INTRODUCTION
Gastric cancer was the fourth most common cancer worldwide and is the second most common cause of death from cancer, with an estimated 951,600 new cases and 723,100 deaths in 2012 [1]. Up to now, the etiology of gastric cancer is still unclear, although multiple factors are thought to play a role in gastric carcinogenesis, including Helicobacter pylori (H. pylori) infection [2], nutrition deficiency, high intake of various traditional salt-preserved foods or salt and chemical carcinogenesis existing in tobacco [3, 4]. However, even when exposed to similar exogenous risk factors, only a subset of individuals will develop gastric cancer, suggesting endogenous genetic variation may also contribute to individual susceptibility to gastric cancer.
The p53 pathway has been shown to be crucial in preventing tumor formation, and the disruption of p53 function commonly leads to the initiation or progression of tumors [5]. The murine double minute protein MDM2 is an established regulator of p53, which can directly bind to p53 protein, inhibit its activity and lead to its degradation via the ubiquitination pathway [6]. As a structural homolog of MDM2, MDM4 has recently emerged as another p53-interacting protein, which directly binds to the p53 transactivation domain, inhibits its transcriptional activity, and thus contributes to tumor formation and progression [7]. Additionally, MDM4 can also interact with MDM2 protein via the RING finger domain and inhibit degradation of the MDM2 protein, regulating the role of MDM2 in inhibiting the p53 activity [7, 8].
The central role of MDM4 in regulating p53 activity and human cancer has been highlighted by many studies. For example, mouse knock-out studies showed that the Mdm4-knockout mice results in p53-dependent embryonic lethality with defects in proliferation and no apoptosis, which were rescued by knocking out the p53 gene, suggesting the biological role of the p53–MDM2–MDM4 interaction and the major function of these molecules during embryonic development [9–11]. Furthermore, the amplification or over-expression of the human MDM4 gene has been observed in a large subset of human tumors, including glioma, stomach, soft tissue sarcoma, head and neck squamous carcinoma, retinoblastoma, melanoma, and breast cancer [12–14]. There is also evidence that over-expression of MDM4 was associated with not only tumor progression but also worse prognosis [13, 15].
Since the p53-MDM4 pathway plays a critical role in response to DNA damage and preventing cancer pathogenesis, we hypothesized that common variants of MDM4 might be associated with gastric cancer risk. Previous studies have investigated three common tagging SNPs (rs11801299 G>A and rs1380576 C>G in 3’-untranslated region [3’-UTR] and rs10900598 G>T in 5′-UTR) of the MDM4 gene with risk of oral cancer, squamous cell carcinoma of oropharynx and squamous cell carcinoma of the head and neck and got some positive findings [16–18]. Recently, it was reported that MDM4 rs1380576 was not associated with gastric cancer risk in a hospital-based Chinese population with a relatively small sample size (642 cases and 720 cancer-free controls) [19]. Here, we reported a relatively large hospital-based case-control study of 1,077 gastric cancer patients and 1,173 cancer-free controls in an Eastern Chinese population to evaluate associations between three common tagging SNPs of MDM4 and gastric cancer risk. To provide additional mechanistic support for the findings, we performed SNP-mRNA expression correlation analysis to unravel the underlying molecular mechanisms.
RESULTS
Characteristics of the study population
The frequency distributions of selected variables between GCa cases and controls are described in Table 1. There was no significant difference in the distributions of age and sex between the cases and the controls (P = 0.733 and P =0.161, respectively) because of frequency matching. Compared with the cases, the controls were more likely to be smokers and drinkers, but these variables (i.e., age, sex, smoking status and drinking status) were further adjusted for in the subsequent multivariate logistic regression analyses. Of the cases, 295 (27.4%) had gastric cardia adenocarcinoma (GCA), and 782 (72.6%) had gastric non-cardia adenocarcinoma (NGCA).
