.05 vs. the doxorubicin alone group. #p < 0.05 vs. the (verapamil 10 μM+doxorubicin 10 μM) group or (EGCG 1 0 μM+doxorubicin 10 μM) group. Experiments were performed for three independent times.
Y6 inhibited the expression of hypoxia-inducible factor-1α(hif-1α) and MDR1 mRNA in BEL-7404/DOX cells
Previous studies have shown that high MDR1-gene expression exists in BEL-7404/DOX cells compared with BEL-7404 cells [21, 22]. Therefore, differences in expressions of hif-1α and MDR1 genes were investigated by quantitative real time polymerase chain reaction (RT-PCR) analysis after drug treatment. The results showed that the expression of hif-1α was significantly lower in the combination drug therapy groups (doxorubicin plus EGCG, verapamil, or Y6) than in the doxorubicin alone group. Expression of hif-1α was 0.90 in BEL-7404/DOX cells treated with doxorubicin (10 μM) alone, but it decreased to 0.65 with verapamil (10 μM), to 0.49 with EGCG (10 μM), to 0.19 with Y6 (10 μM), and to 0.10 with Y6 (15 μM). For the MDR1 gene, the expressions were significantly lower in the combination drug therapy groups (doxorubicin plus EGCG or Y6, excluding verapamil) than that in the doxorubicin alone group. The expression of MDR1 was shown to be 0.71 in BEL-7404/DOX cells treated with doxorubicin (10 μM) alone, but it decreased to 0.39 with EGCG (10 μM), to 0.25 with Y6 (10 μM), and to 0.19 withY6 (15 μM). The differences were statistically significant not only in hif-1α, but also in MDR1, when compared with the doxorubicin group (p < 0.05) (Table 3, Figure 4). These results indicated that treatment of Y6 decreased the expression of hif-1α and MDR1 mRNA.
Table 3: The expression of hif-1α and MDR1 genes in BEL-7404/DOX cells (n = 3)
Gene Names |
2-ΔΔCT ± SDa |
|||||
|---|---|---|---|---|---|---|
Control |
DOX(10)b |
EGCG(10) + DOXc |
VER(10) + DOXc |
Y6(10) + DOXc |
Y6(15) + DOXc |
|
hif-1α |
—— |
0.90 ± 0.07 |
0.49 ± 0.18* |
0.65 ± 0.13* |
0.19 ± 0.10* |
0.10 ± 0.06* |
MDR1 |
—— |
0.71 ± 0.18 |
0.39 ± 0.12& |
0.46 ± 0.15# |
0.25 ± 0.09& |
0.19 ± 0.08& |
The expression of hif-1α and MDR1 genes were measured in BEL-7404/DOX cell by RT-qPCR analysis. a: Values represent means ± SDs of at least three independent experiments performed in triplicate; b:DOX group, 10 μM of DOX alone was used; c:10 μM of DOX combined with 10 μM of VER, 10 μM of EGCG, 10 μM of Y6, or 15 μM of Y6 were used; VER (10)+DOX (10) was used as the positive control; Control group, no drugs were used; *p < 0.05 vs. the DOX alone group on the hif-1α gene; &p < 0.05 vs. the DOX alone group on the MDR1 gene; #p > 0.05 vs. the DOX alone group on the MDR1 gene.
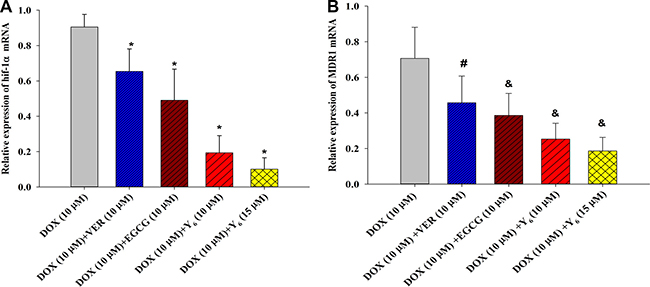
Figure 4: The expression of hif-1α and MDR1 genes were measured in BEL-7404/DOX cells by RT-PCR analysis. (A) *p < 0.05 vs. the doxorubicin alone group on the hif-1α gene; (B) #p > 0.05 vs. the doxorubicin alone group on the MDR1 gene; &p < 0.05 vs. the doxorubicin alone group on the MDR1 gene. Experiments were repeated three independent times.
