INTRODUCTION
Endometriosis is a common gynecological inflam-matory disease with a prevalence of 6~10% in the general female population [1]. It can cause pelvic inflammation, adhesions, infertility and chronic pain [1]. It is estimated that annual costs of endometriosis have exceed $49 billion in the United States [2]. Despite its significant impact on the health-care system, the risk factors of endometriosis remain poorly elucidated.
A number of biologic risk factors, such as a taller height or lesser weight, have been reported to be associated with risk of endometriosis [1, 3-8]. However, most remain inconclusive as risk factors of endometriosis. During the past decades, epidemiological studies [3-7, 9-13] have sought to establish a relationship between a woman’s current body mass index (BMI) and endometriosis, but with varying results. Some studies [5, 9-13] indicated an inverse association between endometriosis and BMI, but others [3, 4, 6] did not observe such relation. Herein, we conducted a meta-analysis to quantitatively assess this association.
RESULTS
Literature search
A total of 3403 records were identified after the search of the databases. After the title and abstract screening, 3380 records which did not meet the pre-specified inclusion criteria were excluded. After the full-text screening, 12 articles [14-25] did not meet the inclusion criteria, of which nine [14-22] did not have useful estimate, two [23, 24] used childhood/early adult BMI as the exposure of interest, and one [25] was an updated study. Moreover, we identified one additional publication were after manually searching the reference lists of selected studies [8]. Finally, a total of 11 publications [3-13] were included in present meta-analysis(Figure 1).
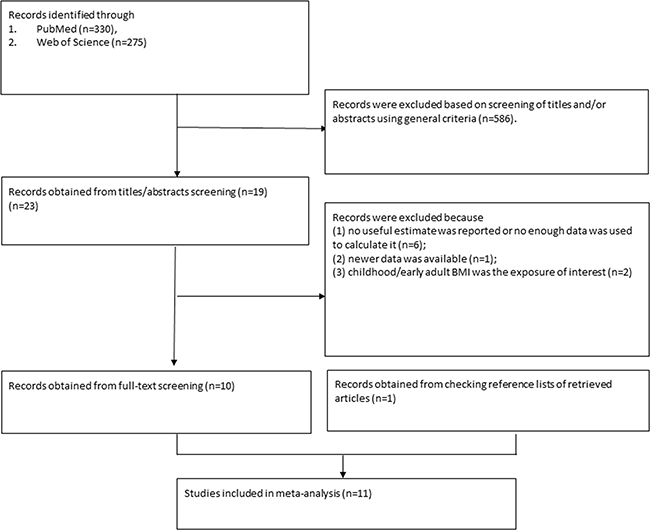
Figure 1: References searched and selection of studies in the meta-analysis.
Description of the studies
There were nine case-control studies [3, 5-9, 11-13] and two cohort studies [4, 10] included in the meta-analysis. Nine studies [3-5, 7, 9-13] reported, or provided necessary data to calculate the study-specific RRs and their corresponding 95% CIs for each 5 kg/m2 increase in BMI associated with endometriosis; five studies [6, 8-10, 12] reported the study-specific RRs and 95% CIs of endometriosis for overweight women compared with normal weight women; and three studies [8-10] reported the study-specific RRs and 95% CIs for obese women compared with normal weight women(Table 1).