Table 1: Frequency distribution of demographic characteristics of gastric cancer cases and cancer-free controls
Variables |
Cases No. (%) |
Controls No. (%) |
P a |
|---|---|---|---|
All subjects |
1,077 (100.0) |
1,173 (100.0) |
|
Age, yr |
0.733 |
||
Range |
21-86 |
22-86 |
|
Meanb |
58.6 ±11.30 |
58.8 ±11.7 |
|
≤ 50 |
225 (20.9) |
259 (22.1) |
|
51-60 |
366 (34.0) |
378 (32.2) |
|
61-70 |
328 (30.4) |
371 (31.6) |
|
>70 |
158 (14.7) |
165 (14.1) |
|
Sex |
0.161 |
||
Males |
771 (71.6) |
808 (68.9) |
|
Females |
306 (28.4) |
365 (31.1) |
|
Smoking status |
<0.0001 |
||
Never |
653 (60.6) |
596 (50.8) |
|
Ever |
424 (39.4) |
577 (49.2) |
|
Drinking status |
0.015 |
||
Yes |
258 (24.0) |
334 (28.5) |
|
No |
819 (76.0) |
839 (71.5) |
|
Pack-years |
<0.0001 |
||
0 |
653 (60.6) |
596 (50.8) |
|
≤ 25 (mean) |
220 (20.4) |
344 (29.3) |
|
> 25 (mean) |
204 (18.9) |
233 (19.9) |
|
Tumor site |
|||
GCA |
295 (27.4) |
— |
|
NGCA |
782 (72.6) |
— |
GCA, gastric cardia adenocarcinoma; NGCA, non-gastric cardia adenocarcinoma.
a Two-sided χ2 test for distributions between cases and controls.
b Data are mean ± SD.
Associations between MDM4 genotypes and risk of gastric cancer
The genotype distributions of the three SNPs among the cases and controls and their associations with gastric cancer risk are summarized in Table 2. The genotype frequencies among the controls were in agreement with the Hardy-Weinberg equilibrium (all P > 0.05). Compared with the CC/CG genotype carriers under the recessive genetic model, the rs1380576 variant GG genotype carriers had significantly decreased risk of gastric cancer (adjusted OR = 0.74, 95% CI = 0.56–0.98). Finally, we performed a mini-meta analysis of rs1380576 with our and another published study (Figure 1). Consistently, we found that rs1380576 in a recessive model was associated with a significantly decreased risk of gastric cancer (the pooled OR= 0.81; 95% CI = 0.68–0.97 for GG vs. CC/CG) based on 1719 cases and 1893 controls in the pooled analysis.
Table 2: Logistic regression analysis of associations between the genotypes of MDM4 and gastric cancer risk
Variants |
Genotypes |
Cases |
Controls |
P a |
Crude OR |
P |
Adjusted OR |
P b |
|---|---|---|---|---|---|---|---|---|
rs10900598 |
||||||||
GG |
547 (50.8) |
604 (51.5) |
0.366 |
1.00 |
1.00 |
|||
GT |
447 (41.5) |
462 (39.4) |
1.07 (0.90-1.27) |
0.457 |
1.08 (0.90-1.28) |
0.409 |
||
TT |
83 (7.7) |
107 (9.1) |
0.86 (0.63-1.17) |
0.326 |
0.87 (0.64-1.19) |
0.379 |
||
GG/GT |
994 (92.3) |
1066 (90.1) |
1.00 |
1.00 |
||||
TT |
83 (7.7) |
107 (9.1) |
0.83 (0.62-1.12) |
0.228 |
0.84 (0.62-1.14) |
0.263 |
||
rs11801299 |
||||||||
GG |
380 (35.3) |
449 (38.3) |
0.657 |
1.00 |
1.00 |
|||
GA |
539 (50.1) |
532 (45.4) |
1.20 (1.00-1.44) |
0.052 |
1.21 (1.01-1.45) |
0.043 |
||
AA |
158 (14.7) |
192 (16.4) |
0.97 (0.76-1.25) |
0.827 |
0.96 (0.75-1.24) |
0.770 |
||
GG/GA |
919 (85.3) |
981 (83.6) |
1.00 |
1.00 |
||||
AA |
158 (14.7) |
192 (16.4) |
0.88 (0.70-1.11) |
0.269 |
1.03 (0.91-1.16) |
0.684 |
||
rs1380576 |
||||||||
CC |
487 (45.2) |
552 (47.1) |
0.044 |
1.00 |
1.00 |
|||
CG |
493 (45.8) |
485 (41.4) |
1.15 (0.97-1.37) |
0.112 |
1.15 (0.97-1.37) |
0.114 |
||
GG |
97 (9.0) |
136 (11.6) |
0.81 (0.61-1.08) |
0.147 |
0.79 (0.59-1.06) |
0.117 |
||
CC/CG |
980 (91.0) |
1037 (88.4) |
1.00 |
1.00 |
||||
GG |
97 (9.0) |
136 (11.6) |
0.76 (0.57-0.99) |
0.045 |
0.74 (0.56-0.98) |
0.034 |
||
CI,confidence interval; OR, odds ratio.
a Chi square test for genotype distributions between cases and controls.
b Adjusted for age, sex, smoking and drinking status in logistic regress models.
