INTRODUCTION
Hepatocellular carcinoma (HCC) is the 5th most common cancer in the world and the 3rd leading cause of tumor-related deaths [1, 2]. Hepatic resection (HR) is a curative treatment for HCC [3], but the incidence of postoperative recurrence is high [6]. Additionally, approximately 70% of cases are not suitable for curative treatment options because they are diagnosed at non-early stages according to the Barcelona Clinic Liver Cancer (BCLC) staging system [4, 5]. At non-early stages, transarterial chemoembolization (TACE) is the recommended treatment option for HCC [7]. The best candidates for TACE are asymptomatic patients with well-preserved liver function and a solitary or limited multifocal HCC without vascular invasion or extrahepatic spread [8, 9]. However, TACE alone results in incomplete tumor necrosis [10], and the 5-year survival rate is only 6%–19% [11, 12].
Intermediate HCC comprises a highly heterogeneous population that differs according to tumor load, age, and liver function. Bolondi et al. [13] proposed a four-stage sub-classification of BCLC-B, and radical therapy was suggested to replace TACE in BCLC-B1 (Table 1). Several studies [14, 15, 16, 17] also suggested that HR was a safe and effective method for intermediate-stage HCC (including BCLC-B1 or BCLC-B2, Table 1). HR combined with TACE was considered the effective method for some intermediate-stage HCC patients, and might improve the effect of the TACE treatment. However, there has been a lack of evidence in whether to perform HR after TACE on non-early HCC patients.
Table 1: Four stages sub-classification of BCLC-B
B1 |
B2 |
B3 |
B4 |
|
|---|---|---|---|---|
Child-Pugh |
5-6-7 |
5-6 |
7 |
8-9* |
Within Ut-7 |
In |
Out |
Out |
Any |
ECOG |
0 |
0 |
0 |
0-1 |
PVT |
No |
No |
No |
No |
1st Option |
TACE |
TACE or TARE |
Best supportive care |
|
Alternative |
Liver Transplantation |
Sorafenib |
Research Trail |
Liver |
TARE= transarterial radioembolization. Ut-7 = this criterion combines the number of nodules and the size of the largest tumor, with the sum being no more than 7. Examples include two tumors up to 5 cm in size (2 + 5 = 7), three tumors up to 4 cm in size (3 + 4 = 7), etc. A single large tumor up to 6 cm in size (1 + 6 = 7) would also meet the criterion.
*, with severe/refractory ascites and/or jaundice; **only if Up-to-7 IN and PSO.
We conducted this retrospective study to clarify the role of HR after TACE in patients with large/multifocal HCC lesions. Therapy-related mortality and liver function change were recorded, and long-term survival of those patients treated with TACE+HR was analyzed. In addition, how to determine suitable patients for HR after TACE was further analyzed.
RESULTS
Baseline status of patients in our study
From January 2006 to December 2010, 110 patients with large/multifocal HCC at our institution were included in this study. A total of 132 patients were excluded from the study because they met the exclusion criteria (Figure 1). TACE was recommended as the initial treatment for all 110 patients. A total of 49 patients were also treated with HR (T+R group), leaving 61 patients who received TACE only (T group). The mean follow-up time was 48.2 months (range: 1–101 months) in the T+R group, and 20.9 months (range: 3–67 months) in the T group. The mean number of TACE procedures before HR was 1.74 ± 1.10 (range: 1–4), and the mean time interval between TACE and HR was 3.0 ± 3.2 months in the T+R group.
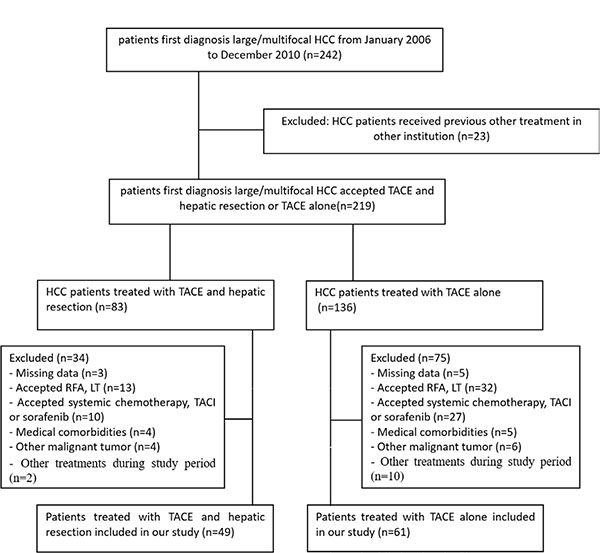
Figure 1: Flow diagram shows exlusion criteria. HCC= Hepatocellular carcinoma; TACE= Transarterial chemoembolization; RFA=Radiofrequency ablation; LT=liver transplantation; TACI=transarterial chemoinfusion.
