INTRODUCTION
Chronic Myeloid Leukemia (CML) is a myelopro- liferative disorder characterized by the presence of the Philadelphia (Ph) chromosome, produced by the reciprocal translocation t(9;22)(q34;q11) [1, 2]. The resulting chimeric BCR/ABL fusion gene encodes for constitutively active tyrosine kinase protein [3]. A small amount of patients (about 5%), displaying typical features of CML, lacks the Ph chromosome by chromosome banding analysis (CBA), meanwhile shows BCR/ABL rearrangement. These cases are reported as Ph-negative (Ph-neg) CML cases and can be identified by fluorescence in situ hybridization (FISH) or by molecular analysis [4].
Two mechanisms have been proposed to explain how this kind of rearrangement can arise: the first one could be a cryptic insertion between chromosomes 9 and 22; the second mechanism could involve two sequential translocations, in which a standard t(9;22) translocation is followed by a reverse translocation with different breakpoints, that reconstitutes the normal morphology of the partner chromosomes. FISH analysis on metaphases shows the fusion BCR/ABL gene on derivative chromosome 22 (der(22)) much more frequently than on derivative chromosome 9 (der(9)) [5].
In the past, some authors reported that this kind of rearrangement was associated with worse prognosis than the classic translocation, when the patients were treated with conventional chemotherapy and/or α-interferon (α-IFN) therapy; while others did not find any differences in terms of outcome [6–8]. Moreover to our knowledge, only 9 Ph-neg CML cases treated with imatinib have been described in 5 previous reports [5, 9–12]. The outcome of 2 patients has not been clearly reported. Two patients achieved at least Major Cytogenetic Response (CgR) after imatinib treatment [9, 11–12], meanwhile 5 patients failed the treatment, suggesting that they were more resistant to imatinib therapy [5, 10]. Among the latter, a recent report [10] described two cases with fusion genes on both the homologous chromosomes 9 (Table 1).
Table 1: Ph-neg cases treated with imatinb and outcome reported in literature
Case |
Sex/Age (years) |
Pattern FISH |
del(9q) |
Location of BCR/ABL rearrangement |
Mechanism of rearrangement |
Therapy |
Cytogenetic response |
Reference |
|---|---|---|---|---|---|---|---|---|
1 |
unknown |
1R2G1F |
NO |
der(9) |
Insertion on der(9) |
imatinib |
NR |
Haigh S. et al, 2004 [5] |
2 |
unknown |
2R1G1F |
NO |
der(22) |
Insertion on der(22) |
imatinib |
NR |
Haigh S. et al, 2004 [5] |
3 |
unknown |
2R1G1F |
NO |
der(9) |
2-step mechanism |
imatinib |
NR |
Haigh S. et al, 2004 [5] |
4 |
M/62 |
1R2G1F |
unknown |
der(9) |
2-step mechanism |
imatinib |
CCgR |
Fugazza G. et al, 2005 [9] |
5 |
M/27 |
1R2G1F (80%) |
NO |
der(9) |
Insertion on der(9) |
hydroxyurea |
NR |
Brahmbhatt M. et al, 2014 [10] |
6 |
M/30 |
1R2G1F |
NO |
der(9) |
Insertion on der(9) |
hydroxyurea |
NR |
Brahmbhatt M. et al, 2014 [10] |
7 |
F/47 |
1R2G1F |
NO |
der(9) |
Insertion on der(9) |
hydroxyurea + α-IFN |
unknown |
Batista A.S. et al, 2005 [11] |
8 |
F/31 |
1R1G1F |
YES |
der(22) |
Insertion on der(22) |
hydroxyurea |
unknown |
Batista A.S. et al, 2005 [11] |
9 |
M/27 |
1R2G1F |
YES |
der(9) |
Insertion on der(9) |
Imatinib |
MCgR |
Bennour A. et al, 2011 [12] |
Abbreviations: R=red signal; G=green signal; F=fusion signal; der(9)= derivative of chromosome 9; der(22)= derivative of chromosome 22; SCT= stem cell transplantation; NR= no response; CCgR= Complete Cytogenetic Response; MCgR= Major Cytogenetic Response.
We report an analysis of the clinical, cytogenetic and molecular characteristics of 6 patients with Ph-neg CML, treated with tyrosine kinase inhibitors (TKIs).
