INTRODUCTION
Colon cancer is among the most common malignant diseases, and its morbidity increases annually [1]. The situation in China may be worse than the global situation [2, 3]. Recently, there have been numerous reports describing correlations between colon cancer prognosis and anatomical location [4] and confirming a significantly worse prognosis and higher risk of recurrence for right-sided colon cancer (RCC) than for left-sided colon cancer (LCC) [5, 6].
According to the cancer stem cell (CSC) theory, only a small number of cells in a tumor have the potential to self-renew and differentiate to maintain malignancy [7–9]. These CSCs are considered tumor-initiating cells (TICs) and have been identified in colorectal cancers and numerous other malignant tumors [10]. The presence of CSCs facilitates tumor relapse and tumor chemotherapy resistance, even when chemotherapies are applied in combination with targeted drugs. These characteristics are similar to the biological features of RCC, which demonstrates a high level of recurrence and a reduced survival rate compared with LCC. Unfortunately, few reports describing the relationship between RCC prognosis and CSCs, particularly CSC markers, are available. However, ATP-binding cassette sub-family G member 2 (ABCG2) and OCT-4 (also known as POU5F1 (POU family of transcription factors, class 5, factor 1)) are accepted CSC markers in numerous cancers and are linked with prognosis. ABCG2 is an ATP-binding cassette (ABC) efflux transporter that was recently accepted by the Food and Drug Administration (FDA) as a critical transporter involved in drug removal from the cell [11]. OCT-4 is a transcription factor that has been shown to play a significant role in tumorigenesis and embryogenesis and is associated with maintenance of stemness and cancer prognosis [12]. In this study, we investigated whether ABCG2 and OCT-4 expression are associated with the clinicopathological features of recurrent and non-recurrent cases of RCC.
RESULTS
Downregulating ABCG2 and OCT-4 inhibits CD133 expression, sphere formation, and tumorigenesis
Because ABCG2 and OCT-4 expression is significantly correlated with RCC recurrence, we suggest that ABCG2 and OCT-4 have a potential role in the development and maintenance of the stem cell–like properties of SW480 cells. Compared with control cells, sh-ABCG2 and sh-OCT-4 cells formed much smaller spheres after 7 days of culture (Figure 1A). Flow cytometry showed that sh-OCT-4 decreased the proportion of CD133+ cells (Figure 1B). To determine whether ABCG2 and OCT-4 are involved in SW480 cell tumorigenesis in vivo, we subcutaneously inoculated cells into the inguinal folds of nude mice. The tumors formed by sh-ABCG2 and sh-OCT-4 cells were visibly smaller than the vector control tumors (Figure 1C). Almost no tumor was formed by the simultaneously downregulated ABCG2 and OCT-4 cells.
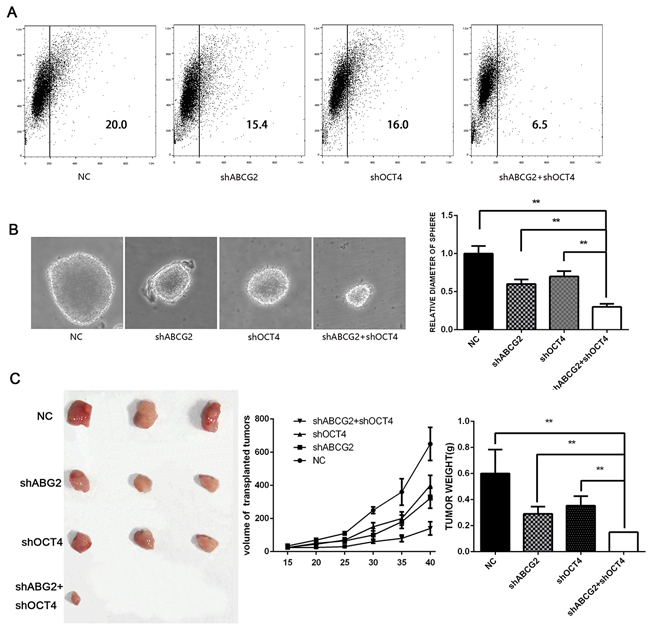
Figure 1: Downregulating ABCG2 and OCT-4 inhibits CD133 expression, sphere formation, and tumorigenic ability. A. Proportion of CD133+ cells among transfected SW480 cells. At 48h after transfection with ABCG2 and OCT-4, the SW480 cells were resuspended, and CD133 was detected on the cell membrane. B. SW480 cells transfected with respective constructs were used for sphere formation and are shown at 14 days along with statistical analysis. C. Lentivirus-based sh-ABCG2-transfected and sh-OCT4-transfected SW480 cells were used for tumor initiation. The BALB/c-nude mice sacrificed six weeks after injection. All data were compared with the NC group.
