INTRODUCTION
Pancreatic cancer has become the seventh leading cause of cancer mortality in the world. In 2012, 337,872 people worldwide were diagnosed with pancreatic cancer, and 330,391 people died of this cancer [1]. The prognosis for patients with pancreatic cancer remains extremely dismal, with a 5-year relative survival rate of only 7% [2]. Less than 20% of patients present with localized disease eligible for curative surgery, and local recurrence (> 20%) and distant metastasis (> 70%) frequently occur after resection. The actuarial 5-year overall survival of patient who present with localized disease is only 20–25% due to the lack of effective adjuvant treatment strategies for this malignancy [3, 4].
The relationships between diabetes and pancreatic cancer are especially complicated and intertwined. About 80% of pancreatic cancer patients have either glucose intolerance or diabetes [5]. Pancreatic cancer is thought to causes diabetes, although the mechanism is not yet completely understood. On the other hand, diabetes appears to be a risk factor for the development of pancreatic cancer [6]. Diabetes may affect treatment outcomes of patients with pancreatic cancer, although the evidence is not consistent [7–9]. Furthermore anti-diabetic medications have been reported to affect pancreatic cancer risk. Sulfonylureas are associated with an increased risk of pancreatic cancer [10]. On the other hand, metformin usage has been found to be associated with a reduced risk of pancreatic cancer [11].
Metformin (1,1-dimethylbiguanide hydrochloride), one of the most widely prescribed drugs for type 2 diabetes mellitus, has been shown to be clinically associated with antitumor effects [12, 13]. Through a number of population, epidemiologic, and cohort studies, metformin has been suggested not only to prevent development of various tumors but also to delay cancer progression in certain tumor types [14–17]. In pancreatic cancer, substantial preclinical studies of metformin support metformin’s ability to inhibit tumorigenesis and their authors have proposed potential mechanisms of the antitumor effect [18–21]. However, a few clinical studies have presented conflicting results and metformin`s effects in pancreatic cancer remain controversial [9, 21–24].
Hence, we hypothesized that the use of metformin might be associated with survival benefits for patients with resectable pancreatic cancer. We conducted a cohort study based on a nationwide population database to evaluate the effect of metformin on the clinical outcomes in patients with pre-existing diabetes and pancreatic cancer treated with curative resection.
RESULTS
Among 28,862 pancreatic cancer patients who were diagnosed between January 1, 2005 and December 31, 2011, 1,919 patients met the eligibility criteria described in Figure 1. Of them, 764 patients were prescribed oral hypoglycemic agents (OHAs) for at least 90 days and classified as the diabetic group; 530 patients received metformin for at least 90 days (Figure 1). The median age was 65 years (interquartile range, 57–70) and 57.7% of the patients were men. The metformin non-user group had more current smokers, a higher prevalence of elevated aspartate transaminase (AST) and a lower prevalence of elevated alanine transaminase (ALT) when compared with the metformin user group. There were no significant difference between the metformin user group and non-user group in terms of age, sex, alcohol drinking behavior, regular exercise, body mass index (BMI), total cholesterol, fasting blood glucose, gamma-glutamyl transpeptidase (rGT), Charlson comorbidity index, treatment methods such as radiotherapy, chemotherapy, or types of surgery (Table 1).
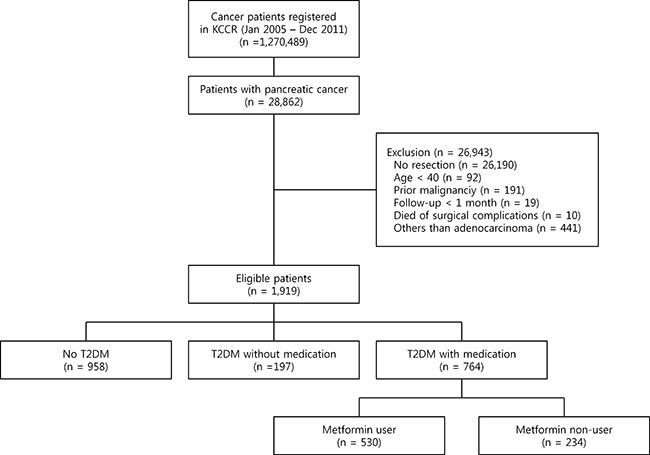
Figure 1: Flow diagram of the study. KCCR, Korea Central Cancer Registry; DM, diabetes mellitus.
