Introduction
Hyponatremia is a common electrolyte disorder in cancer patients, particularly in those who are hospitalized [1]. Although patients with hyponatremia are often asymptomatic, when symptoms do occur they are mainly neurological and include headaches, lethargy, poor concentration, confusion, vomiting, hallucinations and even coma [2]. Mild chronic hyponatremia can lead to marked gait instability, falls, fractures and a higher incidence and duration of hospitalization [3].
The incidence and prevalence of hyponatremia vary depending on the tumor type, clinical setting, and serum sodium cut-off level [4]. The frequency of hyponatremia was estimated at up to 40% in hospitalized patients and 15% in patients with small cell lung cancer (SCLC) [5, 6]. The syndrome of inappropriate anti-diuretic hormone secretion (SIADH) is the main cause of hyponatremia in malignancy [7], with other causes including heart failure, nephritic syndrome, extracellular volume depletion, chemotherapy [5] and target therapies [8]. The onset of hyponatremia has been associated with worst prognosis in several cancers including SCLC, mesothelioma, renal cell carcinoma, gastrointestinal cancer and lymphoma [9, 10, 11, 12, 13, 14, 15, 16]. Furthermore hyponatremia represents a prognostic factor in terminal cancer patients [17], although a prompt correction of serum sodium level is associated to a longer survival and an improvement of clinical condition [18]. Non-small-cell lung cancer (NSCLC) is a poor-prognosis malignancy, which is the leading cause of cancer related death. Often asymptomatic in the early stages, more than half of patients have metastatic disease at time of first diagnosis [19]. The incidence of hyponatremia in NSCLC varies from 1% to 50% [20]. Early recognition and a prompt treatment of this electrolytic imbalance could prevent clinical complications and improve survival [21].
In this study, we aimed to investigate for the first time the prognostic significance of hyponatremia normalization in patients with advanced NSCLC treated with first line therapy.
Results
Patient characteristics
Five hundred and twenty-one patients were treated with first-line therapies at our institutions. Of these, 433 patients (299 males and 134 female) were included in this analysis, whilst 88 were excluded due to lack of complete data.
The median age was 66 years (range 25−86) and the majority were current or former smokers (364 patients, 84%). Histology was adenocarcinoma in 278 patients (64%), squamous carcinoma in 101 patients (23%) and other histology in 54 patients (13%). Tumor stage was III in 112 (26%) patients and IV in 321 patients (74%). Most patients (405, 94%) received first-line chemotherapy whilst 28 (6%) an epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor.
Sixty-nine patients (16%) presented with hyponatremia at the start of first-line therapy (Group A), and 368 patients (85%) were eunatremic (Group B). There were no significant differences in terms of clinicopathological characteristics between the groups (Table 1). Among those in group A, 9 patients (13%) had grade ≥ 2 hyponatremia. Forty-one patients (59%) normalized their serum sodium levels during treatment. Thirteen (18%) patients received saline solution with hyponatremia resolution in 8 (12%) patients, 9 (13%) patients underwent fluid restriction with serum sodium normalization in 4 (6%) patients. Three (4%) patients were suffering from SIADH.
Table 1: Patients’ characteristics
Patients |
Overall 433 (%) |
Serum sodium ≥ 136 364 (84) |
Serum sodium ≤ 135 69 (16) |
p |
Gender Male Female |
299 (69) 134 (31) |
250 (69) 114 (31) |
49 (71) 20 (29) |
0.77 |
Age, years Range |
66 25−86 |
66 25−85 |
67 45−86 |
|
ECOG-PS ≥ 2 ECOG-PS < 2 |
41 (9) 392 (91) |
33 (9) 331 (91) |
8 (12) 61 (88) |
0.50 |
Histology Adenocarcinoma Squamous carcinoma Other |
278 (64) 101 (23) 54 (13) |
238 (65) 80 (22) 46 (13) |
40 (58) 21 (30) 8 (12) |
0.15 |
Tumor Stage Stage III Stage IV |
112 (26) 321 (74) |
96 (26) 268 (74) |
16 (23) 53 (77) |
0.65 |
EGFR mutation status Wild-type Mutated |
388 (90) 45 (10) |
324 (89) 40 (11) |
64 (93) 5 (7) |
0.10 |
Smoking history Former/current smoker Never smokers |
364 (84) 69 (16) |
302 (83) 62 (17) |
62 (90) 7 (10) |
0.20 |
Common sites of metastasis Lung Bone Nervous system Liver |
150 (35) 117 (27) 68 (16) 54 (13) |
123 (34) 102 (28) 62 (17) 47 (13) |
27 (39) 15 (22) 6 (9) 7 (10) |
0.30 |
First-line therapy Platinum-based chemotherapy Non platinum-based EGFR- tyrosine kinase inhibitor |
348 (80) 57 (13) 28 (7) |
289 (79) 48 (13) 27 (8) |
59 (86) 9 (13) 1 (1) |
0.18 |
Response to first-line therapy Partial response Stable disease Progression disease |
162 (37) 113 (26) 158 (37) |
142 (39) 91 (25) 135 (37) |
21 (30) 24 (35) 24 (35) |
0.19 |
Overall Survival (OS)
Median OS from first-line therapy was 13.4 months (95% CI 11.4 to 15.9) in the overall population. Two hundred and eighty one patients (64.9%) died during their follow-up.