A comparison of the clinical and demographic characteristics of the patients in these two groups revealed no significant differences (Table 2).
Table 2: Baseline character of T+R group and T group
T+R group |
T group |
P-Value |
|
|---|---|---|---|
Age (mean ± SD) |
50.31 ± 9.15 |
51.74 ± 12.06 |
0.067# |
Sex |
0.764$ |
||
Male |
45(91.8) |
58 (95.1) |
|
Female |
4(8.2) |
3(4.9) |
|
Etiology |
0.765% |
||
HBV |
44(89.8) |
57(93.4) |
|
HCV |
3(6.1) |
2(3.3) |
|
Other |
2(4.1) |
2(3.3) |
|
Child-Pugh |
0.884$ |
||
A |
47 (95.9) |
57 (93.4) |
|
B |
2 (4.1) |
4 (6.6) |
|
BCLC-B subclassification |
0.018^ |
||
BCLC-B1 |
17(34.7) |
35(57.4) |
|
BCLC-B2 |
32(65.3) |
26(42.6) |
|
AFP(ng/ml) |
0.784^ |
||
≥ 200ng/ml |
33 (67.3) |
45 (73.8) |
|
< 200ng/ml |
16 (32.7) |
16 (26.2) |
|
Tumor diameter (cm) |
7.22 ± 3.18 |
6.80 ± 3.35 |
0.509# |
Number(1/2/3/≥ 4) |
0.998^ |
||
1 |
26 (53.1) |
32 (52.5) |
|
2 |
15 (30.6) |
19 (31.1) |
|
≥3 |
8(16.3) |
10(16.4) |
|
Tumor Capsule |
0.901^ |
||
Yes |
43(87.8) |
54(88.5) |
|
No |
6 (12.2) |
7 (11.5) |
Note. - Unless indicated, data are numbers of patients, and numbers in parentheses are percentages
# Independent-samples t test was used. $ Continuity correction was used. % Fisher exact test was used. ^ Pearson X2 test was used.
Safety of resection after TACE
All of the hepatic resections (100%) in the T+R group were successfully performed. The Clavien-Dindo classification [20] of surgical complications was used to assess post-resection complication. Class I complications occurred in 29 patients, including vomiting (n = 13), abdominal pain (n = 12), pleural effusion (n = 7) and fever (n = 11). Class II complications occurred in 4 patients, including bile leakage (n = 2) and wound infection (n = 2). Only 1 class V complication (gastrointestinal hemorrhage, n = 1) occurred after hepatic resection. At 1 week after resection, liver function was altered from baseline, but it recovered in 1 month (Table 3).
Table 3: Liver function test at 1 weeks and 1 months after hepatic resection compared to baseline liver function test
Baseline Value |
1 week after resection |
P-Value |
1 month after resection |
P-Value |
|
|---|---|---|---|---|---|
AST |
42.94 ± 17.72 |
175.62 ± 145.16 |
0.002 |
45.25 ± 34.43 |
0.798 |
ALT |
38.06 ± 23.78 |
179.06 ± 177.97 |
0.005 |
46.50 ± 34.03 |
0.410 |
TBILI |
15.06 ± 4.8 |
26.09 ± 11.59 |
0.000 |
15.01 ± 8.12 |
0.980 |
ALB |
42.39 ± 4.42 |
34.39 ± 6.64 |
0.000 |
39.62 ± 4.57 |
0.036 |
PT (sec) |
13.29 ± 0.89 |
15.51 ± 1.67 |
0.000 |
13.61 ± 0.77 |
0.063 |
Abbreviations: AST=aspartate aminotransferase; ALT=alanine aminatransferase; TBILI=Total bilirubin; ALB=albumin; PT=prothrombin time.
Overall survival and the 1-yr, 2-yr, and 3-yr survival rates
The median OS time in the T+R group and T group were 47.0 (95% CI 41.4–52.6) months and 20.0 (95% CI 16.4–23.6) months, respectively (P < 0.001, Figure 2). The 1-, 2-, and 3- year survival rates in the T+R group were 89.8%, 79.4%, 59.1%, respectively; the 1-, 2-, and 3- year survival rates in the T group were 75.1%, 61.5%, and 15.1%, respectively.