The results were in bold, if the 95% CI excluded 1 and P < 0.05.
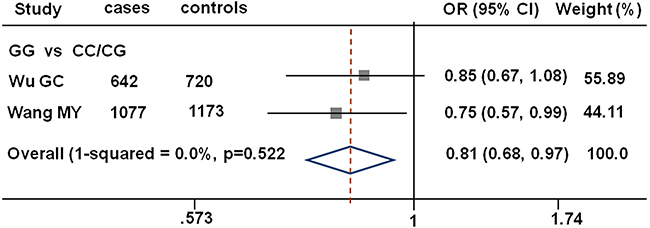
Figure 1: Forest plot showing associations between MDM4 rs1380576 and gastric cancer risk. The ORs and 95% CIs were obtained using GG vs. CC/CG. The axis corresponds to the OR. The diamonds and the horizontal bars represent the overall ORs with 95% CIs given by their width. CI,confidence interval; OR, odds ratio.
Stratification and haplotype analysis
In stratification analyses, as shown in Table 3, by assuming a recessive genetic model, we found that the significantly decreased risk associated with rs1380576 GG variant genotype was more evident in subgroups of males (adjusted OR = 0.72, 95% CI = 0.52-0.99), never-smokers (adjusted OR = 0.57, 95% CI = 0.39-0.84) and subjects with non-cardia cancer (adjusted OR = 0.68, 95% CI = 0.50-0.93). Likewise, we found similar results for those carrying “1-2” risk genotypes among subgroups of older subjects (defined as subjects > 59 years old), males, never-smokers, never-drinkers and subjects with non-cardia cancer.
Table 3: Stratification analysis for associations between rs1380576 G>C variant genotypes and gastric cancer risk
Variables |
Cases/Controls |
Crude OR |
P |
Adjusted OR |
P a |
Phom |
|
|---|---|---|---|---|---|---|---|
CG/GG |
CC |
||||||
Age |
|||||||
≤59 |
507/525 |
46/68 |
0.70 (0.47-1.04) |
0.076 |
0.68 (0.46-1.01) |
0.057 |
0.600 |
>5 |
473/512 |
51/68 |
0.81 (0.55-1.19) |
0.287 |
0.82 (0.55-1.21) |
0.307 |
|
Sex |
|||||||
Females |
283/331 |
23/34 |
0.79 (0.46-1.38) |
0.406 |
0.81 (0.46-1.41) |
0.454 |
0.820 |
Males |
697/706 |
74/102 |
0.74 (0.54-1.01) |
0.057 |
0.72 (0.52-0.99) |
0.044 |
|
Smoking status |
|||||||
Never |
602/524 |
51/72 |
0.62 (0.42-0.90) |
0.012 |
0.57 (0.39-0.84) |
0.005 |
0.103 |
Ever |
378/513 |
46/64 |
0.98 (0.65-1.46) |
0.904 |
0.99 (0.66-1.48) |
0.948 |
|
Drinking status |
|||||||
Never |
745/741 |
74/98 |
0.75 (0.55-1.03) |
0.078 |
0.73 (0.53-1.01) |
0.055 |
0.963 |
Ever |
235/296 |
23/38 |
0.76 (0.44-1.32) |
0.323 |
0.77 (0.45-1.33) |
0.353 |
|
Site |
|||||||
Cardia |
262/1037 |
33/136 |
0.96 (0.64-1.44) |
0.846 |
0.89 (0.59-1.34) |
0.573 |
0.184 |
Non-cardia |
718/1037 |
64/136 |
0.68 (0.50-0.93) |
0.015 |
0.68 (0.50-0.93) |
0.016 |
|
CI, confidence interval; OR, odds ratio.
a Obtained in logistic regression models with adjustment for age, sex, smoking status and drinking status.
Phom derived from the homogeneity test.