ABCG2 and OCT-4 were significantly correlated with RCC recurrence and poor outcomes
A total of 143 RCC patients were enrolled in the study. The patients’ ages ranged from 21 to 93 years (mean ± SD: 57.73 ± 12.32 years). The female-to-male ratio was 1.16:1. In total, 102 patients received chemotherapy, and 41 early-stage patients did not receive chemotherapy. Forty-seven patients experienced recurrence, and 96 patients did not experience recurrence after the operation. ABCG2 is localized to the membrane and cytoplasm, whereas OCT-4 is localized primarily in the cytoplasm. When the ABCG2 and OCT-4 expression status was compared with the clinicopathological variables, there were no correlations with age (p=0.994 vs. p=0.313), gender (p=0.104 vs. p=0.083), tumor size (p=0.073 vs. p=0.491), or tumor shape (p=0.485 vs. p=0.201). However, ABCG2 expression was significantly correlated with the TNM stage (p=0.000), the extent of differentiation (p=0.008), and lymphovascular invasion (p=0.002) of RCC. We did not identify statistical correlations between OCT-4 expression and the TNM stage (p=0.143), the extent of differentiation (p=0.055), or lymphovascular invasion (p=0.063). Our survey revealed significant differences between ABCG2 expression and RCC recurrence compared with non-recurrence (52.5% vs. 47.5%, p=0.002). Interestingly, OCT-4 expression was significantly correlated with RCC recurrence (42% vs. 58%, p=0.002) (Table 1) and was an independent indicator of RCC recurrence.
Table 1: Correlations between ABCG2 and Oct-4 expression and clinicopathologic characteristics in right-sided colon cancer
No. of patients (%) |
ABCG2 (%) |
P-value |
Oct-4 (%) |
P-value |
|
Age(years) |
21-93 |
||||
Mean±SD |
57.73±12.32 |
0.994 |
0.313 |
||
<60 |
74(51.7) |
29(50.8) |
23(46) |
||
≥60 |
69(48.3) |
30(49.2) |
27(54) |
||
Gender |
0.104 |
0.083 |
|||
Male |
66(53.8) |
32(50.2) |
28(56) |
||
Female |
77(43.2) |
27(45.8) |
22(44) |
||
Tumor size(mm) |
0.073 |
0.491 |
|||
≤50 |
10(51) |
23(43.4) |
28(56) |
||
51~99 |
60(42) |
23(43.4) |
20(40) |
||
≥100 |
73(7) |
7(13.2) |
2(4) |
||
pTNM stage |
0.000 |
0.143 |
|||
I |
5(3.5) |
1(1.7) |
1(2) |
||
II |
74(51.7) |
20(33.9) |
21(42) |
||
III |
54(37.8) |
34(57.6) |
22(44) |
||
IV |
10(7) |
4(6.8) |
6(12) |
||
Tumor differentiation |
0.008 |
0.055 |
|||
Well |
15(10.5) |
4(6.8) |
2(4) |
||
Moderate |
62(43.4) |
13(22) |
14(28) |
||
Poor |
66(46.2) |
42(71.2) |
34(68) |
||
Lymphovascular invasion |
0.002 |
0.063 |
|||
Negative |
75(52.4) |
22(37.3) |
22(44) |
||
Positive |
68(47.6) |
37(62.7) |
28(56) |
||
Tumor shape |
0.485 |
0.201 |
|||
Massive |
28(19.6) |
9(15.3) |
9(18) |
||
Ulcerative |
81(56.6) |
34(57.6) |
33(66) |
||
Infiltrating |
34(23.8) |
16(27.1) |
8(16) |
||
Recurrence |
0.002 |
0.000 |
|||
No |
96(67.1) |
31(52.5) |
21(42) |
||
Yes |
47(32.9) |
28(47.5) |
29(58) |
The rates of ABCG2 expression were 32.3% (31/96) and 59.6% (28/47) in the non-recurrent and recurrent cases, respectively (p=0.002). The rates of OCT-4 expression were 21.9% (21/96) and 61.7% (29/47) in the non-recurrent and recurrent cases, respectively (p=0.000).
