INTRODUCTION
Breast cancer is the most frequently diagnosed cancer worldwide and the leading cause of cancer death among females, accounting for 23% of total cancer cases and 14% of cancer deaths [1]. Despite increasing incidence, mortality from breast cancer has declined over the past decade [2]. A considerable proportion of the decrease in mortality is attributed to early diagnostic methods, such as modern digital mammography. However, 13% of breast cancers are undetectable by mammography affected by tumor size and age of patients [3, 4]. Currently used biomarkers with unsatisfactory accuracy, such as cancer antigen CA15-3 and carcinoembryonic antigen (CEA), have been recommended against for accurately diagnosing breast cancer [5, 6]. Therefore, development of new technologies with enhanced sensitivity and specificity to detect and diagnose breast cancer is in critical demand.
Circulating cell-free DNA (cfDNA) is fragmented DNA originating from cancer cells through the processes of necrosis and apoptosis [7]. The cfDNA containing specific mutations, copy number alterations, and structural variants prevail in a wide range of cancers, including pancreatic, ovarian, colorectal, bladder, breast cancers, and other pathologies with rapid cell turnover [8–11]. Through highly sensitive techniques to detect abnormalities of circulating cfDNA, including digital PCR-based [8, 12, 13] and massive sequencing-based technologies [8, 9, 14], it is now feasible to improve early screening and surveillance of breast cancer.
The detection of alternations in circulating cfDNA present in breast cancer patients has led to a wealth of studies that have analyzed the genetic and epigenetic character of these alterations, including microsatellite instability and aberrant DNA methylation in plasma or serum [15]. Many studies have addressed the potential value of circulating cfDNA assays as a repeatable and non-invasive “liquid biopsy” for breast cancer [7, 16]. However, these results of such studies have varied, but have not been systematically reviewed. Hence, the aim of this meta-analysis was to quantitatively evaluate the diagnostic efficiency of circulating cfDNA assays for breast cancer screening.
RESULTS
Database analyses
In primary review, a total of 45 publications dealing with abnormal concentration [8, 17–36], methylation alterations [37–44], microsatellite instability [45–50] and other characteristics [10, 14, 51–58] of plasma or serum DNA for the diagnosis of breast cancer were retrieved. After full-text review, 20 studies [8, 10, 14, 29–35, 42–44, 48–50, 56–59] were excluded because they did not allow the calculation of sensitivity or specificity, included very rare indicators, or consisted of less than 10 breast cancer patients (Figure 1).
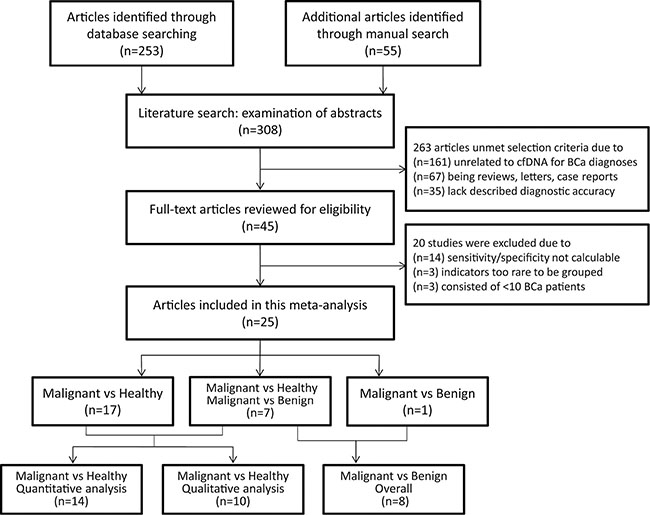
Figure 1: Study identification, inclusion, and exclusion for meta-analysis. BCa = breast cancer; cfDNA = cell-free DNA; vs = versus.
In total, 25 eligible studies [17–28, 36, 38–41, 45–47, 51–55, 60] were included in the analyses (Table 1), comprising 1705 histologically diagnosed breast cancer patients, 1079 healthy controls, and 234 patients with benign breast diseases. A majority of 1959 subjects were from the United States and European countries, with the remaining 979 participants from Asian areas (China, Thailand and Israel) and 80 from Africa (Egypt). Of 25 studies, 15 assessing abnormal concentrations of circulating cfDNA were classified as the quantitative analysis group, while 10 trials evaluating multi-gene methylation alterations, allelic imbalances and genome-wide aberrations represented the group of qualitative analysis. In addition to assessments of 24 studies using health individuals as control [17–28, 38–41, 45–47, 51–55], a diagnostic assessment was also conducted in 8 studies that included benign breast diseases as controls, half of which are quantitative studies while the remain are qualitative [18, 21–23, 36, 39, 40, 45].
Table 1: Summary of included studies
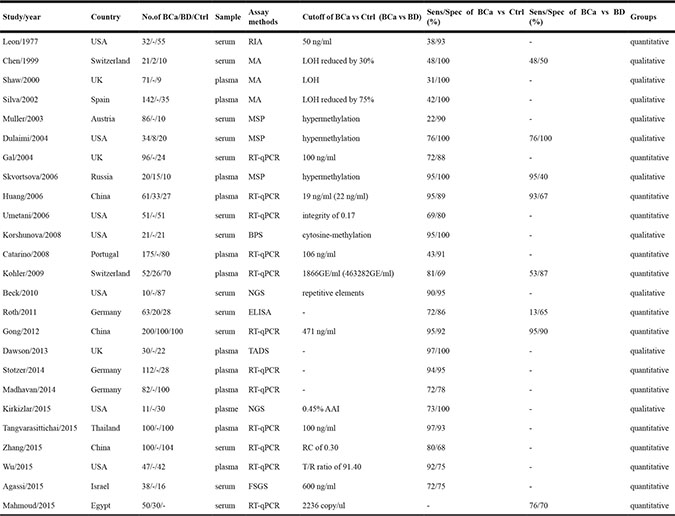
BCa = patients with breast cancers; BD = patients with benign breast diseases; Ctrl = healthy controls; TP = true positive; FP = false positive; FN = false negative; TN = true negative; RIA = radioimmunoassay; MA = microsatellite analysis; LOH = loss of heterozygosity; MSP = methylation specific PCR; RT-qPCR = real-time quantitative PCR; RC = relative concentration; T/R = copy number of telomere relative to copy number of LINE reference sequence; ELISA= enzyme linked immunosorbent assay; BPS = bisulphate pyrosequencing; NGS = next-generation sequencing; AAI = average allelic imbalance; TADS = tagged-amplicon deep sequencing; FSGS = fluorochrome SYBR Gold stain.