Table 1: Baseline characteristics of the patients according to use of metformin as an initial pharmacotherapy
Variable |
Metformin user (n = 530) |
Metformin non-user (n = 234) |
p-value |
|
|---|---|---|---|---|
Demographic |
||||
Age (years) |
Median (Q1, Q3) |
65 (57, 70) |
65 (57, 70) |
0.9033 |
40–49 |
34 (6.4%) |
17 (7.3%) |
0.9072 |
|
50–59 |
140 (26.4%) |
60 (25.6%) |
||
60–69 |
218 (41.1%) |
97 (41.5%) |
||
70– |
138 (26.0%) |
60 (25.6%) |
||
Sex |
Male |
306 (57.7%) |
135 (57.7%) |
0.9910 |
Female |
224 (42.3%) |
99 (42.3%) |
||
*Health risk behavior |
||||
Smoking status |
Never smoker |
225 (68.0%) |
81 (60.0%) |
0.0121 |
Former smoker |
51 (15.4%) |
16 (11.9%) |
||
Current smoker |
55 (16.6%) |
38 (28.1%) |
||
Alcoholic drinking |
Yes |
14 (4.2%) |
9 (6.7%) |
0.2706 |
No |
317 (95.8%) |
126 (93.3%) |
||
Regular exercise |
Yes |
52 (15.7%) |
23 (17.0%) |
0.7236 |
No |
279 (84.3%) |
112 (83.0%) |
||
*Laboratory |
||||
Body mass index (kg/m2) |
Median (Q1, Q3) |
24.0 (21.9, 25.6) |
24.0 (21.8, 25.8) |
0.8515 |
≥ 25 |
116 (35.0%) |
48 (35.6%) |
0.6698 |
|
< 25 |
215 (65.0%) |
87 (64.4%) |
||
Total cholesterol (mg/dl) |
Median (Q1, Q3) |
188 (158, 216) |
186 (161, 217) |
0.6448 |
≥ 240 |
43 (13.0%) |
11 (8.1%) |
0.0898 |
|
< 240 |
288 (87.0%) |
124 (91.9%) |
||
Fasting blood glucose (mg/dl) |
Median (Q1, Q3) |
119 (101, 150) |
108 (96, 121) |
0.0618 |
≥ 126 |
140 (42.3%) |
30 (22.2%) |
0.8127 |
|
< 126 |
191 (57.7%) |
115 (77.8%) |
||
AST (U/L) |
Median (Q1, Q3) |
25 (20, 35) |
25 (20, 32) |
0.6586 |
≥ 51 |
36 (10.9%) |
17 (12.6%) |
0.0091 |
|
< 51 |
295 (89.1%) |
118 (87.4%) |
||
ALT (U/L) |
Median (Q1, Q3) |
24 (17, 37) |
22 (17, 32) |
0.2034 |
≥ 46 |
55 (16.6%) |
13 (9.6%) |
0.0310 |
|
< 46 |
276 (83.4%) |
122 (90.4%) |
||
rGT (U/L) |
Median (Q1, Q3) |
29 (19, 51) |
30 (20, 51) |
0.4752 |
≥ 78 (male), 46 (female) |
61 (18.4%) |
29 (21.5%) |
0.7269 |
|
< 78 (male), 46 (female) |
270 (81.6%) |
106 (78.5%) |
||
Comorbidity |
||||
Charlson comorbidity index |
Median (Q1, Q3) |
6 (5, 8) |
6 (5, 8) |
0.5528 |
0–6 |
278 (52.5%) |
119 (50.9%) |
0.7729 |
|
7–9 |
172 (32.5%) |
82 (35.0%) |
||
≥ 10 |
80 (15.1%) |
33 (14.1%) |
||
Methods of treatment |
||||
Radiotherapy |
Yes |
168 (31.7%) |
80 (34.2%) |
0.4981 |
No |
362 (68.3%) |
154 (65.8%) |
||
Chemotherapy |
Yes |
348 (65.7%) |
165 (70.5%) |
0.1881 |
No |
182 (34.3%) |
69 (29.5%) |
||
Type of surgery |
Whipple/PPPD |
333 (62.8%) |
159 (67.9%) |
0.1732 |
Distal pancreatectomy |
197 (37.2%) |
75 (32.1%) |
||
Antidiabetic medications except metformin |
||||
Kinds of medications |
Sulfonylurea |
457 (86.2%) |
192 (82.1%) |
0.1369 |
Insulin |
326 (61.5%) |
131 (56.0%) |
0.1509 |
|
Thiazolidinedione |
115 (21.7%) |
44 (18.8%) |
0.3636 |
|
DPP-4 inhibitor |
90 (17.0%) |
9 (3.8%) |
< 0.0001 |
|
Others |
332 (62.6%) |
150 (64.1%) |
0.6997 |
|
Q1, the first quartile; Q3, the third quartile; AST, aspartate transaminase; ALT, alanine transaminase; Rgt, gamma-glutamyl transpeptidase; PPPD, pylorus preserving pancreaticoduodenectomy; DPP-4, dipeptidyl peptidase-4.