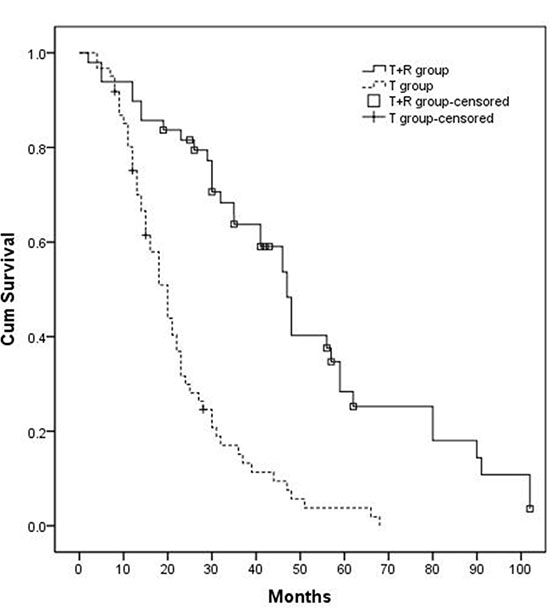
Figure 2: Kaplan-Meier curves of OS in patients with large/multifocal HCC who underwent TACE-resection (T+R group) or TACE (T group). The OS of T+R group (n = 49) was 47.0 months, and the OS of T group (n = 61) was 20.0 months (P < 0.001).
Univariate and multivariate analysis
Univariate analyses revealed that T+R treatment method, single tumor lesion, good tumor response, and lower baseline AFP were associated with better OS. These 4 factors were selected as candidates for multivariate analysis. The multivariate Cox proportional hazard model revealed that T + R treatment method (HR = 0.24, 95% CI = 0.151–0.395. P < 0.001), good tumor response (HR = 0.27, 95% CI = 0.160–0.469. P = 0.043) and lower baseline AFP (HR = 0.59, 95% CI = 0.366–0.965. P < 0.001) were identified as independent prognostic factors for OS (Table 4).
Table 4: Univariate analysis and multivariate analysis of OS
Factors |
No. of Patients |
Median OS |
P-Value |
|---|---|---|---|
Turmor No. |
0.012* |
||
1 |
30 |
48.0 (38.3–57.7) |
|
≧ 1 |
19 |
41.0 (30.7–51.3) |
|
mRECIST before resection |
0.021* |
||
Good Response |
37 |
48.0 (45.9–50.1) |
|
Poor Response |
12 |
35.0 (14.3–55.7) |
|
Baseline AFP |
0.059* |
||
< 200 ng/ml |
16 |
47.0 (12.3–22.8) |
|
≧ 200 ng/ml |
33 |
46.0 (39.7–52.3) |
|
Reduction of AFP& |
0.011^ |
||
> 50% |
22 |
48.0 (43.6–52.5) |
|
< 50% |
11 |
19.0 (5.3–32.7) |
Note-Data in parentheses are 95% CIs.
*Log-rank test was used. &Only 33 patients with baseline AFP value ≥ 200 ng/ml were included in the univariate analyses.
^Breslow test was used.
Sub-group analysis according to tumor response before hepatic resection
Modified RECIST (mRECIST) criteria was used to sub-categorize patients prior to HR. Patients with complete response (CR) or partial response (PR) were defined as the T+R-GR group; patients with stable disease (SD) or progressive disease (PD) were defined as the T + R-PR group. Thirty-seven (37) patients in the T+R group exhibited good response, including 14 patients with CR and 23 patients with PR. Only 12 patients did not exhibit a good response, including 6 patients with SD and 6 patients with PD. The median OS time of the T+R-GR and T+R-PR groups were 48.0 (95% CI 45.9–50.1) months and 35.0 (95% CI 14.3–55.7) months. OS of these 2 sub-groups were compared to the T group (Figure 3A: T+R-GR group vs. T group: P < 0.001; Figure 3B: T+R-PR group vs. T group: P = 0.135.).
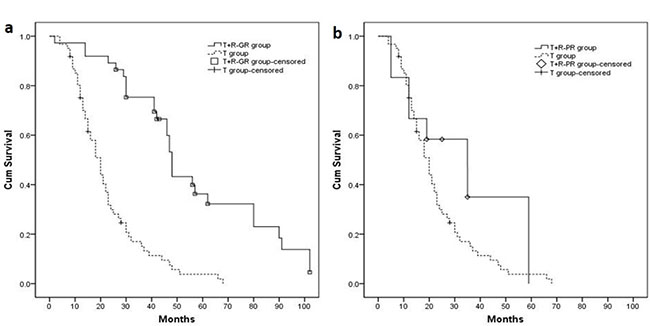
Figure 3: (A) Patients with good response before hepatic resection (T+R–GR group, n = 37, median OS = 48.0 months) had longer median OS compared with T group (P < 0.001). (B) Patients with poor response before hepatic resection (T+R–PR group, n = 12, median OS = 35.0 months) had no significant difference with T group (P = 0.135).