Diagnostic accuracy
Sensitivity and specificity, PLR, NLR, and DOR are indicators applied to estimate diagnostic accuracy. Generated by integrating 24 trials, the overall sensitivity and specificity of cfDNA assays, to distinguish breast cancer patients and healthy individuals, were 0.70 (95% CI, 0.68–0.72) and 0.87 (95% CI, 0.85–0.89), respectively. PLR was 6.22 (95% CI, 4.31–8.99), NLR was 0.25 (95% CI, 0.17–0.36), and DOR was 32.31 (95% CI, 17.35–60.18) (Figure 2A). To determine how methodology affected diagnostic accuracy, we further analyzed two groups that employed quantitative (testing cfDNA concentrations) and qualitative (evaluating multi-gene methylation, allelic imbalances, and genome-wide aberrations) methodologies.
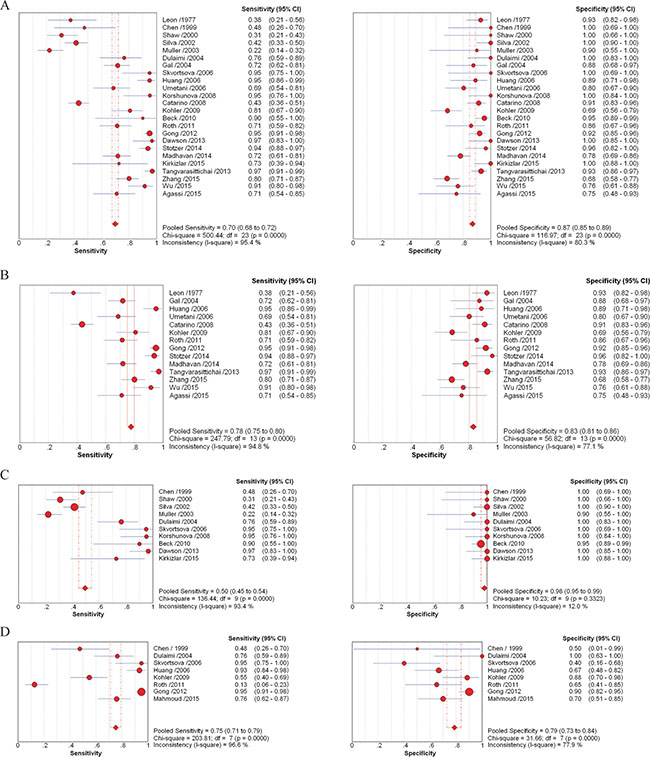
Figure 2: Forest plots of estimates of sensitivity and specificity for different cell-free DNA assay groups. Forest plots of sensitivity and specificity for assays of circulating cell-free DNA in the diagnosis between healthy individuals and breast cancer patients (A), and between benign breast disease and breast cancer patients (D). Forest plots of sensitivity and specificity for methodological groups using quantitative (B) and qualitative (C) analysis of circulating cell-free DNA in the diagnosis of breast cancer. = points estimates of sensitivity and specificity from each study; error bars = 95% CI.
= points estimates of sensitivity and specificity from each study; error bars = 95% CI.
The estimates of sensitivity and specificity of the 14 quantitative analyses of cfDNA for breast cancer diagnosis (Figure 2B) were 0.78 (95% CI, 0.75–0.80) and 0.83 (95% CI, 0.81–0.86), respectively. The value for PLR was 4.83 (95% CI, 3.37–6.91), and NLR was 0.22 (95% CI, 0.13–0.35). The DOR value was 24.40 (95% CI, 12.07–49.31). To further evaluate the diagnostic accuracy of quantitative PCR-based assays, three quantitative studies using radioimmunoassay [54], ELISA [22] and fluorochrome SYBR Gold stain [55] were excluded for further analysis. The value for sensitivity, PLR and DOR increased to 0.79 (95% CI, 0.77–0.82), 5.07 (95% CI, 3.32–7.75) and 31.91 (95% CI, 13.65–74.62); while specificity and NLR declined to 0.83 (95% CI, 0.80–0.85) and 0.17 (95% CI, 0.09–0.32) (Supplementary Table 1).
Figure 2C shows the sensitivity and specificity generated from 10 qualitative analyses, including methylation PCR, microsatellite analysis and sequencing, in diagnosis of breast cancer. The sensitivity and specificity were 0.50 (95% CI, 0.45–0.54) and 0.98 (95% CI, 0.96–0.99); PLR was 16.52 (95% CI, 8.65–31.58), NLR was 0.32 (95% CI, 0.19–0.54), and DOR was 68.45 (95% CI, 19.29–242.85). When we excluded four studies with comparatively low sensitivity [38, 45–47], the sensitivity increased to 0.88 (95% CI, 0.81–0.93), while specificity dropped slightly to 0.98 (95% CI, 0.95–0.99); PLR was 24.46 (95% CI, 11.38–52.58), NLR was 0.14 (95% CI, 0.07–0.28), and DOR was 256.60 (95% CI, 77.40–850.69) (Supplementary Table 1). In addition, the diagnostic accuracy of cfDNA assays between benign diseases and malignant tumors was estimated. Sensitivity and specificity were 0.75 (95% CI, 0.71–0.79) and 0.79 (95% CI, 0.73–0.84), PLR was 2.40 (95% CI, 1.13–5.12), NLR was 0.29 (95% CI, 0.12–0.74), and DOR was 9.49 (95% CI, 1.76–51.03) (Figure 2D).
Furthermore, the observed data, together with the confidence and predictive ellipses, are presented in SROC curves to determine their diagnostic heterogeneity. The satisfactory diagnostic performance for cfDNA assays for diagnosis of cancer patients from healthy individuals was demonstrated by the SROC curve in Figure 3A. The AUC was 0.9314, the LRT_I2 statistic was 78.60%, the LRT_Q (χ2) was 107.52 (p < 0.001), and the Spearman correlation coefficient was -0.061 (p = 0.777), indicating considerable heterogeneity between studies caused by non-threshold effects.