The correlations between ABCG2 and OCT-4 expression and RCC prognosis were investigated by Kaplan-Meier analysis. The median survival periods of the ABCG2- and ABCG2+ groups were 77.65 months and 51.32 months, respectively, whereas the median survival periods of the OCT4- and OCT4+ groups were 68.11 months and 48.92 months. The five-year overall survival rates of the ABCG2-and ABCG2+ groups were 72.0% vs. 31.2%, respectively (p=0.000), whereas the five-year overall survival rates of the OCT-4- and OCT-4+ groups were 56.9% vs. 37.5% (p=0.000). In the recurrent groups, the median survival periods of the ABCG2- and ABCG2+ groups were 76.93 months and 45.41 months, respectively, whereas the median survival periods of the OCT-4- and OCT-4+ groups were 56.67 months and 47.00 months. Furthermore, among the recurrent patients, the five-year overall survival rates of the ABCG2- and ABCG2+ groups were 72% vs. 31.2%, respectively (p=0.000), whereas the five-year overall survival rates of the OCT-4- and OCT-4+ groups were 56.9% vs. 37.5% (p=0.010) (Table 1, 2 and Figure 2).
Table 2: Median survival periods and five-year overall survival in recurrent or nonrecurrent RCC cases with or without chemotherapy
Total cases |
Nonrecurrent cases |
Recurrent cases |
Without chemotherapy cases |
Chemotherapy cases |
|||||||||||
No. of patients (%) |
Median survival period (months) |
Five year OS (%) |
No. of patients (%) |
Median survival periods (months) |
Five year OS (%) |
No. of patients (%) |
Median survival periods (months) |
Five year OS (%) |
No. of patients (%) |
Median survival periods (months) |
Five year OS (%) |
No. of patients (%) |
Median survival periods (months) |
Five year OS (%) |
|
ABCG2- |
58.7 (84/143) |
77.65 |
72.0 |
67.7 (65/96) |
65.00 |
71.3 |
40.4 (19/47) |
76.93 |
69.4 |
75.6 (31/41) |
85.00 |
96.4 |
52 (53/102) |
75.1 |
61.0 |
ABCG2+ |
41.3 (59/143) |
51.32 |
31.2* |
32.3 (31/96) |
63.70 |
52.1 |
59.6 (28/47) |
45.41 |
4.9* |
24.4 (10/41) |
70.73 |
77.1 |
48 (49/102) |
48.68 |
22.6* |
OCT-4- |
65.0 (93/143) |
68.11 |
56.9 |
38.1 (75/96) |
70.92 |
63.2 |
38.3 (18/47) |
56.67 |
38.9 |
75.6 (31/41) |
74.16 |
90.1 |
60.8 (62/102) |
59.43 |
45.7 |
OCT-4+ |
35.0 (50/143) |
48.92 |
37.5* |
21.9 (21/96) |
66.17 |
61.9 |
61.7 (29/47) |
47.58 |
11.4* |
24.4 (10/41) |
65.00 |
85.7 |
39.2 (40/102) |
47.19 |
25.1* |
*p<0.05
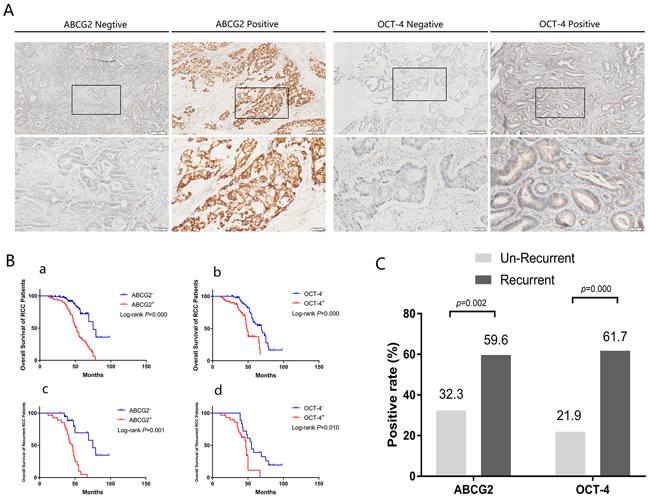
Figure 2: ABCG2 and OCT-4 were significantly correlated with RCC recurrence and predict a poor outcome. A. Representative IHC staining of positive and negative expression of ABCG2 and OCT-4 is presented at 400× magnification and 100× magnification. Images in lower panels show higher magnifications of the areas boxed in upper panels. B. Kaplan-Meier analysis of overall survival in RCC with positive or negative expression of the stem cell marker ABCG2 and OCT-4. Both ACBG2 and OCT-4 were detected have poor prognosis in total groups (143 patients) and recurrent groups (47 patients). a. Overall survival curves of total groups (143 patients) with ABCG2 expression situation. b. Overall survival curves of total groups (143 patients) with OCT-4 expression situation. c. Overall survival curves of recurrent groups (47 patients) with ABCG2 expression situation. d. Overall survival curves of recurrent groups (47 patients) with OCT-4 expression situation. C. The positive expression rate of ABCG2 and OCT-4 in un-recurrent (96 patients) and recurrent groups (47 patients) of RCC.