RESULTS
At diagnosis, we have analysed 398 CML cases, of which 12 (3%) showed normal karyotype by CBA and BCR/ABL rearrangement by RT-PCR analysis. Of these, only 6 recently diagnosed patients were valuable for CBA, FISH and molecular analysis during TKIs therapy.
Detailed baseline genetic characteristics of the patients are presented in Table 2. RT-PCR analysis of the chimeric BCR/ABL transcript showed b3a2 transcript in 5 patients and b2a2 in only 1 patient (n.2). In 4 evaluable patients, no mutation in ABL gene was found at diagnosis and during the following treatment.
Table 2: FISH and molecular features of the Ph-neg patients
Patients |
FISH
|
Location of BCR/ABL fusion gene |
Molecular transcript |
Mutational status |
|---|---|---|---|---|
1 |
1R1G1F |
chr. 9 |
b3a2 |
unknown |
2 |
1R2G1F |
chr. 9 |
b2a2 |
wild-type |
3 |
1R2G1F |
chr. 9 |
b3a2 |
unknown |
4 |
1R2G1F |
chr. 9 |
b3a2 |
wild-type |
5 |
2R1G1F |
chr. 22 |
b3a2 |
wild-type |
6 |
1R1G2F |
chr. 9 and 22 |
b3a2 |
wild-type |
aUsing Vysis LSI BCR/ABL dual-color, dual fusion FISH probe system: R=red signal, G=green signal, F=fusion signal
Dual-Color Dual Fusion (DCDF) FISH analysis revealed BCR/ABL fusion signal on der(9) in 4 patients (n.1-4), on der(22) in 1 patient (n.5) and on both derivative chromosomes in the last one (n.6).
Three patients (n.2-4), beyond BCR/ABL rearrangement on der(9), revealed a red ABL signal and a green BCR signal on the normal chromosomes and a smaller BCR signal on der(22), as consequence of the insertion of the BCR region into ABL region (Figure 1A). The second smaller BCR signal was not observed in 1 patient (n.1) (Figure 1B). The only patient (n.5) with BCR/ABL fusion signal on der(22) showed BCR signal on the normal chromosome 22 and ABL signals on both chromosome 9 (Figure 1C). The classic FISH pattern (2 fusion, 1 ABL and 1 BCR signals) was observed in the last patient (n.6, Figure 1D). Therefore, 4 different FISH signal patterns were observed in 6 patients.
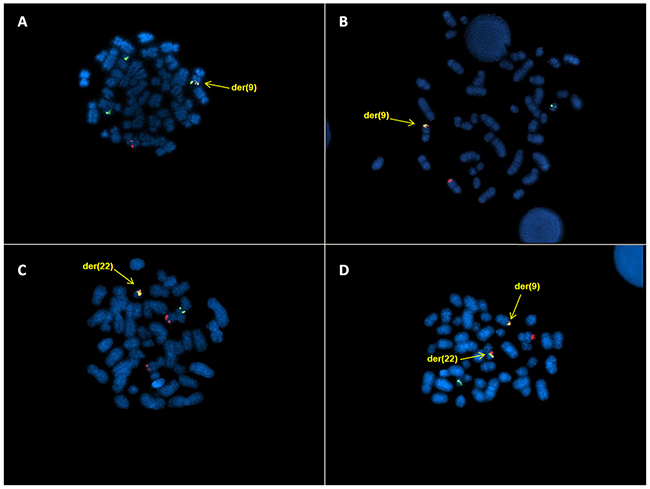
Figure 1: DCDF FISH analysis showing: A. 1R2G1F signal pattern and the BCR/ABL fusion gene on der(9); B. 1R1G1F signal pattern and the BCR/ABL fusion gene on der(9) C. 2R1G1F signal pattern and the BCR/ABL fusion gene on der(22); D. 1R1G2F signal pattern and the BCR/ABL fusion genes on der(9) and der(22).
By Tri-Color Dual Fusion (TCDF) FISH analysis, the rearrangement was confirmed as insertion of the BCR region into the ABL region in the patients with BCR/ABL fusion on der(9) (n.1-4) and insertion of the ABL region into the BCR region in the patient with BCR/ABL fusion on der(22) (n.5). The classic pattern of the last case (n.6) could be explained with 2-step mechanism: the first translocation involved the classic breakpoints and the second translocation with different breakpoints reconstituted the affected chromosomes without modifying the fusion gene.