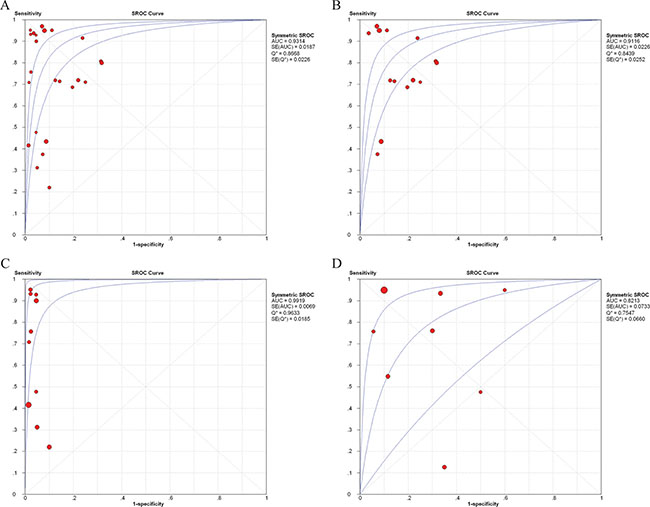
Figure 3: SROC curves for cell-free DNA assays in diagnosis of breast cancer. SROC curves for cell-free DNA assays in diagnosis of breast cancer. SROC curves for cell-free DNA assays in diagnosis between healthy individuals and breast cancer patients (A), and between benign breast disease and breast cancer patients (D). SROC curves for methodological groups using quantitative (B) and qualitative (C) analysis of cell-free DNA in the diagnosis of breast cancer.  = each study in the meta-analysis (the size of each study is indicated by the size of the solid circle); red line = weighted regression; and blue line = unweighted regression. SROC curves summarize the overall diagnostic accuracy. The confidence ellipse indicates that the mean values for sensitivity and specificity were more likely to be in this region. The prediction ellipse (increased uncertainty) indicates that individual values for sensitivity and specificity were more likely to be in this region.
= each study in the meta-analysis (the size of each study is indicated by the size of the solid circle); red line = weighted regression; and blue line = unweighted regression. SROC curves summarize the overall diagnostic accuracy. The confidence ellipse indicates that the mean values for sensitivity and specificity were more likely to be in this region. The prediction ellipse (increased uncertainty) indicates that individual values for sensitivity and specificity were more likely to be in this region.
SROC curves were also applied for the methodological groups. In the quantitative group (Figure 3B), the AUC was 0.9116 (0.9193 specific for qPCR assays), indicating acceptable levels of diagnostic accuracy. The LRT_I2 value was 84.30%, presenting some evident heterogeneity in these studies. The LRT_Q (χ2) was 82.76 (p < 0.001) and Spearman correlation coefficient was −0.191 (p = 0.513), indicating that the heterogeneity was likely the result of non-threshold effects. In the qualitative group, the AUC was 0.9919 (0.9886 when four aforementioned studies were excluded; seen in Figure 3C), suggesting higher diagnostic accuracy compared with the quantitative group. The LRT_ I2 was 49.80% and the LRT_Q (χ2) was 17.94 (p = 0.036), and Spearman correlation coefficient was -0.383 (p = 0.275), revealing no significant heterogeneity.
The SROC curve of cfDNA assays for diagnosis of cancer patients from benign disease populations generated an AUC of 0.8213, the LRT_I2 was 91.20%, the LRT_Q (χ2) was 79.25 (p < 0.001), and the Spearman correlation coefficient was −0.096 (p = 0.821), indicating heterogeneity between studies was caused by non-threshold effects (Figure 3D).
Meta-regression analysis and publication bias
To reveal sources of heterogeneity resulting from non-threshold effects, we assessed major characteristics of these studies, including “publication year (recent 5 years)”, “country (Asian regions)”, “case number (≥ 100 cases)”, “sampling (plasma)”, and “assay methods (microsatellite/ methylation/ sequencing analysis as qualitative analyses; qPCR/ the rest as quantitative analyses)”. These characteristics were used in the meta-regression analyses to assess their effects on the RDOR in the diagnosis of breast cancer. The results suggested that none of the methodological covariates may produce major heterogeneity (p > 0.05) among different groups (Table 2).
Table 2: Weighted meta-regression of effects of methodological characteristics on diagnostic accuracy of cfDNA
Covariates |
Coefficient |
RDOR (95%) |
P value |
Coefficient |
RDOR (95% CI) |
P value |
|---|---|---|---|---|---|---|
Breast cancer versus healthy controls: |
Breast cancer versus benign disease: |
|||||
Country |
0.748 |
2.11 (0.42–10.59) |
0.346 |
2.846 |
17.21 (0.63–472.03) |
0.0802 |
Year |
0.705 |
2.02 (0.49–8.40) |
0.3152 |
−0.472 |
0.62 (0.01–36.47) |
0.7862 |
Case No. |
0.859 |
2.36 (0.50–11.14) |
0.2633 |
3.383 |
29.47 (0.34–2537.67) |
0.1125 |
Sampling |
−0.835 |
0.43 (0.11–1.79) |
0.2346 |
−0.758 |
0.47 (0.01–27.94) |
0.6660 |
Method |
−1.003 |
0.37 (0.08–1.74) |
0.1946 |
−1.641 |
0.19 (0.03–1.32) |
0.0811 |
Breast cancer versus healthy controls: quantitative analysis |
Breast cancer versus healthy controls: qualitative analysis |
|||||
|---|---|---|---|---|---|---|
Country |
1.313 |
3.72 (0.70–19.88) |
0.1136 |
– |
– |
– |
Year |
0.968 |
2.63 (0.47–14.78) |
0.2450 |
2.007 |
7.44 (0.18–308.17) |
0.2491 |
Case No. |
1.414 |
4.11 (0.75–22.55) |
0.0955 |
−0.349 |
0.71 (0.00–115.88) |
0.8787 |
Sampling |
−0.907 |
0.40 (0.07–2.34) |
0.2831 |
−0.712 |
0.49 (0.02–10.14) |
0.6024 |
Method |
−0.005 |
1.00 (0.09–11.36)) |
0.9965 |
1.414 |
4.11 (0.84–20.12) |
0.0741 |
Publication biases in these diagnostic analyses were evaluated using the Deek’s funnel plot asymmetry test. The DOR of all 24 studies to distinguish breast cancer patients from healthy individuals aligned in a fairly symmetric linear regression with a coefficient of 2.71 (95% CI, −19.22–24.64; p = 0.80). A non-zero slope coefficient is suggestive of significant study bias when p < 0.10. Thus, publication bias was not significant in these studies. The test result for 8 studies applying cfDNA to distinguish breast cancer from benign breast diseased patients was also presented. The coefficient was −25.98 (−59.20–7.14) and the p-value was 0.10. This comparatively less symmetric plot of linear regression also indicated no significant publication biases for this group (Figure 4).