Table 1: Characteristics of studies included in the meta-analysis, 2003 to 2016
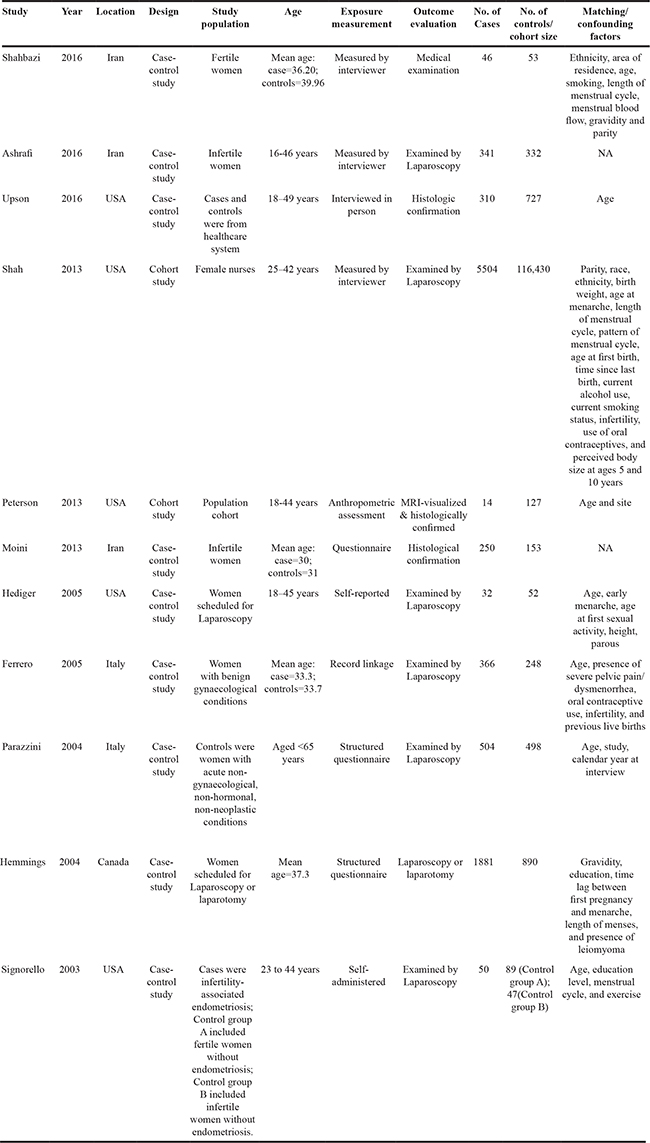
The included studies were conducted in the North America (n=6) [4, 6-8, 10, 11], Europe (n=2) [12, 13], and Asia (n=3) [3, 5, 9]. There was a total of 9,298 women diagnosed with endometriosis. All the endometriosis cases were diagnosed and confirmed by medical examination, such as examined by laparoscopy. The quality scores ranged from 5 to 8 with a median value of 7. There were six studies [7-12] that had ≥7 awarded points, which were defined as high-quality studies (Supplementary Tables 1 and 2).
Overall and subgroup results
As shown in Figure 2, the overall analysis showed a 33% reduction in the risk of endometriosis for each 5 kg/m2 increase in BMI (RR=0.67; 95% CI: 0.53, 0.84), with statistical significant heterogeneity across the studies (P <0.001, I2 =86.9%). No indication of publication bias was detected by Begg’s test (P = 0.602).
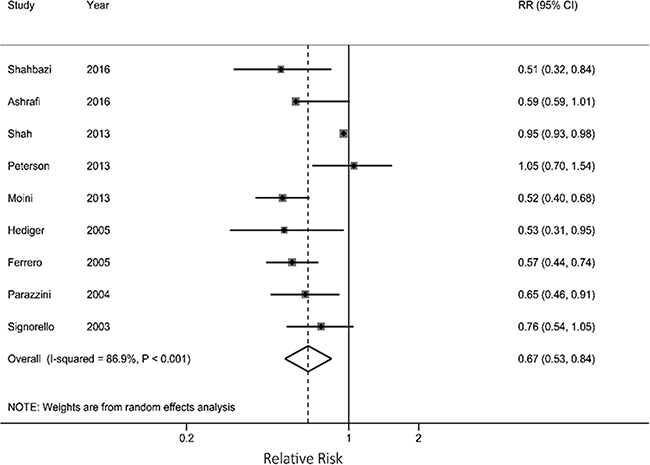
Figure 2: Results from meta-analysis for each 5 kg/m2 increase in current body mass index associated with endometriosis risk.
Compared with normal weight women, the pooled RR for obese women was 0.89 (95% CI: 0.83, 0.96), which was lower than that for overweight women (RR=0.97; 95% CI: 0.91, 1.05) (Figure 3). When we stratified by study design, the pooled RR was 0.95 (95% CI: 0.93, 0.98) for cohort studies, which was higher than that for case-control studies (RR=0.59; 95% CI: 0.52, 0.66). When we restricted the analysis in infertile women, the pooled RR was 0.68 (95% CI: 0.51, 0.89), which was lower than that for fertile women (RR=0.75; 95% CI: 0.39, 1.44). When stratified by study location, studies conducted in North America (RR=0.95; 95% CI: 0.92, 0.97), Europe (RR=0.60; 95% CI: 0.49, 0.74) and Asia (RR=0.55; 95% CI: 0.46, 0.65) all showed significantly inverse association between BMI and endometriosis risk. The strength of the associations attenuated when we pooled the studies adjusted for smoking status, length of menstrual cycle, and age at menarche (Table 2). In sensitivity analyses, we recalculated the pooled RRs by sequentially excluding one study. The eight study-specific RRs ranged from a low of 0.63 (95% CI: 0.54, 0.73) to a high of 0.69 (95% CI: 0.55, 0.87) after omission of Shah et al. and Moini et al., respectively. The results were similar when the analysis was restricted to the high-quality studies (RR=0.67; 95% CI: 0.49, 0.92).