Moreover, 2 patients (n.1 and 3) (33,3%) harbored deletions adjacent to the BCR and/or ABL breakpoints: on both der(9) and der(22) chromosomes in patient n.1 and on der(9) in patient n.3. During the disease progression of the patient n.2, FISH analysis revealed the appearance of a second clone characterized by two fusion BCR/ABL and two BCR signals, that gradually replaced the first clone with only one fusion signal. The clonal evolution was concomitant with the increase of transcript detected by RQ-PCR. By FISH analysis on metaphase, the 2 fusion signals were observed on both chromosomes 9 (Figure 2), suggesting the duplication of BCR/ABL rearrangement, as sign of clonal evolution.
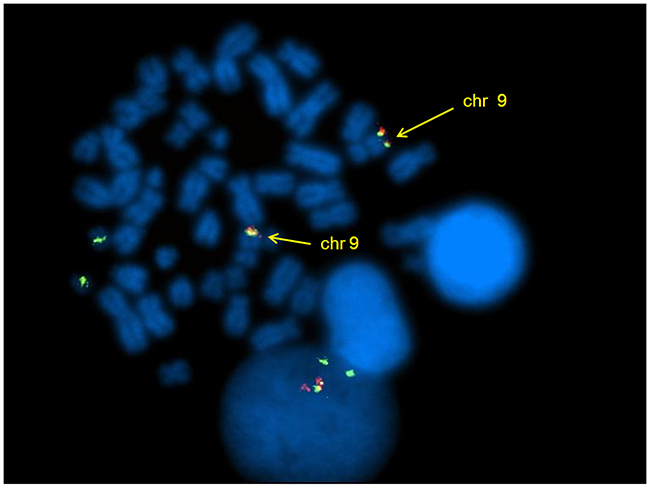
Figure 2: DCDF FISH analysis showing 1R1G2F signal pattern and the BCR/ABL fusion genes on both chromosomes 9.
During imatinib therapy, interphase-FISH analysis revealed decrease of BCR/ABL positive cells in 3 patients (n.1, 3 and 5) and after a median time of 24 months (range 18-30 months) they reached the Complete CgR (CCgR) by FISH, while two patients (n.2 and 6) retained high rate of BCR/ABL positive cells by FISH (81,5% and 79%, respectively). Patient n.2 stopped imatinib because of the appearance of intestinal malignancy and later he died. Patient n.6 failed imatinib therapy, started nilotinib treatment and, at last follow-up, retained Major Molecular Response (MMR).
The last one (n.4) relapsed after 4 years from allogeneic stem cell transplantation (SCT), achieved CCgR and complete MR after 6 months of imatinib therapy. Six years later, she lost MMR, started dasatinib and then nilotinib treatments, but she died in Partial CgR after the second allogeneic SCT.
DISCUSSION
As is known, FISH analysis is an efficient tool to characterize Ph-neg rearrangements, because it allows to detect the occurrence of BCR/ABL rearrangement and to monitor the response to therapy [13].
To our knowledge, here we present the largest monocentric series of the of Ph-neg CML patients treated with TKIs therapies and we confirm their low frequency (3%).
In Ph-neg CML cases, the BCR/ABL fusion gene is often located on der(22) and rarely on der(9), indeed its presence on der(9) has been reported only in 24 Ph-neg CML patients [4–10]. Conversely, in our series 4/6 (66,7%) Ph-neg CML cases showed the rearrangement on der(9) and one displayed 2 fusion signals on both derivative chromosomes 9 and 22, as in the classic translocation.
Regarding the involving mechanisms, previous reports considered the cryptic insertion of chromosome 22, comprising 5’ BCR within the ABL gene or viceversa, more probable than two consecutive translocations. The insertion seems more likely, since it requires only two breaks at 22q11 and one at 9q34, or viceversa, instead of four breaks required in double consecutive translocations [5]. TCDF BCR/ABL FISH analysis allowed us to clarify the mechanisms by which the translocations may take place. We have hypothesized cryptic insertion in 5 cases and two-translocations mechanism in only one case. The observation of 4 different FISH pattern in 6 Ph-neg cases suggested high genetic heterogeneity in this subgroup of CML patients.