DISCUSSION
This research suggests that ABCG2 and OCT-4 play important roles in the malignant biological behavior of colon cancer, while no correlations of ABCG2 and OCT-4 expression were found with the age, gender, tumor size, or tumor shape of RCC patients. ABCG2 expression significantly correlated with TNM stage, tumor differentiation, and lymphovascular invasion. Additionally, the expression of both ABCG2 and OCT-4 was significantly correlated with RCC recurrence and poor outcomes.
Numerous recent studies have reported poor prognosis and a tendency toward recurrence and metastasis in RCC compared with LCC. The clinical characteristics of RCC cells are similar to the biological features of CSCs. However, few studies have demonstrated a relationship between RCC prognosis and CSCs, particularly a correlation with the expression of CSC markers.
According to the CSC theory, only a small population of cancer cells, called TICs, possesses stem cell potency and is responsible for cancer growth, metastasis and recurrence. To date, CSCs have been isolated using flow cytometry from the tumors of numerous types of cancer, such as breast cancer [14, 15], lung cancer [16–18], liver cancer [19, 20], pancreatic cancer [21], prostate cancer [22], and colorectal cancer [23, 24]. CSCs typically express certain CD molecules identified as CSC markers; however, conflicting results have been reported regarding colorectal CSC markers. For example, according to several studies, CD133+ cells possess stem cell potency, and CD133 is a potential CSC marker in colon cancer [25–27]. However, based on other studies, CD133- cells may possess an enhanced capacity to self-renew and differentiate compared with CD133+ colon cancer cells [28, 29]. Thus, here, we focused on the widely accepted CSC markers ABCG2 and OCT-4.
ABCG2, also known as breast cancer resistance protein (BCRP), is an ABC family efflux protein that confers resistance to many drugs and radioactivity. ABCG2 has been described as a stem cell marker for side population cells (SP) in several tumors [30–33]. Similar to stem cells, SP cells possess the stem-like characteristics of self-renewal, differentiation, and tumorigenicity [34, 35]. In our previous study, SP cells were isolated from gallbladder carcinoma cell lines [36] and colon cancer cell lines with high ABCG2 expression, and this small population of cells was able to self-renew and differentiate into both SP cells and non-SP cells. These cells also exhibited increased tumorigenicity and drug resistance.
OCT-4, and octomer-binding family protein, is expressed in early embryos and is a key regulator of stem cell pluripotency [37]. Through its transcription factor activity, OCT-4 possesses the ability to maintain cells in an undifferentiated and pluripotent state by maintaining embryonic stem cell self-renewal [38]. Ponti et al. identified breast carcinoma CD44+/CD24-/low cells exhibiting stem/progenitor cell properties that highly expressed OCT-4. Moreover, as few as 103 CD44+/CD24-/low cells were sufficient to drive tumor formation [39]. As a gatekeeper of embryonic stem cell pluripotency, OCT-4 is also associated with the prognosis of several carcinomas. According to Kosaka et al., high OCT4 expression is an independent prognostic indicator of prostate-specific antigen recurrence [12]. Moreover, OCT-4 overexpression in the absence of SOX2 expression is strongly associated with poor prognosis in cervical cancer [40]. These results are similar to our results.
