INTRODUCTION
Spontaneous hepatitis C virus (HCV) seroclearance occurs in 20-30% of patients after acute infection [1-3]. Subjects with spontaneous HCV seroclearance have been at a much lower risk of HCC development compared to those with persistent HCV viremia [4]. Chronic HCV infection has become one of the leading causes of hepatocellular carcinoma (HCC). It is estimated that a global population of 185 million people are infected with HCV [5], leading to the increasing incidence of HCV-related HCC worldwide [6]. During the past decades, the development of successful antiviral therapy (e.g., achievement of sustained virological responses, SVR) with interferon-based therapy could attain a sustainable HCV seroclearance for years [7], significantly reducing the risk of cirrhosis and HCC development [8-10]. Nevertheless, it is unclear if treatment-induced HCV seroclearance achieved by SVR could decrease the incidence of HCC to a similar rate observed in subjects with self-limited HCV infections. We herein addressed the issue by conducting an age and sex-matched case-control study using a clinical cohort of chronic hepatitis C (CHC) patients with successful antiviral therapy and a community cohort of patients with spontaneous HCV seroclearance.
RESULTS
Patient characteristics
A total of 313 subjects in the SC group (participants who had anti-HCV seropositivity and undetectable HCV RNA at baseline (n = 291) and those who had anti-HCV seropositivity and detectable HCV RNA levels at baseline, but HCV RNA became undetectable after more than one-year follow-up (n = 22)) and 564 age- and sex-matched patients in the SVR group were enrolled for analysis with a follow-up period of 4,224and 2,739 person-years, respectively. The baseline characteristics are shown in Table 1. SVR patients had higher serum levels of aspartate aminotransferase (AST) and alanine aminotransferase (ALT), a lower proportion of favorable IL-28B genotype carriage, and a higher proportion of diabetes, smoking and alcohol consumption. 11.5% of the SVR patients and 6.7% of the SC patients had elevated transaminases. Compared to patients with normal liver function, those with elevated transaminases had a higher proportion of diabetes (18.4% vs. 8.5%, P=0.03).
Table 1: Baseline characteristics in patients with spontaneous or treatment-induced HCV clearance
Treatment-induced |
Spontaneous |
P value |
|
|---|---|---|---|
Age, n (%) |
|||
30-39 |
77 (13.7) |
54 (17.3) |
0.30 |
40-49 |
127 (22.5) |
79 (25.2) |
|
50-59 |
238 (42.2) |
119 (38.0) |
|
60-69 |
122 (21.6) |
61 (19.5) |
|
Sex, n (%) |
|||
Female |
360 (63.8) |
211 (67.4) |
0.29 |
Male |
204 (36.2) |
102 (32.6) |
|
AST (U/L), n (%) |
|||
<40 |
495 (87.8) |
285 (91.1) |
0.02 |
40-79 |
57 (10.1) |
15 (4.8) |
|
>=80 |
8 (1.42) |
6 (1.92) |
|
Unknown |
4 (0.7) |
7 (2.2) |
|
ALT (U/L), n (%) |
|||
<45 |
495 (87.8) |
285 (91.1) |
0.04 |
45-89 |
52 (9.2) |
14 (4.5) |
|
>90 |
14 (2.5) |
7 (2.2) |
|
Unknown |
3 (0.5) |
7 (2.2) |
|
Fibrosis, n (%) |
|||
F0-1 |
226 (40.1) |
NA |
|
F2-4 |
187 (33.2) |
||
Unknown |
151 (26.8) |
||
IL-28B rs8099917, n (%) |
|||
GG |
3 (0.5) |
0 (0.0) |
0.0002 |
GT |
62 (11.0) |
15 (4.8) |
|
TT |
397 (70.4) |
273 (87.2) |
|
Unknown |
102 (18.1) |
25 (8.0) |
|
Diabetes, n (%) |
|||
Yes |
71 (12.6) |
12 (0.6) |
<0.0001 |
No |
490 (86.9) |
299 (95.5) |
|
Unknown |
3 (0.5) |
2 (0.6) |
|
Cigarette smoking, n (%) |
|||
Yes |
134 (23.8) |
16 (5.1) |
<0.0001 |
No |
427 (75.7) |
251 (80.2) |
|
Unknown |
3 (0.5) |
46 (14.7) |
|
Alcohol drinking, n (%) |
|||
Yes |
93 (16.5) |
3 (1.0) |
<0.0001 |
No |
467 (82.8) |
295 (94.3) |
|
Unknown |
4 (0.7) |
15 (4.8) |
Note: SVR: sustained virological response; AST: aspartate aminotransferase; ALT: alanine aminotransferase; IL-28: interleukin-28B genotype.
