INTRODUCTION
Lung cancer remains the most common form of cancer and the leading cause of cancer-related deaths worldwide. Non-small cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancers [1]. Radical surgical resection offers the chance to achieve long-term survival and cure, and is considered as the optimal treatment for patients with operable NSCLC. However, many patients with lung cancer have respiratory deficiencies, impeding surgical resection [2].
Preoperative pulmonary function plays an important role in the evaluation of surgery tolerance, lung reserve, risk of perioperative complications, and short-term outcome [3, 4]. The forced expiratory volume in one second (FEV1) and diffusion lung capacity for carbon monoxide (DLCO) are useful parameters for predicting the perioperative complications and short-term mortality [5, 6] and long-term survival in NSCLC [2, 3]. However, results from previous studies have not yet reached a unified conclusion and are even controversial [4]. Forced vital capacity (FVC) is associated with cardiovascular disease mortality [4]. However, apart from FEV1 and DLCO, few studies have analyzed the effect of FVC and other pulmonary function parameters on the long-term survival of NSCLC patients.
Increasing data have indicated that the prognosis of cancer patients was based not only on tumor-related factors but also on host-related factors, particularly the systemic inflammatory response (SIR). SIR promotes tumor angiogenesis, accelerates tumor cells metastasis, and suppresses antitumor immunity [7]. Mounting evidence supports the prognostic role of systemic inflammatory laboratory parameters, such as neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR), in patients with NSCLC undergoing surgery resection [8–10]. Furthermore, previous reports have revealed that elevated SIR may insult the pulmonary function and increase the peripheral serum white blood cell count, mononuclear cells and lymphocytes were associated with impaired pulmonary function [11, 12]. However, few studies have evaluated the relationship between pulmonary function and SIR biomarkers (NLR, PLR, and LMR) in patients with NSCLC. Thus, considering the available data, we attempted to determine whether preoperative pulmonary functions are associated with SIR biomarkers in patients with NSCLC.
Therefore, in the present study, we determined the optimum preoperative pulmonary function parameters associated with long-term survival in patients with NCSLC by including as many pulmonary function parameters as possible. Furthermore, we investigated for the first time the associations among preoperative pulmonary function, SIR biomarkers, and prognosis in NSCLC.
RESULTS
Clinicopathological characteristics of patients
The baseline clinicopathological characteristics of 358 patients with primary and histologically confirmed NSCLC were summarized in Table 1. There were 247 (69.0%) males and 111 (31.0%) females with the median age of 61 years (range: 25~83 years). Meanwhile, 244 (68.2%) patients had smoking history. After complete resection, 125 (34.9%) patients had adenocarcinoma, 162 (45.3%) patients had squamous cell carcinoma, and 71 (19.8%) patients were not specified. Approximately 38.8% of the patients had lymph node metastasis. The distribution of TNM stages was as follows: stage I, 173 patients; stage II, 87 patients; and stage III, 98 patients. Furthermore, 166 (46.4%) patients had FVC% predicted of < 80%, and 213 (59.5%) patients had FEV1% predicted of < 80%. Impaired vital capacity (VC), FEV1/FVC, peak expiratory flow (PEF), mid-expiratory flow when 50% of FVC has been expired (MEF50%), DLCO, and maximal voluntary ventilation (MVV) were observed in 196, 264, 280, 249, 118, and 123 patients, respectively.
Table 1: Demographics and characteristics of the study population of NSCLC individuals
Variables |
Cases (%) |
|---|---|
Age (years) |
|
≤ 60/ > 60 |
175 (48.9%)/ 183 (51.1%) |
Gender |
|
Male/ Female |
247 (69.0%)/ 111 (31.0%) |
Smoking history |
|
Yes/ None |
244 (68.2%)/ 114 (31.8%) |
Type of resection |
|
Pneumonectomy/ Lobectomy/ bronchial sleeve resection |
92 (25.7%)/ 249 (69.6%)/ 17 (4.7%) |
Histology |
|
Adenocarcinoma/ Squamous cell carcinoma/ Others |
125 (34.9%)/ 162 (45.3%)/ 71 (19.8%) |
T stage |
|
T1/ T2/ T3-4 |
152 (42.5%)/ 185 (51.7%)/ 21 (5.9%) |
Lymph node status |
|
N0/ N+ |
219 (61.2%)/ 139 (38.8%) |
TNM stage |
|
I/ II/ III |
173 (48.3%)/ 87 (24.3%)/ 98 (27.4%) |
VC |
|
Normal/ Impairement |
162 (45.3%)/ 196 (45.7%) |
FVC |
|
Normal/ Impairement |
192 (53.6%)/ 166 (46.4%) |
FEV1 |
|
Normal/ Impairement |
145 (40.5%)/ 213(59.5%) |
FEV1/FVC |
|
Normal/ Impairement |
94 (26.3%)/ 264 (73.7%) |
PEF |
|
Normal/ Impairement |
78 (21.8%)/ 280 (78.2%) |
MEF50% |
|
Normal/ Impairement |
109 (30.4%)/ 249 (69.6%) |
DLCO |
|
Normal/ Impairement |
240 (67.0%)/ 118 (33.0%) |
MVV |
|
Normal/ Impairement |
235 (65.6%)/ 123 (34.4%) |
NLR |
|
<2.14/ ≥2.14 |
196 (54.7%)/ 162 (45.3%) |
PLR |
|
<121.78/ ≥121.78 |
179 (50.0%)/ 179 (50.0%) |
LMR |
|
<3.06/ ≥3.06 |
127 (35.5%)/ 231 (64.5%) |
Cut-point survival analysis for the detection of the optimum cut-points of NLR, PLR, and LMR
The optimum cut-point values of the preoperative NLR, PLR, and LMR for survival prediction were determined through a receiver operating characteristic curve (ROC) curve analysis. For NLR, the optimum cut-point was 2.14 with a maximum joint sensitivity of 55.5% and specificity of 72.9% on the ROC plot. The area under the ROC curve (AUC) was 0.646 (95% CI: 0.589~0.703). For PLR, the optimum cut-point was 121.78 with a maximum joint sensitivity of 57.8% and specificity of 62.1%, and the AUC was 0.628 (95%CI: 0.574~0.692). For LMR, the optimum cut-point was 3.06 with a maximum joint sensitivity of 45.0% and specificity of 82.1%, and the AUC was 0.640 (95% CI: 0.587~0.700) (Figure 1). Therefore, NLR, PLR, and LMR with cut-point values of 2.14, 121.78, and 3.06, respectively, were used as optimum cut-points in the subsequent analysis. For each biomarker, the patients were classified into two groups (NLR [<2.14 and ≥2.14], PLR [<121.78 and ≥121.78], and LMR [<3.06 and ≥3.06]).