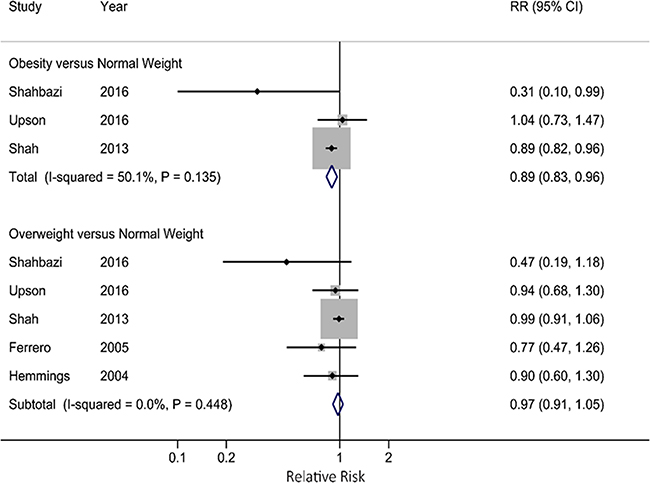
Figure 3: Forest plot for associations of obesity and overweight with endometriosis risk.
Table 2: Summary estimates and corresponding 95% confidence intervals for association of endometriosis risk with each 5 kg/m2 increase in body mass index
Subgroup by study characteristics |
No. of studies |
Pooled |
95% CI |
I2 (%) |
PH1 |
PH2 |
|---|---|---|---|---|---|---|
Overall analysis |
9 |
0.67 |
0.53, 0.84 |
86.9 |
<0.001 |
|
Study design |
<0.001 |
|||||
Case-control study |
7 |
0.59 |
0.52, 0.66 |
0 |
0.678 |
|
Cohort study |
2 |
0.95 |
0.93, 0.98 |
0 |
0.619 |
|
Study location |
0.004 |
|||||
North America |
4 |
0.95 |
0.92, 0.97 |
51.0 |
0.106 |
|
Europe |
2 |
0.60 |
0.49, 0.74 |
0 |
0.548 |
|
Asia |
3 |
0.55 |
0.46, 0.65 |
0 |
0.761 |
|
Study population |
0.595 |
|||||
Infertile women |
4 |
0.68 |
0.51, 0.89 |
83.3 |
<0.0001 |
|
Fertile women |
2 |
0.75 |
0.39, 1.44 |
86.6 |
0.006 |
|
Number of cases |
0.725 |
|||||
<250 |
4 |
0.71 |
0.51, 0.98 |
55.1 |
0.083 |
|
≥250 |
5 |
0.65 |
0.47, 0.89 |
91.9 |
<0.001 |
|
Exposure measurement |
0.175 |
|||||
Measured by interviewers |
4 |
0.76 |
0.55, 1.05 |
83.8 |
<0.001 |
|
Self-reported or others |
5 |
0.60 |
0.52, 0.69 |
0 |
0.459 |
|
Study quality |
0.857 |
|||||
High |
5 |
0.67 |
0.49, 0.92 |
85.0 |
<0.001 |
|
Low |
4 |
0.66 |
0.50, 0.86 |
65.4 |
0.034 |
|
Adjustment for potential confounding factors |
||||||
Smoking status |
0.349 |
|||||
Yes |
2 |
0.73 |
0.40, 1.33 |
84.3 |
0.012 |
|
No |
7 |
0.64 |
0.54, 0.75 |
44.5 |
0.094 |
|
Length of menstrual cycle |
0.234 |
|||||
Yes |
3 |
0.77 |
0.56, 1.06 |
75.2 |
0.018 |
|
No |
6 |
0.62 |
0.52, 0.74 |
46.4 |
0.097 |
|
Parity |
0.923 |
|||||
Yes |
4 |
0.64 |
0.43, 0.96 |
88.0 |
<0.001 |
|
No |
5 |
0.67 |
0.54, 0.84 |
58.7 |
0.046 |
|
Oral contraceptive use |
0.698 |
|||||
Yes |
3 |
0.69 |
0.44, 1.06 |
89.3 |
<0.001 |
|
No |
6 |
0.65 |
0.53, 0.80 |
52.8 |
0.006 |
|
Age at menarche |
0.224 |
|||||
Yes |
2 |
0.76 |
0.43, 1.33 |
76.0 |
0.041 |
|
No |
7 |
0.63 |
0.54, 0.75 |
46.2 |
0.084 |
|
1P value for heterogeneity within each subgroup.