The prognostic significance of the occurrence of cryptic Ph translocations has been discussed in previous reports, but data regarding the outcome of Ph-neg CML patients treated with TKIs are limited. In 4 patients, the CgR by FISH analysis was achieved after a median time of 24.5 months from the start of imatinib (range 7-40 months): 3 patients are still in deep MR, while the other one lost MMR after 6 years of imatinib. Another patient achieved MMR when treated subsequently with nilotinib. Two patients had to change TKI because of resistance to imatinib and of relapse after SCT.
During imatinib treatment, a not responder patient showed duplication of the BCR/ABL rearrangement, confirming that this change is an event of clonal evolution. The duplication of rearrangement appeared as 2 fusion genes on both the homologous chromosomes 9, like in 2 other cases previously reported [10]. Similarly to our case, the 2 patients did not achieve CgR. The amplification of BCR/ABL rearrangement (as the duplication of Ph chromosome) has been always considered a mechanism of resistance to the therapy and, therefore, associated with adverse prognosis, like other additional chromosomal abnormalities [14, 15].
Moreover, deletions of a sizable portion of the der(9) have been described in 10-15% of CML patients. A recent paper asserted that deletions of der(9) do not influence the response and the outcome of CML in early CP treated with imatinib [16]. De Melo et al. [17] reported deletions of ABL or BCR regions in 21% of Ph-neg CML cases, concluding that the loss of genomic material is an effect of any DNA breakage event at chromosome 9q34 and 22q11 regardless of the mechanism of chromosomal rearrangement. In our cases, deletions occur in 2 patients (n.1 and 3): this rate is superior to that found in Ph+ patients [16], although we have to consider the low number of cases. However both our patients achieved MMR.
We have observed that 5 out of 6 Ph-neg CML patients achieved CCgR and/or MMR during TKIs treatment, also as second line of therapy. Clearly, the low number of cases and the heterogeneity of the therapy limit the analysis. We have to highlight that the adverse outcome of 2 patients could be related to their Sokal risk: intermediate and high, respectively. Moreover, the only patient with intermediate Sokal risk showed duplication of BCR/ABL rearrangement during the outcome.
In conclusion, most Ph-neg CML patients benefited from TKIs therapy and achieved CCgR and MMR, showing outcome similar to that of Ph+ CML patients.
Ph-neg rearrangements could be considered variant BCR/ABL rearrangements, although they involve only the chromosomes 9 and 22, differently to “real” variant translocations. As the latter [18], Ph-neg rearrangement does not influence the response to TKIs therapy. We confirm that Ph-neg CML patients do not constitute a “warning” category in imatinib era, in according to ELN 2013 recommendations [19, 20].
Only 2 Ph-neg CML patients failed TKIs therapy; in one a secondary change occurred, confirming that BCR/ABL duplication remains a mechanism of resistance to the therapy, also in TKI era.
MATERIALS AND METHODS
Patients
The clinical features of the Ph-neg patients are reported in Table 3. They were 5 males and 1 female, with a median age of 43.5 years (range 22-68); Sokal risk was low in 4 patients, intermediate in 1 patient and high in the last one; the median WBC count was 102.5*109/l (range 32.9-283); the median PLT count was 324*109/l (range 92-560); epatosplenomegaly was observed in all patients, but one (n.1).
Table 3: Clinical characteristics of the patients
Patients |
Sex |
Agea |
Sokal risk |
Epato-splenomegaly |
Count at diagnosis |
Treatment |
Last |
Follow-up |
|||
|---|---|---|---|---|---|---|---|---|---|---|---|
WBC |
PLT |
Blasts |
Eo/ |
||||||||
1 |
M |
22 |
low |
No |
52,3 |
398 |
1 |
3 |
hydroxyurea, imatinib |
CCgR, MR4 |
156 |
2 |
M |
68 |
intermediate |
Yes |
93 |
92 |
/ |
/ |
hydroxyurea, imatinib |
diedb |
68 |
3 |
M |
44 |
low |
Yes |
32,9 |
250 |
/ |
0,6 |
α-interferon, imatinib |
CCgR, CMR, MR5 |
144 |
4 |
F |
41 |
high |
Yes |
283 |
560 |
/ |
3 |
hydroxyurea, alloSCT, imatinib, dasatinib, nilotinib |
diedc |
105 |
5 |
M |
47 |
low |
Yes |
112 |
469 |
<5 |
0.75 |
imatinib |
MMR, MR4.5 |
132 |
6 |
M |
43 |
low |
Yes |
176 |
184 |
1 |
1,5 |
imatinib, nilotinib |
MMR |
lost |
aat diagnosis; bintestinal malignancy; cin therapy with nilotinib;
Abbreviations: SCT: stem cell transplantation; CCgR= Complete Cytogenetic Response; MR= Molecular Response; CMR= Complete Molecular Response; MMR= Major Molecular Response.
