INTRODUCTION
Colorectal cancer (CRC, including colon cancer and rectal cancer) is the third most commonly diagnosed cancer in males and the second in females, with an estimated 1.4 million cases and 693,900 deaths in 2012. Moreover, the incidence has continued to increase in certain countries where historically, the risk has been low [1]. In 2015, it was projected that 132,700 (69,090 men and 63,610 women) individuals would be newly diagnosed with colorectal cancer, which accounted for a prevalence rate of 8% in both sexes [2]. Almost 694,000 deaths from CRC are estimated to have occurred in 2012, and these accounted for approximately 8.5% of all cancer deaths [3]. With the ever increasing incidence and mortality, CRC is highly regarded as a clinical problem.
Hepatocyte growth factor (HGF, DFNB39, HPTA), which is also called scatter factor, is a gene primarily involved in the regulation of cell growth, motility and morphogenesis [4]. HGF induces complex intracellular signaling networks, which results in cell proliferation and cell survival, and leads to regeneration and homeostasis of various types of epithelial tissues [5]. HGF exerts its biological effects via its tyrosine kinase cell surface receptor, hepatocyte growth factor receptor (HGFR), which is also known as mesenchymal to epithelial transition factor (MET) [6, 7]. When it is aberrantly activated, the HGF-MET pathway is involved in the regulation of proliferation, motility, invasion and metastasis via the phosphorylation and activation of downstream signaling pathways, which consequently, promote tumorigenesis [8].
The expression of HGF plays a critical role in cell proliferation and is involved in CRC [9]. Conclusions from published studies on the correlation between HGF and the survival time of patients with CRC were, for the most part, consistent except for the study conducted by Karabulut, which found a negative correlation between HGF expression and the survival [10]. Studies that have revealed the pathogenic roles of HGF in CRC are not rare, but evidence-based medicine has not verified the prognosis or the survival time of CRC patients in the context of HGF expression. Whether high HGF expression leads to a poor prognosis of CRC remains inconclusive.
Therefore, we systematically evaluated the correlation between HGF and the prognosis and survival of CRC patients and provided clinical guidance for the treatment of CRC.
RESULTS
Search results and study characteristics
The study searches were performed as shown in Figure 1. At first, 329 studies were obtained from multiple databases, and then, 49 duplicates were removed. Of the relevant studies, 247 studies were excluded based on the title and abstract. Subsequently, 33 studies were assessed by screening the full-text, among which 24 articles were excluded. Eventually, nine studies were included in the meta-analysis [5, 10–17].
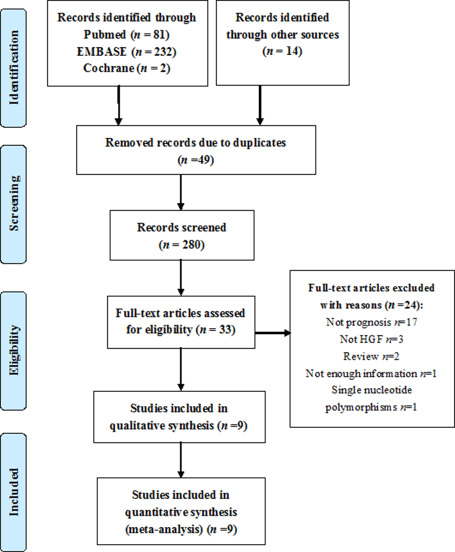
Figure 1: Flow chart of the search strategy.
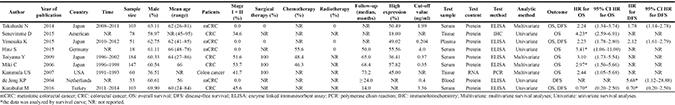
All the studies were published from 2004 to 2016, with incremental tendency since 2015. Most of the studies were conducted in developed countries, such as the USA, Germany, Netherlands, Japan and Turkey. Data on patients in six studies were collected from 1991 to 2014, while other studies did not mention the collection time [5, 13, 17]. Altogether, the nine studies included 777 patients, and each study contained samples that ranged from 18 to 184 patients. The proportion of males was approximately 50–70% in all studies except for the study performed by Kammula (36.5%) [16]. The age of the included patients ranged from 24 to 95 years. Four studies enrolled patients with metastatic CRC (mCRC), while others enrolled patients with stage I-IV CRC. Among the nine studies, five used univariate analyses [5, 10, 12, 13, 17], and four used multivariate analyses [11, 14–16]. The cut-off value of HGF ranged from 0.204 to 4.0 ng/ml in most of the included studies.