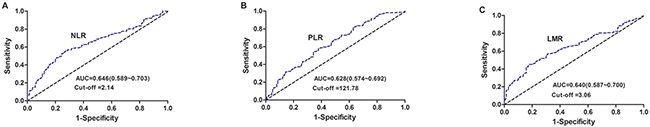
Figure 1: Receiver operating characteristic curve analysis for the cut-points of NLR, PLR, and LMR in NSCLC patients. A. The AUC for NLR was 0.646 (95%CI: 0.589~0.703) with a sensitivity of 55.5% and specificity of 72.9%. B. The AUC for PLR was 0.628 (95%CI: 0.574~0.692) with a sensitivity of 57.8% and specificity of 62.1%. C. The AUC for LMR was 0.640 (95%CI: 0.587~0.700) with a sensitivity of 45.0% and specificity of 82.1%.
Correlation analysis between preoperative pulmonary function and SIR biomarkers
The correlations between preoperative pulmonary function and SIR biomarkers (NLR, PLR, LMR) were analyzed. As shown in Table 2, impaired VC was associated with elevated PLR (x2 = 6.494, P = 0.011). Impaired FEV1 and PEF were associated with decreased LMR (x2 = 4.551, P = 0.034; x2 = 13.383, P <0.001). Lastly, impaired DLCO was associated with elevated NLR (x2 = 4.706, P = 0.030) and PLR (x2 = 5.056, P = 0.025). Notably, impaired FVC was simultaneously associated with elevated NLR (x2 = 4.428, P = 0.035) and PLR (x2 = 6.470, P = 0.011) and decreased LMR (x2 = 4.074, P = 0.044). However, statistically significant association was not observed among other pulmonary function parameters (FEV1/FVC, MEF50%, MVV) and NLR, PLR, or LMR (P > 0.05).
Table 2: Correlation analysis of preoperative pulmonary function parameters and SIR biomarkers in NSCLC patients
Variables |
N |
NLR |
Chi-square test |
PLR |
Chi-square test |
LMR |
Chi-square test |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Low |
High |
x2 |
P |
Low |
High |
x2 |
P |
Low |
High |
x2 |
P |
||
196 |
162 |
179 |
179 |
127 |
231 |
||||||||
VC |
2.430 |
0.119 |
6.494 |
0.011 |
2.748 |
0.097 |
|||||||
Normal |
162 |
96 (59.3%) |
66 (40.7%) |
93 (57.4%) |
69 (42.6%) |
50 (30.9%) |
112 (69.1%) |
||||||
Impairement |
196 |
100 (51.0%) |
96 (49.0%) |
86 (43.9%) |
110 (56.1%) |
77 (39.3%) |
119 (60.7%) |
||||||
FVC |
4.428 |
0.035 |
6.470 |
0.011 |
4.074 |
0.044 |
|||||||
Normal |
192 |
115 (59.9%) |
77 (40.1%) |
108 (56.3%) |
84 (43.8%) |
59 (30.7%) |
133 (69.3%) |
||||||
Impairement |
166 |
81 (48.8%) |
85 (51.2%) |
71 (42.8%) |
95 (57.2%) |
68 (41.0%) |
98 (59.0%) |
||||||
FEV1 |
0.320 |
0.572 |
0.939 |
0.333 |
4.511 |
0.034 |
|||||||
Normal |
145 |
82 (56.6%) |
63 (43.4%) |
77 (53.1%) |
68 (46.9%) |
42 (29.0%) |
103 (71.0%) |
||||||
Impairement |
213 |
114 (53.5%) |
99 (46.5%) |
102 (47.9%) |
111 (52.1%) |
85 (39.9%) |
128 (60.1%) |
||||||
FEV1/FVC |
0.353 |
0.552 |
0.923 |
0.337 |
0.114 |
0.735 |
|||||||
Normal |
94 |
49 (52.1%) |
45 (47.9%) |
51 (54.3%) |
43 (45.7%) |
32 (34.0%) |
62 (66.0%) |
||||||
Impairement |
264 |
147 (55.7%) |
117 (44.3%) |
128 (48.5%) |
136 (51.5%) |
95 (36.0%) |
169 (64.0%) |
||||||
PEF |
0.093 |
0.760 |
0.013 |
0.909 |
13.383 |
0.000 |
|||||||
Normal |
109 |
61 (56.0%) |
48 (44.0%) |
54 (49.5%) |
55 (50.5%) |
14 (17.9%) |
64 (82.1%) |
||||||
Impairement |
249 |
135 (54.2%) |
114 (45.8%) |
125 (50.2%) |
124 (49.8%) |
113 (40.4%) |
167 (59.6%) |
||||||
MEF 50% |
0.093 |
0.760 |
0.646 |
0.412 |
3.388 |
0.066 |
|||||||
Normal |
109 |
61 (56.0%) |
48 (44.0%) |
58 (53.2%) |
51 (46.8%) |
31 (28.4%) |
78 (71.6%) |
||||||
Impairement |
249 |
135 (54.2%) |
114 (45.8%) |
121 (48.6%) |
128 (51.4%) |
96 (38.6%) |
153 (61.4%) |
||||||
DLCO |
4.706 |
0.030 |
5.056 |
0.025 |
0.946 |
0.331 |
|||||||
Normal |
240 |
141 (58.8%) |
99 (41.3%) |
130 (54.2%) |
110 (45.8%) |
81 (33.8%) |
159 (66.3%) |