*Health risk behavior and laboratory data were available for 331 and 135 patients in the metformin user and the non-user group, respectively. Missing data were not imputed.
The cancer-specific survival (5-year, 31.9% vs. 22.2%, p < 0.001 by the log-rank test) was significantly higher in the metformin user group than in the metformin non-user group among the diabetic groups during the follow-up period (Figure 2). In unadjusted analyses, compared to the metformin non-user group, the metformin user group showed a significantly lower risk of cancer-specific mortality (hazard ratio [HR], 0.702; 95% confidence interval [CI], 0.588–0.837). After multivariable adjustments for clinical covariates, the metformin user group still had a significantly lower risk of events as compared with the metformin non-user group (HR, 0.727; 95% CI, 0.611–0.868) (Table 2). In the metformin user group, the adjusted risk for cancer-specific mortality was significantly lower for patients with an medication possession ratio (MPR) ≥ 80% compared to those with an MPR < 80% (HR, 0.595; 95% CI, 0.468–0.757) (Table 2). In the dose-response relationship analysis, we modeled the association between an exposure dose of metformin and cancer-specific mortality using a cubic spline regression model. The negative linear dose-response trend demonstrated a statistically significant decreased cancer-specific mortality with increasing exposure dose of metformin. The cancer-specific mortality was almost 43% lower (HR, 0.668; 95% CI, 0.529–0.845) for those who received more than 1000 mg metformin daily and compared to the metformin non-user group (Figure 3).
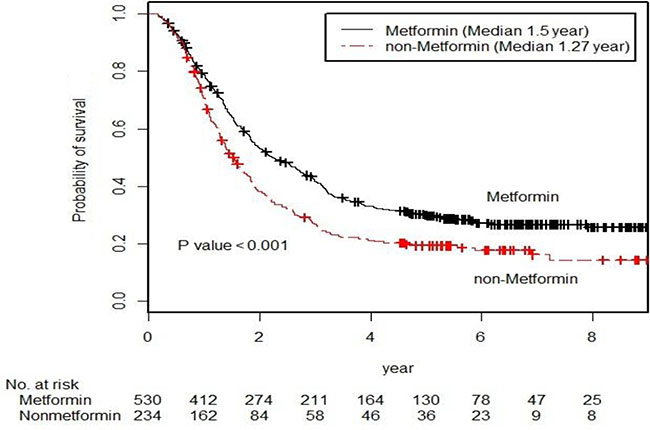
Figure 2: Kaplan-Meier survival curve for the cancer-specific survival of the metformin user group and the metformin non-user group (p-values by the log-rank test).
Table 2: Pancreatic cancer-specific mortality and hazard model according to use of metformin and medication possession ratio (MPR)
Group |
No. of patients |
Pancreatic cancer-specific mortality |
||
|---|---|---|---|---|
No. of event (%) |
Unadjusted HR (95% CI) |
*Adjusted HR (95% CI) |
||
Metformin user |
530 |
373 (70.4%) |
0.702 (0.588–0.837) |
0.727 (0.609–0.868) |
Metformin non-user |
234 |
186 (79.5%) |
1 |
1 |
MPR ≥ 80 in metformin user |
152 |
88 (57.9%) |
0.592 (0.466–0.753) |
0.595 (0.468–0.757) |
MPR < 80 in metformin user |
378 |
285 (75.4%) |
1 |
1 |
HR, hazard ratio; CI, confidence interval.
*Adjusted with age, sex, and Charlson comorbidity index. Hazard ratios were calculated with Cox proportional hazards model for patients with metformin user with reference to patients with metformin non-users. All statistical tests were two-sided.