Eight studies reported the prognostic value of HGF with respect to overall survival (OS) in patients with CRC [5, 10–16]. Among them, four directly reported hazard ratio (HR) [11, 12, 14, 16], while the others provided survival curves (Table 1) [5, 10, 13, 15]. Seven of the eight studies found that the over-expression of HGF was an indicator for poor prognosis and poor OS, while the remaining study revealed no statistical significance between HGF over-expression and OS [10]. Four studies reported the prognostic value of HGF with respect to disease-free survival (DFS) in patients with CRC [10–12, 17]. Of the four studies, 2 studies directly reported HR, while the others reported survival curves [11, 12].
Table 1: The main characteristics and results of the included studies
Outcomes of the meta-analysis
Eight studies containing the data of HGF and OS in CRC patients were included in this research. The combined HR for the over-expression of HGF on OS was 2.50 (95% confidence interval [CI]: 2.12–2.96); it was based on an analysis under fixed effects model (inconsistency index [I2] = 33.1%, χ2 = 10.47, P = 0.164) (Table 2, Figure 2). Random effect model was adopted in studies performing univariate analysis, and in result the adjusted combined HR was 2.51 (95% CI: 1.44–4.35) (Figure 3A). Fixed effect model was adopted in studies performing multivariate analysis (I2 = 0.0%, χ2 = 0.86 P = 0.836), and the combined HR was 2.65 (95% CI: 1.95–3.60) (Figure 3B) and no heterogeneity was observed. Subgroup analyses on patient categories presented that the combined HR of stage I-IV CRC and mCRC were homogenous, and under fixed effect model, the combined HR of stage I-IV CRC patients and mCRC patients were 3.08 (95% CI: 2.30–4.14) (Figure 3C) and 2.26 (95% CI: 1.85–2.78) (Figure 3D), respectively. The studies conducted in Asia presented a homogeneous result (I2 = 0.0%, χ2 = 1.60, P = 0.659) and the combined HR was 2.37 (95% CI: 1.96–2.85) (Figure 3E). The combined HR of the studies conducted outside from Asia was 3.15 (95% CI: 2.15–4.60) (Figure 3F).
Table 2: The results of the meta-analysis
Number of studies |
Patients |
HR (95% CI) |
Heterogeneity |
|||
|---|---|---|---|---|---|---|
I2 |
χ2 |
P |
||||
Overall survival |
||||||
All |
8 |
744 |
2.50 (2.12–2.96) |
33.1% |
10.47 |
0.164 |
Univariate analysis |
4 |
250 |
2.51 (1.44–4.35)* |
68.2% |
9.43 |
0.024 |
Multivariate analysis |
4 |
494 |
2.65 (1.95–3.60) |
0.0% |
0.86 |
0.836 |
Patients |
||||||
I–IV CRC |
5 |
572 |
3.08 (2.30–4.14) |
43.9% |
7.14 |
0.129 |
mCRC |
3 |
172 |
2.26 (1.85–2.78) |
0.0% |
0.49 |
0.784 |
Country |
||||||
Asian |
4 |
485 |
2.37 (1.96–2.85) |
0.0% |
1.60 |
0.659 |
Non-Asian |
4 |
259 |
3.15 (2.15–4.60) |
58.0% |
7.14 |
0.068 |
Disease-free survival |
||||||
All |
4 |
290 |
1.99 (1.59–2.50) |
35.1% |
4.62 |
0.201 |
Univariate analysis |
3 |
187 |
2.07 (1.59–2.70) |
53.4% |
4.30 |
0.117 |
Multivariate analysis |
1 |
103 |
1.78 (1.14–2.78) |
- |
- |
- |
Patients |
||||||
I-IV CRC |
1 |
103 |
0.70 (0.20–2.50) |
- |
- |
- |
mCRC |
3 |
187 |
2.06 (1.64–2.60) |
0.0% |
1.94 |
0.378 |
Country |
||||||
Asian |
2 |
154 |
2.02 (1.60–2.55) |
0.0% |
0.42 |
0.515 |
Non-Asian |
2 |
136 |
1.87 (0.24–14.44)* |
74.6% |
3.94 |
0.047 |
*Results based on a random effects model. -: not applicable.