||||||
Impairement |
118 |
55 (46.6%) |
63 (53.4%) |
49 (41.5%) |
69 (58.5%) |
46 (39.0%) |
72 (61.0%) |
||||||
MVV |
0.090 |
0.764 |
0.310 |
0.578 |
1.031 |
0.310 |
|||||||
Normal |
235 |
130 (55.3%) |
105 (44.7%) |
120 (51.1%) |
115 (48.9%) |
79 (33.6%) |
156 (66.4%) |
||||||
Impairement |
123 |
66 (53.7%) |
57 (46.3%) |
59 (48.0%) |
64 (52.0%) |
48 (39.0%) |
75 (61.0%) |
||||||
Univariate and multivariate analyses for the detection of independent prognostic factors in patients with NSCLC
The follow-up period of the entire cohort ranged from 3 to 84 months (median of 36 months). The one-, three-, and five-year overall survival (OS) rates for the whole study population were 83.5%, 51.1%, and 40.3%, respectively, with the median survival time (MST) of 48 months.
To determine the effect of pulmonary function on the prognosis, we first performed univariate analysis on the clinicopathological variables for prognosis. The results of univariate analysis for OS were showed in Table 3, of the available clinicopathological variables, age (P = 0.025), T stage (P < 0.001), lymph node status (P < 0.001), VC (P < 0.001), FVC (P < 0.001), FEV1 (P = 0.014), MVV (P = 0.016), NLR (P < 0.001), PLR (P = 0.033), and LMR (P < 0.001) were found to be significantly associated with prognosis by log-rank test.
Table 3: Univariate survival analysis of clinicopathological variables with regard to the OS of NSCLC patients
Variables |
N |
5-YSR (%) |
x2 value |
P value |
|---|---|---|---|---|
Age (years) |
4.996 |
0.025 |
||
≤60 |
175 |
44.8 |
||
>60 |
183 |
36.0 |
||
Gender |
0.077 |
0.782 |
||
Male |
247 |
40.9 |
||
Female |
111 |
39.2 |
||
Smoking history |
0.852 |
0.356 |
||
Yes |
244 |
39.3 |
||
None |
114 |
42.5 |
||
Histology |
0.667 |
0.716 |
||
Adenocarcinoma |
125 |
39.6 |
||
Squamous cell carcinoma |
162 |
41.9 |
||
Others |
71 |
37.9 |
||
T stage |
16.805 |
0.000 |
||
T1 |
152 |
50.5 |
||
T2 |
185 |
34.6 |
||
T3-4 |
21 |
18.2 |
||
Lymph node status |
42.664 |
0.000 |
||
N0 |
219 |
53.6 |
||
N+ |
139 |
19.1 |
||
VC |
12.938 |
0.000 |
||
Normal |
162 |
50.7 |
||
Impairement |
196 |
31.9 |
||
FVC |
18.948 |
0.000 |
||
Normal |
192 |
51.2 |
||
Impairement |
166 |
27.7 |
||
FEV1 |
6.076 |
0.014 |
||
Normal |
145 |
48.4 |
||
Impairement |
213 |
34.8 |
||
FEV1/FVC |
0.719 |
0.396 |
||
Normal |
94 |
35.9 |
||
Impairement |
264 |
41.9 |
||
PEF |
0.054 |
0.816 |
||
Normal |
78 |
39.5 |
||
Impairement |
280 |
40.6 |
||
MEF50% |
0.213 |
0.644 |
||
Normal |
109 |
43.5 |
||
Impairement |
249 |
38.8 |
||
DLCO |
0.899 |
0.343 |
||
Normal |
240 |
42.3 |
||
Impairement |
118 |
36.8 |
||
MVV |
5.842 |
0.016 |
||
Normal |
235 |
45.0 |
||
Impairement |
123 |
31.5 |
||
NLR |
20.957 |
0.000 |
||
<2.14 |
196 |
47.7 |
||
≥2.14 |
162 |
31.4 |
||
PLR |
4.545 |
0.033 |
||
<121.78 |
179 |
44.9 |
||
≥121.78 |
179 |
35.8 |
||
LMR |
25.437 |
0.000 |
||
<3.06 |
127 |
27.3 |
||
≥3.06 |
231 |
47.5 |
To determine the independent prognostic factors, we performed a multivariate analysis on the statistically significant factors in the univariate analysis. A Cox proportional hazard model showed that age (hazard ratio (HR) 1.381, 95 % CI 1.082-1.855, P = 0.032), lymph node status (HR 2.468, 95 % CI 1.858-3.277, P = 0.000), FVC (HR 1.689, 95 % CI 1.057-2.699, P = 0.029), and NLR (HR 1.482, 95 % CI 1.029-2.135, P = 0.034) were independent prognostic factors, which were significantly associated with long-term survival of patients with NSCLC (Table 4, Figure 2).