Y6 inhibited the expression of HIF-1α and P-gp proteins in BEL-7404/DOX cells
Since PCR results showed that treatment with Y6 inhibited the mRNA expressions of hif-1α and MDR1 genes compared with the doxorubicin alone group, we investigated whether the expressions of their encoded proteins HIF-1α and P-gp were also inhibited. The expressions of HIF-1α and P-gp proteins were examined by Western blotting analysis. The results showed that the expressions of HIF-1α and P-gp proteins were significantly decreased in the combination drug therapy groups (doxorubicin plus Y6) compared to the doxorubicin alone therapy group (*p < 0.05 and &p < 0.05) (Figure 5).
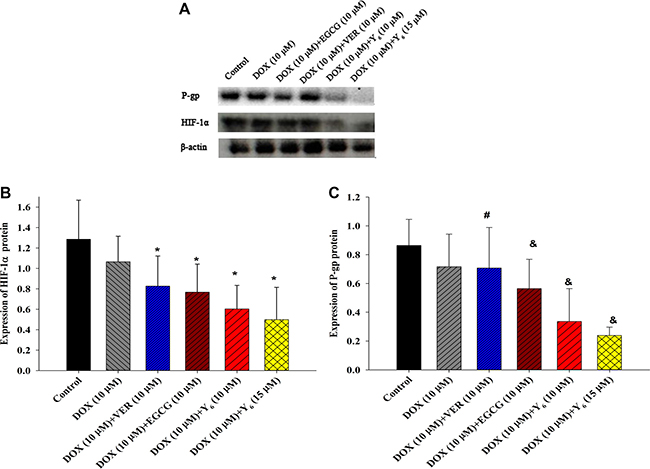
Figure 5: The expression of HIF-1α and P-gp proteins. (A) The expression of HIF-1α and P-gp proteins were measured in BEL-7404/DOX cells by Western blotting analysis. (B) (C) Relative protein levels were quantified by densitometry and showed in the histogram. Values represent means ± SDs of at least three independent experiments performed in triplicate; *p < 0.05 vs. the doxorubicin alone group on the HIF-1α protein; # p > 0.05 vs. the doxorubicin alone group on the P-gp protein; &p < 0.05 vs. the doxorubicin alone group on the P-gp protein. Experiments were repeated three independent times.
DISCUSSION
Nature is an endless source of organic compounds for development of new drugs including resistance reversal agents for cancer. EGCG, from the Chinese green tea, is the effective ingredient for reversing anti-cancer drug resistance. Alkylate modification is one way to remedy the EGCG’s defects of low lipid solubility and stability [23]. Previous studies have shown that the ethylated and propyl derivatives of EGCG had strong reversal effect, while EGCG-methylated derivatives had no effect on the MDR in hepatocellular carcinoma cells [24]. However, with the increase of carbon chain, the toxicity to normal hepatocyte is also increased. Therefore, the propyl derivatives were more appropriate to be the potential anticancer drugs [24]. We synthesized six different EGCG-ethylated derivatives. Among them, Y6 had not only more stable features but also better lipid solubility when compared with EGCG and other EGCG-ethylated derivatives. In this study, we elucidated the effect and mechanism of Y6 in reversing drug resistance in BEL-7404/DOX cells.
By combining non-cytotoxic concentrations of Y6 (below the IC10 values) with doxorubicin, we determined that Y6 potentiated the doxorubicin cytotoxic effects on cancer cells. Our MTT assay result showed that Y6 and EGCG could enhance the cytotoxicity of doxorubicin by reversing multidrug resistance in BEL-7404/DOX cells. In addition, flow cytometric analysis result revealed that Y6 increased cell apoptosis in the late apoptotic stage. The reversal effects of Y6 were higher than that of EGCG both in MTT assay and in flow cytometric analysis experiments. Our study demonstrated that Y6 has excellent ability in increasing cell sensitivity to doxorubicin and inducing apoptotic cell death.