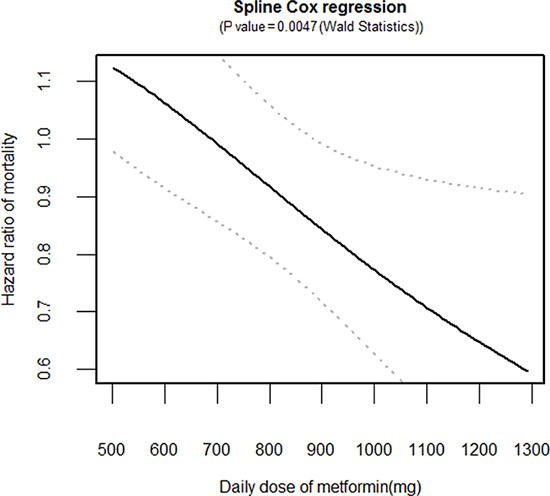
Figure 3: Dose-response relationship between an exposure dose of metformin and cancer-specific mortality. Hazard ratio (solid line) and 95% confidence interval (dashed lines) are from the cubic spline regression model.
In sensitivity analyses, the risks for cancer-specific mortality were consistently lower in the metformin user group when we confined this analysis to those who initiated their prescription during the 6 months before diagnosis, during the 6 months before and after diagnosis, or during the 6 months before and the 12 months after diagnosis. In addition, these lower risks of the metformin user group for cancer-specific mortality were also found in the second sensitivity analysis performed among those whose complete health examination data were available. Similar lower risks of the metformin user group for cancer-specific mortality were shown in the third sensitivity analyses performed among patients treated with chemotherapy, those treated with radiotherapy, or those treated with pancreatic head resection such as Whipple`s procedure or p ylorus-preserving pancreaticoduodenectomy (Table 3).
Table 3: Sensitivity analyses of the association between use of metformin and pancreatic cancer-specific mortality
Variable |
No. of patients |
Pancreatic cancer-specific mortality |
||
|---|---|---|---|---|
*Adjusted HR |
95% CI |
p-value |
||
Initiation of metformin exposure |
||||
During 6 months before diagnosis |
408 |
0.771 |
0.621–0.956 |
0.0178 |
During 6 months before and 6 months after |
574 |
0.744 |
0.616–0.899 |
0.0022 |
During 6 months before and 12 months after |
651 |
0.740 |
0.616–0.889 |
0.0013 |
Complete health examination data |
466 |
0.714 |
0.553–0.923 |
0.0101 |
Chemotherapy |
513 |
0.669 |
0.544–0.821 |
0.0001 |
Radiotherapy |
248 |
0.736 |
0.547–0.990 |
0.0426 |
Whipple`s procedure or PPPD |
492 |
0.796 |
0.644–0.984 |
0.0350 |
Distal pancreatectomy |
272 |
0.576 |
0.419–0.792 |
0.00007 |
HR, hazard ratio; CI, confidence interval; PPPD, pylorus preserving pancreaticoduodenectomy.
*Adjusted with age, sex, and Charlson comorbidity index, hazard ratios were calculated with Cox proportional hazards model for patients with metformin user with reference to patients with metformin non-users. All statistical tests were two-sided.
DISCUSSION
In the present study, we found that those receiving metformin have lower cancer-specific mortality rates than those not receiving metformin in localized resectable pancreatic cancer patients with pre-existing diabetes. In addition, metformin usage was independently predictive of cancer-specific mortality after multivariable adjustment for clinical covariates. This finding is not caused by a difference in treatment methods, because these were balanced between the two pancreatic cancer groups with pre-existing diabetes and our findings remained the same after restricting treatment methods from the analyses. This is the first study showing beneficial effects of metformin in patients with localized resectable pancreatic cancer.