Cytoreduction was started with hydroxyurea in 3 patients: later, 2 patients (n.1 and n.2) were treated with imatinib; meanwhile the other patient (n.4) underwent to allogeneic SCT, after 4 years she relapsed in accelerated phase and started imatinib therapy; then she was treated with dasatinib and afterwards with nilotinib. She died after second allogeneic SCT. Two other patients (n.5 and 6) were treated frontline with imatinb and the last one (n.3) started imatinib after α-IFN treatment.
The study is performed according to Good Clinical Practices and Declaration of Helsinki.
Cytogenetics analysis
Cytogenetic studies were performed on bone marrow samples after short term (24 and/or 48 hours) cultures. Karyotypes were examined after GAW banding techniques. In each case, at least 20 metaphases were analysed and the karyotypes were described according to the criteria of the International System for Human Cytogenetic Nomenclature [21].
FISH analysis
FISH was performed on bone marrow cells prepared according to standard cytogenetic techniques and using LSI BCR/ABL DCDF and LSI BCR/ABL + 9q34 TCDF translocation probes (Vysis Inc, Downers Grove, IL, USA) and ON BCR/ABL t(9;22), triple-color probe (Kreatech Biotechnology, Amsterdam, Netherlands). FISH was carried out according to the manufacturer’s instructions, with slight modifications. FISH analysis was performed at least on 200 nuclei and on metaphases to confirm the interphase FISH pattern and to localize the rearrangement.
Since at diagnosis FISH pattern of the most cases shows only 1 fusion signal by FISH analysis, CgR was defined by the rate of cells with 1 fusion signal. The value of false positive cut-off was calculated on 68 normal controls resulting in 10% (mean+3DS) [22].
RT-PCR and RQ-PCR
Qualitative RT-PCR for BCR/ABL transcript was performed at diagnosis, in order to determine the type of transcript.
The RQ-PCR was performed to monitor the amount of BCR/ABL transcript during the therapy, using the ABIPRISM7700 Sequence Detector (Perkin Elmer, Faster City, CA). ABL was used as housekeeping gene to correct for differences in RNA quality and/or RT efficacy. All experiments were performed in duplicate and the results were expressed as percent ratio to ABL. The BCR/ABL to ABL ratios were additionally multiplied by the conversion factor of the Bologna lab to set the results on the International Scale (IS). Samples yielding an ABL threshold cycle (Ct) greater than 30, which corresponded to fewer than 1,000 ABL transcript copies, were considered to have degraded RNA and were discarded.
The following criteria have been used to define the molecular response (MR) [19, 23]:
MR4= either detectable disease ≤0.01% BCR-ABLIS or undetectable disease in cDNA with ≥10000 ABL1 or ≥24000 GUSB transcripts (numbers of ABL or GUSB transcripts in the same volume of cDNA used to test for BCR-ABL);
MR4,5= either detectable disease ≤0.0032% BCR-ABLIS or undetectable disease in cDNA with ≥32000 ABL1 or ≥77000 GUSB transcripts;
MR5= either detectable disease ≤0.001% BCR-ABLIS or undetectable disease in cDNA with ≥100000 ABL1 or ≥240000 GUSB transcripts.
Authors’ contributions
S.L. performed the research and FISH analysis, interpreted data and wrote the manuscript. C.B., G.Marzocchi and G.A. performed cytogenetic analysis. M.T.B. and S.S. performed molecular analysis. F.C., M.T. and G.G. were involved in patient recruitment and clinical data analysis. G.Martinelli, M.B. and G.R. designed the clinical trials. M.C. critically revised the manuscript. N.T. designed the study and revised the manuscript. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
This study was supported by BolognaAIL, RFO University of Bologna and the European LeukemiaNet.