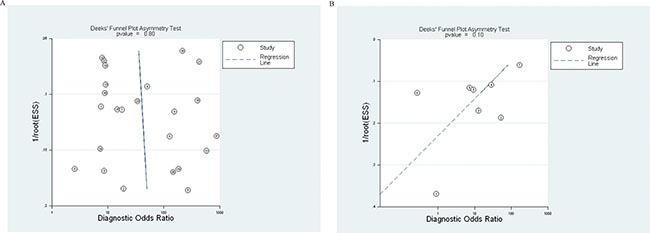
Figure 4: Funnel graphs for the assessment of potential publication bias in cell-free DNA assays. Funnel graphs for the assessment of potential publication bias in cell-free DNA assays to distinguish breast cancer patients vs. healthy individuals (A), and assays to distinguish breast cancer vs benign breast disease patients (B). The funnel graph performs linear regression of log odds ratios on inverse root of effective sample size (ESS). = each study in the meta-analysis; center line = regression line. The results of Deek’s funnel plot asymmetry test for publication bias of studies to distinguish breast cancer patients vs healthy individuals (A) and studies to distinguish breast cancer patients vs benign disease patients (B) were not significant (p = 0.80 and 0.10, respectively).
= each study in the meta-analysis; center line = regression line. The results of Deek’s funnel plot asymmetry test for publication bias of studies to distinguish breast cancer patients vs healthy individuals (A) and studies to distinguish breast cancer patients vs benign disease patients (B) were not significant (p = 0.80 and 0.10, respectively).
DISCUSSION
Our work is the first meta-analysis to calculate the overall accuracy of circulating cell-free DNA assays for detection of breast cancer. The sensitivity and specificity of cfDNA assays based on 24 primary studies were 0.70 and 0.87 respectively, indicating that a correct diagnosis could often be made through these assays. The AUC calculated for SROC curves was 0.9314, well above common standards for diagnosis (> 0.8). Among other serum-based breast cancer markers, CA15-3 exhibited acceptable sensitivity, specificity, and AUC at 0.73, 0.85, and 0.78, respectively [61]; the estimated sensitivity, specificity, and AUC of human epidermal growth factor receptor 2 (HER2) were measured at 0.51, 0.86, and 0.65 [62]. The accuracy of the cfDNA assay appears to be modestly stronger than either of these traditional markers, yet few studies have directly compared the diagnostic performance of cfDNA with other biomarkers.
In grouped analyses, the sensitivity, specificity, and AUC of 14 quantitative studies were slightly lower (0.78, 0.83, and 0.9116) than those of the overall group. When only PCR-based studies were included, the sensitivity and AUC subtly increased, suggesting that qPCR achieved marginally improved diagnostic efficiency compared to the other quantitative assays [22, 54, 55]. However, the limited number of other quantitative studies hindered analysis of their diagnostic values. In the group of 10 qualitative studies, the sensitivity dramatically reduced to 0.50; whereas, the specificity and AUC notably improved to 0.98 and 0.9919. The qualitative group included studies applying various methods to detect epigenetic and genetic abnormalities of cell-free DNA. Among these qualitative methods, microsatellite analyses [45–47] and methylation specific PCR [38] produced very limited sensitivity ranging from 0.22–0.48. By removing these, the remaining studies achieved satisfactory specificity as high as 0.88. The more modern high-throughput molecular methods, including next-generation sequencing and multiplexed PCR [51, 53], have proven to be potent strategies for breast cancer screening.
To further evaluate diagnostic effectiveness, we also analyzed the diagnostic odds ratio (DOR), which is a single indicator of test accuracy [63]. The value of DOR > 10 indicates good discriminatory test performance. In this meta-analysis, the DOR for cfDNA assays to discriminate breast cancer cases from healthy controls was 32.31, while the DOR to distinguish malignant breast tumors and benign breast diseases was much lower at 9.49. The fact that cfDNA may elevate among patients with benign diseases limited the potential of the quantitative cfDNA assay as a tool to discriminate benign hyperplasia and malignancy [21, 22]. The DOR of qualitative assays of cfDNA (68.45) is significantly higher than that of quantitative assays (24.40). The DOR was further improved to 256.60 with 4 earlier published low-sensitive studies excluded. In established studies applying cfDNA for cancer diagnoses, the DOR of quantitative analyses in lung [64], ovarian [65], and hepatocellular cancer (HCC) [66] were 20.33, 26.05, and 16.35, respectively; the DOR of qualitative methods for HCC diagnosis was 19.49. The DOR of qualitative cfDNA analysis for breast cancer was notably higher than that of HCC, and the DOR of quantitative analysis was comparable with those of other cancer types, indicating a strong ability to correctly diagnose breast cancer using cfDNA assays, especially qualitative molecular methods.
PLR and NLR were also presented to measure overall diagnostic accuracy [67]. Likelihood ratios of a PLR > 10 and an NLR < 0.1 indicate high accuracy. The group of quantitative assays had a PLR value of 6.22, suggesting that patients with breast cancer have an approximately 6-fold higher chance of being cfDNA assay-positive compared with healthy controls. The NLR of quantitative analyses was found to be 0.25, implying that the probability for cases with negative test results to have breast cancer is one fourth. These data suggest that circulating DNA assay results should not be used alone as a biomarker to make a breast cancer diagnosis. The qualitative analysis was more promising with a PLR of 16.52 and NLR of 0.32, indicating that an approximately sixteen times greater chance of a breast cancer case being indicated by a positive test result, but a 32% error rate would be present when a healthy individual was determined in the negative test. According to the results, cfDNA assessments may be applied in early detection of breast cancer, but as an auxiliary test it should be combined with cytological or histological examination of breast tissue to ensure correct diagnosis.
Considering the effect of publication bias, the results could have been biased if positive results were more likely to be published. The Deek’s funnel plot asymmetry test compared diseased and healthy groups, or malignant and benign groups, but did not indicate publication bias. Although we found a statistically significant heterogeneity for sensitivity, specificity, PLR, NLR, and DOR among these studies, we found none of the study characteristics including publication year, country, case number and assay types of these studies to represent a major source of heterogeneity. The heterogeneity could have been derived from differences on other methodological characters, such as prospective/retrospective designs and TNM staging of patients enrolled, which were not included in meta-regression analysis due to incomplete information provided by the primary studies. In addition, two studies included less than 20 cancer patients [51, 53], which may have contributed to the poor robustness. Despite significant heterogeneity, the insignificant publication bias suggested that the results of included studies had depended mostly on the objective quality of the research.
