INTRODUCTION
Pituitary adenomas (PAs) are the second most common type of intracranial neoplasm and account for approximately 15% of intracranial tumors, although autopsy studies indicate a higher incidence of 25% [1, 2]. Most PAs are benign and exhibit no expansive properties; however, approximately 30-40% are aggressive PAs that massively invade the surrounding anatomical structures [3]. Surgery has long been the first-line treatment for PAs (except for prolactinomas). However, aggressive PAs are difficult to resect completely and tend to recur due to massive invasion of adjacent tissues (e.g., the cavernous sinus and the dura). Such PAs are generally refractory to repeated surgeries, radiotherapy and alternative medical therapies [4]. Therefore, these aggressive PAs are difficult to manage and are associated with poor prognosis and fatality [5].
Temozolomide (TMZ), a routine chemotherapy for glioblastoma (GBM), it exerts its cytotoxic activity by alkylating DNA at the O6 position of guanine. In recent decades, TMZ has been reported to have significant therapeutical effects on PAs and pituitary carcinomas [6, 7]. However, O-6-methylguanine DNA methyltransferase (MGMT), a DNA repair protein, alters the methylation status of DNA and reverses TMZ-induced alkylation. Until now, MGMT has been recommended as an important predictor of the efficacy of TMZ therapy in GBM [8, 9]. Several clinical studies have demonstrated that the expression and/or promoter methylation of MGMT may have prognostic significance in GBM [10–12]. The low expression of MGMT has also been reported with a higher frequency amongst PAs [13]. Some studies have indicated that MGMT expression is associated with PA patients’ prognoses and responses to TMZ [14–16]. However, in other studies, MGMT expression has not correlated significantly with clinical responses to TMZ or clinical outcomes in patients with PAs [17–20]. It remains uncertain whether discrepancies in these data are mainly due to limited sample sizes or genuine heterogeneity.
Thus, it is necessary to review and systematically assess the precise association of MGMT expression with the prognoses and clinicopathological indicators of PAs patients. To this end, we have performed the following meta-analysis.
RESULTS
Search results and characteristics of included studies
As shown in Figure 1, 43 papers were initially identified in our search. During the initial review of the titles and abstracts, 28 articles not relevant to our goal were excluded. Two reviewers then independently reviewed the remaining 15 articles, 4 of which were excluded for insufficient data. Ultimately, 11 articles that met the criteria were included.
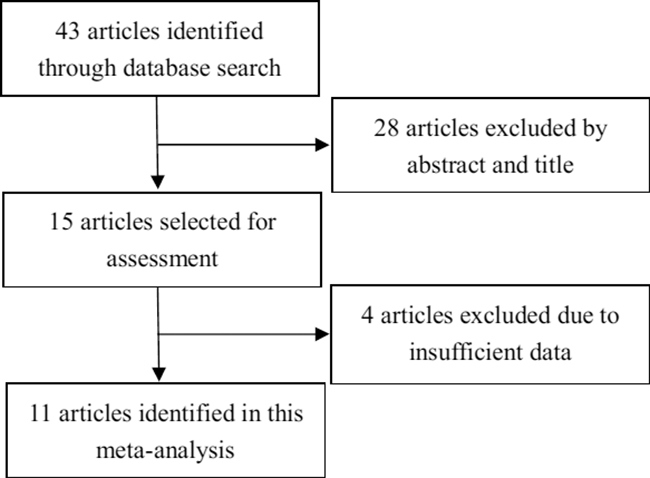
Figure 1: Flow chart depicting the study selection procedure. Eleven studies were included in this meta-analysis according to the inclusion criteria.
The general features of the 11 articles are summarized in Table 1. A total of 454 patients with PA were involved in this meta-analysis, including 180 patients with invasive PAs and 243 patients with noninvasive PAs (the invasion properties of PA were not mentioned for the remaining patients). The classical immunohistochemistry (IHC) method was performed to detect MGMT protein expression in all included studies. The percentage of positive MGMT expression ranged from 5 to 90%. Positive MGMT-expressing patients were defined in three ways. Most investigators scored MGMT expression as positive if any part of the nucleus or cytoplasm was stained. In these studies, MGMT immunoexpression was scored on a 4-tiered scale (1=negative or limited to 10%, 2=10–25%, 3=25–50% and 4=≥50%). Scores of 1 and 2 were combined to form the category of ‘‘low level MGMT expression,’’ while scores of 3 and 4 represented intermediate and high MGMT expression, respectively [26, 27, 31]. There were also differences in the definition of the cut-off value of high MGMT expression; some investigators defined the cut-off value using a score combining the intensity and percentage of MGMT expression, while others used only the percentage of stained cells.