CONFLICTS OF INTEREST
S.S. received compensation as a consultant for Novartis, Bristol-Myers Squibb and Ariad; F.C. has acted as a consultant for and received honoraria from ARIAD Pharmaceuticals, Bristol-Myers Squibb, Novartis and Pfizer; M.T. has acted as consultant and received honoraria from Novartis, BMS and ARIAD; G.G. has acted as a consultant and received honoraria from Bristol-Myers Squibb and Novartis; G.Martinelli served on the speakers’ bureaus of Novartis, Bristol-Myers Squibb, and Pfizer; M.B. has received honoraria from ARIAD Pharmaceuticals, Bristol-Myers Squibb, Novartis and Pfizer, and served on the speakers’ bureaus of Bristol Myers-Squibb and Novartis; M.C. has received honoraria and has been a member of the advisory board for Celgene, Jansen-Cilagand Novartis; G.R. has acted as a consultant for and received honoraria from ARIAD Pharmaceuticals, Bristol-Myers Squibb, Novartis, Pfizer and Roche, and served on the speakers’ bureaus of Bristol Myers-Squibb and Novartis; the remaining Authors had not relevant conflicts of interest to disclose.
REFERENCES
1. Rowley JD. A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and GIEMSA staining. Nature. 1973; 243:290-3.
2. Jabbour E, Kantarjian H. Introduction: chronic myelogenous leukemia (CML). Semin Hematol. 2007; 44:S1-S3.
3. Quintàs-Cardama A, Cortes J. Molecular biology of bcr-abl1-positive chronic myeloid leukemia. Blood. 2009; 113:1619-30.
4. Hagemeijer A, Buijus A, Smit E, Janssen B, Creemers GJ, Van der Plas D, Grosveld G. Translocation of BCR to chromosme 9: a new cytogenetic variant detected by FISH in two Ph-negative, BCR-positive patients with chronic myeloid leukemia. Genes Chromosomes Cancer. 1993; 8:237-45.
5. Haigh S, Cuthbert G. Fluorescence in situ hybridization characterization of different cryptic BCR/ABL rearrangements in chronic myeloid leukemia. Cancer Genet and Cytogenet. 2004; 155:132-7.
6. Verma RS, Chandra P. Clinical significance of reverse BCR/ABL gene rearrangement in Ph-negative chronic myelogenous leukemia. Leukemia Research. 2000; 24:631-5.
7. Michalová K, Zemanová Z, Bkezinová J, Moravcová J, Oltová A, Sobotka J, Kuglík P, Kozak T, Sindelarová L, Jankovská M, Obomilová A, Sieglová Z, Polák J, et al. Location of the BCR/ABL fusion genes on both chromosomes 9q34 in Ph negative Chronic Myeloid Leukemia. Leukemia and Lymphoma. 2002; 43:1695-00.
8. Costa D, Espinet B, Queralt R, Carriò A, Solé F, Colomer D, Cervantes F, Hernandez JA, Besses C, Campo E. Chimeric BCR/ABL gene detected by fluorescence in situ hybridization in three new cases of Philadelphia chromosome-negative chronic myelocytic leukemia. Cancer Genet Cytogenet. 2003; 141:114-9.
9. Fugazza G, Garuti A, Marchelli S, Miglino M, Bruzzone R, Gatti AM, Castello S, Sessarego M. Masked Philadelphia chromosome due to atypical BCR/ABL location in the 9q34 band and duplication on the der(9) in a case of chronic myelogenous leukemia. Cancer Genet Cytogenet. 2005; 163:173-5.
10. Brahmbhatt MM, Trivedi PJ, Patel DM, Shukla SN, Patel PS. Location of the BCR/ABL fusion genes on both chromosomes 9 and 22 in Ph negative young CML patients: an Indian experience. Indian J Hematol and Blood Transfus. 2014;30:241-6.
11. Batista DAS, Hawkins A, Murphy KM, Griffin CA. BCR/ABL rearrangement in two cases of Philadelphia chromosome negative chronic myeloid leukemia: deletion on the derivative chromosome 9 may or not be present. Cancer Genet Cytogenet. 2005; 163:164-7.