Contradictory conclusions on validation of cell-free DNA assays for breast cancer screening have long existed, led by poor method standardization and variable analytical factors. Hence, the present study conducted comprehensive meta-analyses to evaluate the diagnostic accuracy of cfDNA assays. The results show high levels of accuracy of circulating DNA analyses, especially through qualitative assays. The overall accuracy of circulating DNA analysis was higher than the routinely used biomarkers CA15-3 and CA27.29 [68]. The mean sensitivity and specificity (0.88 and 0.98) of 6 studies applying more modern qualitative cfDNA assays were higher than those (0.87 and 0.89) of digital mammography, the current benchmark of breast cancer screening [4]. Although the likelihood ratio (LR) based on 24 studies showed imperfect robustness, the LR of a subgroup of 6 recent qualitative studies was satisfactory [39–41, 51–53]. Thus, these newly-emerging cfDNA tests are highly recommended as a complement to conventional cytological and histological examinations for breast cancer diagnosis.
Our meta-analysis had some limitations. First, it was impossible for us to determine all sources of heterogeneity. We did not include some covariates because the required data were not available from the selected articles. These probable covariates included tumor size, metastasis, TNM staging, flow, and timing of these studies. Second, though we performed a thorough literature search, a smaller number of studies were included in the qualitative analysis group, which might have weakened the statistical significance. Third, the inclusion of only English-language studies might have introduced bias to the analysis. Consequently, further longitudinal studies focusing on advanced molecular methods to characterize cell-free DNA in breast cancer are desired to support the results of our meta-analyses.
MATERIALS AND METHODS
Literature source and search
The studies included in this meta-analysis were independently retrieved and reviewed by two authors (Z Lin and J Neiswender). A systematic literature search was performed in PubMed, Web of Science, and Embase databases to identify eligible studies. Studies from different databases were imported to EndNote for further review. The search terms included “breast cancer”, “breast tumor”, “cell-free DNA”, “circulating DNA”, “plasma DNA”, “serum DNA”, “sensitivity and specificity”, and “accuracy”. No limit on start date for publications was applied, and only studies prior to 20 September 2016 were evaluated. Additional articles were identified by manually reviewing the references of included articles. When necessary, the authors of included articles were contacted for further study details.
Inclusion and exclusion criteria
Studies that met the following criteria were included: (a) cohort studies that evaluated indicators originating from circulating cfDNA in plasma or serum; (b) sufficient data was presented for describing or calculating sensitivity and specificity values. Studies meeting any of the following criteria were excluded: (a) the article included specific evaluation indicators that were studied so rarely that they could not be included in a grouped analysis; (b) reviews, letters, technical reports, case reports, comments; (c) studies consisting of less than 10 breast cancer patients.
Data extraction
Two reviewers (Z Lin and J Neiswender) independently extracted data from the included articles and integrated the final results with assistance from a third author (X Ma). Data extracted from the articles included lead author, publication year, participant characteristics, experimental methods, assay indicators, cutoff values, sensitivity and specificity data. True positive (TP), true negative (TN), false positive (FP), and false negative (FN) were also collected directly or calculated according to the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) in every selected study.
Statistical analysis
Standard methods for meta-analyses of diagnostic tests were performed [69] using statistical software programs (Stata, version 12.0; Stata Corporation; College Station; and Meta-Disc for Windows). To measure the accuracy of cfDNA assays, sensitivity and specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), diagnostic odds ratio (DOR) were yielded by TP, FP, FN, TN from grouped studies. To summarize the overall accuracy, summary receiver operating characteristic (SROC) curves were constructed by the Moses-Shapiro-Littenberg method [70].
The term heterogeneity refers to the degree of variability in results across studies. Statistically significant heterogeneity among these studies was verified using likelihood ratio test (LRT)_I2 statistic [71] and LRT_Q (χ2) statistics. I2 ≥ 50% or P < 0.10 for LRT_Q indicates substantial heterogeneity. Meta-regression analysis was used to explore the sources of heterogeneity [72]. Covariates on DOR including sampling of patients and experimental methods were assessed. The relative DOR (RDOR) was calculated to analyze the change in diagnostic precision in the study per unit in the covariate [73]. The Deek’s funnel plots were used to examine potential presence of publication bias [74]. For each analysis, a result was considered to be statistically significant if the P-value was < 0.05.
CONCLUSIONS
In conclusion, our study is the first comprehensive meta-analysis on the overall accuracy of circulating cell-free DNA assays in breast cancer screening. This study suggested that the diagnostic accuracy of quantitative analysis of circulating DNA is better than conventional tumor biomarkers, and the accuracy of advanced qualitative analysis demonstrated even higher level of discriminatory power in breast cancer detection. Although the high specificity of qualitative methods is encouraging, further research must address ways to make this approach more sensitive through identification of more reliable cfDNA properties associated with breast cancer. Due to lack of robustness, the quantitative cfDNA assays cannot be used alone in cancer diagnosis without parallel cytological or histological examinations. Meanwhile, some modern qualitative assays of circulating cell-free DNA have strong potential to be applied as an independent tool to improve the benchmark for early breast cancer detection and diagnosis.
Abbreviations
AUC: area under the curve; BPS: bisulphate pyrosequencing; CEA: carcinoembryonic antigen; cfDNA: cell-free DNA; CTC: circulating tumor cell; DOR: diagnostic odds ratio; ELISA: enzyme linked immunosorbent assay; FSGS: fluorochrome SYBR Gold stain; HCC: hepatocellular cancer; LRT: likelihood ratio test; LOH: loss of heterozygosity; NGS: next-generation sequencing; NLR: negative likelihood ratio; PLR: positive likelihood ratio; RT-qPCR: real-time quantitative PCR; RDOR: relative diagnostic odds ratio; SROC: summary receiver operating characteristic; TADS: tagged-amplicon deep sequencing.
ACKNOWLEDGMENTS AND FUNDING
Prof. Xiuying Hu is funded by Department of Science and Technology of Sichuan Province, China.
CONFLICTS OF INTERESTS
The authors declare that they have no competing interests.
REFERENCES
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics.CA Cancer J Clin. 2011; 61:69–90.
2. Kohler BA, Sherman RL, Howlader N, Jemal A, Ryerson AB, Henry KA, Boscoe FP, Cronin KA, Lake A, Noone AM, Henley SJ, Eheman CR, Anderson RN, et al. Annual Report to the Nation on the Status of Cancer, 1975–2011, Featuring Incidence of Breast Cancer Subtypes by Race/Ethnicity, Poverty, and State. Journal of the National Cancer Institute. 2015; 107:djv048.