Table 1: Baseline characteristics of studies included in the meta-analysis
Study ID |
Country |
Year |
Num. |
Gender (M/F) |
Age (years) |
Invasiveness Yes/No |
Recurrence Yes/No |
Method |
Cut-off |
|---|---|---|---|---|---|---|---|---|---|
Takeshita A [21] |
Japan |
2009 |
24 |
3/21 |
– |
9/15 |
– |
IHC |
5% |
McCormack A I [22] |
Australia |
2009 |
88 |
– |
– |
46/42 |
13/75 |
IHC |
10% |
Widhalm G [23] |
Austria |
2009 |
45 |
29/16 |
– |
25/20 |
24/21 |
IHC |
50% |
Salehi F [24] |
Canada |
2010 |
8 |
3/5 |
62.4(57–66) |
– |
– |
IHC |
25% |
Fealey M E [25] |
USA |
2010 |
23 |
15/8 |
35.0(17–69) |
– |
2/21 |
IHC |
25% |
Lau Q [26] |
USA |
2010 |
30 |
– |
– |
15/15 |
– |
IHC |
Score>3 |
Zuhur S S [27] |
Turkey |
2011 |
25 |
10/15 |
43.0(23–65) |
10/15 |
– |
IHC |
Score>3 |
Salehi F [28] |
USA |
2011 |
12 |
8/4 |
34.4(17–64) |
7/5 |
2/9 |
IHC |
10% |
Salehi F [29] |
USA |
2012 |
40 |
12/28 |
40.6(15–62) |
16/24 |
11/29 |
IHC |
25% |
McCormack A [30] |
Australia |
2013 |
21 |
15/6 |
55.4(24–79) |
9/12 |
2/19 |
IHC |
10% |
Jiang X [31] |
China |
2013 |
138 |
67/71 |
44±14.5 |
43/95 |
12/126 |
IHC |
Score≥3 |
Num: numbers; M: male; F: female; IHC: immunohistochemistry;
Study assessment
The quality of each eligible study, as assessed with the European Lung Cancer Working Party criteria, is presented in Table 2. The mean final score of the all studies was 67.25%, and the global scores of studies analyzing recurrence and invasiveness were 67.7% and 69.5%, respectively.
Table 2: Clinical and methodological characteristic of included studies
No.of studies |
Design |
Method |
Generalizability |
Results analysis |
Global score(%) |
|
|---|---|---|---|---|---|---|
All studies |
10 |
6.7 |
7.1 |
6.9 |
6.2 |
67.25 |
Invasiveness |
7 |
7.2 |
6.8 |
6.5 |
7.3 |
69.5 |
Recurrence |
7 |
6.6 |
6.9 |
6.5 |
7.1 |
67.75 |
Quantitative data synthesis
There are seven cohort studies that referred to the relationship of MGMT expression with tumor recurrence or invasiveness pituitary adenoma (Table 3). A fixed-effects model was used because there was not significant heterogeneity among the studies (Table 3). The pooled OR from all seven studies on recurrence was 2.09 (95% CI: 1.09–4.02; p=0.026) (Figure 2), indicating that low MGMT expression predicted recurrence and poor survival in PA patients. No heterogeneity was observed (χ2=1.71, p=0.944, I2 =0) (Table 3). However, we found no significant association between MGMT expression and PA invasiveness from all seven included studies regarding to invasion (OR=1.11, 95% CI=0.71–1.75; p=0.646) (Figure 3), indicating that MGMT may be not involved in the invasion of PA. There was no significant heterogeneity among the studies (χ2=8.65, p=0.279, I2 =19.1%) (Table 3). The above results indicate that low expression of MGMT may be related to tumor recurrence but not invasion of patients with PAs.