OCT-4 is rarely expressed in normal tissues. A report showed that ABCG2 may be expressed in the small and large intestine is in contrast to the findings of our study [41]. It is possible that the multidrug resistance protein ABCG2 is only expressed after the activation of quiescent stem cells in normal tissues.
In the present study, ACBG2 and OCT-4 were detected in tissue samples obtained after RCC resections, and the expression of both proteins was significantly correlated with RCC recurrence. These results may be attributable to the fact that ABCG2 belongs to a family of cytomembrane efflux proteins and is involved in the resistance to numerous drugs and radioactivity. Chen et al. also reported an association between ABCG2 overexpression and poor prognosis for hepatocellular carcinoma in elderly patients [42]. However, a different survey found no significant relationship between ABCG2 expression and the clinical outcomes of pediatric sarcomas [43]. ABCG2 may not be enriched in sarcomas, which would explain this result. Another study reported high expression of ABCG2 and OCT-4 in enriched CD90(+)CD133(+) liver CSCs and a close association with chemotherapy drug resistance [44]. However, our experiments identified no significant relationship between OCT-4 expression and the clinicopathological variables of RCC. OCT-4 may act as an ON and OFF switch in CSCs. Furthermore, although OCT-4 expression is typically stable, OCT-4 regulation is complex and primarily depends on the microenvironment [45].
In summary, our study describes the relationship between ABCG2 and OCT-4 expression and the clinicopathological characteristics of RCC patients. ABCG2 and OCT-4 expression was significantly correlated with RCC recurrence, which has a poor prognosis. Therefore, ABCG2 and OCT-4 may be indicators of RCC recurrence. Our results may help to inform the mode of treatment for RCC patients and screen for poor outcomes to allow for timely intervention.
MATERIALS AND METHODS
Cell culture and transfections
The SW480 cell line was obtained from the Center laboratory of the Tianjin Medical University Cancer Institute and Hospital. SW480 cells were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS), L-glutamine, and 1% penicillin/streptomycin at 37°C in a 5% CO2 incubator. The ABCG2 and OCT-4 knockdown lentiviruses (sh-ABCG2 and sh-OCT-4) were constructed by and purchased from Genechem (Shanghai, China). All transfections were carried out according to the manufacturer’s instructions.
Xenograft experiments
A total of 1×105 transfected cells were subcutaneously injected into the right armpit of BALB/c nude mice. The weight of the mice and the diameter of tumors were measured every week.
Spheroid colony-formation assay
The sorted tumor cells were suspended in serum-free DMEM/F12 (1:1 volume, Gibco) supplemented with 20 ng/ml human recombinant EGF (Invitrogen), 20 ng/ml bFGF (Invitrogen) and 5 μg/ml insulin (Sigma), and then cultured in 96-well culture plates. Fresh serum-free DMEM/F12 (described above) was added to each well (0.025 ml/well) every day. After 2–3 weeks, each well was examined using a light microscope, and the total number of spheroid colonies in the 96-well plate was counted. Images of the spheroid colonies were recorded using an inverted microscope (Nikon).
Flow cytometry and FACS analysis
The cells were resuspended in DMEM with 2% FBS at a concentration of 106/100 μl and incubated for 30 min at room temperature with a 100-fold dilution of the anti-CD133/1-phycoerythrin (eBioscience) antibody. After incubation, the samples were washed twice with PBS/2% FBS and resuspended. Then, 4,6-diamidino-2-phenylindole (1 μg/ml, Sigma) was added to the samples to eliminate the dead cells. Flow cytometry analysis was performed using FACSAria (BD Immunocytometry Systems). The cells were routinely sorted twice and then reanalyzed for purity, which was typically >90%.
Patients and specimens
The pathology specimens were obtained at Tianjin Medical University Cancer Institute and Hospital between 2004 and 2012 from patients with histopathologically confirmed RCC who underwent radical right hemicolectomy. The clinical characteristics and outcomes were collected until death or loss of follow-up by telephone and mail. In total, 144 cases were followed (1 patient died immediately postoperatively and was excluded from this study), including 47 cases that were recurrent after surgical resection and 96 cases that were not recurrent after the initial surgery. In addition, 102 patients underwent chemotherapy after the operation. The patients’ ages ranged from 21 to 93 years (mean ± standard deviation (SD): 63.1±11.7 years). The survival time was defined as the time between the original operation and the date of death. The Ethics Committee of Tianjin Medical University Cancer Institute and Hospital approved this survey.