3. Oeffinger KC, Fontham ET, Etzioni R, Herzig A, Michaelson JS, Shih YC, Walter LC, Church TR, Flowers CR, LaMonte SJ, Wolf AM, DeSantis C, Lortet-Tieulent J, et al. Breast Cancer Screening for Women at Average Risk: 2015 Guideline Update From the American Cancer Society. Jama. 2015; 314:1599–1614.
4. Lehman CD, Arao RF, Sprague BL, Lee JM, Buist DS, Kerlikowske K, Henderson LM, Onega T, Tosteson AN, Rauscher GH, Miglioretti DL. National Performance Benchmarks for Modern Screening Digital Mammography: Update from the Breast Cancer Surveillance Consortium. Radiology. 2016:161174.
5. Lumachi F, Norberto L, Marino F, Basso SM, Marzano B, Chiara GB. Hormone receptor rate, MIB-1 score and serum tumour markers CEA and CA 15-3 relationship in elderly women with pT1-2 breast cancer. Anticancer research. 2010; 30:4701–4704.
6. Molina R, Auge JM, Farrus B, Zanon G, Pahisa J, Munoz M, Torne A, Filella X, Escudero JM, Fernandez P, Velasco M. Prospective evaluation of carcinoembryonic antigen (CEA) and carbohydrate antigen 15.3 (CA 15.3) in patients with primary locoregional breast cancer. Clinical chemistry. 2010; 56:1148–1157.
7. Diaz LA, Jr. and Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. Journal of clinical oncology. 2014; 32:579–586.
8. Bettegowda C, Sausen M, Leary RJ, Kinde I, Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, Antonarakis ES, Azad NS, Bardelli A, et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Science translational medicine. 2014; 6:224ra224.
9. Dawson SJ, Rueda OM, Aparicio S, Caldas C. A new genome-driven integrated classification of breast cancer and its implications. The EMBO journal. 2013; 32:617–628.
10. Leary RJ, Sausen M, Kinde I, Papadopoulos N, Carpten JD, Craig D, O'Shaughnessy J, Kinzler KW, Parmigiani G, Vogelstein B, Diaz LA, Jr. and Velculescu VE. Detection of chromosomal alterations in the circulation of cancer patients with whole-genome sequencing. Science translational medicine. 2012; 4:162ra154.
11. Garcia-Murillas I, Schiavon G, Weigelt B, Ng C, Hrebien S, Cutts RJ, Cheang M, Osin P, Nerurkar A, Kozarewa I, Garrido JA, Dowsett M, Reis-Filho JS, et al. Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Science translational medicine. 2015; 7:302ra133.
12. Beaver JA, Jelovac D, Balukrishna S, Cochran RL, Croessmann S, Zabransky DJ, Wong HY, Valda Toro P, Cidado J, Blair BG, Chu D, Burns T, Higgins MJ, et al. Detection of cancer DNA in plasma of patients with early-stage breast cancer. Clinical cancer research. 2014; 20:2643–2650.
13. Oshiro C, Kagara N, Naoi Y, Shimoda M, Shimomura A, Maruyama N, Shimazu K, Kim SJ, Noguchi S. PIK3CA mutations in serum DNA are predictive of recurrence in primary breast cancer patients. Breast cancer research and treatment. 2015; 150:299–307.
14. Forshew T, Murtaza M, Parkinson C, Gale D, Tsui DW, Kaper F, Dawson SJ, Piskorz AM, Jimenez-Linan M, Bentley D, Hadfield J, May AP, Caldas C, et al. Noninvasive identification and monitoring of cancer mutations by targeted deep sequencing of plasma DNA. Science translational medicine. 2012; 4:136ra168.
15. Schwarzenbach H, Hoon DS, Pantel K. Cell-free nucleic acids as biomarkers in cancer patients. Nature reviews Cancer. 2011; 11:426–437.
16. De Mattos-Arruda L, Caldas C. Cell-free circulating tumour DNA as a liquid biopsy in breast cancer. Molecular oncology. 2015.
17. Gal S, Fidler C, Lo YM, Taylor M, Han C, Moore J, Harris AL, Wainscoat JS. Quantitation of circulating DNA in the serum of breast cancer patients by real-time PCR. British journal of cancer. 2004; 90:1211–1215.
18. Huang ZH, Li LH, Hua D. Quantitative analysis of plasma circulating DNA at diagnosis and during follow-up of breast cancer patients. Cancer letters. 2006; 243:64–70.
19. Umetani N, Giuliano AE, Hiramatsu SH, Amersi F, Nakagawa T, Martino S, Hoon DS. Prediction of breast tumor progression by integrity of free circulating DNA in serum. Journal of clinical oncology. 2006; 24:4270–4276.
20. Catarino R, Ferreira MM, Rodrigues H, Coelho A, Nogal A, Sousa A, Medeiros R. Quantification of free circulating tumor DNA as a diagnostic marker for breast cancer. DNA and cell biology. 2008; 27:415–421.
21. Kohler C, Radpour R, Barekati Z, Asadollahi R, Bitzer J, Wight E, Burki N, Diesch C, Holzgreve W, Zhong XY. Levels of plasma circulating cell free nuclear and mitochondrial DNA as potential biomarkers for breast tumors. Molecular cancer. 2009; 8:105.
22. Roth C, Pantel K, Muller V, Rack B, Kasimir-Bauer S, Janni W, Schwarzenbach H. Apoptosis-related deregulation of proteolytic activities and high serum levels of circulating nucleosomes and DNA in blood correlate with breast cancer progression. BMC cancer. 2011; 11:4.
23. Gong B, Xue J, Yu J, Li H, Hu H, Yen H, Hu J, Dong Q, Chen F. Cell-free DNA in blood is a potential diagnostic biomarker of breast cancer. Oncology letters. 2012; 3:897–900.
24. Stotzer OJ, Lehner J, Fersching-Gierlich D, Nagel D, Holdenrieder S. Diagnostic relevance of plasma DNA and DNA integrity for breast cancer. Tumour biology. 2014; 35:1183–1191.
25. Madhavan D, Wallwiener M, Bents K, Zucknick M, Nees J, Schott S, Cuk K, Riethdorf S, Trumpp A, Pantel K, Sohn C, Schneeweiss A, Surowy H, et al. Plasma DNA integrity as a biomarker for primary and metastatic breast cancer and potential marker for early diagnosis. Breast cancer research and treatment. 2014; 146:163–174.