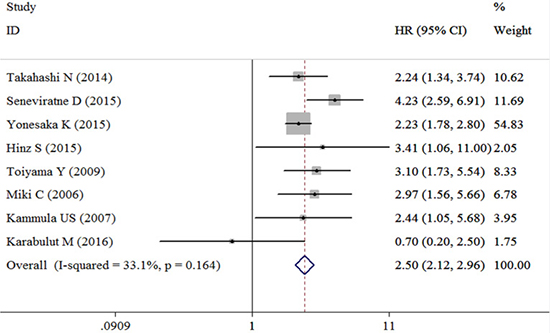
Figure 2: Forest plot evaluating the combined HR between HGF and OS.

Figure 3: Forest plot of the subgroup analysis of HGF and OS ((A): univariate analysis; (B): multivariate analysis; (C): Asian countries; (D): non-Asian countries; (E): I-IV CRC patients; (F): mCRC patients).
Four studies containing data of HGF and DFS in CRC patients were included in this research. The combined HR for the over-expression of HGF on DFS was 1.99 (95% CI: 1.59–2.50) (Table 2, Figure 4). Among these studies three with univariate analysis were homogenous (I2 = 53.4%, χ2 = 4.30, P = 0.117), and the combined HR was 2.07 (95% CI: 1.59–2.70) (Figure 5A). The HR for the studies with multivariate analysis was 1.78 (1.14–2.78) (Figure 5B). The combined HR for the patients with I-IV CRC and mCRC were 0.70 (95% CI: 0.20–2.50) (Figure 5C) and 2.06 (95% CI: 1.64–2.60) (Figure 5D), respectively. Among these four studies, two were conducted in Asian countries and the others were conducted outside from Asia. The combined HR for the Asian studies and the non-Asian studies were 2.02 (95% CI: 1.60–2.55) (Figure 5E) and 1.87 (95% CI: 0.24–14.44) (Figure 5F), respectively.
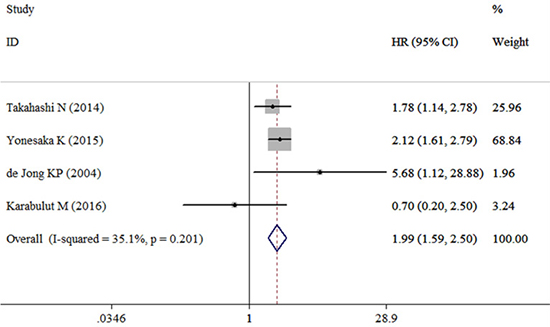
Figure 4: Forest plot evaluating the combined HR between HGF and DFS.
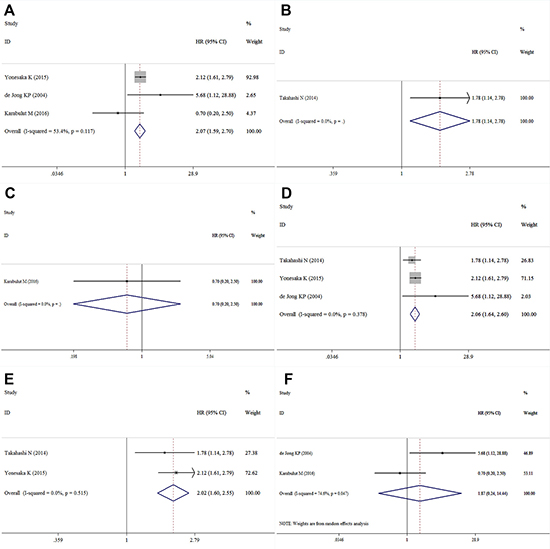
Figure 5: Forest plot of the subgroup analysis of HGF and DFS ((A): univariate analysis; (B): multivariate analysis; (C): Asian countries; (D): non-Asian countries; (E): I–IV CRC patients; (F): mCRC patients).