2P value for heterogeneity between subgroups in meta-regression analysis.
DISCUSSION
To our knowledge, this is the first meta-analysis to assess the relationship between a woman’s current BMI and endometriosis risk. Here, we observed a significant inverse association, which suggested a possible reduced risk of endometriosis for women with higher BMI. The combined estimate was robust across sensitivity analyses.
The natural history of endometriosis remains unknown. Previous study reported that women with more advanced stage of endometriosis trend to have an even lower BMI compared with women with milder disease [11]. Of note, two studies reported an inverse association between childhood body size and risk of endometriosis [23, 24], which was in agreement with the observed association of adult BMI and endometriosis risk. Further elucidation of the role of body size over critical windows may be helpful to better understand the disease.
Several important issues should be taken into further considerations in this study. First, regarding the utilization of BMI, most genetic and molecular effects upon body weight are likely to become obscured. It is argued that clinical categories of the BMI may not provide enough etiological information to reflect the nature of obesity. Other new concept such as “adiposopathy” which was defined as adipose tissue dysfunction will have to be included in any future studies using BMI to associate to endometriosis. Second, it has been suggested that the inverse relationship between obesity and endometriosis is due to diagnostic bias. Obese women with pelvic pain may be less likely to be suggested with an operative intervention, which may subsequently lower the possibility of a laparoscopic diagnosis of endometriosis.
Some limitations should also be taken into considerations. First, residual confounding inherent in the original studies may distort the association between BMI and endometriosis. However, major potential confounders, such as smoking, parity, age at menarche and length of menstrual cycle, were adjusted in most of the original studies. Second, misclassification of BMI may occur, especially for studies with a self-reported height and weight. However, subgroup analysis stratified by exposure measurement method showed similar results between studies with height and weight measured by interviewers and by self-reported. Third, there is a significant heterogeneity across the studies. The observed heterogeneity may originate from various sources, such as different study designs and different study populations. Fourth, although publication bias was not observed in present meta-analysis, the statistical power for publication bias test was lower when the number of studies was limited.
In conclusion, this study suggested that higher BMI may be associated with lower risk of endometriosis. Large cohort studies are needed to confirm this inverse association. Further studies are also need to unveil the underlying biologic mechanism concerning the development of endometriosis.
MATERIALS AND METHODS
Study selection
We followed standard criteria for conducting and reporting of meta-analyses of observational studies [26]. We performed a computerized literature search through November 2016 using the following key words in PubMed and Web of Knowledge: (“body mass index” OR “obesity” OR “overweight” OR “underweight” OR “weight” OR “anthropometric” OR “body size” OR “body figure”) AND (“endometriosis”). The identified publications were reviewed independently for their relevance to the research topic by two authors. We also manually searched the reference lists of relevant publications to identify additional studies. A set of pre-specified inclusion criteria was applied during the review, and discrepancies were resolved by consensus. To be included in the meta-analysis, studies had to: 1) report current BMI as the exposure of interest, 2) report endometriosis as the outcome of interest, 3) use a cohort, case-cohort or case-control design, 4) provide estimates of relative risk (RR), hazard ratio, or odds ratio with confidence intervals (CIs) or standard errors or the data necessary to calculate these.
Because endometriosis is a rare outcome in general female population, the odds ratio in a case-control study is approximate equal to a rate ratio, hazard ratio or relative risk in a cohort study. Thus, we used the relative risk to measure the association between BMI and endometriosis risk. If multiple estimates were provided, priority was given to the multivariable-adjusted risk estimates that were adjusted for the most potential confounding factors in original studies. If more than one study was conducted in the same population, the most recent report was selected for our analysis.