12. Bennour A, Bellâaj H, Ben Youssef Y, Elloumi M, Khelif A, Saad A, Sennana H. Molecular cytogenetic characterization of Philadelphia-negative rearrangements in chronic myeloid leukemia patients. J Cancer Res and Clin Oncol. 2011; 137:1329-36.
13. Lim TH, Tien SL, Lim P, Lim AST. The incidence and patterns of BCR/ABL rearrangements in chronic myeloid leukaemia (CML) using fluorescence in situ hybridization (FISH). Ann Acad Med Singapore. 2005; 34:533-8.
14. Fabarius A, Leitner A, Hochhaus A, Müller MC, Hanfstein B, Haferlach C, Göhring G, Schlegelberger B, Jotterand M, Reiter A, Jung-Munkwitz S, Proetel U, Schwaab J, et al. Impact of additional cytogenetic aberrations at diagnosis on prognosis of CML: long-term observation of 1151 patients from the randomized CML Study IV. Blood. 2011; 118:6760-8.
15. Luatti S, Castagnetti F, Marzocchi G, Baldazzi C, Gugliotta G, Iacobucci I, Specchia G, Zanatta L, Rege-Cambrin G, Mancini M, Abruzzese E, Zaccaria A, Grimoldi MG, et al. Additonal chromosomal abnormalities in Philadelphia-positive clone: adverse prognostic influence on frontline imatinib therapy: a GIMEMA Working Party on CML Analysis. Blood. 2012; 120:761-7.
16. Castagnetti F, Testoni N, Luatti S, Marzocchi G, Mancini M, Kerim S, Giugliano E, Albano F, Cuneo A, Abruzzese E, Martino B, Palandri F, Amabile M, et al. Deletions of the derivative chromosome 9 do not influence the response and the outcome of chronic myeloid leukemia in early chronic phase treated with imatinib mesylate: GIMEMA CML Working Party analysis. J of Clin Oncol. 2010; 28:2748-54.
17. De Melo VA, Milojkovic D, Marin D, Apperley JF, Nacheva EP, Reid AG. Deletions adjacent to BCR and ABL1 breakpoints occur in a substantial minority of chronic myeloid leukemia patients with masked Philadelphia rearrangements. Cancer Genet Cytogenet. 2008; 182:111-5.
18. Marzocchi G, Castagnetti F, Luatti S, Baldazzi C, Stacchini M, Gugliotta G, Amabile M, Specchia G, Sessarego M, Giussani U, Valori L, Discepoli G, Montaldi A, et al. Variant Philadelphia translocations: molecular-cytogenetic characterization and prognostic influence on frontline imatinib therapy, a GIMEMA WP on CML analysis. Blood. 2011; 117:6793-00.
19. Baccarani M, Deininger MW, Rosti G, Hochhaus A, Soverini S, Apperley JF, Cervantes F, Clark RE, Cortes JE, Guilhot F, Hjorth-Hansen H, Hughes TP, Kantarjian HM, et al. European LeukemiaNet recomandation for the management of chronic myeloid leukemia: 2013. Blood. 2013; 122:872-84.
20. Hochhaus A, Mahon FX, le Coutre P, Coriu D, Petrov L, Ossenkoppele GJ, Cross NC, Muller MC, Rea D, Steegmann JL, Castagnetti F, Hellmann A, Rosti G et al. Molecular Response with Nilotinib in patients with Philadelphia Negative (Ph-) Chronic Myeloid Leukemia in Chronic Phase (CML-CP): ENEST1st Sub-Analysis. ASH Meeting. 2015.
21. Shaffer LG, McGowan-Jordan J, Schmid M (eds). ISCN : An International System for Human Cytogenetic Nomenclature. S Karger, Basel; 2013.
22. Testoni N, Marzocchi G, Luatti S, Amabile M, Baldazzi C, Stacchini M, Nanni M, Rege-Cambrin G, Giugliano E, Giussani U, Abruzzese E, Kerim S, Grimoldi MG, et al. Chronic Myeloid Leukemia: a prospective comparison of interphase fluorescence in situ hybridization and chromosome banding analysis for the definition of complete cytogenetic response: a study of the GIMEMA WP. Blood. 2009; 114:4439-43.
23. Cross NCP, White HE, Muller MC, Saglio G and Hochhaus A. Standardized definitions of molecular response in chronic myeloid leukemia. Leukemia. 2002; 26:2172-5.