Hypoxia is a common feature in solid tumors and is generally associated with tumor chemo-resistance [25, 26]. Hypoxia may reduce tumor sensitivity to radiation therapy and chemotherapy by deprivation of the oxygen essential for the cytotoxic actions directly, and lead to increasing resistance by inducing proteomic and genomic changes indirectly [27]. In the study by Du Cetal, hypoxia was shown to enhance tumor development in HCC [28]. In the experiment by Yu G et al, the chemosensitivity to 5-FU or cisplatin in HBx-HepG2 cells was measured by MTT assay in different hypoxic (5% O2, 3% O2, or 1% O2) conditions. The results revealed that under hypoxic conditions, particularly at 1% O2, the cancer killing effects of 5-FU or cisplatin were prominently abrogated [29]. Therefore our experiment adopted the hypoxic condition of 1% O2 to imitate the hypoxic environment of solid tumors in vivo. HIF-1 is a central component of hypoxic adaptation. It has been presented as an important mediator of hypoxia-regulated gene expression and can activate the transcription of genes that are important to cell viability, invasion, metastasis, and angiogenesis [30–32]. Hypoxia may significantly increase the protein expression level of HIF-1α [29]. The activation of HIF-1 is closely related to the increased of expression of MDR1 gene and its product P-gp protein [33–38]. P-gp protein was the first transporter to be identified that associated to MDR. It was also the most representative and widely studied in reversing resistance. Previous study showed that expression of HIF-1α and MDR1/P-gp could be regarded as a predictive marker of chemotherapy resistance, and HIF-1α inhibition was use to reverse multidrug resistance by reducing expression of MDR1/P-gp [39]. Other studies revealed that the expressions of hif mRNA and its encoded protein were decreased after treatment with verapamil [40], but there was no decrease in the level of P-gp protein [41].
To investigate whether the effect of Y6 on the reversal of resistance was associated with decreased P-gp expression via the HIF-1α-mediated pathway, we measured the mRNA expressions of hif-1α and MDR1 genes by quantitative RT-PCR analysis and expressions of their encoded proteins of HIF-1α and P-gp by Western blotting. The BEL-7404/DOX cells were pretreated in hypoxic condition before experiment. The results of our study revealed that expressions of hif-1α gene and its encoded protein were significantly down-regulated upon the treatment with doxorubicin plus Y6. We also observed a significant decreased expression of MDR1 gene and its encoded protein P-gp in parallel with a decrease in HIF-1α expression in BEL-7404/DOX cells. The coincident decline of HIF-1α and MDR1/P-gp expression treated with Y6 suggested a positive correlation between the two proteins. Y6 were more potent in decreasing the expressions of HIF-1α and P-gp both in gene and in protein level than EGCG. The strong reversal effect of Y6 was most likely due to the decline of P-gp transporter.
In conclusion, the present investigation suggested that Y6 might reverse P-gp-mediated MDR. Y6 in combination with doxorubicin could increase the cytotoxicity of doxorubicin in vitro. The mechanisms of Y6 in reversing MDR are likely to be related to the HIF-1α-MDR1/P-gp pathway. Y6, as a novel inhibitor resulting from the modification of natural product, may become an effective drug to overcome MDR in the treatment of hepatocellular carcinoma.