Table 4: Multivariable Cox proportional hazards regression analysis of OS in NSCLC patients
Variables |
B |
Wald |
HR |
95%CI |
P value |
|---|---|---|---|---|---|
Age |
0.323 |
4.589 |
1.381 |
1.028~1.855 |
0.032 |
T stage |
0.206 |
2.812 |
1.228 |
0.966~1.563 |
0.094 |
Lymph node status |
0.903 |
38.953 |
2.468 |
1.858~3.277 |
0.000 |
VC |
-0.016 |
0.005 |
0.984 |
0.616~1.571 |
0.945 |
FVC |
0.524 |
4.797 |
1.689 |
1.057~2.699 |
0.029 |
FEV1 |
0.077 |
0.140 |
1.080 |
0.722~1.614 |
0.708 |
MVV |
0.046 |
0.085 |
1.047 |
0.769~1.426 |
0.770 |
NLR |
0.394 |
4.471 |
1.482 |
1.029~2.135 |
0.034 |
PLR |
0.059 |
0.139 |
1.061 |
0.778~1.447 |
0.709 |
LMR |
0.283 |
2.331 |
1.327 |
0.923~1.907 |
0.127 |
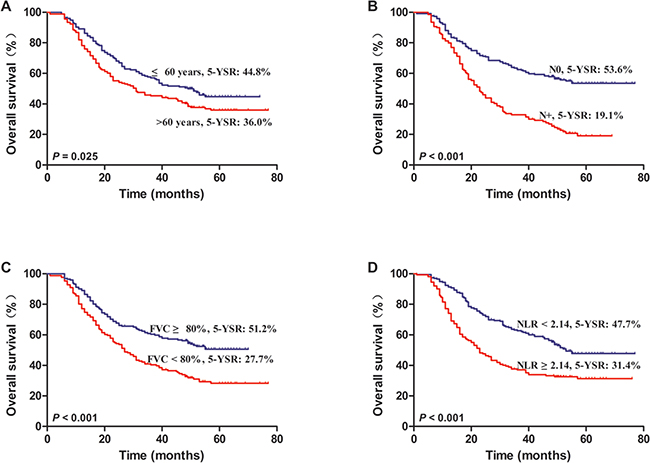
Figure 2: Kaplan–Meier survival curves for NSCLC patients after lung resection. A. Patients with age ≤60 years and >60 years. B. Patients with and without lymph node metastasis. C. Patients with FVC expressed as a percent of predicted ≥80% and FVC% <80%. D. Patients with NLR <2.14 and NLR ≥2.14.
To further evaluate the prognostic value of FVC in different subgroups, the patients were further classified according to age (Figure 3A and 3B), lymph node status (Figure 3C and 3D) and NLR (Figure 3E and 3F). The FVC retained its prognostic value in OS prediction for all the subgroups (P < 0.05). Therefore, it appears that FVC may serve as a powerful prognostic factor for patients with NSCLC in different risk subgroups.
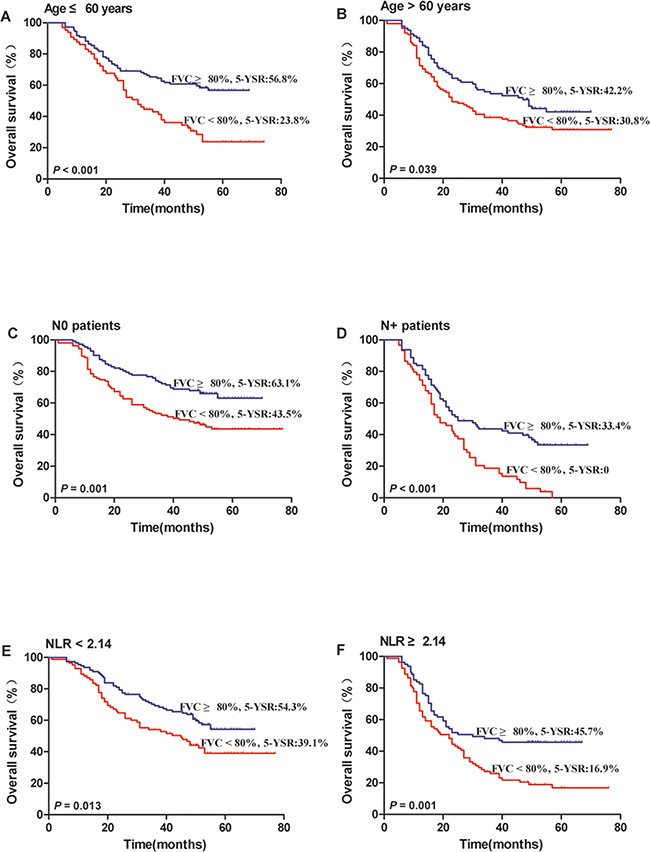
Figure 3: Kaplan–Meier survival analysis of the prognostic value of preoperative FVC in each subgroup. All patients were stratified according to age (A, B), lymph node status (C, D) and NLR (E, F). (A) age ≤60 years subgroup, (B) age >60 years subgroup, (C) no lymph node metastasis subgroup, (D) positive lymph node metastasis subgroup, (E) NLR <2.14 subgroup, and (F) NLR ≥2.14 subgroup.