Median OS was 18.2 months (95% CI 15.1 to 27.2) and 13.0 months (95% CI 10.7 to 15.9) in non-smokers and smokers respectively (p = 0.21). Stratified by gender, median OS was 12.7 months (95% CI 10.5 to 16.5) in males and 16.2 months (95% CI 12.2 to 25.7) in females (p = 0.031). No significant difference was found between patients aged < 70y vs. ≥ 70y (14.4 vs. 13.0 months, p = 0.22). Patients with worse performance status (PS ≥ 2) had a shorter OS compared to those with < 2 (7.2 vs. 14.7 months, p = 0.001).
Based on histology, the median OS was 13.0 (95% CI 9.5 to 17.5) in patients with squamous carcinoma, 14.4 (95% CI 11.6 to 20.2) in patients with adenocarcinoma and 12.7 (95% CI 9.6 to 16.8) in patients with other histologies (p = 0.371). As for EGFR status, patients with EGFR wild-type tumors showed a worst OS compared to mutated tumors (12.7 vs. 20.9 months, p = 0.03).
Stratified by hyponatremia, median OS was 8.8 months (95% CI 6.3 to 12.7) and 15.5 months (95% CI 12.4 to 25.1) in groups A and B respectively (p < 0.001) (Figure 1A).
For patients with hyponatremia (group A), median OS was significantly higher in patients who normalized their sodium levels (11.6 vs. 4.7 months, p =0.0435) (Figure 2A).
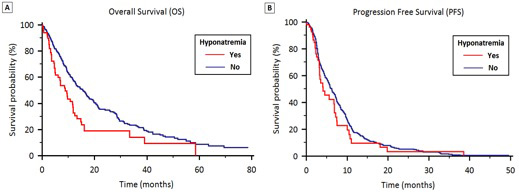
Figure 1: OS (1A) and PFS (1B) stratified by the presence of hyponatremia in patients treated with first-line therapy for locally advanced or metastatic NSCLC.
Progression-Free Survival (PFS)
In the overall study population, median PFS was 5.9 months (95% CI 3.9 to 7.8). Stratified by gender, median PFS was 5.4 months (95% CI 5.0 to 6.1) in males and 6.9 months (95% CI 5.9 to 8.4) in females (p = 0.11).
The median PFS was 6.9 months (95% CI 5.0 to 9.8) and 5.8 months (95% CI 5.3 to 6.5) in non-smokers and smokers (p = 0.49). No significant difference in PFS was found according to aged (< 70y vs. ≥ 70y, 5.8 vs. 6.2 months, p = 0.96), performance status (ECOG-PS ≥ 2 vs. < 2, 4.0 vs. 6.3 months, p = 0.14), or histology (squamous carcinoma vs. adenocarcinoma vs. other histologies: 5.6 vs. 6.3 vs. 4.2 months, respectively). Patients with EGFR wild-type tumors had a worse PFS compared to those with mutated tumors (5.6 vs. 9.4 months, p = 0.02).
Stratified according to hyponatremia, median PFS was 4.1 months (95% CI 3.7 to 4.7) in Group A and 6.3 months (95% CI 5.3 to 8.1) in Group B (p = 0.24) (Figure 1B). In Group A, median PFS was significantly higher in patients who normalized their sodium levels (6.7 vs. 3.3 months, p = 0.011) (Figure 2B).