Sub-group analysis according to baseline AFP level
Thirty-three (33) patients with a high baseline AFP level (≥ 200 ng/ml) in the T+R group were defined as the T+R-highAFP group, and 16 patients with low baseline AFP level (< 200 ng/ml) were defined as the T+R-lowAFP group. The median OS of T+R-highAFP group and T+R-lowAFP group were 46.0 (95% CI 39.7–52.3) months and 47.0 (95% CI 22.8–71.2) months. OS of these 2 sub-groups were compared to the T group (Figure 4A: T+R-highAFP group vs. T group: P < 0.001; Figure 4B: T+R-lowAFP group vs. T group: P < 0.001).
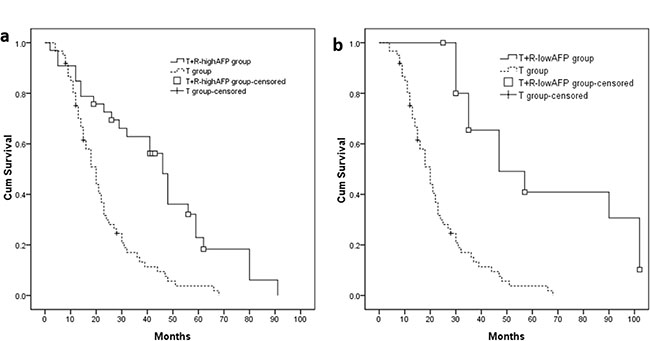
Figure 4: (A, B) Patients with high baseline AFP value (T+R-highAFP group, n = 33, median OS = 46.0 months, P < 0.001) or low baseline AFP value (T+R-lowAFP group, n = 16, median OS = 47.0 months, P < 0.001) had longer median OS compared with T group.
Sub-group analysis according to AFP reduction before hepatic resection
Twenty-two (22) patients that experienced an AFP level decrease > 50% before resection were defined as the T+R-AFP-a group; 11 patients whose AFP reduction was < 50% were defined as the T+R-AFP-b group. The median OS of the T+R-AFP-a and T+R-AFP-b groups were 48.0 (95% CI 43.6–52.5) months and 19.0 (95% CI 5.3–32.7) months. The median OS of these 2 sub-groups were compared to the T group (Figure 5A: T+R-AFP-a group vs. T group: P < 0.001; Figure 5B: T+R-AFP-b group vs. T group: P = 0.247).
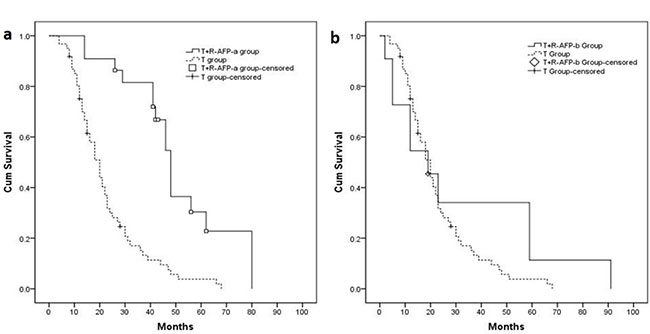
Figure 5: (A) Patients in the T+R-AFP-a group (n = 22, median OS = 48.0 months) had longer median OS compared with T group (P < 0.001). (B) Patients in the T+R-AFP-b group (n = 12, median OS = 19.0 months) had no significant difference with T group (P = 0.247).
DISCUSSION
Large/multifocal HCC is typically treated with TACE [8]. The 1-, 2- and 3-year survival rates of these patients have historically only been 75%, 47%, and 26%, respectively [21], and we found similar results in this retrospective study. HR may be one treatment option when patients did not show a complete response following TACE [24], and sequential resections may prolong survival [22, 23]. However, few studies have examined the safety and long-term efficacy of patients who underwent HR following TACE.
Due to advances in HR techniques and peri/post-operative care, HR for HCC can be performed without mortality, even in cirrhotic patients [25]. In our study, the success rate of resection after TACE was 100%, and only 1 class V complication occurred after resection. AST, ALT, TBILI, ALB and PT-sec levels were altered within 1 week after resection (P < 0.05), but liver function recovered in 1 month (P > 0.05, except the ALB level). We concluded that HR could be safely performed after TACE in patients with large/multifocal HCC.