Table 3: Pooled HR and 95% CI in meta–analysis of association of MGMT expression with chinicopathological indicators
No.of results |
OR |
95% CI |
heterogeneity |
|||
|---|---|---|---|---|---|---|
χ2 |
p |
I2 (%) |
||||
Age |
7 |
0.99 |
0.57–1.74 |
6.78 |
0.342 |
11.5 |
Gender |
7 |
1.08 |
0.63–1.85 |
5.05 |
0.538 |
0 |
Invasiveness |
7 |
1.11 |
0.71–1.75 |
8.65 |
0.279 |
19.1 |
Tumor size |
4 |
0.94 |
0.50–1.78 |
9.32 |
0.025 |
67.8 |
Recurrence |
7 |
2.09 |
1.09–4.02 |
1.71 |
0.944 |
0 |
Functional |
3 |
1.97 |
0.94–3.32 |
2.05 |
0.360 |
2.2 |
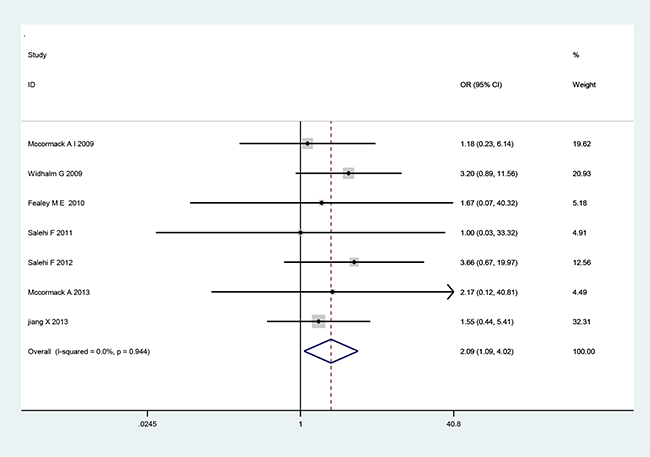
Figure 2: Forest plots for the relationship between MGMT expression and PA tumor recurrence. The pooled OR for all seven studies was 2.09 (95% CI 1.09–4.02; p=0.026). No heterogeneity was observed (χ2=1.71, p=0.944, I2=0).
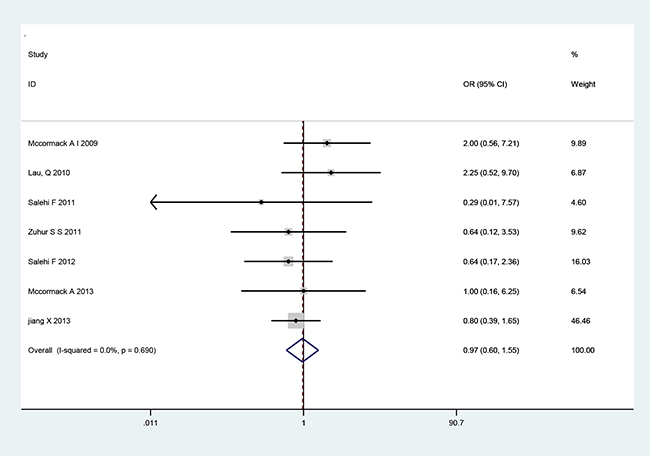
Figure 3: Forest plots for the relationship between MGMT expression and PA tumor invasiveness. The pooled OR for all seven studies was 1.11 (95% CI 0.71–1.75; p=0.646). No heterogeneity was observed (χ2=8.5, p=0.279, I2=19.1).
To gain further insight into the value of MGMT as a biomarker, we investigated the association of MGMT expression with various clinicopathological indicators, including age (greater than the median age), gender, tumor size, and functional status (Table 3). However, no significant relationship was observed between MGMT expression and age, gender, tumor size, or functional status (Figure 4A, 4B, 4C and 4D). To summary, these findings indicate that lower MGMT expression may be used to predict the recurrence of PAs, however, MGMT expression is not related to other clinicopathological indicators, such as invasiveness age, gender, tumor size, or functional status of patients with PAs.