Risk of HCC development among the entire cohort
Nineteen (2.2%) of the 877 patients developed HCC over 6,963 person-years of follow-up, including fourteen (2.5%) SVR patients and 5 (1.6%) SC patients with an incidence of 0.51% and 0.12% per person-year, respectively (P=0.004) (Figure 1 and Table 2). In the univariate analysis, patients who developed HCC were older, had a higher proportion of diabetes and were more likely to be in the SVR group (versus SC) (Table 2). Cox regression analysis of the factors predictive of HCC included treatment induced SVR (versus SC: HR/CI: 5.83/1.27-26.88, P= 0.024), diabetes (HR/CI:3.41/1.21-9.58, P=0.02), and age (HR/CI: 1.07/1.01-1.14, P=0.02) (Table 2, model 1).
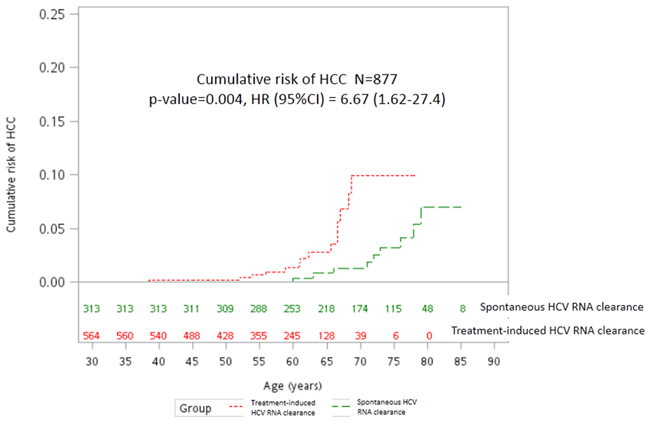
Figure 1: Cumulative risk of HCC between subjects with spontaneous and treatment-induced HCV RNA clearance. HCC: hepatocellular carcinoma; HCV: hepatitis C virus; HR: hazard ratio; CI: confidence intervals.