Data extraction
We used a standardized reporting form to abstract the following data from each publication: the first author’s name, the year of publication, the country in which the study was conducted, the age of the study population, the size of the cohort or number of controls, the number of cases, the assessment method of current BMI, the categories of BMI, the ascertainment of the endometriosis and the RRs and 95% CIs for endometriosis risk associated with those categories, and the potential confounders which were adjusted in multivariable models.
Quality assessment
To assess study quality, a 9-point system on the basis of the Newcastle-Ottawa Scale [27] was used in which a study was judged on 3 broad categories for case-control studies and cohort studies as follows: the selection of study groups, comparability of groups, and ascertainment of either the exposure or outcome of interest. The high-quality study was defined as one with ≥7 points which is a median value of all the included studies.
Statistical analysis
Based on the World Health Organization classification, BMI was categorized into underweight (<18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25–29.9 kg/m2) and obese (≥30 kg/m2). To examine the association between BMI and endometriosis risk, we pooled the study-specific RR for each 5 kg/m2 increase in current BMI. For the studies in which only categorical results were reported, we used the method proposed by Greenland and Longnecker [28] and Orsini et al. [29] to calculate the trend in RR per 5 kg/m2 increase in current BMI. We also calculated the pooled RRs of endometriosis for obese or overweight women compared with normal weight women. We used a fixed effect model to pool the study specific estimates unless significant heterogeneity was observed, then the random effect model proposed by DerSimonian and Laird was used [30].
For further assessing the association of BMI with endometriosis risk, we conducted analyses stratified by study design, study location, exposure assessment method, number of cases, and study quality. We also conducted subgroup analyses stratified by whether the studies adjusted for potentially important confounders or important risk factors, including smoking status, length of menstrual cycle, parity, oral contraceptive use, and age at menarche. In addition, we performed a sensitivity analysis of the influence of individual studies on the summary estimate by repeating the meta-analysis excluding one study at a time.
Heterogeneity among studies was assessed with the Q and I2 statistics, and results were defined as heterogeneous for a P value < 0.10 or an I2 > 50% [31]. Publication bias were evaluated by visual inspection of funnel plot and formal testing by using Begg’s tests [32].
Statistical analyses were conducted using Stata, version 14.0 (StataCorp LP, College Station, Texas). Two-sided P values <0.05 were considered statistically significant.
ACKNOWLEDGMENTS
None.
CONFLICTS OF INTEREST
All the authors declare that they have no conflicts of interest.
GRANT SUPPORT
None.
REFERENCES
1. Giudice LC, Kao LC. Endometriosis. Lancet. 2004; 364: 1789-99.
2. Simoens S, Dunselman G, Dirksen C, Hummelshoj L, Bokor A, Brandes I, Brodszky V, Canis M, Colombo GL, DeLeire T, Falcone T, Graham B, Halis G, et al. The burden of endometriosis: costs and quality of life of women with endometriosis and treated in referral centres. Hum Reprod. 2012; 27: 1292-9.
3. Ashrafi M, Sadatmahalleh SJ, Akhoond MR, Talebi M. Evaluation of risk factors associated with endometriosis in infertile women. Int J Fertil Steril. 2016; 10: 11-21.
4. Peterson CM, Johnstone EB, Hammoud AO, Stanford JB, Varner MW, Kennedy A, Chen Z, Sun L, Fujimoto VY, Hediger ML, Buck Louis GM; ENDO Study Working Group. Risk factors associated with endometriosis: importance of study population for characterizing disease in the ENDO Study. Am J Obstet Gynecol. 2013; 208: 451. e1-11.
5. Moini A, Malekzadeh F, Amirchaghmaghi E, Kashfi F, Akhoond MR, Saei M, Mirbolok MH. Risk factors associated with endometriosis among infertile Iranian women. Arch Med Sci. 2013; 9: 506-14.
6. Hemmings R, Rivard M, Olive DL, Poliquin-Fleury J, Gagne D, Hugo P, Gosselin D. Evaluation of risk factors associated with endometriosis. Fertil Steril. 2004; 81: 1513-21.
7. Signorello LB, Harlow BL, Cramer DW, Spiegelman D, Hill JA. Epidemiologic determinants of endometriosis: a hospital-based case-control study. Ann Epidemiol. 1997; 7: 267-741.
8. Upson K, Sathyanarayana S, Scholes D, Holt VL. Early-life factors and endometriosis risk. Fertil Steril. 2015; 104: 964-71. e5.