Although an antitumor effect of metformin has been shown in preclinical studies and population analyses, several cohort studies have not shown a consistent survival benefit from metformin in pancreatic cancer patients with pre-existing diabetes [9, 21–24]. Sadeghi et al. showed that metformin usage is significantly associated with longer survival in patients with non-metastatic disease only [22], but that benefit was not significant in those with metastatic disease. In a subclass analysis of patients with non-metastatic disease, resectable disease was not associated with a survival benefit, which may be explained by the small sample size, including only 22% of the study population. Choi et al. showed that metformin usage is associated with a longer overall survival for advanced pancreatic cancer patients receiving palliative chemotherapy [9]. However, these studies, based on retrospective analysis of single institution data, were associated with limitations of small sample sizes and uncontrolled selection bias. Recently, a randomized phase 2 trial was performed to find the synergism between metformin and chemotherapeutic agents in metastatic or unresectable locally advanced pancreatic cancer. They reported that metfromin was not beneficial as an add-on to chemotherapy [21]. In fact, the lack of efficacy of metformin in these patients with advanced disease may be explained by a small study sizes and a modest anti-tumor effect of metformin, compounding by heterogeneous prognostic factors, large tumor burden, and a potential difference in tumor biology in metastatic or unresectable locally advanced disease. Unlike these studies, only patients receiving curative resection for localized pancreatic cancer were included in our study to minimize the mixed effect from differing extents of disease. Resectable pancreatic cancer has been considered to be relatively homogeneous population with the similar prognosis [25–27]. Although we could not get more detailed staging information than the Surveillance, Epidemiology, and End Results (SEER) summary stage in the KCCR database, it is quite reasonable to include only resectable pancreatic cancer. It may only have been possible to show these results because we obtained sufficient number of cases of pancreatic cancer from a linked nationwide database. Therefore, our results suggest any prospective randomized trial should be performed with large number of patients and be stratified by treatment options to obtain a homogenous cohort.
Findings from our study should be interpreted in the context of the following limitations. Firstly, confounding by indication is an intractable threat to validity in observational studies, although we used adjusted models to account for confounding factors. Potential confounding may exist due to the lack of some data, such as hemoglobin A1c level, carbohydrate antigen 19–9, tumor size, lymph node status, or surgical margin status [28, 29]. Secondly, immortal person time is a major concern in these types of observational study [30–32]. We conducted sensitivity analyses to minimize immortal person time bias and the difference in metformin initiation time. In sensitivity analyses, the risks for mortality were consistently lower in the metformin user group, even when we confined this analysis to those who initiated metformin prescription during the 6 months before diagnosis, during the 6 months before and after diagnosis, or during the 6 months before and the 12 months after diagnosis. Thirdly, beneficial effects of metformin, which is the most widely used first-line type 2 diabetes medication, may be due to use of metformin among healthier patients with early diabetes. On the contrary, patients using insulin may have poor glucose control from oral medications or a poor performance status which preclude the use of oral medications [28]. In another sensitivity analysis to minimize healthy user bias, we demonstrated that metformin usage was associated with better survival outcome, even when compared with non-diabetic control patients, despite a preponderance of insulin users in the metformin user group. Fourthly, this study included only Korean population, which is quite homogeneous. There may be differences, such as BMI or any other ethnic-specific factors, among Korean patients with pancreatic cancer and others ethnic groups. The SEER research data demonstrated differences among different ethnic groups with pancreatic cancer in the United States with respect to both incidence and survival [33]. For universal applicability, further studies on other ethnic groups are needed. In conclusion, our study indicates that metformin might decrease cancer-specific mortality rates of localized resectable pancreatic cancer patients with pre-existing diabetes, independently of other factors, by means of a dose-response relationship. Considering the high prevalence of diabetes in patients with pancreatic cancer and the lack of effective treatment strategies for this malignancy, well-designed prospective studies are warranted to confirm the survival benefit of metformin in patients with diabetes and resectable pancreatic cancer. If proven beneficial, metformin may be an ideal adjuvant treatment option because it is inexpensive, safe, and well tolerated. We hope that our study can serve as a proof-of-concept for prospective studies of metformin to prevent or defer recurrence and to prolong survival in patients with resectable pancreatic cancer.
MATERIALS AND METHODS
Data sources
The data were provided by the KCCR and they were linked to national claims data from the NHIS through the use of unique personal identification numbers with the consent of KCCR. The data from the KCCR covers nationwide cancer cases in Korea, and it includes patients’ date and site of primary cancer diagnosis [34]. The NHIS covers 98% of the Korean population for the whole lifespan, and the NHIS has comprehensive data sets for diagnoses, treatments, procedures, surgical history, prescription records, and periodic general health examinations data of all insured patients. In addition, we obtained the cause and date of death from the NPR of the Korea NSO, with the use of the unique personal identification numbers. , IRB waived the need for written informed consent from participants, because this study was based on routinely collected administrative data and why This study was approved by the Institutional Review Board of the National Evidence-based Healthcare Collaborating Agency (NECA IRB 14-004-1).