Table 2: Risk factors for HCC development
All patients |
HCC cases |
Crude HR (95% CI) |
Adjusted HR (95% CI) |
Adjusted HR (95% CI) |
|
|---|---|---|---|---|---|
Age, n (%) |
|||||
<55 |
494 |
6 (1.2) |
1.00 (reference) |
1.07 (1.01-1.14) |
1.08 (1.02-1.15) |
≥55 |
383 |
13 (3.4) |
3.06 (1.16-8.05) |
||
Sex, n (%) |
|||||
Female |
571 |
11 (1.9) |
1.00 (reference) |
1.00 |
1.00 |
Male |
306 |
8 (2.6) |
1.46 (0.59-3.62) |
1.19 (0.39-3.65) |
1.14 (0.37-3.46) |
Diabetics, n (%) |
|||||
No |
789 |
13 (1.7) |
1.00 (reference) |
1.00 |
1.00 |
Yes |
83 |
6 (7.2) |
6.10 (2.28-16.33) |
3.41 (1.21-9.58) |
3.23 (1.14-9.19) |
Unknown |
5 |
0 (0) |
-- |
-- |
-- |
IL-28 rs8099917, n (%) |
|||||
TT |
670 |
14 (2.1) |
1.00 (reference) |
||
GG/GT |
80 |
3 (3.8) |
2.26 (0.65-7.91) |
||
Unknown |
127 |
2 (1.6) |
0.91 (0.21-4.04) |
||
Cigarette smoking, n (%) |
|||||
No |
678 |
14 (2.1) |
1.00 (reference) |
1.00 |
1.00 |
Yes |
150 |
4 (2.7) |
1.83 (0.59-5.64) |
1.20 (0.32-4.51) |
1.39 (0.38-5.17) |
Unknown |
49 |
1 (2.0) |
|||
Alcohol drinking, n (%) |
|||||
No |
762 |
16 (2.1) |
1.00 (reference) |
||
Yes |
96 |
3 (3.1) |
2.23 (0.63-7.87) |
||
Unknown |
19 |
0 (0) |
-- |
||
Type of HCV RNA clearance, n (%) |
|||||
Spontaneous |
313 |
5 (1.6) |
1.00 (reference) |
1.00 |
1.00 |
Treatment-induced |
564 |
14 (2.5) |
6.67 (1.62-27.4) |
5.83 (1.27-26.9) |
|
F0-1 (treatment-induced) |
226 |
2 (0.9) |
2.66 (0.38-18.6) |
1.98 (0.26-14.8) |
|
F2-4 (treatment-induced) |
187 |
11 (5.9) |
11.79 (2.77-50.2) |
10.06 (2.20-46.0) |
|
Unknown (treatment-induced) |
151 |
1 (0.7) |
Note: HCC: hepatocellular carcinoma; IL-28B: interleukin 28B genotype; HR: hazard ratio; CI: confidence intervals; F0-1: Metavir fibrosis score 0 or 1; F2-4: Metavir fibrosis score 2, 3 or 4.
Impact of liver fibrosis on HCC development in SVR patients
Four hundred and thirteen (73.2%) of the 564 SVR patients had liver histology available within six months of initiating the antiviral therapy. Of them, eleven (5.9%) of the 187 patients with F2-4 and two (0.9%) of the 226 patients with F0-1 developed HCC (Log-Rank test P=0.01). Compared to SC subjects, only SVR patients with F2-4 (Log-Rank test P<0.001), but not F0-1(Log-Rank test P=0.60), had significantly higher risk of HCC development (Figure 2 and Table 2, model 2). A Cox-regression analysis using liver fibrosis as a variable demonstrated that factors associated with HCC included SVR with F2-4 (versus SC: HR/CI: 10.06/2.20-45.98, P= 0.003), diabetes (HR/CI:3.23/1.14-9.19, P=0.03), and age (HR/CI: 1.08 1.02-1.15, P=0.015), but did not include SVR with F0-1 (versus SC: HR/CI: 1.98/0.26-14.8).
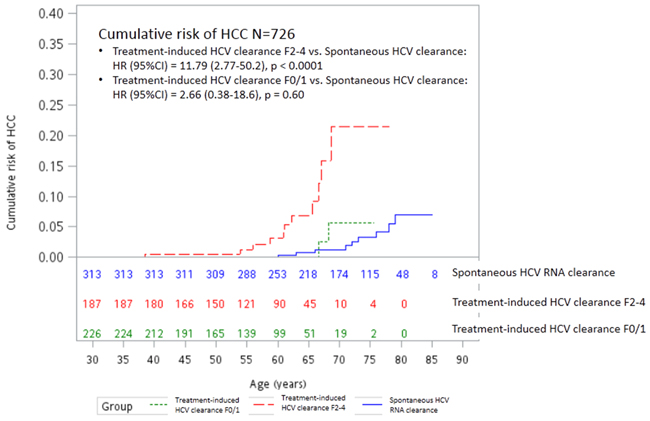
Figure 2: Cumulative risk of HCC between subjects with spontaneous and treatment-induced (Fibrosis score 0 or 1 versus Fibrosis score 2-4) HCV RNA clearance. HCC: hepatocellular carcinoma; HCV: hepatitis C virus; HR: hazard ratio; CI: confidence intervals.