26. Tangvarasittichai O, Jaiwang W, Tangvarasittichai S. The plasma DNA concentration as a potential breast cancer screening marker. Indian journal of clinical biochemistry. 2015; 30:55–58.
27. Zhang L, Song X, Wang X, Xie Y, Wang Z, Xu Y, You X, Liang Z, Cao H. Circulating DNA of HOTAIR in serum is a novel biomarker for breast cancer. Breast cancer research and treatment. 2015; 152:199–208.
28. Wu X, Tanaka H. Aberrant reduction of telomere repetitive sequences in plasma cell-free DNA for early breast cancer detection. Oncotarget. 2015; 6:29795–807. doi: 10.18632/oncotarget.5083.
29. Zanetti-Dallenbach R, Wight E, Fan AX, Lapaire O, Hahn S, Holzgreve W, Zhong XY. Positive correlation of cell-free DNA in plasma/serum in patients with malignant and benign breast disease. Anticancer research. 2008; 28:921–925.
30. Seefeld M, El Tarhouny S, Fan AX, Hahn S, Holzgreve W, Zhong XY. Parallel assessment of circulatory cell-free DNA by PCR and nucleosomes by ELISA in breast tumors. The International journal of biological markers. 2008; 23:69–73.
31. Bechmann T, Andersen RF, Pallisgaard N, Madsen JS, Maae E, Jakobsen EH, Bak Jylling AM, Steffensen KD, Jakobsen A. Plasma HER2 amplification in cell-free DNA during neoadjuvant chemotherapy in breast cancer. Journal of cancer research and clinical oncology. 2013; 139:995–1003.
32. Lehner J, Stotzer OJ, Fersching D, Nagel D, Holdenrieder S. Circulating plasma DNA and DNA integrity in breast cancer patients undergoing neoadjuvant chemotherapy. Clinica chimica acta. 2013; 425:206–211.
33. Nicolini C, Ens C, Cerutti T, Roehe AV, Agnes G, Damin AP, Alexandre CO. Elevated level of cell-free plasma DNA is associated with advanced-stage breast cancer and metastasis. Clinical chemistry and laboratory medicine. 2013; 51:e277–278.
34. Zhong XY, Ladewig A, Schmid S, Wight E, Hahn S, Holzgreve W. Elevated level of cell-free plasma DNA is associated with breast cancer. Archives of gynecology and obstetrics. 2007; 276:327–331.
35. Wang BG, Huang HY, Chen YC, Bristow RE, Kassauei K, Cheng CC, Roden R, Sokoll LJ, Chan DW, Shih Ie M. Increased plasma DNA integrity in cancer patients. Cancer research. 2003; 63:3966–3968.
36. Mahmoud EH, Fawzy A, Ahmad OK, Ali AM. Plasma Circulating Cell-free Nuclear and Mitochondrial DNA as Potential Biomarkers in the Peripheral Blood of Breast Cancer Patients. Asian Pacific journal of cancer prevention. 2015; 16:8299–8305.
37. Silva JM, Dominguez G, Garcia JM, Gonzalez R, Villanueva MJ, Navarro F, Provencio M, San Martin S, Espana P, Bonilla F. Presence of tumor DNA in plasma of breast cancer patients: clinicopathological correlations. Cancer research. 1999; 59:3251–3256.
38. Muller HM, Widschwendter A, Fiegl H, Ivarsson L, Goebel G, Perkmann E, Marth C, Widschwendter M. DNA methylation in serum of breast cancer patients: an independent prognostic marker. Cancer Res. 2003; 63:7641–7645.
39. Dulaimi E, Hillinck J, Ibanez de Caceres I, Al-Saleem T, Cairns P. Tumor suppressor gene promoter hypermethylation in serum of breast cancer patients. Clinical cancer research. 2004; 10:6189–6193.
40. Skvortsova TE, Rykova EY, Tamkovich SN, Bryzgunova OE, Starikov AV, Kuznetsova NP, Vlassov VV, Laktionov PP. Cell-free and cell-bound circulating DNA in breast tumours: DNA quantification and analysis of tumour-related gene methylation. British journal of cancer. 2006; 94:1492–1495.
41. Korshunova Y, Maloney RK, Lakey N, Citek RW, Bacher B, Budiman A, Ordway JM, McCombie WR, Leon J, Jeddeloh JA, McPherson JD. Massively parallel bisulphite pyrosequencing reveals the molecular complexity of breast cancer-associated cytosine-methylation patterns obtained from tissue and serum DNA. Genome research. 2008; 18:19–29.
42. Fiegl H, Millinger S, Mueller-Holzner E, Marth C, Ensinger C, Berger A, Klocker H, Goebel G, Widschwendter M. Circulating tumor-specific DNA: a marker for monitoring efficacy of adjuvant therapy in cancer patients. Cancer research. 2005; 65:1141–1145.
43. Rykova E, Skvortsova TE, Hoffmann AL, Tamkovich SN, Starikov AV, Bryzgunova OE, Permiakova VI, Warnecke JM, Sczakiel G, Vlasov VV, Laktionov PP. [Breast cancer diagnostics based on extracellular DNA and RNA circulating in blood] [Article in Russian]. Biomeditsinskaia khimiia. 2008; 54:94–103.
44. Iqbal S, Vishnubhatla S, Raina V, Sharma S, Gogia A, Deo SS, Mathur S, Shukla NK. Circulating cell-free DNA and its integrity as a prognostic marker for breast cancer. SpringerPlus. 2015; 4:265.
45. Chen X, Bonnefoi H, Diebold-Berger S, Lyautey J, Lederrey C, Faltin-Traub E, Stroun M, Anker P. Detecting tumor-related alterations in plasma or serum DNA of patients diagnosed with breast cancer. Clinical cancer research. 1999; 5:2297–2303.
46. Shaw JA, Smith BM, Walsh T, Johnson S, Primrose L, Slade MJ, Walker RA, Coombes RC. Microsatellite alterations plasma DNA of primary breast cancer patients. Clinical cancer research. 2000; 6:1119–1124.
47. Silva JM, Garcia JM, Dominguez G, Silva J, Miralles C, Cantos B, Coca S, Provencio M, Espana P, Bonilla F. Persistence of tumor DNA in plasma of breast cancer patients after mastectomy. Annals of surgical oncology. 2002; 9:71–76.
