INTRODUCTION
Hepatocellular carcinoma (HCC) accounts for 90% of all liver cancers, with approximately 80% of HCC cases occurring in the Asia-Pacific region [1, 2]. Despite considerable improvements in techniques, complete surgical resection is possible in less than 20% of HCCs. Furthermore, conventional chemotherapy and radiotherapy do not significantly benefit patients with advanced HCC [3, 4].
High linear energy transfer (High-LET) particle therapy produces spread out Bragg’s peaks (SOBP), allowing it to cover tumors with biologically equivalent dose distributions. High-LET radiation has several advantages over low-LET radiation in treating radioresistant human cancers because of its higher relative biological effectiveness (RBE), lower oxygen enhancement ratio (OER), and decreased cell-cycle-dependent radiosensitivity. In addition, because cells are less likely to repair radiation injury, charged particle radiation may exert highly lethal effects even on radioresistant tumors, compared to conventional low-LET X-ray or γ-ray irradiation [5–8].
The Heavy-Ion Medical Accellerator in Chiba (HIMAC) is the first heavy-ion accelerator dedicated to medicine, and it has become the world’s leading heavy-ion cancer treatment facility. Over the past 20 years, HIMAC has treated more than 500 HCC patients and achieved similar or better results than surgery. Because heavy ion radiotherapy is applied in only two fractions, the patient’s quality of life (QOL) is also markedly improved [9, 10]. However, carbon ion radiotherapy alone has some limitations in the treatment of advanced tumors, particularly those located near important organs, and in the treatment of patients with micrometastasis [11, 12]. A recent clinical trial of carbon ion beams combined with gemcitabine achieved promising results for locally advanced pancreatic cancer, with 54% patients achieving 2-year overall survival [13]. We previously investigated whether the elimination of pancreatic cancer stem cells (CSCs) accounted for the high tumor control in that study [14].
Metformin (1,1-dimethylbiguanide hydrochloride), the most widely used treatment for type 2 diabetes, has recently sparked interest as a potential anticancer agent [15]. Metformin provides a synergistic benefit with chemotherapy or radiotherapy against certain cancers [15–23]. We have previously reported that metformin has radiosensitizing effects on HCC cells treated with high-LET neutron irradiation [24]. However, its application is greatly limited, with only three centers, worldwide, currently treating cancer using fast neutrons. This is perhaps because of a lack of funding and/or issues related to regulatory approval. An alternative approach may be to use other forms of high-LET radiation, such as carbon beams, which are widely used in Japan and Europe. In this study, we investigated the potential effects of metformin to sensitize HCC cells to carbon ion beams.
RESULTS
Clonogenic survival of Huh7 and HepG2 cells by γ-ray or carbon ion beams alone or in combination with metformin
Figure 1 shows that the clonogenic survival of Huh7 and HepG2 HCC cells decreased as a function of radiation dose after exposure to γ-ray or carbon ion beams (13 and 70 keV/μm). The average D10 (radiation dose for reducing cell survival to 10%) of Huh7 (HepG2) was 5.2 (5.6) Gy for γ-rays, whereas it was 5.1 (5.2) Gy and 3.1 (3.2) Gy for 13 and 70 keV/μm carbon ion beams, respectively (Table 1). Therefore, the RBE values of carbon ion beams with 13 and 70 keV/μm relative to γ-ray were 1.02 (1.08) and 1.68 (1.75), respectively (Table 1). The radiosensitizing effect of metformin was studied using 5 mM metformin, the dose that reduced cell survival by 25% with a 48 h incubation (data not shown). The D10 of Huh7 (HepG2) for γ-rays combined with metformin was 3.1 (4.6) Gy, whereas the D10s for 13 and 70 keV/μm carbon ion beams were 4.0 (3.9) Gy and 1.8 (2.0) Gy, respectively (Table 1). The radiosensitivity enhancement factor values and dose reduction for IR treatment of metformin -pretreated HCC cells are shown (Table 2).