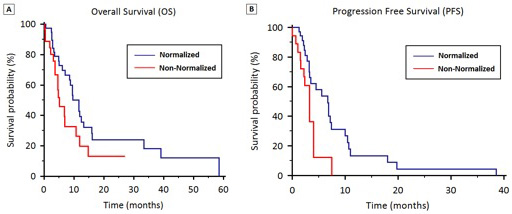
Figure 2: OS (2A) and PFS (2B) stratified by hyponatremia normalization during first-line therapy.
Univariate and Multivariate analyses in the overall study population
Univariate analysis demonstrated that male gender, PS ≥2, tumor stage IV, non-adenocarcinoma histology, wild-type EGFR status and hyponatremia were significantly associated with worse OS (Table 2A). At multivariate analysis, PS, tumor stage, and hyponatremia were predictors of OS (Table 2A).
With respect to PFS, univariate analysis showed that male gender, PS ≥ 2, tumor stage IV and wild-type EGFR status were significantly associated with worse PFS (Table 2B). Multivariate Cox regression analysis revealed that tumor stage IV and wild-type EGFR status were independent prognostic factors for worse PFS (Table 2B).
Table 2A:Univariate and multivariable analysis of predictors of OS in patients treated with first-line therapy for locally advanced or metastatic NSCLC.
OVERALL SURVIVAL IN THE OVERALL POPULATION |
||||
Univariate Cox Regression |
Multivariable Cox regression |
|||
HR (95%CI) |
p-value |
HR (95%CI) |
p-value |
|
Age (≥ 70y vs. < 70y) |
1.16 (0.92−1.47) |
0.222 |
||
Gender (F vs. M) |
0.75 (0.58−0.97) |
0.032 |
0.77 (0.59-1.00) |
0.052 |
ECOG-PS (≥ 2 vs. < 2) |
1.88 (1.27−2.78) |
0.002 |
1.53 (1.00-2.32) |
0.048 |
Smoke status (N vs. Y) |
0.82 (0. 06−1.12) |
0.211 |
||
Tumor Stage (IV vs. III) |
1.57 (1.20−2.05) |
0.001 |
1.70 (1.29-2.24) |
<0.001 |
Histology (AC vs. non-AC) |
0.83 (0.65−1.06) |
0.131 |
0.81 (0.63-1.03) |
0.090 |
EGFR Status (MT vs. WT) |
0.63 (0.42−0.97) |
0.036 |
0.74 (0.48-1.14) |
0.177 |
Hyponatremia (Y vs. N) |
1.71 (1.25−2.34) |
<0.001 |
1.59 (1.14-2.21) |
0.006 |
Significant values are reported in bold.
AC = Adenocarcinoma; CI = confidence interval; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; EGFR = Epidermal growth factor receptor; F = female; HR = hazard ratio; M = male; MT = mutated status; WT = wild-type status
Table 2B: Univariate and multivariable analysis of predictors of PFS in patients treated with first-line therapy for locally advanced or metastatic NSCLC.
PROGRESSION-FREE SURVIVAL IN THE OVERALL POPULATION |
||||
Univariate Cox Regression |
Multivariable Cox regression |
|||
HR (95%CI) |
p-value |
HR (95%CI) |
p-value |
|
Age (≥ 70y vs. < 70y) |
0.99 (0.79−1.25) |
0.964 |
||
Gender (F vs. M) |
0.82 (0.64−0.99) |
0.111 |
0.83 (0.64−1.08) |
0.171 |
ECOG-PS (≥ 2 vs. < 2) |
1.39 (0.90−2.14) |
0.139 |
1.26 (0.80−1.97) |
0.316 |
Smoke status (N vs. Y) |
0.90 (0.67−1.21) |
0.486 |
||
Tumor Stage (IV vs. III) |
1.36 (1.05−1.75) |
0.020 |
1.41 (1.09−1.83) |
0.010 |
Histology (AC vs. non-AC) |
0.91 (0.72−1.15) |
0.433 |
||
EGFR Status (WT vs. MT) |
0.62 (0.42−0.92) |
0.018 |
0.63 (0.42−0.95) |
0.027 |
Hyponatremia (Y vs. N) |
1.23 (0.87−1.73) |
0.245 |
||
Significant values are reported in bold.
AC = Adenocarcinoma; CI = confidence interval; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; EGFR = Epidermal growth factor receptor; F = female; HR = hazard ratio; M = male; MT = mutated status; WT = wild-type status
Univariate and Multivariate analyses in patients with hyponatremia (Group A)
Univariate and multivariate analysis showed that tumor IV disease and failure to normalize sodium neutralization were significantly associated with worse OS (Table 3A).