The results were in bold, if the 95% CI excluded 1 or P < 0.05.
By using the SAS PROC HAPLOTYPE program, we inferred all the possible haplotypes based on the observed genotype data, of which three common (>10%) haplotypes (GAC, GGG, and TGC) represented 99.4% of all haplotypes for the cases and 97.3% for the controls (Table 4). When the most common haplotype GAC was used as the reference, none of the haplotypes were associated with a significant risk of gastric cancer in logistic regression models, either in a univariate model or a multivariate model with adjustment for age, sex, smoking and alcohol use.
Table 4: Haplotype analysis for genotypes of MDM4 and Gastric Cancer risk
Haplotypesa |
Haplotype frequencies |
Crude OR |
P |
Adjusted OR |
P a |
|||
|---|---|---|---|---|---|---|---|---|
Cases |
Controls |
|||||||
N |
% |
N |
% |
|||||
G-A-C |
848 |
48.8 |
889 |
51.2 |
1.00 |
1.00 |
||
G-G-G |
684 |
48.4 |
730 |
51.6 |
1.01 (0.88-1.16) |
0.933 |
1.01 (0.87-1.16) |
0.945 |
T-G-C |
608 |
48.3 |
652 |
51.8 |
1.00 (0.87-1.16) |
0.987 |
1.01 (0.87-1.17) |
0.889 |
a Obtained in logistic regression models with adjustment for age, sex, smoking status and drinking status. CI, confidence interval; OR, odds ratio.
Genotype-phenotype correlation analysis
In addition, we also performed the genotype-phenotype correlation analysis for the MDM4 rs1380576 C>G SNP by using publically available genotyping data and mRNA expression data of MDM4 from the 270 lymphoblastoid cell lines. We found that the GG genotype, compared with the CC genotype, appeared to be correlated with significantly decreased mRNA expression, which is consistent for Asians (n = 89, P = 0.005), Chinese (n = 44, P = 0.032), and all populations (n = 269, P <0.0001) (Figure 2). We searched the GTEx database (http://www.gtexportal.org/home) [20] and performed eQTL analysis of rs1380576 for the stomach tissue, and we found that the GG genotype was associated with an increased mRNA expression level of MDM4, compared with the CC genotype (Figure 2). However, the eQTL result of the allelic effect in this database was in the different direction from the results from lymphoblastoid cell lines.
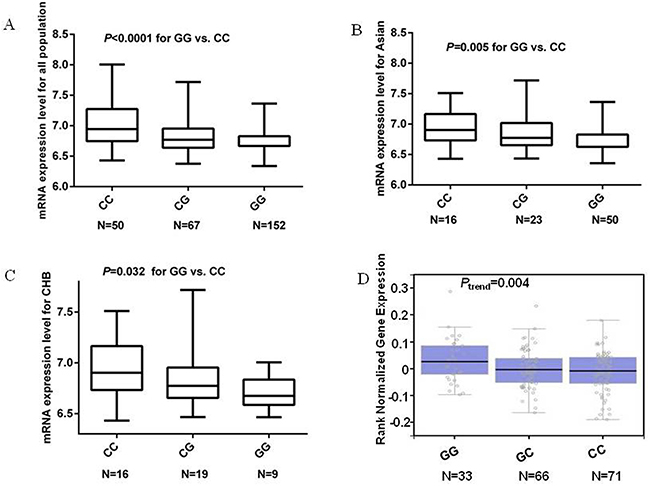
Figure 2: mRNA expression level of the MDM4 gene in EBV-transformed lymphoblastoid cell lines. A. mRNA expression in 269 HapMap cell lines. B. mRNA expression in 89 HapMap cell lines of unrelated Asian. C. mRNA expression in 44 HapMap cell lines of unrelated Han Chinese in Beijing, China. D. eQTL results from the GTEx for rs1380576 in stomach tissue.
DISCUSSION
We investigated the associations of three tagging SNPs of MDM4 with risk of gastric cancer in this large, ethnic-specific single institutional case-control study. We found a significant association of the rs1380576 variant GG genotype in MDM4 with a decreased gastric cancer risk under a recessive genetic model, especially among subgroups of males, never-smokers and subjects with non-cardia cancer. Given the role of MDM4 in activating gene expression and influencing the p53 activity, it is biologically plausible that the MDM4 SNPs may modulate risk of gastric cancer.