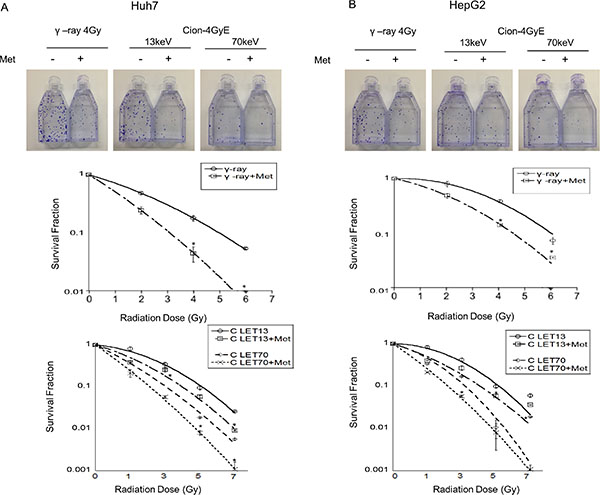
Figure 1: (A, B) Surviving fraction of Huh7 and HepG2 cells with and without metformin after γ-ray and carbon radiation. Huh7 and HepG2 cells were plated immediately after carbon ion beam and γ -ray irradiation alone or in combination with metformin (Met, 5 mM) for 48 h. The graphs show the mean +/− SEM calculated from 3 independent experiments.
Table 1: RBE values at D10 level for Huh7 and HepG2 cells after γ-ray, carbon ion alone or in combination with metformin
Cell Type |
γ-ray |
C-ion |
RBE |
||
|---|---|---|---|---|---|
13kev |
70kev |
13kev |
70kev |
||
Huh7 |
5.2Gy |
5.1Gy |
3.1Gy |
1.02 |
1.68 |
+metfromin |
3.1 Gy |
4.0Gy |
1.8 Gy |
||
HepG2 |
5.6Gy |
5.2Gy |
3.2Gy |
1.08 |
1.75 |
+ metformin |
4.6Gy |
3.9Gy |
2.0Gy |
||
Table 2: Radiosensitivity enhancement factor (REF) and dose reduction values
Cell type |
Radiation type |
REF value |
Dose reduction (%) |
|---|---|---|---|
Huh7 |
γ-ray |
1.68 |
40.4 |
C-ion 13keV |
1.28 |
21.6 |
|
C-ion 70keV |
1.72 |
41.9 |
|
HepG2 |
γ-ray |
1.22 |
17.9 |
C-ion 13keV |
1.33 |
25.0 |
|
C-ion 70keV |
1.60 |
37.5 |
The radiosensitivity enhancement factor (REF) corresponding to 90% cell killing was calculated to quantify the radiosensitization due to metformin using the equation. REF = radiation dose for 90% killing/radiation dose in the presence of metformin for 90% killing.
Induction of DNA damage by γ-ray and carbon ion beams alone or in combination with metformin
To elucidate the involvement of DNA damage in metformin-induced radiosensitization, we analyzed the formation of γH2AX. The number of γH2AX foci was initially increased 6 h after irradiation with either 4 Gy γ-ray or 4 GyE carbon ion beams, but then substantially declined after 24 h. Metformin treatment alone only slightly increased γH2AX expression, but markedly increased the effects of γ-rays and carbon ion beams on γH2AX (Figure 2). Notably, γH2AX foci formation by 4 GyE carbon ion irradiation was similar to, or slightly greater than, that caused by 4 Gy γ-ray irradiation either with or without metformin treatment.
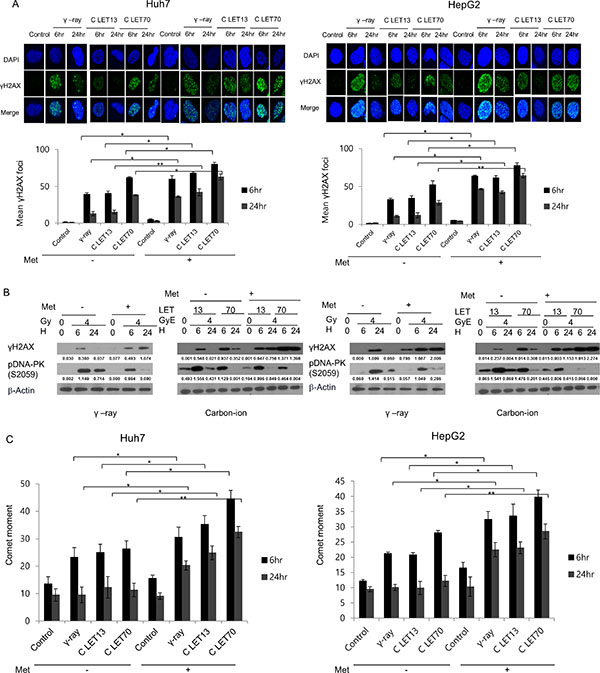
Figure 2: (A) Immunofluorescence staining for γH2AX phosphorylation in Huh7 and HepG2 cells after γ-ray and carbon ion beam alone or in combination with metformin (Met, 5 mM) at 6 and 24 h. (B) Cell lysates were immunoblotted with the indicated antibodies. (C) Neutral comet assay in Huh7 and HepG2 cells after γ-rays and carbon ion beams alone or in combination with metformin (Met, 5 mM) at 6 and 24 h. Values represent the means +/− SD of 3 experiments; *p < 0.05, **p < 0.001.