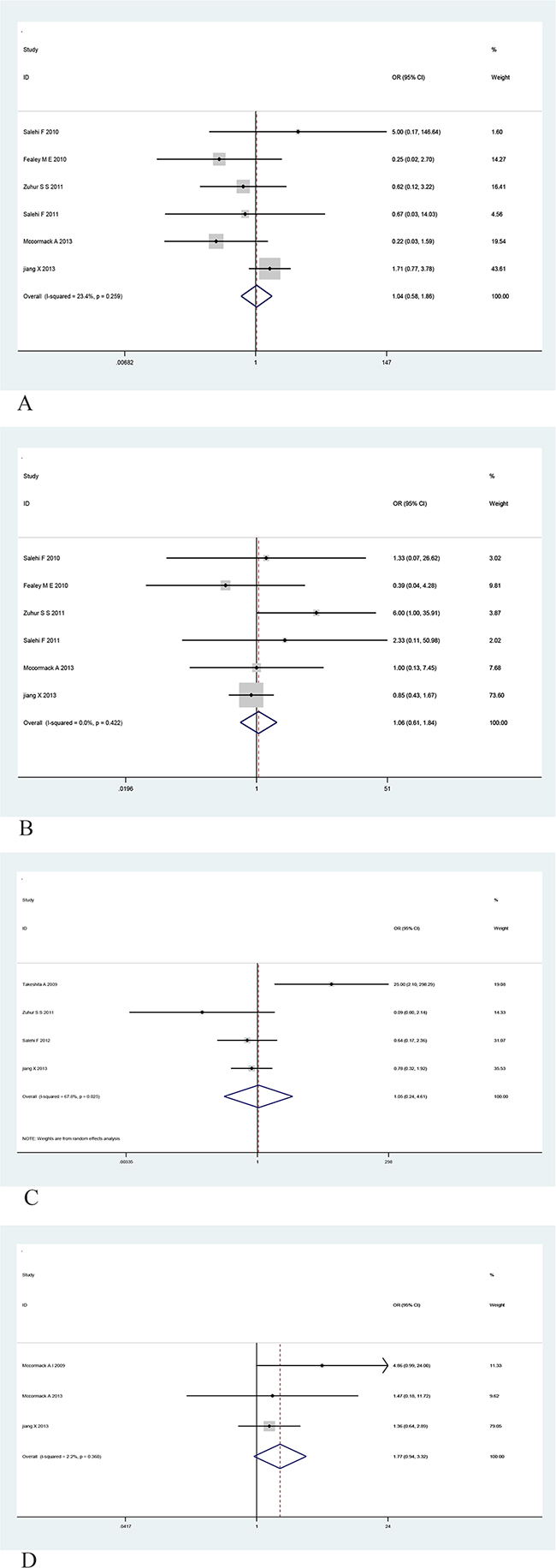
Figure 4: Forest plots for the relationship between MGMT expression and age, gender, tumor size and functional status of PA.
Publication bias
In the present meta-analysis, the publication bias among studies with regard to tumor recurrence was investigated with Begg’s and Egger’s tests. No publication bias was observed (p=0.124, 0.610, respectively). The shapes of Begger’s and funnel plots did not reveal any obvious asymmetry (Figure 5). Sensitivity analysis was performed to assess the effects of individual studies on the pooled ORs through omission of each individual study. No individual study significantly affected the pooled ORs (Figure 6), indicating that the results were statistically robust.
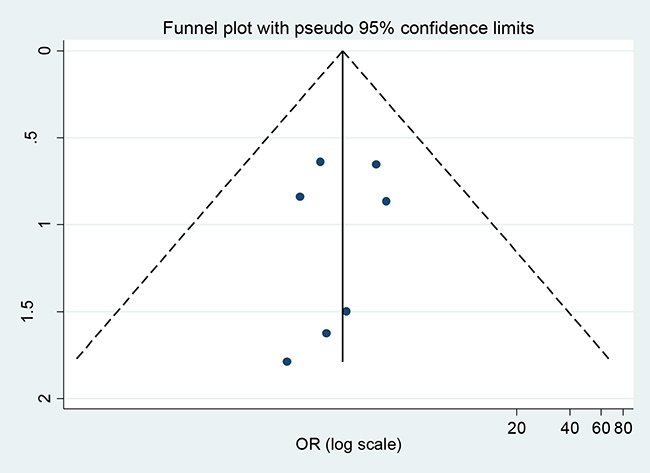
Figure 5: Begger’s funnel plots of the association between MGMT expression and PA tumor recurrence.
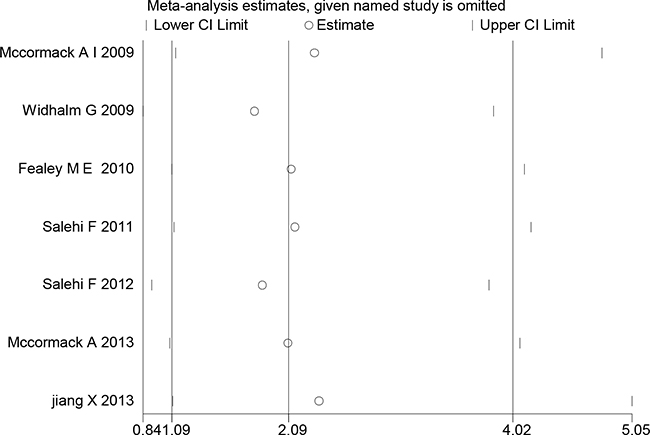
Figure 6: Sensitivity analysis of the association between MGMT expression and PA tumor recurrence. Results were computed through the omission of each study in turn. Meta-analysis fixed-effect estimates were used. The two ends of the dotted lines represent the 95% CI.