MATERIALS AND METHODS
Materials
EGCG (purity 97%) from green tea was obtained from Hangzhou Hetian biotechnology Co., Ltd. (Hangzhou, Zhejiang, China). Y6 (purity 96.87%) was synthesized by our research group and was dissolved in dimethylsulfoxide (DMSO) to form a stock solution of 10 mM and stored at −20°C. Verapamil hydrochloride was obtained from Shanghai Hefeng Pharmaceutical Co., Ltd. (Shanghai, China). Doxorubicin was purchased from Shanxi Pude Pharmaceutical Co., Ltd. (Datong, Shanxi, China). DMSO was purchased from Guangzhou Xingang Co., Ltd. (Guangzhou, Guangdong, China). 3-(4, 5-dimethylthiazol-2-y1)-2, 5-diphenyltetrazolium bromide was purchased from Sigma (St. Louis, MO). An Annexin V-FITC/ Propidium iodide (PI) staining assay kit was obtained from Sony Biotechnology Inc. (San Jose, CA). Total Ribonucleic Acid (RNA) extraction reagent kit (RNAiso plus) and Prime ScriptTM RT reagent Kit with cDNA Eraser for Perfect Real Time were obtained from TAKARA Bio Inc. (Dalian, China). The anti-HIF-1α, anti-P-gp antibody, and anti-rabbit IgG (H+L) (Dy Light 680 Conjugate) were purchased from Cell Signaling Technology (Beverly, Massachusetts). β-actin polyclonal antibody was obtained from Bioworld Technology, Inc. (St. Louis Park, MN). RPMI-1640 medium was obtained from Hyclone Laboratories (Logan, UT), and fetal bovine serum (FBS) was from Hangzhou Sijiqing Co., Ltd. (Hangzhou, Zhejiang). Penicillin-Streptomycin Liquid (PS; 10,000 U/mL) and trypsin-EDTA Solution (containing 0.25% trypsin and 0.02% EDTA) were obtained from Beijing Solarbio Science&Technology Co., Ltd. (Beijing, China).
Hypoxia cell culture
Cells were cultured under normoxic condition (2% O2, 5% CO2, 74% N2) for 24 h and subsequently under hypoxic condition (1% O2, 5%CO2, 94% N2) for 48 h in the incubator (NU4950, NUAIRE company, USA) [29].
Cell culture
The parental HCC cell line BEL-7404 and the drug resistant HCC cell line BEL-7404/DOX were obtained from the Department of Physiopathology, Guangxi Medical University (Nanning, Guangxi, China). The doxorubicin-resistant cell line BEL-7404/DOX had been developed by exposure of cells to doxorubicin at gradually increasing doses. BEL-7404 and BEL-7404/DOX cells were maintained in RPMI-1640 medium containing 10% (v/v) FBS and 1% (v/v) PS at 37°C in a normoxic incubator (Forma Scientific, Inc., Marietta, OH). BEL-7404/DOX cells were cultured with doxorubicin (0.1 μg/mL) to maintain drug resistance. For subculturing, cells were dissociated by using 0.25% trypsin.
Cell viability assay
The cytotoxicity of drugs to HCC cells (BEL-7404 and BEL-7404/DOX) was measured by the MTT assay [39]. Cells were seeded in 96-well plates at a density of 5000 cells/well for 48 h. The cells were treated with various concentrations of Y6, EGCG, verapamil, and doxorubicin, or doxorubicin in combination with Y6, EGCG, and verapamil for 48 h in hypoxic condition. Then MTT (20 μL; 5 mg/mL) was added to each well for an additional 4 h. The medium was removed and 100 μL of DMSO was added to each well to dissolve the formazan crystals that had formed. The optical density at 490nm was measured on a multi-well plate reader (Bio-Rad, Hercules, CA). The percentage of cell survival was calculated using the background-corrected absorbance, as follows: % Cell viability = (ODexperimental/ODcontrol) × 100.
All experiments were repeated 3 times. IC50 values are drug concentrations at which there is 50% reduction in optical density. The fold of drug resistance was calculated as the IC50 of BEL-7404/DOX divided by the IC50 of BEL-7404. The degree of reversal of drug resistance (reversal fold or RF) was calculated as the IC50 with doxorubicin alone divided by the IC50 with doxorubicin in combination with other drugs (Y6, EGCG, and verapamil).
Annexin-V-FITC/PI staining assay
The Bel-7404/DOX cells were incubated for 48 h, and then were treated with medium alone, medium with doxorubicin alone, doxorubicin+Y6, doxorubicin+EGCG, or doxorubicin+verapamil for 48 h. Subsequently, the cells were subjected to flow cytometry analysis. The Bel-7404/DOX cells were suspended in Annexin V Binding Buffer at a concentration 0.25~1.0 × 107 cells/ml. Five μL of FITC and 10 μL of PI were added to 100μL of cell suspension per tube in turn. The cells were mixed in vortex and incubated for 15 minutes at room temperature (25°C) in the dark. Annexin V Binding Buffer (400 μL) was added to each tube. The mixed cell suspensions were analyzed by flow cytometry. The fluorescence of the sample was detected by Epics XL Flow Cytometry (Beckman Coulter) (Ex = 488 nm and Em = 525 nm for Annexin V-FITC; Ex = 488 nm and Em = 620 nm for PI).