Immunohistochemistry staining
Tumor samples (5-μm sections) were formalin-fixed and paraffin-embedded. The samples were immersed in a 0.3% hydrogen peroxide solution in methanol for 30 min to block endogenous peroxidase. Then, the samples were blocked in a blocking solution containing 10% normal rabbit serum at room temperature for 90 min, followed by phosphate-buffered saline for 15 min. Next, the sections were incubated overnight at 4°C with the following primary antibody dilutions: 1:100 anti-ABCG2 (SANTA CRUZ, Dallas, Texas, USA) and 1:60 anti-OCT-4 (GeneTex, San Antonio, Texas, USA). Finally, the sections were incubated with 3,3’-diaminobenzidine (DAB) substrate solution (eBioscience) for 3 min for color development, counterstained with 0.1% hematoxylin, dehydrated in ethanol and cleared in xylene. The negative controls were obtained by performing all of the above steps except the primary antibody incubation.
Immunohistochemistry evaluation
A semi-quantitative evaluation system was employed to obtain the staining scores. The staining intensity was classified into four grades: 0, no staining; 1, weak staining; 2, moderate staining; and 3, strong staining. The percentage of stained cells was graded at four levels: 0, no tumor cells; 1, <10% tumor cells; 2, 10-50% tumor cells; and 3, >50% tumor cells. Scores <4 were defined as negative staining, and scores ≥4 were defined as positive staining [13]. Two pathologists who were blinded to the patient prognosis and other clinicopathological variables performed the repeated measurements. The relationships among ABCG2 and OCT-4 expression, clinicopathological features, tumor recurrence and overall survival were analyzed.
Statistical analysis
Student’s t-test and the Chi-square test were used to calculate the significance of the clinicopathological data. Kaplan-Meier analysis was performed to evaluate overall survival. Log-rank tests were used to compare the marker-negative and marker-positive cases. The Cox hazard regression model was employed for the multivariate analysis. All data analyses and graph creation were performed with GraphPad Prism software (Version 7.0), and p<0.05 was considered statistically significant.
Abbreviations
Right-sided colon cancer (RCC), left-sided colon cancer (LCC), cancer stem cells (CSCs), tumor-initiating cells (TICs), ATP-binding cassette sub-family G member 2 (ABCG2), POU family of transcription factors, class 5, factor 1 (POU5F1), ATP-binding cassette (ABC), Food and Drug Administration (FDA), fetal bovine serum (FBS)
ACKNOWLEDGMENTS
We are grateful to Dr. Fei Zhang for experimental assistance.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
GRANT SUPPORT
This research was supported by grants from the National Nature Science Foundation of China (No. 81101870), the National Key Clinical Specialist Construction Programs of China (No.2013-544), the Key Programs of National Health and Family Planning Commission of Tianjin (No.16KG127).
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65: 87-108.
2. Shin A, Jung KW, Won YJ. Colorectal cancer mortality in Hong Kong of China, Japan, South Korea, and Singapore. World J Gastroenterol. 2013; 19: 979-983.
3. Ng SC, Wong SH. Colorectal cancer screening in Asia. Br Med Bull. 2013; 105: 29-42.
4. Benedix F, Kube R, Meyer F, Schmidt U, Gastinger I, Lippert H. Comparison of 17,641 patients with right- and left-sided colon cancer: differences in epidemiology, perioperative course, histology, and survival. Dis Colon Rectum. 2010; 53: 57-64.
5. Yahagi M, Okabayashi K, Hasegawa H, Tsuruta M, Kitagawa Y. The Worse Prognosis of Right-Sided Compared with Left-Sided Colon Cancers: a Systematic Review and Meta-analysis. J Gastrointest Surg. 2016; 20: 648-655.
6. Papagiorgis PC, Zizi AE, Tseleni S, Oikonomakis IN, Nikiteas NI. The pattern of epidermal growth factor receptor variation with disease progression and aggressiveness in colorectal cancer depends on tumor location. Oncol Lett. 2012; 3: 1129-1135.
7. Noguchi K, Konno M, Nishida N, Kawamoto K, Koseki J, Eguchi H, Doki Y, Mori M, Satoh T, Ishii H. [Therapeutic Implication Targeting for Cancer Stem Cells]. Gan To Kagaku Ryoho. 2015; 42: 1036-1039.