The main limitations of TACE are incomplete necrosis due to the dual blood supply around the capsule, multiple collateral circulation, or recanalization of the embolized artery [26]. Furthermore, patients with large HCC rarely achieve complete remission by undergoing TACE alone. To overcome these limitations and eliminate residual cancer cells left behind by TACE, a combined treatment regimen of HR following TACE was performed at our institution. The median OS of patients who underwent TACE and HR treatment was longer than that of patients who underwent TACE alone (47.0 months vs. 20.0 months, P < 0.05). Moreover, our data showed that the 1- , 2- and 3- year survival rates in the T+R group were higher than those of patients who underwent HR alone (89.8%, 79.4%, 59.1% vs. 83%, 62%, 49%) [27]. Therefore, HR following TACE is suggested to be more safe and effective for large/multifocal HCC patients.
It was difficult to select suitable patients for the combined HR + TACE treatment. We assumed that tumor response after TACE, baseline AFP value, and reduction of AFP level after TACE might be independent prognosis factors that impact the survival in patients who underwent HR after TACE.
Detection of arterial lesions by enhanced CT/MR was used to evaluate viable tumors according to mRECIST [19, 28]. mRECIST is correlated with long-term survival [29, 30], and it was used to determine the amount of tumor necrosis induced by TACE. Furthermore, Lei’s study [31] suggested that mRECIST may represent selection criterion for intermedian-HCC for resection. Adachi E et al. [32] reported that preoperative TACE resulted in better disease-free survival rates when complete tumor necrosis was induced. However, other studies [33, 34] have found that the extent of tumor necrosis was actually not a favorable prognostic factor. In this study, patients with good tumor response in the T+R group experienced longer OS than those in the T group (P<0.001). In contrast, the OS of patients with poor tumor response in the T+R group was not significantly different with that of the T group (P = 0.135). We presumed that the tumor response before HR might be a valuable prognostic factor for large/multifocal HCC patients.
AFP values correlated with tumor activity, and it has been shown that AFP is an important determinant of the degree of malignancy of HCC in cytological studies [35, 36, and 37]. A close relationship exists between the level of serum AFP and HCC recurrence/metastasis [38, 39]. AFP values have been considered a significant independent predictor of large/multifocal patients [40]. We presumed that a reduction in the AFP level after TACE might indicate the preoperative TACE had reduced tumor activity, which in turn might lead to better results in the following resection procedure. In our study, patients with AFP reduction > 50% before HR showed longer OS than TACE alone (P < 0.001). In contrast, patients with AFP reduction < 50% did not show a difference in OS from the T group (P = 0.247).
There were several limitations of our study. First, our study was retrospective, and therapeutic options (TACE-HR vs. TACE alone) in patients with large/multifocal HCC were individually determined on the basis of the attending physician’s preference. Second, the numbers of patients were limited, especially in the T+R-PR (12 patients) and T+R-AFP-b groups (11 patients). The low patient numbers in these groups might have led to the observed differences in OS. We suggest that an adequately powered, prospective, randomized trial of TACE combined with resection is needed for further study.
In conclusion, HR following TACE can be safely performed and is more effective than TACE alone. Large/multifocal HCC patients with poor tumor response, higher baseline AFP level, and AFP decrease less than 50% before HR might not be suitable for HR after TACE.
MATERIALS AND METHODS
Study design and patient selection
This study protocol was approved by the ethics committee of our hospital and written informed consent was obtained from the patients. We reviewed the electronic medical records of 242 consecutive patients diagnosed with intermediate HCC from January 2006 to December 2010 (all follow-ups were completed by December 2014) at the third affiliated Hospital of Sun Yat-sen University. HCC was diagnosed according to noninvasive criteria in accordance with the European Association for the Study of the Liver/American Association for the study of Liver Disease guidelines.
The inclusion criteria for the study population were as follows: (a) patients between 18 and 75 years of age, (b) an ECOG performance status of 0, (c) Child-Pugh class A or B liver disease, and (d) single large HCC (> 5 cm) or multifocal HCC without vascular invasion or extra-hepatic spread [12, 17]. Patients were excluded from this study if they had any of the following: (a) previously undergone local-regional therapies (radiofrequency ablation, percutaneous ethanol injection, or iodine 125 seed implantation), hepatic resection, and liver transplantation as an initial treatment, or TACE at other institutions; (b) underwent sorafenib therapy, systemic chemotherapy, or transarterial chemoinfusion (TACI) during our study; (c) underwent other treatment methods (radiofrequency ablation, percutaneous ethanol injection, or iodine 125 seed implantation, et al.) in addition to TACE+HR or TACE during this study; (d) had serious medical comorbidities; or (e) currently had or had a history of malignant tumors in addition to HCC.