DISCUSSION
Aggressive PAs are notoriously difficult to manage and are associated with poor prognosis because the therapeutic options are limited and the tumors are generally refractory to standard therapy [32]. Despite the use of multimodal therapies, including repeated surgeries, radiotherapy and alternative medical therapies, postoperative recurrence often occurs [33, 34]. Therefore, it is essential to identify novel molecular markers that will allow the early prediction of PA recurrence and/or invasiveness and the early application of multi-modal therapy to prevent tumor recurrence.
As a salvage therapy, TMZ has recently been shown to have significant efficacy for the treatment of aggressive PAs and pituitary carcinomas [6]. However, some patients acquire TMZ resistance after treatment. MGMT has been suggested as a biomarker that predicts the response to TMZ and the prognosis in GBM [9, 12]. Despite the large number of studies on this subject, the prognostic value of MGMT for predicting PA patient survival is controversial, due to the small sample sizes and genuine heterogeneity of the published studies [23, 27, 30].
In the present meta-analysis, we evaluated the association of MGMT expression with features of human PAs. Ultimately, 11 independent case-control studies with a total of 454 PA patients were included. Our meta-analysis results suggested that lower expression of MGMT was associated with PA recurrence, suggesting that MGMT expression could be used as a marker of poor prognosis and tumor recurrence for patients with PA. Widhalm et al. reported that low MGMT expression was observed more frequently in patients with progressive tumors than in tumor-free subjects [23]. Consistent with this, Salehi et al. also demonstrated that 92% of silent subtype 3 PAs exhibited low MGMT immunoreactivity [28]. Although the exact function of MGMT in the pathogenesis and progression of PAs is not yet fully understood, it may be that low MGMT expression causes the upregulation of gene sets involved in DNA repair and transcription, thus increasing mutagenesis, which further drives the tumorigenic process and increases cellular proliferation [37]. Until now, no study has demonstrated that any molecular marker could be used routinely to predict PA recurrence. Here, we provided the first meta-analysis of the relationship between MGMT expression and biological characteristics of PAs, and found that low expression of MGMT may be associated with PA recurrence, suggesting that TMZ therapy may have promising efficacy for recurrent PAs with low MGMT expression.
Nevertheless, our meta-analysis also had some limitations. MGMT expression in the included studies was measured by IHC, which depends strongly upon methodological factors. The different sources and concentrations of primary and secondary antibodies may have severely reduced the reliability and applicability of the results regarding MGMT expression. Furthermore, there were large differences in the definitions of cut-off values; after all, there are no criteria available for such cut-offs. In addition, the present research was restricted to articles published in English and Chinese, because articles in other languages such as Japanese were not accessible to the readers. Last but not least, this meta-analysis used a retrospective study design that may have led to subject selection bias and thus may have reduced the reliability of our results.
In conclusion, the present meta-analysis indicates that MGMT expression is indeed associated with PA recurrence, but not with invasiveness, age, gender, tumor size or functional status. Thus, MGMT expression may be used as a molecular marker for the early prediction of PA recurrence and the identification of candidates for TMZ therapy. However, due to the limitations mentioned above, further large-scale studies are still required to confirm our findings.
MATERIALS AND METHODS
Search strategy
A literature search was carried out with the databases of CISCOM, CINAHL, Web of Science, PubMed, Google Scholar, EBSCO, Cochrane Library, and CBD up to July 2016. There was no language restriction. The following keywords and MeSH terms were used: “pituitary”, “pituitary adenoma”, “pituitary adenomas”, “pituitary tumor”, “pituitary tumors”, “pituitary macroadenoma’’, ‘‘MGMT’’ or ‘‘O6methylguanine DNA methyl transferase’’, etc. We also performed a manual search to find other potential articles.
Selection criteria
The following criteria were used for the selection of studies for this meta-analysis: (1) the study was designed as a clinical cohort study or case-control study; (2) the study related to the expression of MGMT in human PAs; (3) all patients had confirmed diagnoses of PA; (4) the study provided sufficient information about the MGMT expression and clinical characteristics of the PAs; and (5) the invasiveness of the PAs was assessed by imaging characteristics or intraoperative observation. If a study did not meet the inclusion criteria, it was excluded. The most recent publication or the publication with the largest sample size was included when the authors published several studies using the same subjects. Any disagreements were resolved through discussions and subsequent consensus.