Western blot results for γH2AX expression (Figure 2B) were in agreement with the results of γH2AX foci formation (Figure 2A). The expression of γH2AX upon irradiation with either 4 Gy γ -rays of 4 GyE carbon beams was enhanced by metformin. As shown in Figure 2B, irradiation with γ -rays or carbon ion beams increased the expression of phospho-DNA-PK, and metformin suppressed the radiation-induced increase in phospho-DNA-PK. The results of the comet assay indicated that metformin alone caused only slight DNA damage, while combined treatment further increased significant DNA damage, as indicated by an increase in comet movement (Figure 2C).
Apoptosis is induced by γ-rays and carbon ion beams alone or in combination with metformin
Apoptosis induced by irradiation with or without metformin was investigated by assessing the levels of cleaved caspase-3 and cleaved PARP1 by western blot. As shown in Figure 3, 4 Gy γ-ray or 4 GyE carbon ion beam irradiation induced apoptosis in HCC cells, as indicated by increased cleaved caspase-3 and PARP1. Treatment with metformin alone only slightly increased apoptosis, while combined treatment further increased apoptosis.
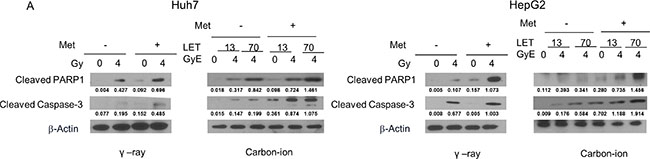
Figure 3: Western blotting analysis of apoptosis-related proteins in Huh7 and HepG2 cells 48 h after γ-rays and carbon ion beams alone or in combination with metformin (Met, 5 mM). β-actin was used as an internal control.
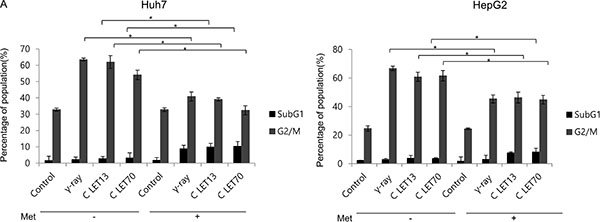
Figure 4: Alteration of cell cycle progression 24 h after γ-ray and carbon ion beams alone or in combination with metformin (Met, 5 mM). Huh7 and HepG2 cells were treated with metformin (Met, 5 mM) and/or 4Gy γ-ray and 4 GyE of carbon ion beams for 24 h. Values represent the means +/− SD of 3 experiments; *p < 0.05, **p < 0.001.
Changes in cell cycle phase distribution by γ-rays and carbon ion beams alone or in combination with metformin
We next examined the effects of γ-ray and carbon ion beam irradiation alone or in combination with metformin on cell cycle progression using flow cytometry (Figure 4). The most obvious changes in cell cycle distribution following irradiation were increases in G2/M phase cells. Metformin induced G1 arrest and decreased subsequent G2/M arrest after γ-ray, LET 13 carbon-ion, and LET 70 carbon-ion irradiation. Sub-G1 cells, which represent apoptotic cells, were only moderately increased by metformin, but markedly increased by the combined treatment of metformin and γ-ray or carbon ion beam irradiation.
Changes in tumor cell migration and invasion by γ-rays and carbon ion beams alone or in combination with metformin
We next estimated the effects of γ-rays or carbon ion beams alone or combined with metformin on the invasiveness and migration of HCC cells. Irradiation inhibited cell migration toward wound sites, while metformin only slightly inhibited cell migration and invasion (Figure 5A, 5B). However, metformin enhanced the negative effect of γ-rays and carbon ion beams on HCC cell migration. In a Matrigel invasion assay, irradiation with γ-rays or carbon ion beams decreased cell invasiveness, and the combination treatment was more effective in inhibiting tumor cell invasion than irradiation alone, especially with 70keV/μm carbon ion beams. Furthermore, metformin enhanced the effects of γ-rays and carbon ion beams on the decreased expression of vimentin, which is used widely as a marker of the epithelial-mesenchymal transition (EMT) that occurs during metastasis (Figure 5C).