DISCUSSION
To date, most of the studies that investigated the beneficial effects of SVR upon HCV-related HCC prevention compared to patients who failed viral eradication or described the differential benefits of antiviral therapy on different SVR subgroups [10]. The magnitude of HCC risk reduction by antiviral treatment has seldom been compared with self-limited HCV infection. Subjects with self-limited HCV infection share common environmental and behavioral risk factors with CHC patients. Selecting subjects with spontaneous HCV clearance as the comparator may have the advantage of eliminating selection bias when evaluating liver-related clinical outcomes [11]. In the current study, we demonstrated that successful viral eradication with antivirals greatly reduced HCV-related HCC occurrence. Nevertheless, the risk remains higher compared to subjects with spontaneous viral clearance, especially among those with pre-existing significant hepatic fibrosis.
Spontaneous seroclearance of HBV DNA and HBsAg are important predictors of reduced HBV-related HCC risk [12]. It has been suggested that HCC occurrence is rare in chronic hepatitis B (CHB) patients who experienced HBsAg seroclearance after nucleoside analogue therapy [13]. On the other hand, compared with inactive CHB carriers, there remains a higher risk for HCC even if the virus was suppressed with oral antiviral therapy [14]. Genetic instability due to HBV viral integration may play a role in the hepatic carcinogenesis. Unlike HBV infection, HCV replication could be completed aborted, which decreases the HCC risk by 75% compared to patients with viral persistence [10]. The results have raised concerns regarding the clinical outcome of treatment-induced SVR compared to self-limited HCV infections. We observed that there remains a six-fold HCC risk for those with successful viral eradication. Notably, the risk was similar to controls in SVR patients without significant fibrosis. The American Association for the Study of Liver Disease has advocated that SVR patients with mild liver disease should receive the same medical care that is recommended for patients who were never infected with HCV [15]. One of the major treatment concerns is prioritization and justification of patients with F2. We have proved that CHC patients with F2 would carried a HCC risk of 6.6 fold as compared to those with F0-1 if they were left with persist viremia [16]. The World Health Organization (WHO) has updated patients with a METAVIR score ≥ 2 as having “significant fibrosis”. Consequently, patients with significant fibrosis had a ten-fold risk of HCC even though they could benefit from successful HCV eradication. Careful monitoring of HCC development remains warranted in these patient groups [16]. Age was an independent risk factor for HCC, which echoes our previous finding that attention should also be paid to elderly patients who achieved an SVR [9]. The issue of the impact of diabetes on HCC is conflicting. A recent meta-analysis has shown that CHC patients carried higher risks of HCC if they possess DM [17]. While the threat decreases with successful HCV eradication, it still remains in diabetic patients. As a consequence, diabetic patients should be viewed as a high priority for treatment [18] with persistent follow-up visits after antiviral therapy.
The role of Il-28B genetic variants in the interferon-based antiviral treatment response is well documented. However, it impact on HCV related HCC remains elusive. We have demonstrated that unfavorable Il-28B genotype, rs8099917 TG/GG genotype, was associated with HCC risk in untreated cohort [3]. On the other hand, we recently demonstrated that it no longer played a role in the post-treatment cohort [19]. In the current study, Il-28B genetic variants also did not determine HCC occurrence in patients with viral clearance. This is in line with the hypothesis that successful viral eradication may overcome the poor genetic predispositions in terms of HCC occurrence. Further studies in different ethnicities are warranted to validate our finding.