Risk groups based on the prognostic model
Multivariate survival analysis identified four independent risk factors, namely, age of >60 years, positive lymph node metastasis, impaired FVC, and elevated NLR. To identify the patients with poor long-term survival, we constructed a new risk prognostic model based on the four independent prognostic factors. The patients were then assigned into the following three groups: Group I, low-risk group, patients with zero or one risk factor; Group II, intermediate-risk group, patients with two risk factors; Group III, high-risk group, patients with three or four risk factors. Among the patients, 141 were included in the low-risk group, 123 were part of the intermediate group, and 94 were included in the high-risk group. The five-year OS rates for the low-, intermediate-, and high-risk groups were 60.9%, 35.9%, and 15.3%, respectively. Based on the prognostic model, the five-year OS rates of the three groups were statistically different (P < 0.001) (Figure 4).
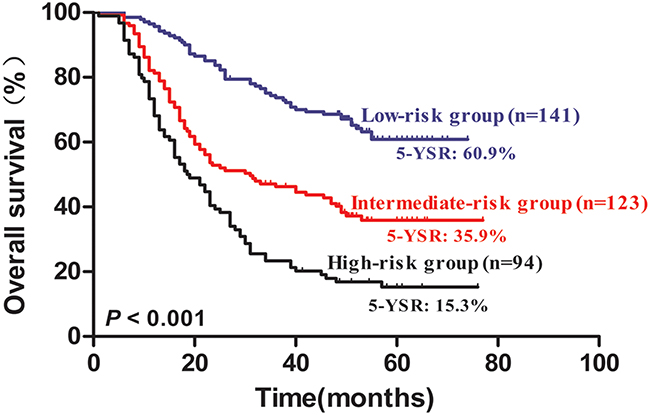
Figure 4: Kaplan–Meier survival curves for 358 NSCLC patients according to risk groups. The five-year OS rates for low-, intermediate-, and high-risk groups were 60.9%, 35.9%, and 15.3%, respectively(P < 0.001).
DISCUSSION
In the present study, the associations among the preoperative pulmonary function, serum SIR biomarkers (NLR, PLR, and LMR), and prognosis were investigated in 358 patients with NSCLC. Our results first demonstrated that preoperative pulmonary function were associated with SIR biomarkers, and the FVC was simultaneously associated with the three SIR biomarkers. These results implied that increased systemic inflammatory activity may play an important role in impaired pulmonary function in NSCLC patients. Furthermore, only FVC was demonstrated to be an independent prognostic predictor in all pulmonary function parameters. Subgroup analysis results further showed that FVC retained its prognostic value in OS prediction in all the risk subgroups and may be considered as a potent prognostic factor in NSCLC patients.
For many years, SIR has been shown to be related to the presence of cancer, tumor progression, and prognosis in NSCLC [13, 14]. Inflammatory mediators and cytokines produced by tumors or a host’s innate response can exert immunomodulatory effects through apoptosis inhibition, DNA damage, and angiogenesis promotion [15]. Moreover, recent data showed that elevated SIR may contribute to impaired pulmonary function not only in patients with pulmonary disease and smokers but also in healthy individuals and non-smokers [11, 12, 16, 17]. The systemic effects of cancer-related inflammation can be quantified using routinely available serum SIR biomarkers, such as NLR, PLR, and LMR. In recent, mounting data reported that objective, inexpensive and stage-independent predictors, NLR, PLR and LMR were associated with long-term survival in NSCLC [9, 10, 18, 19]. Thus, these biomarkers are becoming increasingly important in clinical applications. Meanwhile, peripheral white cell count, mononuclear cells, and lymphocytes were reported to be inversely associated with pulmonary function [12]. However, as the ratios of absolute neutrophil, lymphocyte, and monocytes counts, the relationship between SIR biomarkers and preoperative pulmonary function remains unclear. Therefore, based on these facts, we included these SIR biomarkers in our analysis and explored the relationship between preoperative pulmonary function and SIR biomarkers, respectively. Results showed that NLR, PLR, and LMR were associated with VC, FVC, FEV1, and DLCO. Notably, FVC was simultaneously related to three SIR biomarkers. However, the mechanism underlying the relationship between pulmonary function and SIR remains unclear. In the study of Yang et al. [20], elevated serum white blood cell count is demonstrated to be associated with decreased pulmonary function. McKeever et al. [12] further reported that elevated circulating granulocytes, mononuclear cells, and lymphocytes were inversely associated with FVC and FEV1. A considerable number of resident macrophages can be found in the innate protection system of lung tissues for protection against air pollution, tobacco smoking, and microorganisms. Lung protective macrophages was activated to generate various cytokines when microorganisms or particles were inhaled into lung tissue. These cytokines then regulated local inflammatory response and entered the circulation to activate the SIR [16, 21]. The SIR induced endothelial dysfunction and then contributed to pulmonary vascular filtration, lung tissue injury [22], obstructive and restrictive lung diseases, and impaired pulmonary function [16, 23]. These findings implied the possible role of inflammatory cells and cytokines in impaired pulmonary function.
In clinical practice, preoperative pulmonary functions, especially FEV1 and DLCO, are regularly used to evaluate the operability and to predict the risk of perioperative complications and mortality in thoracic surgery [3]. Percent predicted values, which adjust with gender, age, weight, height and race of patients, are thought to be more useful than absolute values [3, 24]. At present, however, previous studies have different conclusions regarding the prognostic value of pulmonary function on the long-term survival in NSCLC after lung resection. In the present study, by including more pulmonary function parameters, we explored the optimum preoperative pulmonary function parameters for prognosis prediction and subsequently observed that preoperative FVC was the only independent prognostic predictor in NSCLC. Our results were consistent with those of Guo et al. [4] and Burney et al. [25]. Although FEV1 and DLCO have been interpreted as evidence for assessing the chronic obstructive lung disease (COPD) and many cardiovascular diseases [25, 26], few studies found impaired FEV1 and DLCO were related to poor prognosis [3, 27]. Possible explanations may be improved ventilator function after resection of the obstructive disease and better perioperative care and therapy. Besides, in some studies, the pulmonary function parameters were expressed as absolute values instead of as percentage of predicted values, and additional pulmonary function parameters (such as FVC and FEV1/FVC) were not incorporated in the analysis for prognosis. Finally, different studies adopted various criteria points to stratify severity of pulmonary function. All these factors mentioned above might lead to different conclusions. On the other hand, the FVC, forced vital capacity, is mainly determined by level of physical energy, respiratory muscle function, airway resistance, and elasticity of thorax and lung tissues [4]. According to Guo et al. [4], FVC does not only reflect the pulmonary function but also the status of the entire body. The possible mechanism, to some degree, could explain why preoperative FVC was an independent prognostic predictor in NSCLC.