Transarterial chemoembolization and hepatic resection procedure
The clinical treatment strategy of HCC patients was determined by a multidisciplinary liver tumor conference at our hospital. Large/multifocal HCC patients who received TACE as their initial treatment were included in this study. Patients were suggested to undergo HR after 1 – 4 TACE procedures, and the indications for surgery in our department were as follows: (a) a Child-Pugh A/B classification, total bilirubin < 51.30 μmol/L, serum total protein > 60 g/L or serum albumin > 30 g/L, and a prothrombin time ≤ 17 s; (b) no obvious presence of hepatic decompensation; (c) anatomic resection was our preferred surgical method for hepatic resection for multiple nodules in one segment or in neighboring segments; (d) an appropriate residual liver volume evaluated by CT; and (e) no contraindications for major surgery indicated by a multidisciplinary evaluation. Patients in accordance with these indications were suggested to receive HR after TACE. If patients and their relatives agreed to perform HR, resection would be performed at our institute by 4 surgeons who had 10–15 years of experience in the procedure. Patients who refused HR underwent TACE alone.
TACE was performed with a 5-French catheter (Cook, Bloomington, Indiana, USA) or microcatheter (Renegade, Boston Scientific, Natick, MA, USA, or Progreat, Terumo, Tokyo, Japan) as selectively as possible through the lobar, segmental, or subsegmental arteries, depending on the tumor distribution and hepatic functional reserve. Initially, an emulsion of lipiodol (Lipiodol Ultrafluido, Guerbet, France) and doxorubicin hydrochloride was administered into the feeder vessels. The volume of lipiodol ranged from 2 to 20 ml, and the amount of doxorubicin ranged from 20 to 60 mg. 300–500 μm gelatin sponge particles (Cook, Bloomington, Indiana, USA) were mixed with contrast material, and then administered into the feeder vessels until stasis of the arterial flow was achieved. A solution of lobaplatin at a concentration of 0.5 mg/ml was infused into the tumor feeder vessels at a rate of 5 ml/min. The total amount of lobaplatin used ranged from 20 to 50 mg depending on the patient’s body weight.
Hepatic resection was performed under general anesthesia via an L-shaped laparotomy or bilateral subcostal incision with a midline extension. Intraoperative ultrasound (US) was routinely performed to evaluate the tumor burden, liver remnant, and the resection margin. An intermittent Pringle’s maneuver [18] for 20 min and a 5-minute clamp-free interval were used to reduce blood loss during resection. Two Jackson–Pratt drains (size 10) were placed after surgery.
Follow-up and repeated TACE
The clinical, laboratory, and radiologic records were reviewed. Additionally, laboratory liver function tests, including serum total bilirubin, albumin and prothrombin time, at 1 week and 1 month after HR were used to evaluate the safety of HR after TACE.
The tumor response was evaluated with contrast-enhanced CT/MR imaging according to the modified Response Evaluation Criteria in Solid Tumors (mRECIST) [19]. Baseline tumor measurements were performed in both groups. In the T+R group, CT/MR exam was performed at 1 month after resection and every 3 months thereafter. During the follow-up period, patients in both groups with recurrent tumor, according to contrast-enhanced CT/MR images, underwent repeated TACE if the Child-Pugh status remained at class A or B and there was no evidence of hepatic decompensation (e.g., uncontrolled ascites or hepatic encephalopathy). Contrast-enhanced CT/MR was performed at 1 month after the initial TACE procedure in the T group, and again every 1 – 2 months. Further TACE therapy was based on mRECIST on contrast-enhanced CT/MR imaging, and decided by consensus.
Furthermore, we compared and analyzed the overall survival (OS) between the T+R group and the T group for the total study population. OS was defined as the time from the date of the first TACE procedure until death or the last follow-up.
Statistical analysis
All statistical analyses were performed using SPSS version 16.0 (SPSS Inc, Chicago, IL, USA). Pearson chi-square tests, continuity correction, independent-samples t-tests, and Fisher’s exact tests were used to determine significant differences between the groups. A Wilcoxon signed rank test was used to determine the difference in liver function test values before and after resection. The OS was calculated for both groups using Kaplan-Meier methods. Univariate analyses were performed by the log-rank test, and variables with a P value of less than 0.05 at univariate analysis were entered into a multivariate analysis. Multivariate analysis by Cox proportional hazards model was performed to identify independent prognostic factors. A P-value of less than 0.05 was considered significant.
ACKNOWLEDGMENTS
None.
CONFLICTS OF INTEREST
The authors have declared that they have no conflicts of interest.
GRANT SUPPORT
This work was supported by the National Natural Science Foundation of China (nos. 81172193 and 81571774).
REFERENCES
1. El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007; 132:2557–2576.
2. Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, Feuer EJ, Thun MJ. Cancer statistics, 2005. CA Cancer J Clin. 2005; 55:10–30.
3. Forner A, Reig ME, de lope CR, Bruix J. Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Did. 2010; 30:61–74.
4. Bruix J, Sherman M. Practice Guidelines Committee. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology. 2005; 42:1208–1236.