The following criteria were used for the diagnosis of invasive PAs: (1) PAs of grades III and IV and stages C, D, and E were considered invasive according to Hardy classification; (2) invade to adjacent structures such as the parasellar region, cavernous sinus and hypothalamus could be seen on MRI and CT scans; (3) tumor cell invasion was pathologically confirmed in the sellar bone or adjacent dura mater; and (4) the sellar bone and dura mater were invaded and damaged, and tumors penetrated the sphenoid sinus or invaded the parasellar vascular and nervous crossroads. If a tumor did not meet these criteria, it was considered to be a non-invasive PA.
Data extraction
Two reviewers collected the following data independently using a purpose-designed form: the name of the first author, language of publication, publication date, country of the population studied, sample size, source of subjects, histology, detection methods, MGMT expression, invasion of PAs, recurrence of PAs, etc. Disagreement between the two reviewers was settled by a third reviewer.
Quality assessment
We evaluated the methodological quality of the included studies by reading and scoring each publication according to the quality scale for biological prognostic factors established by the European Lung Cancer Working Party [38]. This scale is widely used to assess the scientific design, laboratory methodology, generalizability and result analysis of studies. A total of 10 points could be attained in each category, so the maximum total score was 40 points. All reviewers compared their calculated scores and, if necessary, reached a consensus score during a meeting. The final scores represent the percentage of the maximum achievable score, ranging from 0 to 100 percent. Therefore, higher scores indicate better methodological quality.
Statistical analysis
Meta-analysis was performed with STATA 12.0 software (Stata Corp, College Station, TX, USA). Crude odds ratios (ORs) or standard mean differences (SMDs) with corresponding 95% confidence intervals (95% CIs) were calculated. OR and 95% CI could be extracted directly from included studies. The Z test was used to estimate the statistical significance of the ORs. Cochran’s Q-statistic and I2 tests were performed to evaluate potential heterogeneity among the studies. A random-effects model was used if the Q-test yielded a P value <0.05 or the I2 test yielded a value >50%, indicating significant heterogeneity; otherwise, if heterogeneity was not significant, a fixed-effects model was used. Subgroup and meta-regression analyses also were used to explore potential sources of heterogeneity. A sensitivity analysis was carried out, in which each study was omitted in turn, to evaluate the influence of each study on the overall estimate. Funnel plots and Egger’s linear regression test were used to investigate publication bias. All P-values were two-sided, and P<0.05 was considered statistically significant.
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (Grant 81502639) and the Beijing Municipal Natural Science Foundation (7154231). The funding institution had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
CONFLICTS OF INTEREST
The authors have declared that no competing interests exist.
REFERENCES
1. Asa SL, Ezzat S. The pathogenesis of pituitary tumours. Nat Rev Cancer. 2002; 2:836–49. doi: 10.1038/nrc926.
2. Ezzat S, Asa SL. Mechanisms of disease: the pathogenesis of pituitary tumors. Nat Clin Pract Endocrinol Metab. 2006; 2:220–30. doi: 10.1038/ncpendmet0159.
3. Colao A, Grasso LF, Pivonello R, Lombardi G. Therapy of aggressive pituitary tumors. Expert Opin Pharmacother. 2011; 12:1561–70. doi: 10.1517/14656566.2011.568478.
4. Patil CG, Prevedello DM, Lad SP, Vance ML, Thorner MO, Katznelson L, Laws ER Jr. Late recurrences of Cushing’s disease after initial successful transsphenoidal surgery. J Clin Endocrinol Metab. 2008; 93:358–62. doi: 10.1210/jc.2007-2013.
5. Di Ieva A, Rotondo F, Syro LV, Cusimano MD, Kovacs K. Aggressive pituitary adenomas--diagnosis and emerging treatments. Nat Rev Endocrinol. 2014; 10:423–35. doi: 10.1038/nrendo.2014.64.
6. Syro LV, Ortiz LD, Scheithauer BW, Lloyd R, Lau Q, Gonzalez R, Uribe H, Cusimano M, Kovacs K, Horvath E. Treatment of pituitary neoplasms with temozolomide: a review. Cancer. 2011; 117:454–62. doi: 10.1002/cncr.25413.
7. Raverot G, Sturm N, de Fraipont F, Muller M, Salenave S, Caron P, Chabre O, Chanson P, Cortet-Rudelli C, Assaker R, Dufour H, Gaillard S, François P, et al. Temozolomide treatment in aggressive pituitary tumors and pituitary carcinomas: a French multicenter experience. J Clin Endocrinol Metab. 2010; 95:4592–99. doi: 10.1210/jc.2010-0644.