RT-PCR analysis
The BEL-7404/DOX cells were incubated in hypoxia (1% O2) for 48 h, and then were treated with medium alone, medium with doxorubicin alone, doxorubicin+Y6, doxorubicin+EGCG, or doxorubicin+verapamil for 48 h. Subsequently, the cells were subjected to RT-PCR analysis. Total mRNA of the BEL-7404/DOX cells was extracted using Trizol reagent according to manufacturer’s instructions, and resuspended in RNase water. The content and purity of total RNA in suspension was measured by ultraviolet spectrophotometer (wave length 260 nm), and the integrity of RNA was checked with the Bioanalyser 2100. One μg of total RNA was reverse transcribed (RT) to cDNA by using the PrimeScript and RT Primer Mix (including Oligo dt Primer and Random 6 mers). For PCR amplification of cDNA, 2 μL of cDNA liquid was added to 23 μL of PCR reaction media (including SYBR Premix Ex Taq, PCR Forward/Reverse Primer, and double distilled water). PCRs were carried out in the conditions according to the instructions: initial denaturation (95°C for 30 s) followed by 40 cycles of denaturation (95°C for 5s), and hybridization-extension (60°C for 31s) using Applied Biosystems7300 Real Time PCR System. The gene names (including target genes hif-1α and MDR1 and housekeeping gene ACTB), access number, primer sequence, and amplifying size are listed in Table 4. In the results, transcription levels of target genes were normalized to those of ACTB to compensate for difference in efficiency of reverse transcription and input RNA amounts; they were expressed relative to the control. The relative quantitation of target gene expression was calculated by the comparative ΔΔCT method.
Table 4: Primers used for the RT-qPCR studies
Gene |
Genebank access number |
Primer sequence (5′–3′) |
Amplicon size (bp) |
|---|---|---|---|
hif-1α |
NM_001243084.1 [42] |
Forward- GACACAGAAGCAAAGAACCCA Reverse- CATCAGTGGTGGCAGTGGTA |
241 |
MDR1 |
NM_000927.4 [43] |
Forward-CTCTTTGCCACAGGAAGCCT Reverse- CATTGCGGTCCCCTTCAAGA |
187 |
ACTB |
NM_001101.3 [44] |
Forward-GGCATCCTCACCCTGAAGTA Reverse-GCACACGCAGCTCATTGTAG |
102 |
Western blot analysis
The BEL-7404/DOX cells were incubated in hypoxia (1% O2) for 48 h, and then were treated with medium alone, medium with doxorubicin alone, doxorubicin+Y6, doxorubicin+EGCG, or doxorubicin+verapamil for 48 h. Subsequently, the cells were subjected to Western blot analysis. Cells with different treatments were lysed in an ice-cold buffer containing 50 mM Tris (pH 7.4), 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, sodium orthovanadate, sodium fluoride, EDTA, leupeptin, 1× protease inhibitor cocktail, and 1×phosphatase inhibitor cocktail at 4°C for 30 min. Cell lysates were centrifuged at 4°C with 13,000 rpm/min for 10 min. Supernatant of cell lysates was transferred to the Eppendorf tube and measured for protein concentrations.
Twenty-five micrograms of protein liquid from each sample were placed in electrophoresis tanks in a 10–12% SDS polyacrylamide gel. The cell lysates were separated on the SDS polyacrylamide gel and then transferred electrophoretically onto a nitrocellulose membrane, which was pre-hybridized in methyl alcohol. The membrane was submerged in 5% skim milk for 1 h, and then transferred to a solution containing 20 mM Tris-HCl (pH 7.4), 150 mM NaCl, 0.05% Tween-20 (TBST buffer) and a primary antibody and incubated overnight at 4°C. After washing with the TBST buffer, the membrane was submerged in TBST buffer containing a Dy Light 680 secondary antibody in the dark at room temperature for 2 h. The membrane was washed with TBST buffer and then exposed to Infrared Imaging System to visualize the bands.