One of the major limitations of the current study was that we used patients with self-limited HCV infections as healthy controls. Indeed, the incidence of spontaneous HCV clearance in CHC patients is rare [20]. It has been reported that up to 17% of patients who have self-limited HCV infections cleared the virus after more than 1 year of exposure [21]. Yet However, only 22 (7.56%) of the spontaneous sero-converters in our study had delayed HCV clearance, and the impact of viral persistence on the hosts in terms of liver fibrosis and hepatic carcinogenesis may not be significant. Furthermore, different characteristics between the two groups may interfere with the association with HCC. Spontaneous resolvers are usually younger [11, 20]. It has been recently suggested that that SC in the CHC population possessed four characteristics, younger age, female gender, low HCV RNA levels and HBV coinfection [20]. Among them, age [9], sex and HBV dual infection sex are known as risk factors for HCV related HCC. HBV dual infection was not included in the study. We attempted to adjust the impact of the potential confounders by conducting the age- and sex-matched study. Patients with HBs Ag seropositivity were excluded. However, we failed to exclude subjects with occult HBV infection (OBI) in the current study. The influence of OBI in HCC remains conflicting. Nevertheless, it has been suggested that it has less impact on HCV related HCC [22].
In conclusion, treatment-induced SVR greatly reduces the risk of HCC development. Nevertheless, patients with pre-existing significant hepatic fibrosis remain to be at a higher risk compared to subjects with self-limited HCV infections. All patients who are chronically infected with HCV should be treated, except for individuals with anticipated short-life expectancies [15]. Direct antiviral agents (DAAs) are currently the mainstream of treatment toward CHC. However, it not unaffordable and inaccessible for the majority patients and countries. Treatment at early stage can reduce HCC risk as much as population with spontaneous viral clearance. HCV should be eradicated before the development of significant fibrosis on the host. The results may provide important information for decision-making regarding the prioritization of the current high-cost DAAs for CHC in resource-limited countries.
MATERIALS AND METHODS
Subjects with spontaneous HCV seroclearance, namely the SC group, were selected from the Risk Evaluation of Viral Load Elevation and Associated Liver Disease/Cancer (R.E.V.E.A.L) HCV cohort. The R.E.V.E.A.L-HCV study is a community-based cohort and enrolled participants from seven townships in Taiwan during 1991-1992. The details of the study participants, interview, blood collection and laboratory examinations have been described previously in detail. [4, 23] Sex- and age- matched CHC patients with treatment-induced SVR after antiviral therapy, namely the SVR group, were consecutively enrolled at a medical center and three regional hospitals between1991 and2011. Patients who had evidence of underlying HCC, hepatitis B virus (defined as HBs antigen seropositivity, Abbott, North Chicago, IL) or human immunodeficiency virus (HIV) co-infections were excluded from both groups. In the SVR group, patients receiving either conventional interferon or peginterferon with or without ribavirin were enrolled if they achieved an SVR, defined as seronegativity of HCV RNA throughout a 24-week post-treatment follow-up period. The subjects with spontaneous HCV RNA seroclearance was defined as participants who had anti-HCV seropositivity and undetectable HCV RNA at baseline and participants who had anti-HCV seropositivity and detectable HCV RNA levels at baseline, but HCV RNA became undetectable after more than one-year follow-up [3]. HCV antibody was detected using third-generation enzyme immunoassay (Abbott Laboratories, North Chicago, IL). Serum levels of HCV RNA in the SC group were measured using the COBASTaqMan HCV test, version 2 (Roche Diagnostics, Indianapolis, NJ, detection limit: 25 IU/mL). In the SVR group, the HCV RNA levels were detected using a qualitative real-time polymerase chain reaction (PCR) (COBAS AMPLICOR Hepatitis C Virus Test, ver. 2.0; Roche, Branchburg, NJ, USA, detection limit: 50 IU/ml) and a quantification branched DNA assay (Versant HCV RNA 3.0, Bayer, Tarrytown, New Jersey, USA; quantification limit: 615 IU/ml). Liver histology, which was obtained within one year of starting antiviral therapy in the SVR group, was graded and staged according to the scoring system described by Scheuer [24]. All of the participants provided their written informed consent for the intervention. This study was approved by the Institutional Review Board of the College of Public Health, National Taiwan University, Taipei, Taiwan and Kaohsiung Medical University Hospital, Kaohsiung, Taiwan, and conformed to the guidelines of the International Conference on Harmonization for Good Clinical Practice.