Begg’s test was used to evaluate the publication bias. The results of Begg’s test for both OS and DFS revealed no publication bias (POS = 0.536, PDFS = 1.000) (Table 3, Figure 6A, and Figure 6B). A sensitivity analysis was then used to evaluate the influence of potential publication bias (Figure 7A, and Figure 7B). The funnel plots for publication bias were basically symmetric, which indicated the stability of the results (Figure 8A, and Figure 8B).
Table 3: Results of Begg’s and Egger’s tests
Number of studies |
Begg’s test |
Egger’s test |
|||
|---|---|---|---|---|---|
Z value |
P |
t value |
P |
||
Overall survival |
|||||
All |
8 |
0.62 |
0.536 |
0.23 |
0.827 |
Univariate analysis |
4 |
0.34 |
0.734 |
0.02 |
0.987 |
Multivariate analysis |
4 |
–0.34 |
1.000 |
0.27 |
0.814 |
Patients |
|||||
CRC |
5 |
2.20 |
0.027 |
–6.97 |
0.006 |
mCRC |
3 |
1.04 |
0.296 |
1.49 |
0.377 |
Country |
|||||
Asian |
4 |
1.02 |
0.308 |
1.87 |
0.202 |
Non-Asian |
4 |
1.02 |
0.308 |
–1.79 |
0.215 |
Disease-free survival |
|||||
All |
4 |
–0.34 |
1.000 |
–0.24 |
0.832 |
Univariate analysis |
3 |
–0.00 |
1.000 |
–0.13 |
0.916 |
Multivariate analysis |
1 |
- |
- |
- |
- |
Patients |
|||||
CRC |
1 |
- |
- |
- |
- |
mCRC |
3 |
0.00 |
1.000 |
0.83 |
0.559 |
-: not applicable.
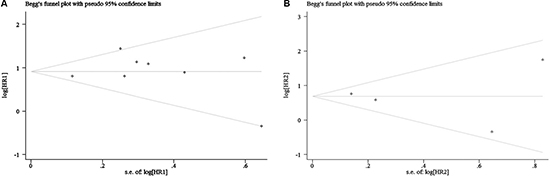
Figure 6: Funnel plot for all the included studies ((A): OS; (B): DFS).
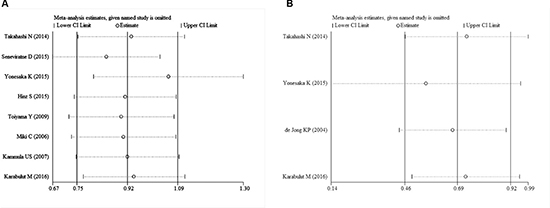
Figure 7: Sensitivity analysis for the included studies ((A): OS; (B): DFS).
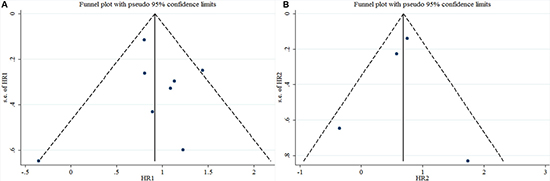
Figure 8: Funnel plots for the included studies ((A): OS; (B): DFS).
DISCUSSION
This analysis provided evidence that over-expression of HGF could be a prognostic indicator in CRC. The over-expression of HGF contributed to lower OS and DFS in CRC patients via the targeting of HGF/MET signaling pathways. HGF, as the only known ligand for the MET proto-oncogene product of receptor tyrosine kinase, is converted into an active form that dimerizes and activates the MET receptor [18]. An active HGF/MET signal, which is involved in a number of malignancies, is associated with a poor prognosis and serves as an early predictor of further metastasis [19]. Toiyama reported that a high HGF level was associated with CRC development, lymphatic or distant invasive phenotypes and survival [14]. Therefore, poor prognosis is associated with a high HGF level. Similar results were also found in patients with gastric cancer [20], liver cancer [21], glioma [22], non-small cell lung cancer [23, 24], breast cancer [25, 26] and thyroid cancer [27, 28]. Additionally, two previous meta-analyses demonstrated that MET was associated with a poor prognosis of colorectal cancer [29] and gastric cancer [30].