5. El-Serag HB, Marrero JA, Rudolph L, Reddy KR. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology. 2008; 134:1752–1763.
6. Minagawa M, Makuuchi M, Takayama T, Kokudo N. Selection criteria for repeat hepatectomy in patients with recurrent hepatocellular carcinoma. Ann Surg. 2003; 238:703–710.
7. Forner A, Llovet JM, Bruix J. Lancet 2012; 379:1245–1255.
8. Bruix J, Sala M, Llovet JM. Chemoembolization for hepatocellular carcinoma. Gastroenterology. 2004: 127:S179–S188.
9. Bruix J, Reig M, Sherman M. Evidence-Based Diagnosis, Staging, and Treatment of Patients With Hepatocellular Carcinoma. Gastroenterology. 2016; 150:835–853.
10. Sasaki Y, Imaoka S, Kasugai H, Fujita M, Kawamoto S, Ishiguro S, Kojima J, Ishikawa O, Ohigashi H, Furukawa H, Koyama H, Iwanaga T. A new approach to chemoembolization therapy for hepatoma using ethiodized oil, cisplatin, and gelatin sponge. Cancer. 1987; 60:1194–1203.
11. O'Suilleabhain CB, Poon RT, Yong JL, Ooi GC, Tso WK, Fan ST. Factors predictive of 5-year survival after transarterial chemoembolization for inoperable hepatocellular carcinoma. Br J Surg. 2003; 90:325–331.
12. Ono Y, Yoshimasu T, Ashikaga R, Inoue M, Shindou H, Fuji K, Araki Y, Nishimura Y. Long-term results of lipiodol-transcatheter arterial embolization with cisplatin or doxorubicin for unresectable hepatocellular carcinoma. Am J Clin Oncol. 2000; 23:564–568.
13. Bolondi L, Burroughs A, Dufour JF, Galle PR, Mazzaferro V, Piscaglia F, Raoul JL, Sangro B. Heterogeneity of patients with intermediate (BCLC B) Hepatocellular Carcinoma: proposal for a subclassification to facilitate treatment decisions. Semin Liver Dis. 2012; 32:348–359.
14. Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology. 2003; 37:429–442.
15. Shuto T, Hirohashi K, Kubo S, Tanaka H, Yamamoto T, Ikebe T, Kinoshita H. Efficacy of major hepatic resection for large hepatocellular carcinoma. Hepatogastroenterology. 1999; 46:413–416.
16. Hanazaki K, Kajikawa S, Shimozawa N, Shimada K, Hiraguri M, Koide N, Adachi W, Amano J. Hepatic resection for large hepatocellular carcinoma. Am J Surg. 2001; 181:347–353.
17. Ng KK, Vauthey JN, Pawlik TM, Lauwers GY, Regimbeau JM, Belghiti J, Ikai I, Yamaoka Y, Curley SA, Nagorney DM, Ng IO, Fan ST, Poon RT; International Cooperative Study Group on Hepatocellular Carcinoma. Is hepatic resection for large or multinodular hepatocellular carcinoma justified? Results from a multi-institutional database. Ann Surg Oncol. 2005; 12:364–373.
18. Pringle JH. Notes on the arrest of hepatic hemorrhage due to trauma. Ann Surg. 1908; 48:541–549.
19. Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010; 30:52–60.
20. Dindo D, Demartines N, Clavien PA. Classification of surgical complications. A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004; 240:205–213.
21. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003; 362:1907–1917.
22. Lau WY, Leung TW, Lai BS, Liew CT, Ho SK, Yu SC, Tang AM. Preoperative systemic chemoimmunotherapy and sequential resection for unresectable hepatocellular carcinoma. Ann Surg. 2001; 233:236–241.
23. Ku Y, Iwasaki T, Tominaga M, Fukumoto T, Takahashi T, Kido M, Ogata S, Takahashi M, Kuroda Y, Matsumoto S, Obara H. Reductive surgery plus percutaneous isolated hepatic perfusion for multiple advanced hepatocellular carcinoma. Ann Surg. 2004; 239:53–60.
24. Choi SB, Kim KS, Park YN, Choi JS, Lee WJ, Seong J, Han KH, Lee JT. The efficacy of hepatic resection after neoadjuvant transarterial chemoembolization (TACE) and radiation therapy in hepatocellular carcinoma greater than 5 cm in size. J Korean Med Sci. 2009; 24:242–247.
25. Fan ST, Lo CM, Liu CL, Lam CM, Yuen WK, Yeung C, Wong J. Hepatectomy for hepatocellular carcinoma: toward zero hospital deaths. Ann Surg. 1999; 229:322–330.