Study subjects
The study population included 28,862 adults who had a primary diagnosis of pancreatic cancer (the International Classification of Diseases, 10th revision (ICD-10), C25) recorded by the KCCR between January 1, 2005 and December 31, 2011. Only individuals, who had undergone curative resection for localized pancreatic cancer, were eligible. The Whipple`s procedure, p ylorus-preserving pancreaticoduodenectomy, or distal pancreatectomy was defined as curative resection. Those, whose surgery was aborted due to findings of unresectability at the time of surgery, were excluded. The individuals with the age of 40 or less were excluded due to the possibility of genetic or family syndrome. The individuals were excluded if they had a history of invasive cancers other than pancreatic cancer or a follow-up duration of less than 1 month, died from complications following surgery, or were not diagnosed with pancreatic adenocarcinoma. The death from complications following surgery was defined as the death without evidence of disease progression within 1 month of the surgery date.
Exposure and follow-up
The drug exposures of interest were metformin and non-metformin (sulfonylurea, insulin, thiazolidinedione, dipeptidyl peptidase 4 inhibitors, or other antidiabetic drugs). Drug exposure was defined as OHAs or insulin medication administration in the same class for at least 90 days in the period from 6 months before first diagnosis of pancreatic cancer to last follow-up. Participants were classified as patients with pre-existing diabetes and pancreatic cancer if they had a diagnosis of non-insulin-dependent diabetes (ICD-10, E11) and received the drug exposure on the basis of the above-mentioned definition. The MPR was defined as the total days the target medication was prescribed divided by the total days between prescriptions. An MPR of more than 80% was considered acceptable adherence. Cohort entry date for each patient was the date of first diagnosis of pancreatic cancer, and the exit date (censoring date) was the earliest of: a) date of death; b) date of recording of clinical event; c) 5 years after cohort entry or d) the end of the study period (December 31, 2013).
Statistical analysis
In the main analysis, we compared the baseline characteristics and medications for OHA for patients with and without use of metformin. We further compared the event rates for cancer-specific mortality during the follow-up period between patients with and without metformin by cumulative probability curves derived from Kaplan-Meier estimates; the same analysis was applied for MPR groups among patients with metformin. After unadjusted analyses were initially performed, we conducted adjusted analyses, including the following potential confounders: age, sex, and Charlson comorbidity index [35]. We performed further analyses to compare metformin users with non-diabetic control patients to minimize healthy user bias. We performed cubic spline regression analysis to characterize a dose-response relationship between an exposure dose of metformin and cancer-specific mortality [36]. All analyses were performed with SAS software, version 9.3 (SAS Institute, Cary, NC). All reported p-values were 2-sided and a 5% or lower p-value was considered to be statistically significant.
Sensitivity analyses
We conducted sensitivity analyses by the following restrictions. In the first sensitivity analysis, we restricted analysis among those whose exposure of metformin initiated from 180 days before pancreatic cancer diagnosis through the date of pancreatic cancer diagnosis to control immortal time bias [37]. In addition, information from the periodic general health examination was available for 61% of patients. These data included smoking status, alcohol drinking behavior, regular exercise, BMI, total cholesterol, fasting blood glucose, AST, ALT, and rGT. We performed the second sensitivity analyses among those with these data available. In the third sensitivity analyses, we restricted analysis among patients who received chemotherapy, radiotherapy, or pancreatic head resection such as Whipple`s procedure or p ylorus-preserving pancreaticoduodenectomy to obtain a homogeneous cohort. All of these patients received an adjuvant treatment and thus may regarded as representing a cohort that had more advanced diseases and better performance status.
Abbreviations
BMI: body mass index; CI: confidence interval; ALT: alanine transaminase; AST: aspartate transaminase; rGT: gamma-glutamyl transpeptidase; HR: hazard ratio; ICD: International Classification of Diseases; KCCR: Korea Central Cancer Registry; MPR: medication possession ratio; NHIS: National Health Insurance Service; NPR: National Population Registry; NSO; National Statistical Office; OHA: oral hypoglycemic agent; SEER: Surveillance, Epidemiology, and End Results.
ACKNOWLEDGMENTS
This work was selected for an oral presentation in the 26th World Congress of the International Association of Surgeons, Gastroenterologists and Oncologists (IASGO 2016) to be held at the Sheraton Grande Walkerhill Hotel, Seoul, Korea, from September 8–10, 2016.
CONFLICTS OF INTEREST
All authors have nothing to disclose any potential conflicts that are relevant to the manuscript.