Univariate analysis showed that adenocarcinoma histotype and sodium normalization were significantly associated with longer PFS (Table 3B). Multivariate Cox regression analysis confirmed the prognostic value of sodium normalization (Table 3B).
Table 3A: Univariate and multivariable analysis of predictors of OS in the 69 patients with hyponatremia at the start of first-line therapy for locally advanced or metastatic NSCLC.
OVERALL SURVIVAL IN HYPONATREMIC PATIENTS |
||||
Univariate Cox Regression |
Multivariable Cox regression |
|||
HR (95%CI) |
p-value |
HR (95%CI) |
p-value |
|
Age (≥ 70y vs. < 70y) |
1.18 (0.64−2.20) |
0.598 |
||
Gender (F vs. M) |
0.99 (0.54−1.84) |
0.990 |
||
ECOG-PS (≥ 2 vs. < 2) |
1.29 (0.62−2.67) |
0.530 |
||
Smoke status (N vs. Y) |
0.67 (0.23−1.64) |
0.462 |
||
Tumor Stage (IV vs. III) |
2.59 (1.25−5.39) |
0.011 |
2.76 (1.31−5.81) |
0.008 |
Histology (AC vs. non-AC) |
1.20 (0.69−2.08) |
0.520 |
||
EGFR Status (MT vs. WT) |
0.90 (0.22−3.72) |
0.889 |
||
Sodium Normalization (N vs. Y) |
1.81 (1.01−3.31) |
0.047 |
1.96 (1.05−3.66) |
0.035 |
Significant values are reported in bold.
AC = Adenocarcinoma; CI = confidence interval; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; EGFR = Epidermal growth factor receptor; F = female; HR = hazard ratio;
M = male; MT = mutated status; WT = wild-type status
Table 3B: Univariate and multivariable analysis of predictors of PFS in the 69 patients with hyponatremia at the start of first-line therapy for locally advanced or metastatic NSCLC.
PROGRESSION-FREE SURVIVAL IN HYPONATREMIC PATIENTS |
||||
Univariate Cox Regression |
Multivariable Cox regression |
|||
HR (95%CI) |
p-value |
HR (95%CI) |
p-value |
|
Age (≥ 70y vs. < 70y) |
0.91(0.44−1.85) |
0.789 |
||
Gender (F vs. M) |
0.64 (0.30−1.36) |
0.250 |
||
ECOG-PS (≥ 2 vs. < 2) |
1.53 (0.66−3.54) |
0.322 |
||
Smoke status (N vs. Y) |
0.52 (0.15−1.74) |
0.288 |
||
Tumor Stage (IV vs. III) |
1.55 (0.73−3.30) |
0.259 |
||
Histology (AC vs. non-AC) |
0.56 (0.28−1.11) |
0.097 |
0.70 (0.33−1.48) |
0.359 |
EGFR Status (MT vs. WT) |
0.62 (0.08−4.57) |
0.641 |
||
Sodium Normalization (N vs. Y) |
2.61 (1.22−5.57) |
0.014 |
2.22 (1.02−5.04) |
0.047 |
Significant values are reported in bold.
AC = Adenocarcinoma; CI = confidence interval; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; EGFR = Epidermal growth factor receptor; F = female; HR = hazard ratio;
M = male; MT = mutated status; WT = wild-type status
Discussion
Hyponatremia is the most common electrolyte disorder encountered in cancer patients [22, 23, 24, 25].
Hyponatremia has been identified as a negative prognostic factor in a number of different malignancies[5, 9 ,26, 27, 28, 29, 30, 31, 32]. In the lung cancer population hyponatraemia is a negative prognostic factor in hospitalized patients and those with advanced-stage disease treated with erlotinib [33, 34]. Furthermore it has been shown to negatively correlate with the performance status [20] as well as tumour status and inflammation in completely resected NSCLC [35]. It is important for physicians to determine and validate prognostic factors in order to optimize and personalize the management of NSCLC. Therefore we evaluated the prognostic value of hyponatremia in 433 NSCLC patients treated with first-line chemotherapy or targeted therapy. We observed a significant difference between eunatremic and hyponatremic patients in OS (15.5 vs. 8.8 months, respectively) but not PFS (6.3 vs. 4.1 months p = 0.24). Futhermore, for the first time, we showed the prognostic significance of hyponatremia normalization in patients with advanced NSCLC treated with first line therapy.