9. Shahbazi S, Shahrabi-Farahani M. Evaluation of the correlation between body mass index and endometriosis among Iranian fertile women. Gynecol Endocrinol. 2016; 32: 157-60.
10. Shah DK, Correia KF, Vitonis AF, Missmer SA. Body size and endometriosis: results from 20 years of follow-up within the Nurses’ Health Study II prospective cohort. Hum Reprod. 2013; 28: 1783-92.
11. Hediger ML, Hartnett HJ, Louis GM. Association of endometriosis with body size and figure. Fertil Steril. 2005; 84: 1366-74.
12. Ferrero S, Anserini P, Remorgida V, Ragni N. Body mass index in endometriosis. Eur J Obstet Gynecol Reprod Biol. 2005; 121: 94-8.
13. Parazzini F, Chiaffarino F, Surace M, Chatenoud L, Cipriani S, Chiantera V, Benzi G, Fedele L. Selected food intake and risk of endometriosis. Hum Reprod. 2004; 19: 1755-9.
14. Cramer DW, Wilson E, Stillman RJ, Berger MJ, Belisle S, Schiff I, Albrecht B, Gibson M, Stadel BV, Schoenbaum SC. The relation of endometriosis to menstrual characteristics, smoking, and exercise. JAMA. 1986; 255: 1904-8.
15. Lafay Pillet MC, Schneider A, Borghese B, Santulli P, Souza C, Streuli I, de Ziegler D, Chapron C. Deep infiltrating endometriosis is associated with markedly lower body mass index: a 476 case-control study. Hum Reprod. 2012; 27: 265-72.
16. Mamdouh HM, Mortada MM, Kharboush IF, Abd-Elateef HA. Epidemiologic determinants of endometriosis among Egyptian women: a hospital-based case-control study. J Egypt Public Health Assoc. 2011; 86: 21-6.
17. Burghaus S, Klingsiek P, Fasching PA, Engel A, Haberle L, Strissel PL, Schmidt M, Jonas K, Strehl JD, Hartmann A, Lermann J, Boosz A, Thiel FC, et al. Risk factors for endometriosis in a German case-control study. Geburtshilfe Frauenheilkd. 2011; 71: 1073-9.
18. McCann SE, Freudenheim JL, Darrow SL, Batt RE, Zielezny MA. Endometriosis and body fat distribution. Obstet Gynecol. 1993; 82: 545-9.
19. Darrow SL, Vena JE, Batt RE, Zielezny MA, Michalek AM, Selman S. Menstrual cycle characteristics and the risk of endometriosis. Epidemiology. 1993; 4: 135-42.
20. Yi KW, Shin JH, Park MS, Kim T, Kim SH, Hur JY. Association of body mass index with severity of endometriosis in Korean women. Int J Gynaecol Obstet. 2009; 105: 39-42.
21. Matalliotakis IM, Cakmak H, Fragouli YG, Goumenou AG, Mahutte NG, Arici A. Epidemiological characteristics in women with and without endometriosis in the Yale series. Arch Gynecol Obstet. 2008; 277: 389-93.
22. McLeod BS, Retzloff MG. Epidemiology of endometriosis: an assessment of risk factors. Clin Obstet Gynecol. 2010; 53: 389-96.
23. Vitonis AF, Baer HJ, Hankinson SE, Laufer MR, Missmer SA. A prospective study of body size during childhood and early adulthood and the incidence of endometriosis. Hum Reprod. 2010; 25: 1325-34.
24. Nagle CM, Bell TA, Purdie DM, Treloar SA, Olsen CM, Grover S, Green AC. Relative weight at ages 10 and 16 years and risk of endometriosis: a case-control analysis. Hum Reprod. 2009; 24: 1501-6.
25. Missmer SA, Hankinson SE, Spiegelman D, Barbieri RL, Marshall LM, Hunter DJ. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am J Epidemiol. 2004; 160: 784-96.
26. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000; 283: 2008-12.
27. Wells G, Shea B, O’connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. 2000.
28. Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992; 135: 1301-9.
29. Orsini N, Li R, Wolk A, Khudyakov P, Spiegelman D. Meta-analysis for linear and nonlinear dose-response relations: examples, an evaluation of approximations, and software. Am J Epidemiol. 2012; 175: 66-73.
30. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986; 7: 177-88.
31. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002; 21: 1539-58.
32. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50: 1088-101.