FUNDING
This work was supported by the National R&D Program through the Korea Institute of Radiological and Medical Sciences funded by the Ministry of Science, ICT & Future Planning, Republic of Korea (No. 1711031807/50541-2016, No. 1711031802/50432-2016); and the National Evidence based Healthcare Collaborating Agency, Republic of Korea (No. NECA-P-14-006).
Authors’ contributions
Won Il Jang, Mi-Sook Kim, Min Jung Ko, and Shin Hee Kang: study concept and design, acquisition of data, analysis and interpretation of data, drafting of the initial manuscript, critical revision of the final manuscript, and final approval of the version to be published; Ae Jung Jo, Yun Jung Kim, Ha Jin Tchoe, Chan Mi Park, Hyo Jeong Kim, Jin A Choi, and Hyung Jin Choi: acquisition of data, analysis and interpretation of data, critical revision of the final manuscript, and final approval of the version to be published; Eun-Kyung Paik, Young Seok Seo, Hyung Jun Yoo, Jin-Kyu Kang, Chul Ju Han, Yeon Ju Kim, and Sang Beom Kim: study concept and design, analysis and interpretation of data, critical revision of the final manuscript, and final approval of the version to be published.
REFERENCES
1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359–386.
2. Siegel RL, Miller KD, Jemal A. Cancer Statistics. CA Cancer J Clin. 2015; 65:5–29.
3. Vincent A, Herman J, Schulick R, Hruban RH, Goggins M. Pancreatic cancer. Lancet. 2011; 378:607–620.
4. Van den Broeck A, Sergeant G, Van Steenbergen W, Aerts R, Topal B. Patterns of recurrence after curative resection of pancreatic ductal adenocarcinoma. Eur J Surg Oncol. 2009; 35:600–604.
5. Permert J, Ihse I, Jorfeldt L, von Schenck H, Arnqvist HJ, Larsson J. Pancreatic cancer is associated with impaired glucose metabolism. Eur J Surg. 1993; 159:101–7.
6. Wang F, Herrington M, Larsson J, Permert J. The relationship between diabetes and pancreatic cancer. Mol Cancer. 2003; 2:4.
7. Raghavan SR, Ballehaninna UK, Chamberlain RS. The impact of perioperative blood glucose levels on pancreatic cancer prognosis and surgical outcomes: an evidence-based review. Pancreas. 2013; 42:1210–1217.
8. Toriola AT, Stolzenberg-Solomon R, Dalidowitz L, Linehan D, Colditz G. Diabetes and pancreatic cancer survival: a prospective cohort-based study. Br J Cancer. 2014; 111:181–185.
9. Choi Y, Kim TY, Oh DY, Lee KH, Han SW, Im SA, Kim TY, Bang YJ. The Impact of Diabetes Mellitus and Metformin Treatment on Survival of Patients with Advanced Pancreatic Cancer Undergoing Chemotherapy. Cancer Res Treat. 2016; 48:171–179.
10. Bosetti C, Rosato V, Li D, Silverman D, Petersen GM, Bracci PM, Neale RE, Muscat J, Anderson K, Gallinger S, Olson SH, Miller AB, et al. Diabetes, antidiabetic medications, and pancreatic cancer risk: an analysis from the International Pancreatic Cancer Case-Control Consortium. Ann Oncol. 2014; 25:2065–2072.
11. Wang Z, Lai ST, Xie L, Zhao JD, Ma NY, Zhu J, Ren ZG, Jiang GL. Metformin is associated with reduced risk of pancreatic cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Res Clin Pract. 2014; 106:19–26.
12. Kourelis TV, Siegel RD. Metformin and cancer: new applications for an old drug. Med Oncol. 2012; 29:1314–1327.
13. Quinn BJ, Kitagawa H, Memmott RM, Gills JJ, Dennis PA. Repositioning metformin for cancer prevention and treatment. Trends Endocrinol Metab. 2013; 24:469–480.
14. Zhang P, Li H, Tan X, Chen L, Wang S. Association of metformin use with cancer incidence and mortality: a meta-analysis. Cancer Epidemiol. 2013; 37:207–218.
15. Margel D, Urbach DR, Lipscombe LL, Bell CM, Kulkarni G, Austin PC. Metformin use and all-cause and prostate cancer-specific mortality among men with diabetes. J Clin Oncol. 2013; 31:3069–3075.
16. Kumar S, Meuter A, Thapa P, Langstraat C, Giri S, Chien J. Metformin intake is associated with better survival in ovarian cancer: a case-control study. Cancer. 2013; 119:555–562.