Our results are consistent with those reported in SCLC by Hansen et al. that showed that hyponatremia was associated with a lower median OS in a retrospective study of 453 SCLC patients undergoing chemotherapy. The study showed also that patients who did not fully correct serum sodium values within the first two cycles of chemotherapy had a worse outcome [25]. Hence, our results showed that the correction of sodium levels was associated with significantly higher OS (11.6 vs. 4.7 months) and PFS (6.7 vs. 3.3 months) in patients with NSCLC treated with first-line therapy. Lack of hyponatremia normalization was associated with worse OS and PFS at univariate and multivariate analyses. Thus suggesting that an early detection, a careful monitoring and supportive therapy of hyponatremia can help to improve the medical case and prognosis.
It is therefore important to achieve international consensus about the optimal investigation, diagnosis and management of hyponatraemia in order to optimize the outcome of NSCLC patients.
There are limitations to this study. First, it is a retrospective analysis, which is therefore susceptible to bias in data selection and analysis. A prospective study would be useful to validate these results.
Secondly, the management of hyponatremia was not standardized in all patients and therefore it is not possible to certain whether failure to normalize serum sodium was a reflection of the overall clinical scenario or sub-optimal medical management. Finally, concurrent drugs cannot be fully accounted for could influence the cause and course of hyponatremia.
Our results confirm the prognostic value of low serum sodium in NSCLC patients treated with first-line therapy and underline the importance of a prompt and effective correction of hyponatremia in lung cancer patients.
Materials and Methods
Study population and data collection
The study population included adult patients with histologically or cytologically confirmed diagnosis of locally advanced or metastatic NSCLC treated with first-line chemotherapy or targeted therapy at two institutions (Università Politecnica Marche, Italy and Chelsea & Westminster Hospital, UK) between 1st May 2006 and 31th January 2015. Tumor stage was assessed according to the tumor-node-metastasis (TNM) system and included patients with stage IIIB, IV and IIIA not suitable for surgery, as defined in AJCC version 7. Data were retrospectively collected from patients’ medical records.
Treatment with first-line chemotherapy or targeted therapy was continued until evidence of disease progression, unacceptable adverse events, or death. Follow-up generally consisted of regular physical examination and laboratory assessment (haematology and serum biochemistry), and imaging using computed tomography (CT) or magnetic resonance imaging (MRI) according to local procedures every 8-12 weeks.
Overall survival (OS) was defined as the time from beginning of first-line treatment to death, irrespective of cause. Progression free survival (PFS) was defined as the time from beginning of treatment to progression or to death from any cause, whichever occurred first. Patients without tumour progression or death at the time of the data cut-off for the analysis or at the time of receiving an additional anticancer therapy were censored at their last date of tumour evaluation.
Statistical Analysis
PFS and OS were estimated using Kaplan-Meier method with Rothman’s 95% confidence intervals (CI) and compared across the groups using the log-rank test. Patients with a stable disease (SD), partial remission, and a complete remission were considered as responders.
Hyponatremia was assessed within one week prior to starting first-line therapy, and after each treatment cycle. Potential factors associated with outcome were evaluated, including patients’ age (≥ 70y vs. < 70y), gender, tumor stage, histology, EGFR mutational status, Eastern Cooperative Oncology Group performance status (PS) and smoking history. Data about concomitant medications were not available.
Cox proportional hazards models were applied to explore patients’ characteristics predictors of survival in univariate- and multivariable analysis. Variables not fitting at univariate analysis were excluded from the multivariate model. No-multicollinearity of the grouped co-variates was checked. Significance level in the univariate model for inclusion in the multivariate final model was more liberally set at a 0.2 level [36, 37]. The likelihood ratio test was conducted to evaluate the improvement in prediction performance gained by backward elimination of variables from the prognostic model [38]. All other significance levels were set at a 0.05 value and all P values were two-sided. Statistical analyses were performed using MedCalc version 11.4.4.0 (MedCalc Software, Broekstraat 52, 9030 Mariakerke, Belgium). The research was carried out in accordance with the ethical committee of our institution. All patients gave their written consent to all the diagnostic-therapeutic procedures.
Acknowledgments
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector. The study was realized with Authors’ University Funding (Università Politecnica delle Marche, Ancona, Italy).
Conflicts of Interest grant support
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector. The study was realized with solely Authors’ University Funding (Università Politecnica Marche).