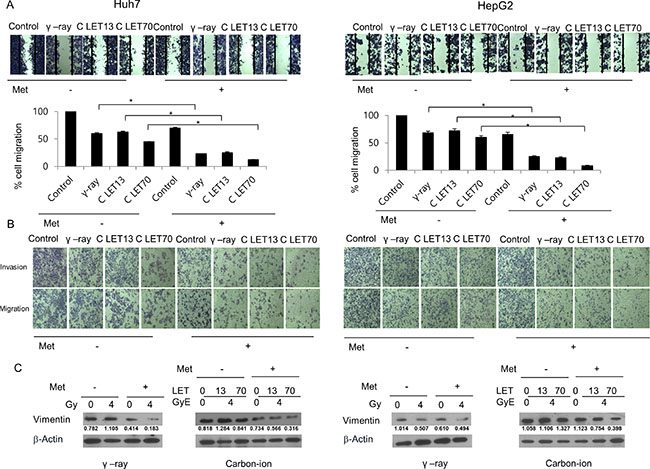
Figure 5: Migration and invasion analysis by a wound-healing scratch assay (A), a transwell chamber assay (B), and western blot (C) after treatment for 24 h. Carbon ion beam in combination with metformin (Met, 5 mM) inhibited HCC cell migration and invasion compared with γ-rays.
Activation of the AMPK/mTOR/Akt pathway by γ-rays and carbon ion beams alone or in combination with metformin
Finally, we examined the effect of metformin and irradiation alone or in combination on activation of the AMPK/mTOR/Akt signaling pathway using western blots. Treatment of HCC cells for 48 h with 5 mM metformin or irradiation alone increased the expression of p-AMPK and decreased p-mTOR and p-Akt levels. The combined treatment of metformin and irradiation was more effective than metformin or irradiation alone at increasing p-AMPK and decreasing p-mTOR and p-Akt (Figure 6), with the combination of metformin with 70keV/μm carbon ion beams being most effective at altering the expression of these proteins.
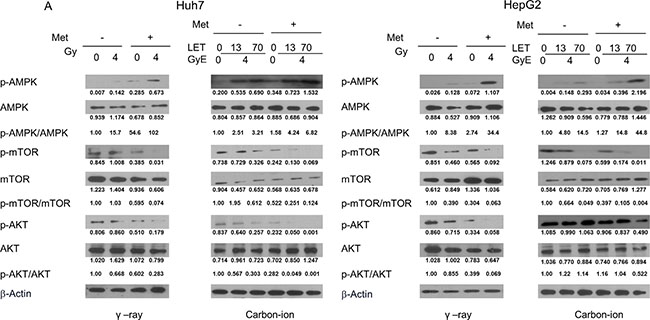
Figure 6: Western blotting analysis of AMPK/mTOR/Akt pathway-related proteins in Huh7 and HepG2 cells 48 h after γ-rays and carbon ion beams alone or in combination with metformin.
DISCUSSION
Based on cell survival curves, we estimated that the Huh7 (HepG2) RBE value of carbon ion beams with 13 and 70keV/um relative to γ-rays was 1.02 (1.08) and 1.68 (1.75), respectively. LET 13 carbon-ion mono-beams were used because they are representative of the radiation to which normal tissues adjacent to tumors are exposed, while LET 70 beams were used because they are close to the Bragg peak to target tumor volume. With low-LET radiation, including protons and γ-rays, cell survival curves of cancer cells hardly differ between the control group and carbon 13keV/μm LET. However, cells exposed to carbon 70keV/μm LET have differences in cell survival between the Co-60 γ-ray and high-LET groups. In the present study, 70keV/μm carbon ion beam irradiation was more effective than Co-60 γ-ray and 13keV/μm carbon ion beam irradiation in causing clonogenic cell death in HCC cells in vitro. Metformin enhanced the effects of ionizing radiation, including Co-60 γ-ray and 13 and 70 keV/μm carbon ion beams to induce clonogenic cell death, apoptosis, γH2AX formation, and inhibition of cell migration in HCC cells.
Metformin is the most frequently used treatment for type II diabetes with few side effects [25–27]. Metformin acts as a chemosensitizer or radiosensitizer in various tumor cell types, including HCC cells [15–19, 25, 26]. In the present study, we first demonstrated that metformin sensitizes HCC cells not only to low-LET photon beams but also to high-LET carbon ion beams. This finding is consistent with previous reports that metformin radiosensitizes HCC cells to high-LET neutron irradiation [24].