Therefore, HGF-targeted therapy should be considered since the over-expression of HGF was confirmed to be the cause of poor prognosis in CRC. HGF inhibitors could potentially be effective in the inhibition of the HGF/MET axis in CRC patients with over-expression of HGF [12]. A randomized phase Ib/II trial indicated that anti-HGF monoclonal antibodies improved the overall remission rate and DFS in patients with mCRC [31]. Van Cutsem reported that combination therapy (including rilotumumab, an HGF inhibitor) showed promising activity compared with single-agent panitumumab in patients with chemo-refractory tumors (overall response rate was 31% versus 21%; PFS was 5.2 versus 3.7 months) [31]. Moreover, Yonesaka found that drug resistance to anti-EGFR antibody therapy caused by HGF might lead to a decreased OS [12]. Anti-EGFR antibody treatment was superior as a front-line therapy for patients with mCRC [32]. Hua demonstrated that an HGF inhibitor restored the sensitivity of the anti-EGFR antibody, which infers that HGF-targeted therapy might promote OS of CRC patients [33]. The data discussed above support the concept that the over-expression of HGF is a prognostic indicator in CRC, even though normal expression of HGF contributes to organ growth and development due to its capacity to aid in the regeneration of damaged liver, kidney and lung tissue and because of its protective function in the heart and brain [34].
A subgroup analysis showed that the results between different features were consistent, which means that the eligible studies were homogeneous. For example, the combined HR of the patients with stage I-IV CRC and the patients with mCRC were 3.08 and 2.26, respectively. The HRs of the stage I-IV CRC patients and the mCRC patients were not significantly different in terms of OS. The combined HR in the studies that used univariate analysis was 2.51, while that for the studies that used multivariate analysis was 2.65. The HRs of the CRC patients from non-Asian countries and those from Asian countries were not different. These results suggest that HGF had the same effect on the survival time of patients from different countries.
There are several limitations to this study: (1) The classification criteria for the over-expression and normal expression of HGF vary in the included studies, which might have been a confounding factor. However, the effects of different cut-off value would not be substantial. The reasons were as follow: The cut-off value of HGF ranged from 0.204 to 4.0 ng/ml in most of the eligible articles. Most of the eligible articles used scientific methods to classify HGF levels. The definition of the high expression of HGF is still controversial under current circumstance, similar to other genes in cancer, such as the carcino-embryonic antigen in CRC [35]. (2) Some HRs and their 95% CIs were calculated from the data extracted in survival curves (Kaplan-Meier curves), which might have caused subjective bias [36]. (3) Although we attempted to collect all relevant data on the patients included in these studies, some data might still be missing. For example, the treatment given to patients in some of the included studies was not reported in detail. The treatment played an important role in survival time, and different treatments might affect the prognosis of patients with over-expression of HGF. (4) The number of the included studies was small, especially in the study that reported DFS as its main outcome.
In conclusion, this meta-analysis demonstrated that HGF is associated with a poor prognosis (including poor OS and DFS) in patients with CRC. In this case, HGF may be a promising, new therapeutic target for CRC and may enable clinical practitioners to better predict patient prognosis through the detection of HGF levels in patients. However, this conclusion should be further confirmed by large-sample cohort studies.
MATERIALS AND METHODS
Search strategy
One reviewer (CXL) conducted a systematic literature search of PubMed, EMBASE, and the Cochrane Central Register of Controlled Trials databases from their inception until March 14th, 2016. The search included the following terms: colorectal cancer (including colon cancer and rectal cancer), hepatocyte growth factor (HGF, DFNB39, HPTA), and prognosis. We combined the term appropriately with MeSH terms. Details of the search strategies are shown in the Supplementary Appendix File 1. The meta-analysis was conducted in accordance with the PRISMA statement [37, 38]; the PRISMA 2009 Checklist is reported in the Supplementary Appendix File 2.
Study selection
Studies that explored the correlation between HGF and the prognosis of patients with CRC were included. The eligible studies included cross-sectional studies, cohort studies, or randomized controlled trials. HR, its 95% CI, or the relevant information (such as a survival curve, namely Kaplan-Meier curves) was provided. The studies were also reported as full articles, which were published in English. The included patients were diagnosed with colorectal cancer (including colon cancer or rectal cancer), and patients were considered regardless of race, region, clinical stage, histological type, and treatment regimen. The prognostic outcomes included OS, DFS, and relapse-free survival (RFS). Studies published in abstract form were considered only if sufficient outcome data could be retrieved from the abstract or as a result of contact with the authors.