Genetic testing
Interleukin 28B (IL-28B) rs8099917 was selected as the single-nucleotide polymorphism (SNP) to be tested for its association with HCC. The genetic testing was determined using methods that have been described previously [3, 25-27].
Statistical analyses
The baseline characteristics of the clinical cohort and R.E.V.E.A.L.-HCV cohort were compared by chi-squared tests. The cumulative risk of the incidence of HCC among patients who occurred spontaneous HCV RNA clearance (R.E.V.A.E.L.-HCV cohort) and treatment-induced RNA clearance was estimated by Kaplan-Meier method, and the statistical significance of the difference was examined by log-rank tests. Cox proportional hazards models were used to estimate multivariate-adjusted hazard ratios (HR) with 95 percent confidence intervals (95% CI) for HCC risk for various groups with RNA clearance after adjustment for other risk factors. Statistical significance levels were determined by a 2-sided P-value of 0.05. The proportionality assumption (non-changing HRs over time) of Cox models was examined, and the assumption was not violated. All analyses were performed using the SAS statistical software package (release 9.1; SAS Institute Inc., Cary, NC).
Abbreviations
ALT: alanine aminotransferase; AST: aspartate aminotransferase; CHC: chronic hepatitis C; DAAs: direct antiviral agents; HCC: hepatocellular carcinoma; HCV: hepatitis C virus; HIV: human immunodeficiency virus; IL-28B: interleukin 28B; SVR: sustained virological response; SNP: single-nucleotide polymorphism; SC: seroclearance.
Authors’ contributions
Conception and design: Ming-Lung Yu, Wan-Long Chuang and Chien-Jen Chen.
Acquisition of data: Ming-Lun Yeh, Ching-I Huang,
Zu-Yau Lin, Shinn-Cherng Chen, Hwai-I Yang, Jee-Fu Huang, Sheng-Nan Lu, Chia-Yen Dai, Chin-Lan Jen, Yong Yuan, Gilber L’Italien and Li-Yu Wang
Data analysis and interpretation: Mei-Hsuan Lee, Yu-Ju Lin, Soa-Yu Chan, Pei-Chien Tsaiand Ming-Lung Yu.
Manuscript drafting and critical revising: Chung-Feng Huang, Mei-Hsuan Lee and Ming-Lung Yu.
Approval of the final version of the manuscript: Mei-Hsuan Lee and Ming-Lung Yu.
ACKNOWLEDGMENTS
This study was supported by Kaohsiung Medical University Hospital (KMUH103-10V04), Kaohsiung Medical University (MOST 104-2628-B-010-001-MY3) and National Science Council of Taiwan (NSC 102-2314-B-010-057-MY2).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Yu ML, Dai CY, Huang CF, Lee JJ, Yeh ML, Yeh SM, Kuo HT, Huang JF, Chang JM, Chen HC, Juo SH, Hwang SJ, Chuang WL, et al. High hepatitis B virus surface antigen levels and favorable interleukin 28B genotype predict spontaneous hepatitis C virus clearance in uremic patients. J Hepatol. 2014;60:253-9.
2. Dai CY, Ho CK, Huang JF, Hsieh MY, Hou NJ, Lin ZY, Chen SC, Wang LY, Chang WY, Yu ML, Chuang WL. Hepatitis C virus viremia and low platelet count: a study in a hepatitis B & C endemic area in Taiwan. J Hepatol. 2010;52:160-6.