Metformin markedly enhanced and also prolonged radiation-induced γH2AX formation such that a significant amount of γH2AX was still present even at 24 h after irradiation. There were no differences in the induction of γH2AX between γ-ray and carbon ion beams alone or in combination with metformin. Metformin also markedly enhanced the DNA damage effects of γ-rays and carbon ion beams and decreased the phospho-DNA-PK expression caused by both γ-ray and carbon ion beams. These findings are consistent with previous reports that the combination of metformin and radiation yielded greater numbers of γH2AX foci than radiation alone, and the reduced phospho-DNA-PKcs is important for metformin to induce radiosensitivity in prostate cancer cells [17, 18]. Taken together, these findings suggest the metformin increases and prolongs DNA damage caused by irradiation with r-rays and high-LET carbon ion beams.
Metformin treatment also enhanced the effects of irradiation on the expression of cleaved caspase-3 and PARP, indicating potentiation of radiation-induced apoptosis in HCC cells. These results are consistent with those of previous reports that metformin potentiated radiation-induced caspase-9/-3 cleavage and PARP-dependent cell death in nasopharyngeal and breast cancer cells [29, 30]. These data may partially explain why carbon ion beams combined with metformin more effectively destroy HCC cells than γ-rays combined with metformin.
AMPK activators and metformin treatment have been shown to inhibit HCC cell proliferation and induce cell cycle arrest at the G1-S checkpoint [30–32]. In this study, metformin alone increased the number of cells in G1 phase, while decreasing the number of cells in G2/M phase. Treatment with γ-ray or carbon ion alone increased the number of cells in G2/M phase and reduced the number of cells in G1 phase. Combined metformin and radiation (γ-ray or carbon ion) treatment reduced G2-M and increased subG1 distribution in comparison to radiation alone, possibly via an enhanced DNA damage response and increased DNA double strand breaks by metformin. Such slowing of cell cycle progression may account for the suppression of cell migration and invasion observed with the combination of metformin and carbon ion beams. The suppression of cell migration and invasion by metformin suggests that metformin may reduce metastasis in clinical settings. Because the radiosensitizing effects of metformin are intended to enhance tumor cell death, experiments with mouse models should be conducted first to examine the possible complications in clinical applications.
We also observed that the combination of metformin with γ-rays and carbon beams was more effective than γ-rays or carbon ion beams alone in increasing the expression of p-AMPK and suppressing the expression of p-mTOR. This finding is consistent with recent reports that the AMPK/mTOR pathway may be the target of the anti-proliferative effects of metformin in combination with ionizing radiation in esophageal and gastric cancer cells [33–36]. Other recent reports have also shown that metformin inhibits the expression of N-cadherin, MMP-2, and MMP-9 proteins in an AMPK/p53-dependent manner in melanoma and suppresses MMP-9 in HCC cells [37, 38].
Importantly, it has been recently reported that metformin is cytotoxic to cancer stem cells (CSCs) and enhances the effects of chemotherapy and radiation against CSCs in many types of human cancer, including HCC, glioblastoma, breast, pancreas, lung, and esophageal cancer [31, 35,39–44]. Based on our previous observation that carbon ion beams are effective in killing CSCs [15, 45, 46], in future studies we plan to elucidate the combination of metformin and carbon ion beams against CSCs.
In conclusion, metformin sensitized HCC cells to γ-ray irradiation and carbon ion beams, as indicated by the increase in clonogenic cell death, DNA damage, apoptosis, and cell cycle arrest and the inhibition of tumor cell migration and invasion. Enhanced activation of AMPK and down-regulation of mTOR and Akt appeared to play a role in the metformin-induced sensitization of cancer cells to γ-rays and carbon ion beams.
MATERIALS AND METHODS
Antibodies and chemicals
Anti-phospho-DNA-PK, anti-cleaved PARP1, anti-cleaved caspase-3, anti-phospho-AMPK, anti-AMPK, anti-phospho-mTOR, anti-mTOR, anti-phospho-AKT and anti-AKT were all purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-vimentin and anti-b-Actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Anti-γ H2AX was obtained from Millipore (Billerica, MA, USA). Metformin (1-(diaminomethylidene)-3, 3-dimethylguanidine) was purchased from Sigma-Aldrich Chemical Corp (St. Louis, MO, USA).