8. Mery B, Rancoule C, Guy JB, Espenel S, Wozny AS, Simonet S, Vallard A, Alphonse G, Ardail D, Rodriguez-Lafrasse C, Magne N. Cancer stem cells: Radiotherapeutic features and therapeutic targets. Bull Cancer. 2016; 103: 48-54.
9. Yang M, Liu P, Huang P. Cancer stem cells, metabolism, and therapeutic significance. Tumour Biol. 2016; 37: 5735-5742.
10. Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001; 414: 105-111.
11. Mao Q, Unadkat JD. Role of the breast cancer resistance protein (BCRP/ABCG2) in drug transport--an update. AAPS J. 2015; 17: 65-82.
12. Kosaka T, Mikami S, Yoshimine S, Miyazaki Y, Daimon T, Kikuchi E, Miyajima A, Oya M. The prognostic significance of OCT4 expression in patients with prostate cancer. Hum Pathol. 2016; 51: 1-8.
13. Aas T, Borresen AL, Geisler S, Smith-Sorensen B, Johnsen H, Varhaug JE, Akslen LA, Lonning PE. Specific P53 mutations are associated with de novo resistance to doxorubicin in breast cancer patients. Nat Med. 1996; 2: 811-814.
14. Bozorgi A, Khazaei M, Khazaei MR. New Findings on Breast Cancer Stem Cells: A Review. J Breast Cancer. 2015; 18: 303-312.
15. Lamb R, Lisanti MP, Clarke RB, Landberg G. Co-ordination of cell cycle, migration and stem cell-like activity in breast cancer. Oncotarget. 2014; 5: 7833-7842. doi: 10.18632/oncotarget.2312.
16. Lundin A, Driscoll B. Lung cancer stem cells: progress and prospects. Cancer Lett. 2013; 338: 89-93.
17. Suresh R, Ali S, Ahmad A, Philip PA, Sarkar FH. The Role of Cancer Stem Cells in Recurrent and Drug-Resistant Lung Cancer. Adv Exp Med Biol. 2016; 890: 57-74.
18. Yan X, Luo H, Zhou X, Zhu B, Wang Y, Bian X. Identification of CD90 as a marker for lung cancer stem cells in A549 and H446 cell lines. Oncol Rep. 2013; 30: 2733-2740.
19. Mikhail S, He AR. Liver cancer stem cells. Int J Hepatol. 2011; 2011: 486954.
20. Sun JH, Luo Q, Liu LL, Song GB. Liver cancer stem cell markers: Progression and therapeutic implications. World J Gastroenterol. 2016; 22: 3547-3557.
21. Rasheed Z, Wang Q, Matsui W. Isolation of stem cells from human pancreatic cancer xenografts. J Vis Exp. 2010; 43: 2169.
22. Vidal SJ, Quinn SA, de la Iglesia-Vicente J, Bonal DM, Rodriguez-Bravo V, Firpo-Betancourt A, Cordon-Cardo C, Domingo-Domenech J. Isolation of cancer stem cells from human prostate cancer samples. J Vis Exp. 2014; 14:85.
23. Jiao ZY, Cao HT, Li YM. Possible Role of Cancer Stem Cells in Colorectal Cancer Metastasizing to the Liver. Curr Stem Cell Res Ther. 2016; 11: 440-443.
24. Mikhail S, Zeidan A. Stem cells in gastrointestinal cancers: The road less travelled. World J Stem Cells. 2014; 6: 606-613.
25. Jing F, Kim HJ, Kim CH, Kim YJ, Lee JH, Kim HR. Colon cancer stem cell markers CD44 and CD133 in patients with colorectal cancer and synchronous hepatic metastases. Int J Oncol. 2015; 46: 1582-1588.
26. Lee MR, Ji SY, Mia-Jan K, Cho MY. Chemoresistance of CD133(+) colon cancer may be related with increased survivin expression. Biochem Biophys Res Commun. 2015; 463: 229-234.
27. Zhou JY, Chen M, Ma L, Wang X, Chen YG, Liu SL. Role of CD44(high)/CD133(high) HCT-116 cells in the tumorigenesis of colon cancer. Oncotarget. 2016; 7: 7657-7666. doi: 10.18632/oncotarget.7084.