48. Schwarzenbach H, Muller V, Milde-Langosch K, Steinbach B, Pantel K. Evaluation of cell-free tumour DNA and RNA in patients with breast cancer and benign breast disease. Molecular bioSystems. 2011; 7:2848–2854.
49. Higgins MJ, Jelovac D, Barnathan E, Blair B, Slater S, Powers P, Zorzi J, Jeter SC, Oliver GR, Fetting J, Emens L, Riley C, Stearns V, et al. Detection of tumor PIK3CA status in metastatic breast cancer using peripheral blood. Clinical cancer research. 2012; 18:3462–3469.
50. Shaw JA, Page K, Blighe K, Hava N, Guttery D, Ward B, Brown J, Ruangpratheep C, Stebbing J, Payne R, Palmieri C, Cleator S, Walker RA, Coombes RC. Genomic analysis of circulating cell-free DNA infers breast cancer dormancy. Genome research. 2012; 22:220–231.
51. Beck J, Urnovitz HB, Mitchell WM, Schutz E. Next generation sequencing of serum circulating nucleic acids from patients with invasive ductal breast cancer reveals differences to healthy and nonmalignant controls. Molecular cancer research. 2010; 8:335–342.
52. Dawson SJ, Tsui DW, Murtaza M, Biggs H, Rueda OM, Chin SF, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B, Rajan S, Humphray S, Becq J, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. The New England journal of medicine. 2013; 368:1199–1209.
53. Kirkizlar E, Zimmermann B, Constantin T, Swenerton R, Hoang B, Wayham N, Babiarz JE, Demko Z, Pelham RJ, Kareht S, Simon AL, Jinnett KN, Rabinowitz M, et al. Detection of Clonal and Subclonal Copy-Number Variants in Cell-Free DNA from Patients with Breast Cancer Using a Massively Multiplexed PCR Methodology. Transl Oncol. 2015; 8:407–416.
54. Leon SA, Shapiro B, Sklaroff DM, Yaros MJ. Free DNA in the serum of cancer patients and the effect of therapy. Cancer research. 1977; 37:646–650.
55. Agassi R, Czeiger D, Shaked G, Avriel A, Sheynin J, Lavrenkov K, Ariad S, Douvdevani A. Measurement of circulating cell-free DNA levels by a simple fluorescent test in patients with breast cancer. American journal of clinical pathology. 2015; 143:18–24.
56. Klevebring D, Neiman M, Sundling S, Eriksson L, Darai Ramqvist E, Celebioglu F, Czene K, Hall P, Egevad L, Gronberg H, Lindberg J. Evaluation of exome sequencing to estimate tumor burden in plasma. PloS one. 2014; 9:e104417.
57. Andronas M, Dlay SS, Sherbet GV. Oestrogen and progesterone receptor expression influences DNA ploidy and the proliferation potential of breast cancer cells. Anticancer research. 2003; 23:3029–3039.
58. Ellis MJ, Ding L, Shen D, Luo J, Suman VJ, Wallis JW, Van Tine BA, Hoog J, Goiffon RJ, Goldstein TC, Ng S, Lin L, Crowder R, et al. Whole-genome analysis informs breast cancer response to aromatase inhibition. Nature. 2012; 486:353–360.
59. Silva JM, Dominguez G, Villanueva MJ, Gonzalez R, Garcia JM, Corbacho C, Provencio M, Espana P, Bonilla F. Aberrant DNA methylation of the p16INK4a gene in plasma DNA of breast cancer patients. British journal of cancer. 1999; 80:1262–1264.
60. Kamel AM, Teama S, Fawzy A, El Deftar M. Plasma DNA integrity index as a potential molecular diagnostic marker for breast cancer. Tumour biology. 2016; 37:7565–7572.
61. Thriveni K, Deshmane V, Ramaswamy G, Krishnamoorthy L. Diagnostic Significance of CA15-3 with Combination of HER-2/neu Values at 85th Percentiles in Breast Cancer. Indian journal of clinical biochemistry. 2013; 28:136–140.
62. Leyland-Jones B, Smith BR. Serum HER2 testing in patients with HER2-positive breast cancer: the death knell tolls. The Lancet Oncology. 2011; 12:286–295.
63. Glas AS, Lijmer JG, Prins MH, Bonsel GJ, Bossuyt PM. The diagnostic odds ratio: a single indicator of test performance. Journal of clinical epidemiology. 2003; 56:1129–1135.
64. Zhang R, Shao F, Wu X, Ying K. Value of quantitative analysis of circulating cell free DNA as a screening tool for lung cancer: a meta-analysis. Lung cancer. 2010; 69:225–231.
65. Zhou Q, Li W, Leng B, Zheng W, He Z, Zuo M, Chen A. Circulating Cell Free DNA as the Diagnostic Marker for Ovarian Cancer: A Systematic Review and Meta-Analysis. PloS one. 2016; 11:e0155495.
66. Liao W, Mao Y, Ge P, Yang H, Xu H, Lu X, Sang X, Zhong S. Value of quantitative and qualitative analyses of circulating cell-free DNA as diagnostic tools for hepatocellular carcinoma: a meta-analysis. Medicine. 2015; 94:e722.
67. Deeks JJ. Systematic reviews in health care: Systematic reviews of evaluations of diagnostic and screening tests. Bmj. 2001; 323:157–162.
68. Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, Somerfield MR, Hayes DF, Bast RC, Jr. and American Society of Clinical O. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. Journal of clinical oncology. 2007; 25:5287–5312.
69. Deville WL, Buntinx F, Bouter LM, Montori VM, de Vet HC, van der Windt DA, Bezemer PD. Conducting systematic reviews of diagnostic studies: didactic guidelines. BMC medical research methodology. 2002; 2:9.
70. Walter SD. Properties of the summary receiver operating characteristic (SROC) curve for diagnostic test data. Statistics in medicine. 2002; 21:1237–1256.
71. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Statistics in medicine. 2002; 21:1539–1558.
72. Thompson SG, Higgins JP. How should meta-regression analyses be undertaken and interpreted? Statistics in medicine. 2002; 21:1559–1573.
73. Suzuki S, Moro-oka T, Choudhry NK. The conditional relative odds ratio provided less biased results for comparing diagnostic test accuracy in meta-analyses. Journal of clinical epidemiology. 2004; 57:461–469.
74. Song F, Gilbody S. Bias in meta-analysis detected by a simple, graphical test. Increase in studies of publication bias coincided with increasing use of meta-analysis. Bmj. 1998; 316:471.