Cell culture
The HCC cell lines Huh7 and HepG2 were purchased from Japanese Collection of Research Bioresources (JCRB, Tokyo, Japan) and cultured in Dulbecco’s minimal essential medium (DMEM; GIBCO, Gaithersburg, MD, USA) supplemented with heat-inactivated 10% fetal bovine serum (FBS; GIBCO), 0.1 mM non-essential amino acids, glutamine, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), and antibiotics at 37°C in a 5% CO2 humidified incubator.
Irradiation
Cells were plated in 60-mm plastic dishes and incubated at 37°C under a humidified 5% CO2 atmosphere. Cells at 70–80% confluence were irradiated with a 137Cs γ-ray source (Atomic Energy of Canada, Ltd., Ontario, Canada) at a dose rate of 3.81 Gy/min. Carbon ions were provided by 290 MeV/nucleon beams at NIRS-HIMAC. Cells were irradiated with mono-peak C-ions with a dose-averaged LET of 13 or 70keV/μm at a dose rate of 2.0–5.0 Gy/min. The dose unit in the literature was expressed as Gray equivalent (GyE), which is determined for the clinical situation as previously described [47].
Colony-forming assay
Cells were seeded into 6-well plates in triplicate and irradiated with or without metformin. After incubation for 14–20 days, colonies were stained with 0.4% crystal violet (Sigma, St. Louis, MO, USA), and the plating efficiency (PE) was determined. The PE of Huh7 and HepG2 were 0.48 ± 0.18, 0.45 ± 0.15, respectively. The surviving cell fraction was calculated as follows: surviving fraction = colonies counted/(cells seeded × PE/100). The radiosensitizing effect of metformin was calculated by dividing the radiation dose (Gy) for radiation alone by the dose for radiation plus metformin at a surviving fraction of 10%.
Flow cytometry
Cells were cultured, harvested at the indicated times, stained with propidium iodide (1 μg/ml, Sigma) according to the manufacturer’s protocol, and then analyzed using a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA). A minimum of 10,000 cells was counted for each sample, and data analysis was performed using CellQuest software (BD Biosciences).
Immunocytochemistry
γH2AX expression was assessed using an immunocytochemistry assay method [46]. Cells were grown on chambered slides for 1 day, pretreated with metformin for 6 or 24 h, and then irradiated. Cells were then fixed with 4% paraformaldehyde and permeabilized with 0.5% Triton X-100 in PBS. Detection was performed after the slides were blocked in 4% FBS in PBS for 1 h and stained with a 1:100 dilution of a primary antibody against γ-H2AX (Millipore, Billerica, MA, USA) and a 1:500 dilution of FITC-labeled secondary antibodies.
Western blotting
HCC cells in the exponential growth phase were pretreated with metformin for 8 h and then irradiated for 6 or 24 h. The cells were then lysed with RIPA buffer, and proteins were separated by SDS-polyacrylamide gel electrophoresis (PAGE) and transferred to nitrocellulose membranes. The membranes were blocked with 1% (v/v) nonfat dry milk in Tris-buffered saline with 0.05% Tween20 and incubated with antibodies. Blots were reacted with primary antibodies at 1:1000 dilution and secondary antibodies at 1:5000 dilution. Immunoreactive protein bands were visualized by Enhanced Chemi-luminescence (Amersham Biosciences) and scanned.
Neutral comet assay
The radiation-induced DNA double-strand breaks were determined by the comet assay (single cell gel electrophoresis assay) as previously described [24]. Cells were plated in 100-mm tissue culture dishes at 1 × 106/dish and incubated overnight. After metformin exposure for 8 h, the cells were irradiated and incubated for 6 or 24 h, and a neutral comet assay was performed using a comet assay kit (Trevigen, Gaithersburg, MD, USA). The cells were lysed at 4°C for 1 h in lysis buffer (2.5 M NaCl, 100 mM ethylenediaminetetraacetic acid, 10 mM Tris-HCl, 1% N-lauroylsarcosine, pH 7) and subjected to neutral electrophoresis at 4°C. The slides were stained with ethidium bromide and examined for fluorescence emission using an excitation filter of 515–560 nm and a barrier filter of 590 nm. DNA damage was assessed via computer-assisted image analysis (Komet analysis software, ver. 3.1; Kinetic Imaging, Liverpool, UK).