Statistical analysis
The results of each experiment were represented as the mean ± standard deviation (SD) and analyzed in SPSS 20.0 software. One-Way ANOVA method was used in multiple comparison tests. A difference was considered significant when p value was less than 0.05.
ACKNOWLEDGMENTS AND FUNDING
The authors would like to thank Dr. Xiao-Cong Kuang (College of Basic Medicine, Guangxi Medical University, Nanning, P.R. China) for providing the BEL-7404 cells and BEL-7404/DOX cells. We thank Dr. Yangmin Chen (MediMedia Managed Markets, an ICON company, Yardley, PA) for editorial support.
This work was supported by the Natural Science Foundation of China (No. 81160532 ) (http://isisn.nsfc.gov.cn/egrantweb/), the Ph.D. Programs Foundation of Ministry of Education of China (No. 20134503110007), the Innovation Project of Guangxi Graduate Education and the Ray Biotech Co. (Atlanta, GA).
CONFLICTS OF INTEREST
There is no conflicts of interest.
REFERENCES
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.
2. Rampone B, Schiavone B, Martino A, Viviano C, Confuorto G. Current management strategy of hepatocellular carcinoma. World J Gastroenterol. 2009; 15:3210–3216.
3. Dong X, Mumper RJ. Nanomedicinal strategies to treat multidrug-resistant tumors: current progress. Nanomedicine (Lond). 2010; 5:597–615.
4. Zinzi L, Capparelli E, Cantore M, Contino M, Leopoldo M, Colabufo NA. Small and innovative molecules as new strategy to revert MDR. Front Oncol. 2015:48.
5. Zhang YK, Wang YJ, Gupta P, Chen ZS. Multidrug resistance proteins (MRPs) and cancer therapy. AAPS J. 2015; 17:802–812.
6. Vargas JR, Stanzl EG, Teng NN, Wender PA. Cell-penetrating, guanidinium-rich molecular transporters for overcoming efflux-mediated multidrug resistance. Mol Pharm. 2014; 11:2553–2565.
7. Ambudkar SV, Kimchi-Sarfaty C, Sauna ZE, Gottesman MM. P-glycoprotein: from genomics to mechanism. Oncogene. 2003; 22:7468–7485.
8. Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP–dependent transporters. Nat Rev Cancer. 2002; 2:48–58.
9. Sauna ZE, Smith MM, Müller M, Kerr KM, Ambudkar SV. The mechanism of action of multidrug-resistance-linked P-glycoprotein. J Bioenerg Biomembr. 2001; 33:481–491.
10. Wang B, Peng Xx, Sun R, Li J, Zhan Xr, Wu Lj, Wang Sl, Xie T. Systematic review of β-elemene injection as adjunctive treatment for lung cancer. Chin J Integr Med. 2012; 18:813–823.
11. Wu CP, Calcagno AM, Ambudkar SV. Reversal of ABC drug transporter-mediated multidrug resistance in cancer cells: evaluation of current strategies. Curr Mol Pharmacol. 2008; 1:93–105.
12. Thomas H, Coley HM. Overcoming multidrug resistance in cancer: an update on the clinical strategy of inhibiting p-glycoprotein. Cancer Control. 2003; 10:159–159.
13. Bissett D, Kerr D, Cassidy J, Meredith P, Traugott U, Kaye S. Phase I and pharmacokinetic study of D-verapamil and doxorubicin. Br J Cancer. 1991; 64:1168.
14. Fischer V, Rodríguez-Gascón A, Heitz F, Tynes R, Hauck C, Cohen D, Vickers AE. The Multidrug Resistance Modulator Valspodar (PSC 833) Is Metabolized by Human Cytochrome P450 3A Implications for Drug-Drug Interactions and Pharmacological Activity of the Main Metabolite. Drug Metab Dispos. 1998; 26:802–811.