MDM4, located on chromosome1q32, is a member of the mouse double minute oncoprotein family, which includes the full-length MDM2, MDM4 and their derivate minor forms. MDM4 was initially described as an MDM2 homologue with high similarity with the primary structural level [7, 21]. However, unlike MDM2, MDM4 does not have appreciable ubiquitin ligase activity. Careful mouse genetic studies indicate that MDM4 contributes more to inhibition of p53-mediated transcriptional transactivation while MDM2 contributes more to degradation of p53 [22]. Additionally, it has been shown that through their RING domains, MDM4 binds MDM2 and enhances the ability of MDM2 to regulate p53. Although MDM4 inhibits p53 function, homozygous Myc-tagged MDM4 transgene expression was embryonic lethal, and this could not be rescued with deletion of p53, suggesting a p53-independent function of MDM4 in development. Currently, the development of molecules that block p53-MDM2/MDM4 interactions is considered a promising strategy to combat cancers that contain inactive wild-type p53.
In the present study, the rs1380576 GG variant genotype in MDM4 was found to be associated with a decreased gastric cancer risk under a recessive genetic model, which only showed some trend but did not reach statistically significance in another Chinese study. The discrepancy may be due to the different population selected. Our patients and controls came from Eastern China, while in another Chinese study, patients and controls came from North of China. The second reason may be due to the different sample size. In the stratification analysis, we found that risk effect of the rs1380576 variant GG genotype was more obvious in subgroups of older subjects, males, never smokers, never drinkers and cancers of non-cardia. This is consistent with the notion that susceptible individuals are likely to have had a light exposure. Never smokers and never drinkers were defined as those who may have exposed to low levels of such exposures as a result of being exposed to passive smoking or other unknown carcinogens in the environment, and, therefore, genetic variation may play a major role in carcinogenesis in such subgroups. Variation by sex may reflect disparate acquisition of risk factors such as H pylori infection and Barrett’s esophagus [23, 24]. Furthermore, variation by sex or a late age onset of gastric cancer in women compared with that in men may also reflect a protective effect of estrogen in women [25, 26]. Gastric cancer can generally be classified into two categories: cardia gastric cancer (CGC) arising in the area of the stomach adjoining the esophageal-gastric junction, and non-CGC (NCGC) arising from more distal regions of the stomach. Several investigators have suggested that cardia gastric cancer may have distinct risk factors, clinical features and biological behavior compared with non-CGC [22, 27, 28]. Risk factors for CGC include obesity, GORD and Barrett’s esophagus, a metaplastic condition that can result from GORD [8, 9, 29, 30]]. NCGC, however, is strongly associated with H. pylori infection [14], and the influence of socioeconomic status (SES) also differs. While indicators of low SES such as household crowding, low income, low education and increased number of siblings are positively associated with NCGC, they do not appear to be associated with CGC [16, 17]. In the present study, the combined risk genotypes were more evident in patients with NCGC, which may be due to different biological entities and tumor site-specific etiologies. It may also be due to the relatively small sample size in each subgroup, and, therefore, larger studies with a more stringent design are needed to validate our findings.
To further identify molecular mechanisms underlying our findings, we performed genotype-phenotype correlation analysis of mRNA expression levels for MDM4 by using publically avaialble genotyping and mRNA expression data. The observed significantly decreased expression of the MDM4 gene associated with the rs1380576 GG variant genotype may have lead gastric cancer susceptibility in this study population. The eQTL results of the allelic effect from GTEx with stomach tissue were in the different direction from the results of lymphoblastoid cell lines, which may be due to the tissue specificity. Therefore, other studies with larger numbers of gastric tissues are needed to validate our findings. As rs1380576 is a tag SNP, this SNP is in fact in LD with several other potential functional SNPs in MDM4 that may affect the protein function at the translational level. Thus, fine mapping of this gene will be necessary to identify additional causal variants in the future.