28. Vincent Z, Urakami K, Maruyama K, Yamaguchi K, Kusuhara M. CD133-positive cancer stem cells from Colo205 human colon adenocarcinoma cell line show resistance to chemotherapy and display a specific metabolomic profile. Genes Cancer. 2014; 5: 250-260. doi: 10.18632/genesandcancer.23.
29. Lim SH, Jang J, Park JO, Kim KM, Kim ST, Park YS, Lee J, Kim HC. CD133-positive tumor cell content is a predictor of early recurrence in colorectal cancer. J Gastrointest Oncol. 2014; 5: 447-456.
30. Niess H, Camaj P, Renner A, Ischenko I, Zhao Y, Krebs S, Mysliwietz J, Jackel C, Nelson PJ, Blum H, Jauch KW, Ellwart JW, Bruns CJ. Side population cells of pancreatic cancer show characteristics of cancer stem cells responsible for resistance and metastasis. Target Oncol. 2015; 10: 215-227.
31. Wang M, Wang Y, Zhong J. Side population cells and drug resistance in breast cancer. Mol Med Rep. 2015; 11: 4297-4302.
32. Feng L, Wu JB, Yi FM. Isolation and phenotypic characterization of cancer stem-like side population cells in colon cancer. Mol Med Rep. 2015; 12: 3531-3536.
33. Lee MR, Ju HJ, Kim BS, Ko YH, Kim WS, Kim SJ. Isolation of side population cells in B-cell non-Hodgkin’s lymphomas. Acta Haematol. 2013; 129: 10-17.
34. Szepesi A, Matula Z, Szigeti A, Varady G, Szabo G, Uher F, Sarkadi B, Nemet K. ABCG2 is a selectable marker for enhanced multilineage differentiation potential in periodontal ligament stem cells. Stem Cells Dev. 2015; 24: 244-252.
35. Boesch M, Zeimet AG, Reimer D, Schmidt S, Gastl G, Parson W, Spoeck F, Hatina J, Wolf D, Sopper S. The side population of ovarian cancer cells defines a heterogeneous compartment exhibiting stem cell characteristics. Oncotarget. 2014; 5: 7027-7039. doi: 10.18632/oncotarget.2053.
36. Hu J, Li J, Wang M, Du Z, Chen M, Qin R. Isolation of side population cells from gallbladder carcinoma of human being and the expression of ABCG2 gene. Chinese-German Journal of Clinical Oncology. 2007; 6: 469-473.
37. Nichols J, Zevnik B, Anastassiadis K, Niwa H, Klewe-Nebenius D, Chambers I, Scholer H, Smith A. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 1998; 95: 379-391.
38. Niwa H, Miyazaki J, Smith AG. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat Genet. 2000; 24: 372-376.
39. Ponti D, Costa A, Zaffaroni N, Pratesi G, Petrangolini G, Coradini D, Pilotti S, Pierotti MA, Daidone MG. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 2005; 65: 5506-5511.
40. Kim BW, Cho H, Choi CH, Ylaya K, Chung JY, Kim JH, Hewitt SM. Clinical significance of OCT4 and SOX2 protein expression in cervical cancer. BMC Cancer. 2015; 15: 1015.
41. Dlugosz A, Janecka A. ABC transporters in the development of multidrug resistance in cancer therapy. Curr Pharm Des. 2016; 22: 4705-4716.
42. Chen YL, Chen PM, Lin PY, Hsiau YT, Chu PY. ABCG2 Overexpression Confers Poor Outcomes in Hepatocellular Carcinoma of Elderly Patients. Anticancer Res. 2016; 36: 2983-2988.
43. Zambo I, Hermanova M, Zapletalova D, Skoda J, Mudry P, Kyr M, Zitterbart K, Sterba J, Veselska R. Expression of nestin, CD133 and ABCG2 in relation to the clinical outcome in pediatric sarcomas. Cancer Biomark. 2016; 17: 107-116.
44. Jia Q, Zhang X, Deng T, Gao J. Positive correlation of Oct4 and ABCG2 to chemotherapeutic resistance in CD90(+)CD133(+) liver cancer stem cells. Cell Reprogram. 2013; 15: 143-150.
45. Zeineddine D, Hammoud AA, Mortada M, Boeuf H. The Oct4 protein: more than a magic stemness marker. Am J Stem Cells. 2014; 3: 74-82.