15. Pusztai L, Wagner P, Ibrahim N, Rivera E, Theriault R, Booser D, Symmans FW, Wong F, Blumenschein G, Fleming DR. Phase II study of tariquidar, a selective P-glycoprotein inhibitor, in patients with chemotherapy–resistant, advanced breast carcinoma. Cancer. 2005; 104:682–691.
16. Nobili S, Landini I, Giglioni B, Mini E. Pharmacological strategies for overcoming multidrug resistance. Current drug targets. 2006; 7:861–879.
17. Srivastava V, Negi AS, Kumar J, Gupta M, Khanuja SP. Plant-based anticancer molecules: a chemical and biological profile of some important leads. Bioorg Med Chem. 2005; 13:5892–5908.
18. Kathawala RJ, Gupta P, Ashby CR, Chen ZS. The modulation of ABC transporter-mediated multidrug resistance in cancer: A review of the past decade. Drug Resist Updat. 2015; 18:1–17.
19. Liang G, Tang A, Lin X, Li L, Zhang S, Huang Z, Tang H, Li QQ. Green tea catechins augment the antitumor activity of doxorubicin in an in vivo mouse model for chemoresistant liver cancer. Int J Oncol. 2010; 37:111.
20. Lee MJ, Maliakal P, Chen L, Meng X, Bondoc FY, Prabhu S, Lambert G, Mohr S, Yang CS. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans formation of different metabolites and individual variability. Cancer Epidemiol Biomarkers Prev. 2002; 11:1025–1032.
21. Liang G, Zhang S, Huang Z, Tang A. MDR-reversing effect of two components of catechin on human hepatocellular carcinoma BEL-7404/Adr in vitro. [Article in Chinese] Ai zheng. 2004; 23:401–405.
22. Li JS. Effect of N, N-Bis(2-Chloroethyl) docos-13-Enamide on The MDR Reversal of BEL-7404 and Related Mechanism. Guangxi Medical University. 2016.
23. Lambert JD, Sang S, Hong J, Kwon SJ, Lee MJ, Ho CT, Yang CS. Peracetylation as a means of enhancing in vitro bioactivity and bioavailability of epigallocatechin-3-gallate. Drug Metab Dispos. 2006; 34:2111–2116.
24. Ou BN. The Structural Modification of (−)-Epigallocatechin Gallate and The study of Reversal Activity of Its Derivatives on Multidrugs Resistance of Human Hepatocellular Carcinoma. Guangxi Medical University. 2011.
25. Brown JM. Tumor hypoxia in cancer therapy. Methods in enzymology. 2007; 435:295–321.
26. Huang L, Ao Q, Zhang Q, Yang X, Xing H, Li F, Chen G, Zhou J, Wang S, Xu G. Hypoxia induced paclitaxel resistance in human ovarian cancers via hypoxia-inducible factor 1α. J Cancer Res Clin Oncol. 2010; 136:447–456.
27. Harrison L, Blackwell K. Hypoxia and anemia: factors in decreased sensitivity to radiation therapy and chemotherapy? Oncologist. 2004; 9:31–40.
28. Du C, Weng X, Hu W, Lv Z, Xiao H, Ding C, Owusu-anash KG, Xie H, Zhou L, Wu J. Hypoxia-inducible MiR-182 promotes angiogenesis by targeting RASA1 in hepatocellular carcinoma. J Exp Clin Cancer Res. 2015; 34:1.
29. Yu G, Chen X, Chen S, Ye W, Hou K, Liang M. Arsenic trioxide reduces chemo-resistance to 5-fluorouracil and cisplatin in HBx-HepG2 cells via complex mechanisms. Cancer Cell Int. 2015; 15:1.
30. Chen MC, Hsu WL, Hwang PA, Chou TC. Low molecular weight fucoidan inhibits tumor angiogenesis through downregulation of HIF-1/VEGF signaling under hypoxia. Mar Drugs. 2015; 13:4436–4451.