Rossana Berardi and Tom Newsom-Davis have received consulting fee or honoraria from Otsuka.
The other authors declare that they have no competing interests.
References
1. Doshi SM, Shah P, Lei X, Lahoti A, Salahudeen AK. Hyponatremia in hospitalized cancer patients and its impact on clinical outcomes. Am J Kidney Dis. 2012; 59: 222-8.
2. Schwartz, E., Fogel, R. L., Chokas, W. V., and Panariello, V. A.: Unstable osmolar homeostasis with and without renal sodium wastage. Amer J Med. 1962; 33: 39.
3. Renneboog B, Musch W, Vandemergel X, Manto MU, Decaux G. Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits. Am J Med 2006; 119: 71-78.
4. Adrogué HJ, Madias NE. Hyponatremia. N Engl J Med. 2000; 342: 1581-1589.
5. Berghmans T., Paesmans M., Body J. A prospective study on hyponatraemia in medical cancer patients: epidemiology, aetiology and differential diagnosis. Support Care Cancer. 2000; 8: 192-197.
6. Sørensen JB, Andersen MK, Hansen HH. Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) in malignant disease. J Intern Med 1995; 238: 97-110.
7. List AF, Hainsworth JD, Davis BW, Hande KR, Greco FA, Johnson DH. The syndrome of inappropriate secretion of antidiuretic hormone (SIADH) in small-cell lung cancer. J Clin Oncol. 1986; 4: 1191e8.
8. Berardi R, Santoni M, Rinaldi S, Nunzi E, Smerilli A, Caramanti M, Morgese F, Torniai M, Savini A, Fiordoliva I, Onofri A, Pistelli M, Taccaliti A, Cascinu S. Risk of Hyponatraemia in Cancer Patients Treated with Targeted Therapies: A Systematic Review and Meta-Analysis of Clinical Trials. PLoS One. 2016 May 11;11(5):e0152079.
9. Rawson NS, Peto J. An overview of prognostic factors in small cell lung cancer. A report from the Subcommittee for the Management of Lung Cancer of the United Kingdom Coordinating Committee on Cancer Research. Br J Cancer. 1990; 61: 597-604.
10. Berardi R, Caramanti M, Fiordoliva I, Morgese F, Savini A, Rinaldi S, Torniai M, Tiberi M, Ferrini C, Castagnani M, Rovinelli F, Onofri A, Cascinu S. Hyponatraemia is a predictor of clinical outcome for malignant pleural mesothelioma. Support Care Cancer. 2015; 23: 621-6.
11. Schutz FA, Xie W, Donskov F, Schutz FA, Xie W, Donskov F, Sircar M, McDermott DF, Rini BI, Agarwal N, Pal SK, Srinivas S, Kollmannsberger C, et al. The impact of low serum sodium on treatment outcome of targeted therapy in metastatic renal cell carcinoma: results from the International Metastatic Renal Cell Cancer Database Consortium. Eur Urol. 2014; 65: 723-30.
12. Kim HS, Yi SY, Jun HJ, Lee J, Park JO, Park YS, Jang J, Kim HJ, Ko Y, Lim HY, Kang WK. Clinical outcome of gastric cancer patients with bone marrow metastases. Oncology. 2007; 73: 192-7.
13. Dhaliwal HS, Rohatiner AZ, Gregory W, Richards MA, Johnson PW, Whelan JS, Gallagher CJ, Matthews J, Ganesan TS, Barnett MJ. Combination chemotherapy for intermediate and high grade non-Hodgkin’s lymphoma. Br J Cancer. 1993; 68: 767-74.
14. Choi JS, Bae EH, Ma SK, Kweon SS, Kim SW. Prognostic impact of hyponatraemia in patients with colorectal cancer. Colorectal Dis. 2015; 17: 409-16.
15. Farid SG, Prasad KR. Prognostic impact of hyponatraemia in patients with colorectal cancer. Colorectal Dis. 2015; 17: 451.
16. Castillo JJ, Glezerman IG, Boklage SH, Chiodo J 3rd, Tidwell BA, Lamerato LE, Schulman KL. The occurrence of hyponatremia and its importance as a prognostic factor in a cross-section of cancer patients. BMC Cancer. 2016 Jul 29;16:564.
17. Yoon J, Ahn SH, Lee YJ, Kim CM. Hyponatremia as an independent prognostic factor in patients with terminal cancer. Support Care Cancer. 2015; 23: 1735-40.