Wound-healing scratch assay
Cells were seeded onto six-well plates (Corning) at 2.5 × 104 cells/well with 3 ml of DMEM with 10% FBS. After metformin pre-incubation for 8 h, the cells were irradiated. On day 2, the monolayers were mechanically disrupted with a sterile 200-μl pipette tip. The assay was performed in duplicate. Wells were imaged after incubation for 24 h, and cell migration was measured using a Nikon Eclipse Ti microscope with a DS-Fi1 camera. Cells were counted using ImageJ software (United States National Institutes of Health, Bethesda, MD, USA).
Invasion assay
The invasive ability of cells was measured using a transwell invasion assay kit (Chemicon, Millipore, GA, USA), according to the manufacturer’s protocol. Briefly, cells were seeded onto the membrane of the upper chamber of the transwell at a concentration of 4 × 105 cells/ml in 150 μl of serum-free RPMI and were left untreated or treated with the indicated doses of metformin, radiation, or a combination of both. The medium in the lower chamber contained 10% FBS as a chemoattractant. After incubation for 24 h, cells that passed through the Matrigel-coated membrane were stained with the Cell Stain Solution containing crystal violet and imaged.
Statistical analyses
All analyses were determined by Student’s t-test. Differences were considered significant if the p value was less than 0.05 or 0.001.
ACKNOWLEDGMENTS AND FUNDING
This work was supported by a National Research Foundation of Korea (NRF) grant (No. NRF-2014R1A1A3053958, NRF-2014029534) funded by the Korean government (MSIP) and a grant from the Korea Institute of Radiological and Medical Sciences (KIRAMS), funded by Ministry of Science, ICT and Future Planning, Republic of Korea (50576-2016, 50476-2016).
CONFLICTS OF INTEREST
The authors declare that they have no competing interests.
REFERENCES
1. Waly Raphael S, Yangde Z, Yuxiang C. Hepatocellular carcinoma: focus on different aspects of management. ISRN Oncol. 2012; 2012:421673.
2. Bosch FX, Ribes J, Díaz M, Cléries R. Primary liver cancer: Worldwide incidence and trends. Gastroenterology. 2004; 127:S5–S16.
3. Giannini EG, Farinati F, Ciccarese F, Pecorelli A, Rapaccini GL, Di Marco M, Benvegnu L, Caturelli E, Zoli M, Borzio F, et al. Prognosis of untreated hepatocellular carcinoma. Hepatology. 2015; 61:184–190.
4. Bruix J, Raoul JL, Sherman M, Mazzaferro V, Bolondi L, Craxi A, Galle PR, Santoro A, Beaugrand M, Sangiovanni A, et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: subanalyses of a phase III trial. J Hepatol. 2012; 57:821–829.
5. Skarsgard LD. Radiobiology with heavy charged particles: a historical review. Phys Med. 1998; 14:1–19.
6. Okayasu R. Repair of DNA damage induced by accelerated heavy ions—a mini review. Int J Cancer. 2012; 130:991–1000.
7. Kamada T, Tsujii H, Blakely EA, Debus J, De Neve W, Durante M, Jäkel O, Mayer R, Orecchia R, Pötter R, et al. Carbon ion radiotherapy in Japan: an assessment of 20 years of clinical experience. The Lancet Oncology. 2015; 16:e93–e100.
8. Kanai T, et al. Biophysical characteristics of himac clinical irradiation system for heavy-ion radiation therapy. Int J Radiat Oncol Biol Phys. 1999; 44:201–210.
9. Kato H, Tsujii H, Miyamoto T, Mizoe JE, Kamada T, Tsuji H, Yamada S, Kandatsu S, Yoshikawa K, Obata T, et al. Results of the first prospective study of carbon ion radiotherapy for hepatocellular carcinoma with liver cirrhosis. Int J Radiat Oncol Biol Phys. 2004; 59:1468–1476.
10. Imada H, Kato H, Yasuda S, Yamada S, Yanagi T, Hara R, Kishimoto R, Kandatsu S, Minohara S, Mizoe JE, et al. Compensatory enlargement of the liver after treatment of hepatocellular carcinoma with carbon ion radiotherapy - relation to prognosis and liver function. Radiother Oncol. 2010; 96:236–242.
11. Wakai T, Shirai Y, Sakata J, Valera VA, Korita PV, Akazawa K, Ajioka Y, Hatakeyama K. Appraisal of 1 cm hepatectomy margins for intrahepatic micrometastases in patients with colorectal carcinoma liver metastasis. Ann Surg Oncol. 2008; 15:2472–2481.