31. Denko NC, Fontana LA, Hudson KM, Sutphin PD, Raychaudhuri S, Altman R, Giaccia AJ. Investigating hypoxic tumor physiology through gene expression patterns. Oncogene. 2003; 22:5907–5914.
32. Prabhakar NR, Semenza GL. Adaptive and maladaptive cardiorespiratory responses to continuous and intermittent hypoxia mediated by hypoxia-inducible factors 1 and 2. Physiol Rev. 2012; 92:967–1003.
33. Comerford KM, Wallace TJ, Karhausen J, Louis NA, Montalto MC, Colgan SP. Hypoxia-inducible factor-1-dependent regulation of the multidrug resistance (MDR1) gene. Cancer Res. 2002; 62:3387–3394.
34. Li J, Shi M, Cao Y, Yuan W, Pang T, Li B, Sun Z, Chen L, Zhao RC. Knockdown of hypoxia-inducible factor-1α in breast carcinoma MCF-7 cells results in reduced tumor growth and increased sensitivity to methotrexate. Biochem Biophys Res Commun. 2006; 342:1341–1351.
35. Song X, Liu X, Chi W, Liu Y, Wei L, Wang X, Yu J. Hypoxia-induced resistance to cisplatin and doxorubicin in non-small cell lung cancer is inhibited by silencing of HIF-1α gene. Cancer Chemother Pharmacol. 2006; 58:776–784.
36. Riganti C, Doublier S, Aldieri E, Orecchia S, Betta PG, Gazzano E, Ghigo D, Bosia A. Asbestos induces doxorubicin resistance in MM98 mesothelioma cells via HIF-1α. Eur Respir J. 2008; 32:443–451.
37. Riganti C, Doublier S, Viarisio D, Miraglia E, Pescarmona G, Ghigo D, Bosia A. Artemisinin induces doxorubicin resistance in human colon cancer cells via calcium-dependent activation of HIF-1α and P-glycoprotein overexpression. Br J Pharmacol. 2009; 156:1054–1066.
38. Doublier S, Belisario DC, Polimeni M, Annaratone L, Riganti C, Allia E, Ghigo D, Bosia A, Sapino A. HIF-1 activation induces doxorubicin resistance in MCF7 3-D spheroids via P-glycoprotein expression: a potential model of the chemo-resistance of invasive micropapillary carcinoma of the breast. BMC Cancer. 2012; 12:1.
39. Chen J, Ding Z, Peng Y, Pan F, Li J, Zou L, Zhang Y, Liang H. HIF-1α inhibition reverses multidrug resistance in colon cancer cells via downregulation of MDR1/P-glycoprotein. PloS One. 2014; 9:e98882.
40. Liao XM, Tang WX, Lai JJ, Pu SP, Sun SL. Effect of Calcium Signal on Expression of HIF-lα in Breast Cancer Cell Line and Invasion and Metastasis of Cells. J Fourth Mil Med Univ. 2009; 1:49–52.
41. Liu DL, Li YJ, Yang DH, Wang CR, Xu J, Yao N, Zhang XQ, Chen ZS, Ye WC, Zhang DM. Ganoderma lucidum derived ganoderenic acid B reverses ABCB1-mediated multidrug resistance in HepG2/ADM cells. Int J Oncol. 2015; 46:2029–2038.
42. Ghafoory S, Mehrabi A, Hafezi M, Cheng X, Breitkopf-Heinlein K, Hick M, Huichalaf M, Herbel V, Saffari A, Wölfl S. Nuclear accumulation of CDH1 mRNA in hepatocellular carcinoma cells. Oncogenesis. 2015; 4:e152.
43. Li A, Song J, Lai Q, Liu B, Wang H, Xu Y, Feng X, Sun X, Du Z. Hypermethylation of ATP-binding cassette B1 (ABCB1) multidrug resistance 1 (MDR1) is associated with cisplatin resistance in the A549 lung adenocarcinoma cell line. Int J Exp Pathol. 2016.
44. Zhang W, Lei XJ, Wang YF, Wang DQ, Yuan ZY. Role of Kir2.1 in human monocyte-derived foam cell maturation. J Cell Mol Med. 2016; 20:403–412.