3. Lee MH, Yang HI, Lu SN, Lin YJ, Jen CL, Wong KH, Chan SY, Chen LC, Wang LY, L’Italien G, Yuan Y, Chen CJ. Polymorphisms near the IFNL3 gene associated with HCV RNA spontaneous clearance and hepatocellular carcinoma risk. Sci Rep. 2015;5:17030.
4. Lee MH, Yang HI, Lu SN, Jen CL, Yeh SH, Liu CJ, Chen PJ, You SL, Wang LY, Chen WJ, Chen CJ. Hepatitis C virus seromarkers and subsequent risk of hepatocellular carcinoma: long-term predictors from a community-based cohort study. J Clin Oncol. 2010;28:4587-93.
5. Mohd Hanafiah K, Groeger J, Flaxman AD, Wiersma ST. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57:1333-42.
6. de Martel C, Maucort-Boulch D, Plummer M, Franceschi S. World-wide relative contribution of hepatitis B and C viruses in hepatocellular carcinoma. Hepatology. 2015;62:1190-200.
7. Yu ML, Lee CM, Chen CL, Chuang WL, Lu SN, Liu CH, Wu SS, Liao LY, Kuo HT, Chao YC, Tung SY, Yang SS, Kao JH, et al. Sustained hepatitis C virus clearance and increased hepatitis B surface antigen seroclearance in patients with dual chronic hepatitis C and B during posttreatment follow-up. Hepatology. 2013;57:2135-42.
8. Yu ML, Lin SM, Chuang WL, Dai CY, Wang JH, Lu SN, Sheen IS, Chang WY, Lee CM, Liaw YF. A sustained virological response to interferon or interferon/ribavirin reduces hepatocellular carcinoma and improves survival in chronic hepatitis C: a nationwide, multicentre study in Taiwan. Antivir Ther. 2006;11:985-94.
9. Huang CF, Yeh ML, Tsai PC, Hsieh MH, Yang HL, Hsieh MY, Yang JF, Lin ZY, Chen SC, Wang LY, Dai CY, Huang JF, Chuang WL, et al. Baseline gamma-glutamyl transferase levels strongly correlate with hepatocellular carcinoma development in non-cirrhotic patients with successful hepatitis C virus eradication. J Hepatol. 2014;61:67-74.
10. Morgan RL, Baack B, Smith BD, Yartel A, Pitasi M, Falck-Ytter Y. Eradication of hepatitis C virus infection and the development of hepatocellular carcinoma: a meta-analysis of observational studies. Ann Intern Med. 2013;158:329-37.
11. Innes H, Hutchinson SJ, Obel N, Christensen PB, Aspinall EJ, Goldberg D, Krarup H, McDonald SA, McLeod A, Weir A, Omland LH. Liver mortality attributable to chronic hepatitis C virus infection in Denmark and Scotland - using spontaneous resolvers as the benchmark comparator. Hepatology. 2016;63:1506-16.
12. Liu J, Yang HI, Lee MH, Lu SN, Jen CL, Batrla-Utermann R, Wang LY, You SL, Hsiao CK, Chen PJ, Chen CJ; R.E.V.E.A.L.-HBV Study Group. Spontaneous seroclearance of hepatitis B seromarkers and subsequent risk of hepatocellular carcinoma. Gut. 2014;63:1648-57.
13. Kim GA, Lim YS, An J, Lee D, Shim JH, Kim KM, Lee HC, Chung YH, Lee YS, Suh DJ. HBsAg seroclearance after nucleoside analogue therapy in patients with chronic hepatitis B: clinical outcomes and durability. Gut. 2014;63:1325-32.
14. Cho JY, Paik YH, Sohn W, Cho HC, Gwak GY, Choi MS, Lee JH, Koh KC, Paik SW, Yoo BC. Patients with chronic hepatitis B treated with oral antiviral therapy retain a higher risk for HCC compared with patients with inactive stage disease. Gut. 2014;63:1943-50.
15. HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C. http://www.hcvguidelines.org/. Accessed on Oct 22, 2016.
16. Yu ML, Huang CF, Yeh ML, Tsai PC, Huang CI, Hsieh MH, Hsieh MY, Lin ZY, Chen SC, Huang JF, Dai CY, Chuang WL. Time-degenerative factors and the risk of hepatocellular carcinoma after antiviral therapy among hepatic C virus patients: a model for prioritization of treatment. Clin Cancer Res. 2016.
17. Chen J, Han Y, Xu C, Xiao T, Wang B. Effect of type 2 diabetes mellitus on the risk for hepatocellular carcinoma in chronic liver diseases: a meta-analysis of cohort studies. Eur J Cancer Prev. 2015;24:89-99.
18. AASLD/IDSA HCV Guidance Panel. Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology 2015;62:932-54.
19. Huang CF, Huang CY, Yeh ML, Wang SC, Chen KY, Ko YM, Lin CC, Tsai YS, Tsai PC, Lin ZY, Chen SC, Dai CY, Huang JF, et al. Genetics variants and serum levels of MHC class I chain-related A in predicting hepatocellular carcinoma development in chronic hepatitis C patients post antiviral treatment. EBioMedicine. 2017;15:81-9.
20. Bulteel N, Partha Sarathy P, Forrest E, Stanley AJ, Innes H, Mills PR, Valerio H, Gunson RN, Aitken C, Morris J, Fox R, Barclay ST. Factors associated with spontaneous clearance of chronic hepatitis C virus infection. J Hepatol. 2016;65:266-72.
21. Grebely J, Page K, Sacks-Davis R, van der Loeff MS, Rice TM, Bruneau J, Morris MD, Hajarizadeh B, Amin J, Cox AL, Kim AY, McGovern BH, Schinkel J, et al. The effects of female sex, viral genotype, and IL28B genotype on spontaneous clearance of acute hepatitis C Virus infection. Hepatology. 2014;59:109-20.
22. Huang X, Hollinger FB. Occult hepatitis B virus infection and hepatocellular carcinoma: a systematic review. J Viral Hepat. 2014;21:153-62.
23. Lee MH, Yang HI, Jen CL, Lu SN, Yeh SH, Liu CJ, You SL, Sun CA, Wang LY, Chen WJ, Chen CJ; R.E.V.E.A.L.-HCV Study Group. Community and personal risk factors for hepatitis C virus infection: a survey of 23,820 residents in Taiwan in 1991-2. Gut. 2011;60:688-94.
24. Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol. 1991;13:372-4.
25. Yu ML, Huang CF, Huang JF, Chang NC, Yang JF, Lin ZY, Chen SC, Hsieh MY, Wang LY, Chang WY, Li YN, Wu MS, Dai CY, et al. Role of interleukin-28B polymorphisms in the treatment of hepatitis C virus genotype 2 infection in Asian patients. Hepatology. 2011;53:7-13.
26. Huang CF, Huang JF, Yang JF, Hsieh MY, Lin ZY, Chen SC, Wang LY, Juo SH, Chen KC, Chuang WL, Kuo HT, Dai CY, Yu ML. Interleukin-28B genetic variants in identification of hepatitis C virus genotype 1 patients responding to 24 weeks peginterferon/ribavirin. J Hepatol. 2012;56:34-40.
27. Huang CF, Yu ML, Kao JH, Tseng TC, Yeh ML, Huang JF, Dai CY, Lin ZY, Chen SC, Wang LY, Juo SH, Chuang WL, Liu CH. Profound week 4 interferon responsiveness is mandatory for hepatitis C genotype 1 patients with unfavorable IL-28B genotype. J Clin Virol. 2013;56:293-8.