8. Thomas RP, Recht L, Nagpal S. Advances in the management of glioblastoma: the role of temozolomide and MGMT testing. Clin Pharmacol. 2013; 5:1–9.
9. Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005; 352:997–1003. doi: 10.1056/NEJMoa043331.
10. Hegi ME, Liu L, Herman JG, Stupp R, Wick W, Weller M, Mehta MP, Gilbert MR. Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMT activity. J Clin Oncol. 2008; 26:4189–99. doi: 10.1200/JCO.2007.11.5964.
11. Miyazaki M, Nishihara H, Terasaka S, Kobayashi H, Yamaguchi S, Ito T, Kamoshima Y, Fujimoto S, Kaneko S, Katoh M, Ishii N, Mohri H, Tanino M, et al. Immunohistochemical evaluation of O6 -methylguanine DNA methyltransferase (MGMT) expression in 117 cases of glioblastoma. Neuropathology. 2014; 34:268–76. doi: 10.1111/neup.12091.
12. Pollack IF, Hamilton RL, Sobol RW, Burnham J, Yates AJ, Holmes EJ, Zhou T, Finlay JL. O6-methylguanine-DNA methyltransferase expression strongly correlates with outcome in childhood malignant gliomas: results from the CCG-945 Cohort. J Clin Oncol. 2006; 24:3431–37. doi: 10.1200/JCO.2006.05.7265.
13. McCormack AI, Wass JA, Grossman AB. Aggressive pituitary tumours: the role of temozolomide and the assessment of MGMT status. Eur J Clin Invest. 2011; 41:1133–48. doi: 10.1111/j.1365-2362.2011.02520.x.
14. Kovacs K, Scheithauer BW, Lombardero M, McLendon RE, Syro LV, Uribe H, Ortiz LD, Penagos LC. MGMT immunoexpression predicts responsiveness of pituitary tumors to temozolomide therapy. Acta Neuropathol. 2008; 115:261–62. doi: 10.1007/s00401-007-0279-5.
15. Arya S, Majaid MA, Shwetha SD, Sravani K, Arivazhagan A, Sampath S, Santosh V. Implications of MGMT methylation status in pituitary adenoma. Pathol Res Pract. 2014; 210:407–11. doi: 10.1016/j.prp.2014.02.010.
16. Widhalm G, Wolfsberger S, Preusser M, Woehrer A, Kotter MR, Czech T, Marosi C, Knosp E. O(6)-methylguanine DNA methyltransferase immunoexpression in nonfunctioning pituitary adenomas: are progressive tumors potential candidates for temozolomide treatment? Cancer. 2009; 115:1070–80. doi: 10.1002/cncr.24053.
17. Hirohata T, Asano K, Ogawa Y, Takano S, Amano K, Isozaki O, Iwai Y, Sakata K, Fukuhara N, Nishioka H, Yamada S, Fujio S, Arita K, et al. DNA mismatch repair protein (MSH6) correlated with the responses of atypical pituitary adenomas and pituitary carcinomas to temozolomide: the national cooperative study by the Japan Society for Hypothalamic and Pituitary Tumors. J Clin Endocrinol Metab. 2013; 98:1130–36. doi: 10.1210/jc.2012-2924.
18. Bush ZM, Longtine JA, Cunningham T, Schiff D, Jane JA Jr, Vance ML, Thorner MO, Laws ER Jr, Lopes MB. Temozolomide treatment for aggressive pituitary tumors: correlation of clinical outcome with O(6)-methylguanine methyltransferase (MGMT) promoter methylation and expression. J Clin Endocrinol Metab. 2010; 95:E280–90. doi: 10.1210/jc.2010-0441.
19. Takeshita A, Inoshita N, Taguchi M, Okuda C, Fukuhara N, Oyama K, Ohashi K, Sano T, Takeuchi Y, Yamada S. High incidence of low O(6)-methylguanine DNA methyltransferase expression in invasive macroadenomas of Cushing’s disease. Eur J Endocrinol. 2009; 161:553–59. doi: 10.1530/EJE-09-0414.
20. McCormack AI, McDonald KL, Gill AJ, Clark SJ, Burt MG, Campbell KA, Braund WJ, Little NS, Cook RJ, Grossman AB, Robinson BG, Clifton-Bligh RJ. Low O6-methylguanine-DNA methyltransferase (MGMT) expression and response to temozolomide in aggressive pituitary tumours. Clin Endocrinol (Oxf). 2009; 71:226–33. doi: 10.1111/j.1365-2265.2008.03487.x.