In summary, the present study investigated the associations between three MDM4 tagging SNPs and gastric cancer risk with a relatively large sample size. Several limitations of our study need to be addressed. Firstly, this hospital-based case-control study may have selection bias and information bias, which may be minimized by frequency-matching cases and controls as well as the adjustment for potential confounding factors in the final analyses. Second, only three tagging SNPs of MDM4 were investigated in the present study, which did not cover all SNPs and may have missed some important variants within MDM4. Third, other risk factors, especially the H. pylori infection status, were not available for further analysis due to the nature of the retrospective study design. This limitation should be overcome in our future studies. Fourth, we were not able to measure the expression of MDM4 in mRNA and protein levels due to the lack of clinic tissues/samples. Finally, although our sample size was relatively large, the small sample size in subgroup analyses may have limited statistical power. Hence, our findings need to be confirmed by studies with larger sample sizes. Despite these limitations, our findings are biologically plausible and provide some novel clues for the role of MDM4 in the development of gastric cancer.
MATERIALS AND METHODS
Study population
Patients with newly diagnosed, histopathologically confirmed, and untreated primary gastric adenocarcinoma were recruited between January 2009 and March 2011 at Fudan University Shanghai Cancer Center. All patients came from Eastern China, including Shanghai City, Jiangsu Province, Zhejiang Province and the surrounding regions. Participants who had gastric adenosquamous, squamous cell carcinoma, neuroendocrine tumor, stromal tumor, metastasized cancer from other organs or any histopathologic diagnosis other than gastric adenocarcinoma were excluded from this study. Cancer-free controls were recruited from a large prospective cohort recruited for Taizhou longitudinal study (TZL) at the same time period in the Eastern China with the selection criteria including no individual history of cancer. These cancer-free Han Chinese controls were frequency matched to cases on age (± 5 years) and sex [31]. A structured questionnaire was used to obtain the following information from each of the participants during personal interviews: age, sex, ethnicity, smoking and alcohol consumption. After interview, each participant donated a sample of approximately 10-mL blood, of which 1 mL was used for genomic DNA extraction. This research protocol was approved by the Institutional Review Board of FUSCC and the experiment on humans was performed in accordance with relevant guidelines and regulations.
SNP Genotyping
Genomic DNA extraction and genotyping were conducted as described previously [32], with a successful genotyping rate of 99.5% by using the TaqMan probe assays (Applied Biosystems, Foster City, CA, USA) on a 7900 HT sequence detector system (Applied Biosystems) according to the manufacturer’s protocol. More than 10% of the samples were retested for each polymorphism randomly, and the results were 100% concordant.
Genotype and mRNA expression data of lymphoblastoid cell lines from hapmap database
To explore the functionality of the MDM4 rs1380576 C>G SNP, we used publically available data on genotypes and transcript expression levels of MDM4 from 270 lymphoblastoid cell lines from all populations (45 Han Chinese in Beijing, China +45 Japanese in Tokyo, Japan +90 Utah residents with Northern and Western European ancestry +90 Yoruba in Ibadan, Nigeria) available online for the genotype–phenotype correlation analysis [33]. The genotyping data were from the HapMap phase II release 23 data set consisting of 3.96 million SNP genotypes from these 270 individuals.
Statistical analysis
Differences in the distributions of demographic characteristics, selected variables, and frequencies of genotypes between cases and controls were tested by the Student’s t-test (for continuous variables) or Chi-square-test (for categorical variables). Hardy-Weinberg equilibrium (HWE) was evaluated by a goodness-of-fit Chi-square test to compare the observed genotype frequencies with the expected among the controls. Univariate and multivariate logistic regression models were used to evaluate associations between genotypes and risk of gastric cancer by odds ratios (ORs) and their 95% confidence intervals (CIs) with the adjustment for possible confounders. Risk genotypes of studied SNPs were combined to create a genetic score of the number of the observed risk genotypes, and this score was used for further analyses. All statistical analyses were performed by using Statistical Analysis Software (v.9.3 SAS Institute, Cary, NC), and all P values were two-sided with a 0.05 significance level.
Abbreviations
SNPs, single nucleotide polymorphisms; GCa, gastric cancer; OR, odds ratio; CI, confidence interval; MDM4, murine double minute protein4; HWE, Hardy-Weinberg equilibrium; GCA, gastric cardia adenocarcinoma; NGCA, non-cardia adenocarcinoma.
ACKNOWLEDGMENTS
We would like to thank the tissue bank staff members for their continued support in providing the stored blood samples.
CONFLICTS OF INTEREST
No potential conflicts of interest were disclosed.
GRANT SUPPORT
This study was supported by the funds from the National Natural Science Foundation of China (81302101), China Recruitment Program of Global Experts at Fudan University, and Ministry of Health (201002007). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
REFERENCES
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J and Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87-108.