Data extraction
Duplicate studies from different databases were identified, and the remaining abstracts were read for eligibility by two independent authors (LMG and MJL); the third author was involved in the reconciliation of studies with inconsistent results (CXL). The full texts of potentially eligible studies were retrieved and reviewed independently by two authors (HCY and ZQY). Any disagreements were recorded and resolved by consensus under the guidance of the third author (CXL).
The eligible studies were reviewed, and data were extracted independently by two authors (HCY and ZQY). The study information (the first author, the year of publication), study participants (the characteristics and sample sizes), characteristics of HGF, and prognostic outcomes (OS, DFS, and RFS) were extracted. If data from any of the above categories were not reported in the study, the item was recorded as “NR” (not reported).
Data synthesis and analysis
The eligible studies were divided into the OS and DFS groups for the analysis. RFS was treated as DFS. The HGF value was classified as “over-expression” (high expression) or “low-expression” (normal expression).
The HR and its 95% CI were used to measure the effect of the over-expression of HGF on survival. If the HR and its 95% CI were given explicitly in the studies, we used the crude values. If these indexes were not given explicitly, they were calculated from the available numerical data (or survival curve, namely Kaplan-Meier curves) using the methods reported by Tierney et al. [39].
The heterogeneity of the individual HR was calculated using Chi-square tests. A heterogeneity test with inconsistency index statistic and Q statistic was also performed. If HRs among the studies were homogenous, a fixed effects model was used for analysis; if not, a random effects model was used. Subgroup analyses were performed according to different countries (Asian and non-Asian countries) and methods of analysis (univariate analysis, multivariate analysis). Some studies enrolled patients with metastatic colorectal cancer, while others enrolled patients with stage I-IV CRC. Subgroup analyses were also conducted according to the patient categories (I–IV CRC and mCRC).
A P value ≤ 0.05 was considered statistically significant. An observed HR >1 implied a worse prognosis in the case of HGF over-expression compared with the low expression of HGF. Publication bias was evaluated using Begg’s test and funnel plots. All analyses were performed using STATA version 12.0.
ACKNOWLEDGMENTS AND FUNDING
This study was funded by the National Natural Science Foundation of China (81403296, 81373786), the Outstanding Youth Foundation of Guangdong Province Colleges and Universities (YQ2015041), the Young Talents Foundation of Guangzhou University of Chinese Medicine (QNYC20140101), the Torch Plan of Guangzhou University of Chinese Medicine (XH20140105), the Science and Technology Program of Guangdong (2014A020212274), and the Science and Technology Program of Guangzhou (201510010044).
CONFLICTS OF INTEREST
All the authors declare no conflicts of interest.
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108.
2. Favoriti P, Carbone G, Greco M, Pirozzi F, Pirozzi REM, Corcione F. Worldwide burden of colorectal cancer: a review. Updates in Surgery. 2016; 68:7–11.
3. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359-E386.
4. Blumenschein GJ, Mills GB, Gonzalez-Angulo AM. Targeting the hepatocyte growth factor-cMET axis in cancer therapy. J Clin Oncol. 2012; 30:3287–3296.
5. Seneviratne D, Ma J, Tan X, Kwon Y, Muhammad E, Melhem M, DeFrances MC, Zarnegar R. Genomic Instability Causes HGF Gene Activation in Colon Cancer Cells, Promoting Their Resistance to Necroptosis. Gastroenterology. 2015; 148:181–191.
6. Trusolino L, Bertotti A, Comoglio PM. MET signalling: principles and functions in development, organ regeneration and cancer. Nat Rev Mol Cell Bio. 2010; 11:834–848.
7. Gherardi E, Birchmeier W, Birchmeier C, Woude GV. Targeting MET in cancer: rationale and progress. Nat Rev Cancer. 2012; 12:89–103.
8. Salgia R. Role of c-Met in cancer: emphasis on lung cancer. Semin Oncol. 2009; 36:S52-S58.
9. Elliott VA, Rychahou P, Zaytseva YY, Evers BM. Activation of c-Met and upregulation of CD44 expression are associated with the metastatic phenotype in the colorectal cancer liver metastasis model. Plos One. 2014; 9:e97432.