Furthermore, the present study extended these findings by revealing that NLR and PLR were inversely associated with pulmonary function and survival, and LMR was positively related to pulmonary function and survival. Previous studies reported that lymphocytes with targets for tumor-associated antigens could contribute to tumor suppression [28]. However, elevated neutrophils may influence the cytolytic activity of the lymphocytes, and increased NLR reflects the pro-tumor capacity of the host [29]. Circulatory mononuclear cells enter the tumor tissue and differentiate into tumor-associated macrophages (TAMs) [30]. TAMs could secrete pro-angiogenic factors to induce angiogenesis under certain conditions to promote tumor growth and metastasis [31]. Besides, previous studies revealed that increased NLR and PLR and decreased LMR were associated with poor prognosis in NSCLC [18]. Our results also demonstrated that impaired preoperative pulmonary function was associated with increased NLR and PLR, decreased LMR, and poor prognosis. Based on these data, we assumed that increased SIR led to poor prognosis in NSCLC patients with impaired preoperative pulmonary function. However, the mechanisms that connect pulmonary function, inflammatory response, and long-term prognosis of NSCLC patients require further investigation.
In the present study, age, lymph node status, preoperative FVC, and NLR were demonstrated to be the independent prognostic predictors for the long-term survival in NSCLC patients. We further construct a novel and comprehensive prognostic model based on the four independent risk factors. This prognostic model classified NSCLC patients into low-, intermediate- and high-risk groups according to the number of risk factors. For NSCLC patients in the high risk group, anti-inflammatory treatment, improvement of pulmonary function and caring, and chemotherapy may be more important for prolonging the long-term survival. As a supplement of the TNM stage in the prognostic prediction, the model of risk stratification may help clinicians to make individualized treatment protocol and improve the prognostic prediction based on the TNM stage.
The current study had some limitations. First, the study was a retrospective one in nature with a relatively small sample population, and thus leaving some groups small in the statistical analysis. Second, although we concluded that FVC was simultaneously associated with NLR, PLR, and LMR, other pulmonary function parameters didn’t simultaneously related to NLR, PLR, and LMR. Third, other systemic inflammatory immune indexes, such as C-reactive protein or albumin, which were known as prognostic factors, were absent in our retrospective study. Finally, considering the the association among preoperative pulmonary function, SIR and prognosis, we indirectly speculated the association between pulmonary function and prognosis by regulating the SIR. We recognized that all results in the current study need an in-depth analysis, and to be explored in one large population.
In conclusion, our study first revealed that impaired preoperative pulmonary function was related to increased NLR and PLR and decreased LMR. Furthermore, preoperative FVC may be the optimum prognostic factor for long-term survival of patients with NSCLC independent of age, lymph node status and NLR. The introduction of a new and comprehensive prognostic model may assist clinicians to make individualized treatment protocol according to different risk groups.
PATIENTS AND METHODS
Patients and characteristics
From January 2007 to December 2008, a retrospective analysis was conducted on 358 histologically confirmed NSCLC patients, who underwent curative lung resection (bronchial sleeve resection, lobectomy, or pneumonectomy) and systematic lymph nodes dissection at the Tianjin Medical University Cancer Institute and Hospital. The eligibility criteria for the current study were as follows: histologically confirmed primary NSCLC, absence of distant metastasis, complete surgical resection and systematic node dissection, no clinical evidence of infection or other inflammatory conditions, without any preoperative chemotherapy or radiotherapy, with preoperative pulmonary function tests, and complete clinical and follow-up data.
Pathological diagnosis and tumor grade were reviewed and confirmed by two independent pathologists. Tumor stage and histological classification were described according to the 7th TNM staging system of the Union for International Cancer Control (UICC) for NSCLC [32]. The main adjuvant treatment for patients after operations was chemotherapy or radiotherapy, either alone or in combination according to the NSCLC guidelines [32]. Based on the medical records, the following characteristics of each patients were collected: sex, age, smoking history, surgical procedure, histological type, T stage, lymph node status, TNM stage, pulmonary function, complete blood cell counts, and other miscellaneous information. This study was approved by the Ethical Committees of the Tianjin Medical University Cancer Institute and Hospital, and all patients have written informed consent. The methods were carried out in accordance with the relevant guidelines and regulations.
Pulmonary function tests
Pulmonary function tests were performed by one trained technician within the week prior to the surgery and reported as percent of predicted values. All patients underwent pulmonary function tests performed on a Masterscreen-PFT (Cardinal healthcare Germany 234 GmBH). The VC, FVC, FEV1, and FEV1/FVC were measured by spirometry, which was performed according to the American Thoracic Society (ATS) standards, and the lung volumes were performed utilizing body plethysmography in all patients [33]. The diffusion lung capacity for carbon monoxide corrected for alveolar volume were measured by using the carbon monoxide single-breath technique, utilizing an inhalational mixture of 0.3% carbon monoxide (CO) and 10% helium [34]. The patients were instructed to hold their breath for 10 seconds followed by a complete and consistent exhalation. The DLCO testing was repeated after 5 minutes. Two acceptable DLCO tests within 2 mL/min/mmHg were obtained, the mean value was reported for each patient. The observed values were all expressed as a percent of predicted values for a normal patient according to age, sex, height, and weight. Pulmonary function values <80% of the predicted were defined as impaired lung function [4].