17. Coyle C, Cafferty FH, Vale C, Langley RE. Metformin as an adjuvant treatment for cancer: a systematic review and meta-analysis. Ann Oncol. 2016 Sep 28. pii: mdw410. [Epub ahead of print].
18. Tan XL, Bhattacharyya KK, Dutta SK, Bamlet WR, Rabe KG, Wang E. Metformin Suppresses Pancreatic Tumor Growth With Inhibition of NFκB/STAT3 Inflammatory Signaling. Pancreas. 2015; 44:636–647.
19. Kisfalvi K, Moro A, Sinnett-Smith J, Eibl G, Rozengurt E. Metformin inhibits the growth of human pancreatic cancer xenografts. Pancreas. 2013; 42:781–785.
20. Jalving M, Gietema JA, Lefrandt JD, Mathot RA, Weterman MJ, Beeker A. Metformin: taking away the candy for cancer? Eur J Cancer. 2010; 46:2369–2080.
21. Kordes S, Pollak MN, Zwinderman AH, Mathôt RA, Weterman MJ, Beeker A, Punt CJ, Richel DJ, Wilmink JW. Metformin in patients with advanced pancreatic cancer: a double-blind, randomised, placebo-controlled phase 2 trial. Lancet Oncol. 2015; 16:839–847.
22. Sadeghi N, Abbruzzese JL, Yeung SC, Hassan M, Li D. Metformin use is associated with better survival of diabetic patients with pancreatic cancer. Clin Cancer Res. 2012; 18:2905–2912.
23. Hwang AL, Haynes K, Hwang WT, Yang YX. Metformin and survival in pancreatic cancer: a retrospective cohort study. Pancreas. 2013; 42:1054–1059.
24. Chaiteerakij R, Petersen GM, Bamlet WR, Chaffee KG, Zhen DB, Burch PA, Leof ER, Roberts LR, Oberg AL. Metformin Use and Survival of Patients With Pancreatic Cancer: A Cautionary Lesson. J Clin Oncol. 2016; 34:1898–904.
25. Hartwig W, Werner J, Jäger D, Debus J, Büchler MW. Improvement of surgical results for pancreatic cancer. Lancet Oncol. 2013; 14:e476–485.
26. Neoptolemos JP, Moore MJ, Cox TF, Valle JW, Palmer DH, McDonald AC. Effect of adjuvant chemotherapy with fluorouracil plus folinic acid or gemcitabine vs observation on survival in patients with resected periampullary adenocarcinoma: the ESPAC-3 periampullary cancer randomized trial. JAMA. 2012; 308:147–156.
27. Oettle H, Neuhaus P, Hochhaus A, Hartmann JT, Gellert K, Ridwelski K. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013; 310:1473–1481.
28. Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012. 35:1364–1379.
29. Berger AC, Garcia M Jr, Hoffman JP, Regine WF, Abrams RA, Safran H. Postresection CA 19-9 predicts overall survival in patients with pancreatic cancer treated with adjuvant chemoradiation: a prospective validation by RTOG 9704. J Clin Oncol. 2008; 26:5918–5922.
30. Suissa S. Immortal time bias in pharmaco-epidemiology. Am J Epidemiol. 2008; 167:492–499.
31. Suissa S, Azoulay L. Metformin and the risk of cancer: time-related biases in observational studies. Diabetes Care. 2012; 35:2665–2673.
32. McCandless LC. Statin use and fracture risk: can we quantify the healthy-user effect? Epidemiology. 2013; 24:743–752.
33. Howlader N, Noone AM, Krapcho M, Garshell J, Miller D, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, et al. (Accessed 1 Feb 2016) SEER Cancer Statistics Review, 1975–2012. In: National Cancer Institute. Bethesda, MD, US. 2015. http://seer.cancer.gov/csr/1975_2012/.
34. Jung KW, Won YJ, Kong HJ, Oh CM, Cho H, Lee DH. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2012. Cancer Res Treat. 2015; 47:127–141.
35. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987; 40:373–383.
36. Greenland S. Dose-response and trend analysis in epidemiology: alternatives to categorical analysis. Epidemiology. 1995; 6:356–365.
37. Zhou Z, Rahme E, Abrahamowicz M, Pilote L. Survival bias associated with time-to-treatment initiation in durg effectiveness evaluation: A comparison of methods. Am J Epidemiol. 2005; 162:1016–1023.