10. Mehmet Karabulut, Gunaldi Ham, Hakan Seyit, Faruk Msn. clinical significance of serum hepatocyte growth factor(HGF) and its receptor cMET levels in colorectal cancer patients. Acta Medica Mediterr. 2016; 179–187.
11. Takahashi N, Yamada Y, Furuta K, Honma Y, Iwasa S, Takashima A, Kato K, Hamaguchi T, Shimada Y. Serum levels of hepatocyte growth factor and epiregulin are associated with the prognosis on anti-EGFR antibody treatment in KRAS wild-type metastatic colorectal cancer. Brit J Cancer. 2014; 110:2716–2727.
12. Yonesaka K, Satoh T, Ueda S, Yoshida T, Takeda M, Shimizu T, Okamoto I, Nishio K, Tamura T, Nakagawa K. Circulating hepatocyte growth factor is correlated with resistance to cetuximab in metastatic colorectal cancer. Anticancer Res. 2015; 35:1683–1689.
13. Hinz S, Tepel J, Roder C, Kalthoff H, Becker T. Profile of serum factors and disseminated tumor cells before and after radiofrequency ablation compared to resection of colorectal liver metastases - a pilot study. Anticancer Res. 2015; 35:2961–2967.
14. Toiyama Y, Miki C, Inoue Y, Okugawa Y, Tanaka K, Kusunoki M. Serum hepatocyte growth factor as a prognostic marker for stage II or III colorectal cancer patients. Int J Cancer. 2009; 125:1657–1662.
15. Miki C, Inoue Y, Hiro J, Ojima E, Araki T, Uchida K, Kusunoki M. Combined Measurement of Hepatocyte Growth Factor and Carcinoembryonic Antigen as a Prognostic Marker for Patients with Dukes A and B Colorectal Cancer: Results of a Five-Year Study. Dis Colon Rectum. 2006; 49:1710–1718.
16. Kammula US, Kuntz EJ, Francone TD, Zeng Z, Shia J, Landmann RG, Paty PB, Weiser MR. Molecular co-expression of the c-Met oncogene and hepatocyte growth factor in primary colon cancer predicts tumor stage and clinical outcome. Cancer Lett. 2007; 248:219–228.
17. de Jong KP, Hoedemakers RM, Fidler V, Bijzet J, Limburg PC, Peeters PM, de Vries EG, Slooff MJ. Portal and systemic serum growth factor and acute-phase response after laparotomy or partial hepatectomy in patients with colorectal liver metastases: a prognostic role for C-reactive protein and hepatocyte growth factor. Scand J Gastroenterol. 2004; 39:1141–1148.
18. Joo MK, Park JJ, Chun HJ. Recent updates of precision therapy for gastric cancer: Towards optimal tailored management. World J Gastroenterol. 2016; 22:4638–4650.
19. Takeuchi H, Bilchik A, Saha S, Turner R, Wiese D, Tanaka M, Kuo C, Wang HJ, Hoon DS. c-MET expression level in primary colon cancer: a predictor of tumor invasion and lymph node metastases. Clin Cancer Res. 2003; 9:1480–1488.
20. Hao NB, Tang B, Wang GZ, Xie R, Hu CJ, Wang SM, Wu YY, Liu E, Xie X, Yang SM. Hepatocyte growth factor (HGF) upregulates heparanase expression via the PI3K/Akt/NF-kappaB signaling pathway for gastric cancer metastasis. Cancer Lett. 2015; 361:57–66.
21. Chau GY, Lui WY, Chi CW, Chau YP, Li AF, Kao HL, Wu CW. Significance of serum hepatocyte growth factor levels in patients with hepatocellular carcinoma undergoing hepatic resection. European Journal of Surgical Oncology (EJSO). 2008; 34:333–338.
22. Guo YF, Wang XB, Tian XY, Li Y, Li B, Huang Q, Zhang M, Li Z. Tumor-derived hepatocyte growth factor is associated with poor prognosis of patients with glioma and influences the chemosensitivity of glioma cell line to cisplatin in vitro. World J Surg Oncol. 2012; 10:128.
23. Naumnik W, Naumnik B, Niklinska W, Ossolinska M, Chyczewska E. Clinical Implications of Hepatocyte Growth Factor, Interleukin-20, and Interleukin-22 in Serum and Bronchoalveolar Fluid of Patients with Non-Small Cell Lung Cancer. Adv Exp Med Biol. 2016.