2. Hu Z, Ajani JA and Wei Q. Molecular epidemiology of gastric cancer: current status and future prospects. Gastrointest Cancer Res. 2007; 1:12-19.
3. Ladeiras-Lopes R, Pereira AK, Nogueira A, Pinheiro-Torres T, Pinto I, Santos-Pereira R and Lunet N. Smoking and gastric cancer: systematic review and meta-analysis of cohort studies. Cancer Causes Control. 2008; 19:689-701.
4. Palli D. Epidemiology of gastric cancer: an evaluation of available evidence. J Gastroenterol. 2000; 35:84-89.
5. Raia V, Schilling M, Bohm M, Hahn B, Kowarsch A, Raue A, Sticht C, Bohl S, Saile M, Moller P, Gretz N, Timmer J, Theis F, et al. Dynamic mathematical modeling of IL13-induced signaling in Hodgkin and primary mediastinal B-cell lymphoma allows prediction of therapeutic targets. Cancer Res. 2011; 71:693-704.
6. Landers JE, Cassel SL and George DL. Translational enhancement of mdm2 oncogene expression in human tumor cells containing a stabilized wild-type p53 protein. Cancer research. 1997; 57:3562-3568.
7. Shvarts A, Steegenga WT, Riteco N, van Laar T, Dekker P, Bazuine M, van Ham RC, van der Houven van Oordt W, Hateboer G, van der Eb AJ and Jochemsen AG. MDMX: a novel p53-binding protein with some functional properties of MDM2. EMBO J. 1996; 15:5349-5357.
8. Yang P, Zhou Y, Chen B, Wan HW, Jia GQ, Bai HL and Wu XT. Overweight, obesity and gastric cancer risk: results from a meta-analysis of cohort studies. Eur J Cancer. 2009; 45:2867-2873.
9. Ye W, Chow WH, Lagergren J, Yin L and Nyren O. Risk of adenocarcinomas of the esophagus and gastric cardia in patients with gastroesophageal reflux diseases and after antireflux surgery. Gastroenterology. 2001; 121:1286-1293.
10. Migliorini D, Lazzerini Denchi E, Danovi D, Jochemsen A, Capillo M, Gobbi A, Helin K, Pelicci PG and Marine JC. Mdm4 (Mdmx) regulates p53-induced growth arrest and neuronal cell death during early embryonic mouse development. Molecular and cellular biology. 2002; 22:5527-5538.
11. Parant J, Chavez-Reyes A, Little NA, Yan W, Reinke V, Jochemsen AG and Lozano G. Rescue of embryonic lethality in Mdm4-null mice by loss of Trp53 suggests a nonoverlapping pathway with MDM2 to regulate p53. Nature genetics. 2001; 29:92-95.
12. Riemenschneider MJ, Buschges R, Wolter M, Reifenberger J, Bostrom J, Kraus JA, Schlegel U and Reifenberger G. Amplification and overexpression of the MDM4 (MDMX) gene from 1q32 in a subset of malignant gliomas without TP53 mutation or MDM2 amplification. Cancer research. 1999; 59:6091-6096.
13. Bartel F, Schulz J, Bohnke A, Blumke K, Kappler M, Bache M, Schmidt H, Wurl P, Taubert H and Hauptmann S. Significance of HDMX-S (or MDM4) mRNA splice variant overexpression and HDMX gene amplification on primary soft tissue sarcoma prognosis. International journal of cancer Journal international du cancer. 2005; 117:469-475.
14. Kamangar F, Dawsey SM, Blaser MJ, Perez-Perez GI, Pietinen P, Newschaffer CJ, Abnet CC, Albanes D, Virtamo J and Taylor PR. Opposing risks of gastric cardia and noncardia gastric adenocarcinomas associated with Helicobacter pylori seropositivity. J Natl Cancer Inst. 2006; 98:1445-1452.
15. Sampieri K, Mencarelli MA, Epistolato MC, Toti P, Lazzi S, Bruttini M, De Francesco S, Longo I, Meloni I, Mari F, Acquaviva A, Hadjistilianou T, Renieri A, et al. Genomic differences between retinoma and retinoblastoma. Acta Oncol. 2008; 47:1483-1492.