21. Salehi F, Scheithauer BW, Moyes VJ, Drake WM, Syro LV, Manoranjan B, Sharma S, Horvath E, Kovacs K. Low immunohistochemical expression of MGMT in ACTH secreting pituitary tumors of patients with Nelson syndrome. Endocr Pathol. 2010; 21:227–29. doi: 10.1007/s12022-010-9138-3.
22. Fealey ME, Scheithauer BW, Horvath E, Erickson D, Kovacs K, McLendon R, Lloyd RV. MGMT immunoexpression in silent subtype 3 pituitary adenomas: possible therapeutic implications. Endocr Pathol. 2010; 21:161–65. doi: 10.1007/s12022-010-9120-0.
23. Lau Q, Scheithauer B, Kovacs K, Horvath E, Syro LV, Lloyd R. MGMT immunoexpression in aggressive pituitary adenoma and carcinoma. Pituitary. 2010; 13:367–79. doi: 10.1007/s11102-010-0249-0.
24. Zuhur SS, Tanik C, Karaman Ö, Velet S, Çil E, Öztürk FY, Özkayalar H, Müslüman AM, Altuntaş Y. MGMT immunoexpression in growth hormone-secreting pituitary adenomas and its correlation with Ki-67 labeling index and cytokeratin distribution pattern. Endocrine. 2011; 40:222–27. doi: 10.1007/s12020-011-9485-y.
25. Salehi F, Scheithauer BW, Kros JM, Lau Q, Fealey M, Erickson D, Kovacs K, Horvath E, Lloyd RV. MGMT promoter methylation and immunoexpression in aggressive pituitary adenomas and carcinomas. J Neurooncol. 2011; 104:647–57. doi: 10.1007/s11060-011-0532-6.
26. Salehi F, Scheithauer BW, Kovacs K, Horvath E, Syro LV, Sharma S, Manoranjan B, Cusimano M. O-6-methylguanine-DNA methyltransferase (MGMT) immunohistochemical expression in pituitary corticotroph adenomas. Neurosurgery. 2012; 70:491–96. doi: 10.1227/NEU.0b013e318230ac63.
27. McCormack A, Kaplan W, Gill AJ, Little N, Cook R, Robinson B, Clifton-Bligh R. MGMT expression and pituitary tumours: relationship to tumour biology. Pituitary. 2013; 16:208–19. doi: 10.1007/s11102-012-0406-8.
28. Jiang XB, Hu B, Fan X, He DS, Mao ZG, Chen M, Zhu YH, Wang HJ. [The expression of MGMT in pituitary adenomas and its association with tumour aggressiveness]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2013; 44:462–65. Article in Chinese.
29. Heaney A. Management of aggressive pituitary adenomas and pituitary carcinomas. J Neurooncol. 2014; 117:459–68. doi: 10.1007/s11060-014-1413-6.
30. Hornyak M, Couldwell WT. Multimodality treatment for invasive pituitary adenomas. Postgrad Med. 2009; 121:168–76. doi: 10.3810/pgm.2009.03.1989.
31. Buchfelder M. Management of aggressive pituitary adenomas: current treatment strategies. Pituitary. 2009; 12:256–60. doi: 10.1007/s11102-008-0153-z.
32. Erickson D, Scheithauer B, Atkinson J, Horvath E, Kovacs K, Lloyd RV, Young WF Jr. Silent subtype 3 pituitary adenoma: a clinicopathologic analysis of the Mayo Clinic experience. Clin Endocrinol (Oxf). 2009; 71:92–99. doi: 10.1111/j.1365-2265.2008.03514.x.
33. George DH, Scheithauer BW, Kovacs K, Horvath E, Young WF Jr, Lloyd RV, Meyer FB. Crooke’s cell adenoma of the pituitary: an aggressive variant of corticotroph adenoma. Am J Surg Pathol. 2003; 27:1330–36. doi: 10.1097/00000478-200310000-00005.
34. Steels E, Paesmans M, Berghmans T, Branle F, Lemaitre F, Mascaux C, Meert AP, Vallot F, Lafitte JJ, Sculier JP. Role of p53 as a prognostic factor for survival in lung cancer: a systematic review of the literature with a meta-analysis. Eur Respir J. 2001; 18:705–19. doi: 10.1183/09031936.01.00062201.