24. Chen Y, Du M, Wang J, Xing P, Zhang Y, Li F, Lu X. MiRNA-200a expression is inverse correlation with hepatocyte growth factor expression in stromal fibroblasts and its high expression predicts a good prognosis in patients with non-small cell lung cancer. Oncotarget. 2016; 7:48432–48442. doi: 10.18632/oncotarget.10302.
25. Yang H, Zhang C, Cui S. Expression of hepatocyte growth factor in breast cancer and its effect on prognosis and sensitivity to chemotherapy. Mol Med Rep. 2015; 11:1037–1042.
26. Veenstra C, Pérez-Tenorio G, Stelling A, Karlsson E, Mirwani SM. Met and its ligand HGF are associated with clinical outcome in breast cancer. Oncotarget. 2016; 7:37145–37159. doi: 10.18632/oncotarget.9268.
27. Belfiore A, Gangemi P, Costantino A, Russo G, Santonocito GM, Ippolito O, Di Renzo MF, Comoglio P, Fiumara A, Vigneri R. Negative/low expression of the Met/hepatocyte growth factor receptor identifies papillary thyroid carcinomas with high risk of distant metastases. J Clin Endocrinol Metab. 1997; 82:2322–2328.
28. Jung KH, Park BH, Hong S. Progress in cancer therapy targeting c-Met signaling pathway. Arch Pharm Res. 2012; 35:595–604.
29. Gao H, Guan M, Sun Z, Bai C. High c-Met expression is a negative prognostic marker for colorectal cancer: a meta-analysis. Tumour Biol. 2015; 36:515–520.
30. Peng Z, Zhu Y, Wang Q, Gao J, Li Y, Li Y, Ge S, Shen L. Prognostic significance of MET amplification and expression in gastric cancer: a systematic review with meta-analysis. Plos One. 2014; 9:e84502.
31. Van Cutsem E, Eng C, Nowara E, Wieboda-Sadlej A, Tebbutt NC, Mitchell E, Davidenko I, Stephenson J, Elez E, Prenen H, Deng H, Tang R, McCaffery I, et al. Randomized Phase Ib/II Trial of Rilotumumab or Ganitumab with Panitumumab versus Panitumumab Alone in Patients with Wild-type KRAS Metastatic Colorectal Cancer. Clin Cancer Res. 2014; 20:4240–4250.
32. Tamburini E, Rudnas B, Gianni L, Drudi F, Nicoletti S, Santelmo C, Ridolfi C, Pasini G, Fabbri P, Papi M. E02*Anti-EGFR or Bevacizumab in first line treatment of RAS wild type metastatic colorectal neoplasm (RwtMCRC): meta-analysis of randomized clincal trials. Aiom. 2015; S365.
33. Yang H, Wang R, Peng S, Chen L, Li Q, Wang W. Hepatocyte growth factor reduces sensitivity to the epidermal growth factor receptor-tyrosine kinase inhibitor, gefitinib, in lung adenocarcinoma cells harboring wild-type EGFR. Oncotarget. 2016; 7:16273–16281. doi: 10.18632/oncotarget.7586.
34. Nakamura T, Mizuno S. The discovery of Hepatocyte Growth Factor (HGF) and its significance for cell biology, life sciences and clinical medicine. Proceedings of the Japan Academy, Series B. 2010; 86:588–610.
35. Liu Z, Zhang Y, Niu Y, Li K, Liu X, Chen H, Gao C. A systematic review and meta-analysis of diagnostic and prognostic serum biomarkers of colorectal cancer. Plos One. 2014; 9:e103910.
36. Xiao J, Hu CP, He BX, Chen X, Lu XX, Xie MX, Li W, He SY, You SJ, Chen Q. PTEN expression is a prognostic marker for patients with non-small cell lung cancer: a systematic review and meta-analysis of the literature. Oncotarget. 2016; 36:57832–57840. doi: 10.18632/oncotarget.11068.
37. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int J Surg. 2010; 8:336–341.
38. Zeng X, Zhang Y, Kwong JS, Zhang C, Li S, Sun F, Niu Y, Du L. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med. 2015; 8:2–10.
39. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007; 8:16.