26. Gotohda N, Kinoshita T, Konishi M, Nakagohri T, Takahashi S, Furuse J, Ishii H, Yoshino M. New indication for reduction surgery in patients with advanced hepatocellular carcinoma with major vascular involvement. World J Surg. 2006; 30:431–438.
27. Lin CT, Hsu KF, Chen TW, Yu JC, Chan DC, Yu CY, Hsieh TY, Fan HL, Kuo SM, Chung KP, Hsieh CB. Comparing hepatic resection and transarterial chemoembolization for Barcelona Clinic Liver Cancer (BCLC) stage B hepatocellular carcinoma: change for treatment of choice? World J Surg. 2010; 34:2155–2161.
28. Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M, Talwalkar J, Gores GJ; Panel of Experts in HCC-Design Clinical Trials. Panel of Experts in HCC-Design Clinical Trials. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008; 100:698–711.
29. Gillmore R, Stuart S, Kirkwood A, Hameeduddin A, Woodward N, Burroughs AK, Meyer T. EASL and mRECIST responses are independent prognostic factors for survival in hepatocellular cancer. J Hepatol. 2011; 55:1309–1316.
30. Shim JH, Lee HC, Kim SO, Shin YM, Kim KM, Lim YS, Suh DJ. Which response criteria best help predict survival of patients with hepatocellular carcinoma following chemoembolization? A validation study of old and new models. Radiology. 2012; 262:708–718.
31. Lei JY, Zhong JJ, Yan LN, Zhu JQ, Wang WT, Zeng Y, Li B, Wen TF, Yang JY; - Liver Surgery Group. Response to transarterial chemoembolization as a selection criterion for resection of hepatocellular carcinomas. Br J Surg. 2016 Jun; 103:881–890.
32. Adachi E, Matsumata T, Nishizaki T, Hashimoto H, Tsuneyoshi M, Sugimachi K. Effects of preoperative transcatheter hepatic arterial chemoembolization for hepatocellular carcinoma. The relationship between postoperative course and tumor necrosis. Cancer. 1993; 72:3593–3598.
33. Harada T, Matsuo K, Inoue T, Tamesue S, Inoue T, Nakamura H. Is preoperative hepatic arterial chemoembolization safe and effective for hepatocellular carcinoma? Ann Surg. 1995; 224:4–9.
34. Choi GH, Kim DH, Kang CM, Kim KS, Choi JS, Lee WJ, Kim BR. Is preoperative transarterial chemoembolization needed for a resectable hepatocellular carcinoma? World J Surg. 2007; 31:2370–2377.
35. Li P, Wang SS, Liu H, Li N, McNutt MA, Li G, Ding HG. Elevated serum alpha fetoprotein levels promote pathological progression of hepatocellular carcinoma. World J Gastroenterol. 2011; 17:4563–4571.
36. Um SH, Mulhall C, Alisa A, Ives AR, Karani J, Williams R, Bertoletti A, Behboudi S. Alpha-fetoprotein impairs APC function and induces their apoptosis. J Immunol. 2004; 173:1772–1778.
37. Li MS, Ma QL, Chen Q, Liu XH, Li PF, Du GG, Li G. Alpha-fetoprotein triggers hepatoma cells escaping from immune surveillance through altering the expression of Fas/FasL and tumor necrosis factor related apoptosis-inducing ligand and its receptor of lymphocytes and liver cancer cells. World J Gastroenterol. 2005; 11:2564–2569.
38. Chan SL, Mo FK, Johnson PJ, Hui EP, Ma BB, Ho WM, Lam KC, Chan AT, Mok TS, Yeo W. New utility of an old marker: serial alpha-fetoprotein measurement in predicting radiologic response and survival of patients with hepatocellular carcinoma undergoing systemic chemotherapy. J Clin Oncol. 2009; 27:446–452.
39. Baig JA, Alam JM, Mahmood SR, Baig M, Shaheen R, Sultana I, Waheed A. Hepatocellular carcinoma (HCC) and diagnostic significance of A-fetoprotein (AFP). J Ayub Med Coll Abbottabad. 2009; 21:72–75.
40. Farinati F, Vanin V, Giacomin A, Pozzan C, Cillo U, Vitale A, Di Nolfo AM, Del Poggio P, Benvegnu' L, Rapaccini G, Zoli M, Borzio F, Giannini EG, Caturelli E, Trevisani F; Italian Liver Cancer (ITA.LI.CA) group. Italian Liver Cancer (ITA.LI.CA) group. BCLC-B hepatocellular carcinoma and transcatheter arterial chemoembolization: a 20-year survey by the Italian Liver Cancer group. Liver Int. 2015; 35:223–231.