Blood cell count analysis
Preoperative venous blood samples were collected for laboratory full blood count analysis within one week before surgery. Blood cell counts were analyzed using a standard Coulter counter (Model XE2100; Sysmex Co, Kobe, Japan). The NLR was defined as the ratio between the absolute neutrophil and absolute lymphocyte counts. PLR was defined as the ratio between the absolute platelet and absolute lymphocyte counts. LMR was defined as the ratio between the absolute lymphocyte and absolute mononuclear cells counts. The optimum cut-off points of NLR, PLR, and LMR were adopted by the ROC curves for survival prediction.
Prognostic model and risk groups
The prognostic model was established based on the risk factors according to the multivariate survival proportional hazard model. We further divided the patients into low-, intermediate- and high-risk groups according to the number of risk factors in the model. Comparisons of survival among different risk groups were analyzed by using the log-rank test.
Follow-up
After surgery, all the patients were followed up through hospital visits, telephone or correspondence every 3–6 months. Patients alive, all death and cases lost to follow up at the last follow-up were censored. The OS was defined as the duration from the date of surgery to death or the last follow-up. The deadline of the follow-up was December 2013, and the follow-up rate was 90.2%.
Statistical analysis
All statistical analyses were conducted using the SPSS statistical package 17.0 (SPSS, Inc, Chicago, IL). The associations between pulmonary function and clinicopathological factors and SIR were calculated using the Chi-square test, and displayed in cross-tables. An ROC curve for survival prediction was used to verify the optimum cut-off points of NLR, PLR, and LMR. The AUC was measured to estimate the diagnostic accuracy. Univariate survival analyses were estimated using the Kaplan-Meier method, and compared via the log-rank test. Multivariate analyses with Cox proportional hazards model were performed to identify the independent prognostic variables. Two-sided P-value < 0.05 was considered as statistically significant.
Abbreviations
NSCLC: Non-small cell lung cancer
VC: Vital capacity
FVC: Forced vital capacity
FEV1: Forced expiratory volume in one second
PEF: Peak expiratory flow
MEF50%: Mid-expiratory flow when 50% of FVC has been expired
DLCO: Diffusion lung capacity for carbon monoxide
MVV: Maximal voluntary ventilation
SIR: Systemic inflammatory response
NLR: Neutrophil-to-lymphocyte ratio
PLR: Platelet-to-lymphocyte ratio
LMR: Lymphocyte-to-monocyte ratio
ROC: Receiver operating characteristic curve
OS: Overall survival
5-YSR: 5-year survival rate
HR: Hazard ratio
CI: Confidence interval
ACKNOWLEDGMENTS
We greatly appreciate Huiqin Liu and Wen Li (The editorial department of Journal of Cancer Biology & Medicine, Tianjin Medical University Cancer Institute and Hospital) for their excellent technical assistance.
CONFLICTS OF INTEREST
The authors declare no conflict of interest.
REFERENCES
1. Jemal A, Siegel R, Xu J, Ward, E. Cancer statistics, 2010. CA Cancer J Clin. 2010; 60:277-300.
2. Santambrogio L, Nosotti M, Baisi A, Ronzoni G, Bellaviti N, Rosso L. Pulmonary lobectomy for lung cancer: a prospective study to compare patients with forced expiratory volume in 1 s more or less than 80% of predicted. Eur J Cardiothorac Surg. 2001; 20:684-687.
3. Liptay MJ, Basu S, Hoaglin MC, Freedman N, Faber LP, Warren WH, Hammoud ZT, Kim AW. Diffusion lung capacity for carbon monoxide (DLCO) is an independent prognostic factor for long-term survival after curative lung resection for cancer. J Surg Oncol; 2009; 100:703-707.
4. Guo X, Cao H, Xu J, Yu J, Zheng C, Meng L, Du J. Forced vital capacity predicts long-term survival for curative-resected NSCLC. Med Oncol. 2014; 31:146.
5. Wang J, Olak J, Ultmann RE, Ferguson MK. Assessment of pulmonary complications after lung resection. Ann Thorac Surg. 1999; 67:1444-1447.
6. Markos J, Mullan BP, Hillman DR, Musk AW, Antico VF, Lovegrove FT, Carter MJ, Finucane KE. Preoperative assessment as a predictor of mortality and morbidity after lung resection. Am Rev Respir Dis. 1989; 139:902-910.
7. Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis. 2009; 30:1073-1081.
8. Cannon NA, Meyer J, Iyengar P, Ahn C, Westover KD, Choy H, Timmerman R. Neutrophil-lymphocyte and platelet-lymphocyte ratios as prognostic factors after stereotactic radiation therapy for early-stage non-small-cell lung cancer. J Thorac Oncol. 2015; 10:280-285.
9. Song YJ, Wang LX, Hong YQ, Lu ZH, Tong Q, Fang XZ, Tan J. Lymphocyte to monocyte ratio is associated with response to first-line platinum-based chemotherapy and prognosis of early-stage non-small cell lung cancer patients. Tumour Biol. 2015; 37:5285-5293.