16. Brewster DH, Fraser LA, McKinney PA and Black RJ. Socioeconomic status and risk of adenocarcinoma of the oesophagus and cancer of the gastric cardia in Scotland. Br J Cancer. 2000; 83:387-390.
17. de Martel C, Forman D and Plummer M. Gastric cancer: epidemiology and risk factors. Gastroenterol Clin North Am. 2013; 42:219-240.
18. Yu H, Wang LE, Liu Z, Wei S, Li G, Sturgis EM and Wei Q. Polymorphisms of MDM4 and risk of squamous cell carcinoma of the head and neck. Pharmacogenet Genomics. 2011; 21:388-396.
19. Wu GC and Zhang ZT. Genetic association of single nucleotide polymorphisms in P53 pathway with gastric cancer risk in a Chinese Han population. Med Oncol. 2015; 32:401.
20. Gibson G. Human genetics. GTEx detects genetic effects. Science. 2015; 348:640-641.
21. Shvarts A, Bazuine M, Dekker P, Ramos YF, Steegenga WT, Merckx G, van Ham RC, van der Houven van Oordt W, van der Eb AJ and Jochemsen AG. Isolation and identification of the human homolog of a new p53-binding protein, Mdmx. Genomics. 1997; 43:34-42.
22. Lehmann K and Schneider PM. Differences in the molecular biology of adenocarcinoma of the esophagus, gastric cardia, and upper gastric third. Recent Results Cancer Res. 2010; 182:65-72.
23. de Martel C and Parsonnet J. Helicobacter pylori infection and gender: a meta-analysis of population-based prevalence surveys. Dig Dis Sci. 2006; 51:2292-2301.
24. Cook MB, Wild CP and Forman D. A systematic review and meta-analysis of the sex ratio for Barrett’s esophagus, erosive reflux disease, and nonerosive reflux disease. Am J Epidemiol. 2005; 162:1050-1061.
25. Camargo MC, Goto Y, Zabaleta J, Morgan DR, Correa P and Rabkin CS. Sex hormones, hormonal interventions, and gastric cancer risk: a meta-analysis. Cancer Epidemiol Biomarkers Prev. 2012; 21:20-38.
26. Derakhshan MH, Liptrot S, Paul J, Brown IL, Morrison D and McColl KE. Oesophageal and gastric intestinal-type adenocarcinomas show the same male predominance due to a 17 year delayed development in females. Gut. 2009; 58:16-23.
27. Peleteiro B, Cavaleiro-Pinto M, Barros R, Barros H and Lunet N. Is cardia cancer aetiologically different from distal stomach cancer? Eur J Cancer Prev. 2011; 20:96-101.
28. Xue L, Zhang X, Li Y, Yang H, Li X, Mi J, Wang H, Wang J and Yan X. Differences of immunophenotypic markers and signaling molecules between adenocarcinomas of gastric cardia and distal stomach. Hum Pathol. 2011; 42:594-601.
29. Hoyo C, Cook MB, Kamangar F, Freedman ND, Whiteman DC, Bernstein L, Brown LM, Risch HA, Ye W, Sharp L, Wu AH, Ward MH, Casson AG, et al. Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: a pooled analysis from the International BEACON Consortium. Int J Epidemiol. 2012; 41:1706-1718.
30. Whiteman DC, Sadeghi S, Pandeya N, Smithers BM, Gotley DC, Bain CJ, Webb PM and Green AC. Combined effects of obesity, acid reflux and smoking on the risk of adenocarcinomas of the oesophagus. Gut. 2008; 57:173-180.
31. Wang X, Lu M, Qian J, Yang Y, Li S, Lu D, Yu S, Meng W, Ye W and Jin L. Rationales, design and recruitment of the Taizhou Longitudinal Study. BMC public health. 2009; 9:223.
32. Wang M, Zhang R, He J, Qiu L, Li J, Wang Y, Sun M, Yang Y, Wang J, Yang J, Qian J, Jin L, Ma H, et al. Potentially functional variants of PLCE1 identified by GWASs contribute to gastric adenocarcinoma susceptibility in an eastern Chinese population. PLoS One. 2012; 7:e31932.
33. Holm K, Melum E, Franke A and Karlsen TH. SNPexp - A web tool for calculating and visualizing correlation between HapMap genotypes and gene expression levels. BMC Bioinformatics. 2010; 11:600.