18. Balachandran K, Okines A, Gunapala R, Morganstein D, Popat S. Resolution of severe hyponatraemia is associated with improved survival in patients with cancer. BMC Cancer. 2015; 15: 163.
19. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013; 63: 11-30.
20. Sengupta A, Banerjee SN, Biswas NM, Jash D, Saha K, Maji A, Bandyopadhyaya A, Agarwal S. The Incidence of Hyponatraemia and Its Effect on the ECOG Performance Status among Lung Cancer Patients. J Clin Diagn Res. 2013; 7: 1678-82.
21. Castillo JJ, Grohé C, Verbalis JG. Cancer-Related Hyponatremia: Enhancing Recognition and Improving Management. Clin Lung Cancer. 2016; 17: e29.
22. Wakui A. Electrolyte abnormalities associated with cancer: a review. Gan To Kagaku Ryoho 1986; 13: 2031-2038.
23. Palmer BF, Gates JR, Lader M. Causes and management of hyponatremia. Ann Pharmacother. 2003; 37: 1694-1702.
24. Raftopoulos H. Diagnosis and management of hyponatremia in cancer patients. Support Care Cancer. 2007; 15: 1341-1347.
25. Onitilo AA, Kio E, Doi SA. Tumor-related hyponatremia. Clin Med Res. 2007; 5: 228 -237
26. Hansen O, Sørensen P, Hansen KH. The occurrence of hyponatremia in SCLC and the influence on prognosis: a retrospective study of 453 patients treated in a single institution in a 10-year period. Lung Cancer. 2010; 68: 111-4.
27. Castillo JJ, Vincent M, Justice E. Diagnosis and management of hyponatremia in cancer patients. Oncologist. 2012; 17: 756-65
28. Jeppesen AN, Jensen HK, Donskov F, Marcussen N, von der Maase H. Hyponatremia as a prognostic and predictive factor in metastatic renal cell carcinoma. Br J Cancer. 2010; 102: 867-72.
29. Tiseo M, Buti S, Boni , Mattioni R, Ardizzoni A. Prognostic role of hyponatremia in 564 small cell lung cancer patients treated with topotecan. Lung Cancer. 2014; 86: 91-5.
30. Berardi R, Caramanti M, Castagnani M, Guglielmi S, Marcucci F, Savini A, Morgese F, Rinaldi S, Ferrini C, Tiberi M, Torniai M, Rovinelli F, Fiordoliva I, et al. Hyponatremia is a predictor of hospital length and cost of stay and outcome in cancer patients. Support Care Cancer. 2015; 23: 3095-101.
31. Grohé C, Berardi R, Burst V. Hyponatraemia-SIADH in lung cancer diagnostic and treatment algorithms. Crit Rev Oncol Hematol. 2015; 96: 1-8.
32. Selmer C, Madsen JC, Torp-Pedersen C, Gislason GH, Faber J. Hyponatremia, all-cause mortality, and risk of cancer diagnoses in the primary care setting: A large population study. Eur J Intern Med. 2016 Aug 13. pii: S0953-6205(16)30236-9.
33. Zarzecka M, Kubicki P, Kozielski J. Hyponatraemia - evaluation of prevalence in patients hospitalized in the Pulmonary Department and prognostic significance in lung cancer patients. Pneumonol Alergol Pol. 2014; 82: 18-24.
34. Svaton M, Fiala O, Pesek M, Bruha F, Mukensnabl P, Racek J, Minarik M, Bortlicek Z. Predictive and Prognostic Significance of Sodium Levels in Patients with NSCLC Treated by Erlotinib. Anticancer Res. 2014; 34: 7461-5.
35. Kobayashi N, Usui S, Yamaoka M, Suzuki H, Kikuchi S, Goto Y, Sakai M, Sato Y. The influence of serum sodium concentration on prognosis in resected non-small cell lung cancer. Thorac Cardiovasc Surg. 2014; 62: 338-43.
36. Fagerland MW, Hosmer DW, Bofin AM. Multinomial goodness-of-fit tests for logistic regression models. Stat Med. 2008; 27: 4238-4253.
37. Mickey RM, Greenland S. The impact of confounder selection criteria on effect estimation. Am J Epidemiol. 1989; 129: 125-137.
38. Pepe MS, Kerr KF, Longton G, Wang Z. Testing for improvement in prediction model performance. Stat Med. 2013; 32: 1467−1482.