10. Zhang H, Zhang L, Zhu K, Shi B, Yin Y, Zhu J, Yue D, Zhang B, Wang C. Prognostic Significance of Combination of Preoperative Platelet Count and Neutrophil-Lymphocyte Ratio (COP-NLR) in Patients with Non-Small Cell Lung Cancer: Based on a Large Cohort Study. PLoS One. 2015; 10:e126496.
11. Yang HF, Kao TW, Wang CC, Peng TC, Chang YW, Chen WL. Serum white blood cell count and pulmonary function test are negatively associated. Acta Clin Belg. 2015; 70:419-424.
12. McKeever T, Saha S, Fogarty AW. The association between systemic inflammatory cellular levels and lung function: a population-based study. PLoS One. 2011; 6:e21593.
13. O'Callaghan DS, O'Donnell D, O'Connell F, O'Byrne KJ. The role of inflammation in the pathogenesis of non-small cell lung cancer. J Thorac Oncol. 2010; 5:2024-2036.
14. Pinato DJ, Shiner RJ, Seckl MJ, Stebbing J, Sharma R, Mauri FA. Prognostic performance of inflammation-based prognostic indices in primary operable non-small cell lung cancer. Br J Cancer. 2014; 110:1930-1935.
15. Zhang H, Xia H, Zhang L, Zhang B, Yue D, Wang C. Clinical significance of preoperative neutrophil-lymphocyte vs platelet-lymphocyte ratio in primary operable patients with non-small cell lung cancer. Am J Surg. 2015; 210:526-535.
16. Aronson D, Roterman I, Yigla M, Kerner A, Avizohar O, Sella R, Bartha P, Levy Y, Markiewicz W. Inverse association between pulmonary function and C-reactive protein in apparently healthy subjects. Am J Respir Crit Care Med. 2006; 174:626-632.
17. van Rooyen Y, Schutte AE, Huisman HW, Eloff FC, Du Plessis JL, Kruger A, van Rooyen JM. Inflammation as Possible Mediator for the Relationship Between Lung and Arterial Function. Lung. 2016; 194:107-115.
18. Wang L, Long W, Li PF, Lin YB, Liang Y. An Elevated Peripheral Blood Monocyte-to-Lymphocyte Ratio Predicts Poor Prognosis in Patients with Primary Pulmonary Lymphoepithelioma-Like Carcinoma. PLoS One. 2015; 10:e126269.
19. Cannon NA, Meyer J, Iyengar P, Ahn C, Westover KD, Choy H, Timmerman R. Neutrophil-lymphocyte and platelet-lymphocyte ratios as prognostic factors after stereotactic radiation therapy for early-stage non-small-cell lung cancer. J Thorac Oncol. 2015; 10:280-285.
20. Yang HF, Kao TW, Wang CC, Peng TC, Chang YW, Chen WL. Serum white blood cell count and pulmonary function test are negatively associated. Acta Clin Belg. 2015; 70:419-424. doi: 10.1179/2295333715Y.0000000051.
21. Riediker M, Cascio WE, Griggs TR, Herbst MC, Bromberg PA, Neas L, Williams RW, Devlin RB. Particulate matter exposure in cars is associated with cardiovascular effects in healthy young men. Am J Respir Crit Care Med. 2004; 169:934-940.
22. Clapp BR, Hingorani AD, Kharbanda RK, Mohamed-Ali V, Stephens JW, Vallance P, MacAllister RJ. Inflammation-induced endothelial dysfunction involves reduced nitric oxide bioavailability and increased oxidant stress. Cardiovasc Res. 2004; 64:172-178.
23. Sin DD, Man SF. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation. 2003; 107:1514-1519.
24. Win T, Jackson A, Sharples L, Groves AM, Wells FC, Ritchie AJ, Laroche CM. Relationship between pulmonary function and lung cancer surgical outcome. Eur Respir J. 2005; 25:594-599.
25. Burney PG, Hooper R. Forced vital capacity, airway obstruction and survival in a general population sample from the USA. Thorax. 2011; 66:49-54.
26. Suda R, Tanabe N, Ishida K, Kato F, Urushibara T, Sekine A, Nishimura R, Jujo T, Sugiura T, Shigeta A, Sakao S, Tatsumi K. Prognostic and pathophysiological marker for patients with chronic thromboembolic pulmonary hypertension: Usefulness of diffusing capacity for carbon monoxide at diagnosis. Respirology. 2016; doi:10.1111/resp.12883.
27. Bousamra MN, Presberg KW, Chammas JH, Tweddell JS, Winton BL, Bielefeld MR, Haasler GB. Early and late morbidity in patients undergoing pulmonary resection with low diffusion capacity. Ann Thorac Surg. 1996; 62:968-974, 974-975.
28. Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002; 3:991-998.
29. Pillay J, Kamp VM, van Hoffen E, Visser T, Tak T, Lammers JW, Ulfman LH, Leenen LP, Pickkers P, Koenderman L. A subset of neutrophils in human systemic inflammation inhibits T cell responses through Mac-1. J Clin Invest. 2012; 122:327-336.
30. Siveen K S, Kuttan G. Role of macrophages in tumour progression. Immunol Lett. 2009; 123:97-102.
31. Gordon S, Martinez FO. Alternative activation of macrophages: mechanism and functions. Immunity. 2010; 32:593-604.
32. Goldstraw P, Crowley J, Chansky K, Giroux DJ, Groome PA, Rami-Porta R, Postmus PE, Rusch V, Sobin L. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol. 2007; 2:706-714.
33. Standardization of Spirometry, 1994 Update. American Thoracic Society. Am J Respir Crit Care Med. 1995; 152:1107-1136.
34. Jensen RL, Crapo RO. Diffusing capacity: how to get it right. Respir Care. 2003; 48:777-782.