12. Vigano L, Capussotti L, De Rosa G, De Saussure WO, Mentha G, Rubbia-Brandt L. Liver resection for colorectal metastases after chemotherapy: impact of chemotherapy-related liver injuries, pathological tumor response, and micrometastases on long-term survival. Ann Surg. 2013; 258:731–740; discussion 741–732.
13. Yamada S, Terashima K, Shinoto M, Yasuda S, et al. Pancreatic Cancer. Carbon-Ion Radiotherapy Principles, Practices, and Treatment 221–228. Springer 2014.
14. Sai S, Wakai T, Vares G, Yamada S, Kamijo T, Kamada T, Shirai T. Combination of carbon ion beam and gemcitabine causes irreparable DNA damage and death of radioresistant pancreatic cancer stem-like cells in vitro and in vivo.Oncotarget. 2015; 6:5517–5535. doi: 10.18632/oncotarget.3584.
15. Skinner HD, McCurdy MR, Echeverria AE, Lin SH, Welsh JW, O’Reilly MS, Hofstetter WL, Ajani JA, Komaki R, Cox JD, et al. Metformin use and improved response to therapy in esophageal adenocarcinoma. Acta Oncol. 2013; 52:1002–1009.
16. Fasih A, Elbaz HA, Huttemann M, Konski AA, Zielske SP. Radiosensitization of pancreatic cancer cells by metformin through the AMPK pathway. Radiat Res. 2014; 182:50–59.
17. He L, Sabet A, Djedjos S, Miller R, Sun X, Hussain MA, Radovick S, Wondisford FE. Metformin and insulin suppress hepatic gluconeogenesis through phosphorylation of CREB binding protein. Cell. 2009; 137:635–646.
18. Foretz M, Hebrard S, Leclerc J, Zarrinpashneh E, Soty M, Mithieux G, Sakamoto K, Andreelli F, Viollet B. Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state. J Clin Invest. 2010; 120:2355–2369.
19. Zhang T, Zhang L, Zhang T, Fan J, Wu K, Guan Z, Wang X, Li L, Hsieh JT, He D, Guo P. Metformin sensitizes prostate cancer cells to radiation through EGFR/p-DNA-PKCS in vitro and in vivo. Radiat Res. 2014; 181:641–649.
20. Zannella VE, Dal Pra A, Muaddi H, McKee TD, Stapleton S, Sykes J, Glicksman R, Chaib S, Zamiara P, Milosevic M, et al. Reprogramming metabolism with metformin improves tumor oxygenation and radiotherapy response. Clin Cancer Res. 2013; 19:6741–6750.
21. Liu J, Hou M, Yuan T, Yi G, Zhang S, Shao X, Chen J, Jia X, He Z. Enhanced cytotoxic effect of low doses of metformin combined with ionizing radiation on hepatoma cells via ATP deprivation and inhibition of DNA repair. Oncol Rep. 2012; 28:1406–1412.
22. Hundal RS, Krssak M, Dufour S, Laurent D, Lebon V, Chandramouli V, Inzucchi SE, Schumann WC, Petersen KF, Landau BR, Shulman GI. Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes. 2000; 49:2063–2069.
23. Inzucchi SE, Maggs DG, Spollett GR, Page SL, Rife FS, Walton V, Shulman GI. Efficacy and metabolic effects of metformin and troglitazone in type II diabetes mellitus. N Engl J Med. 1998; 338:867–872.
24. Kim EH, Kim MS, Cho CK, Jung WG, Jeong YK, Jeong JH. Low and high linear energy transfer radiation sensitization of HCC cells by metformin. J Radiat Res. 2014; 55:432–442.
25. Madiraju AK, Erion DM, Rahimi Y, Zhang XM, Braddock DT, Albright RA, Prigaro BJ, Wood JL, Bhanot S, MacDonald MJ, et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature. 2014; 510:542–546.
26. Inzucchi SE, Lipska KJ, Mayo H, Bailey CJ, McGuire DK. Metformin in patients with type 2 diabetes and kidney disease: a systematic review. JAMA. 2014; 312:2668–2675.
27. Goodwin PJ, Parulekar WR, Gelmon KA, Shepherd LE, Ligibel JA, Hershman DL, Rastogi P, Mayer IA, Hobday TJ, Lemieux J, et al. Effect of metformin vs placebo on and metabolic factors in NCIC CTG MA.32. J Natl Cancer Inst. 2015; 107.
28. Song CW, et al. Metformin kills and radiosensitises cancer cells and preferentially kills cancer stem cells. Scientific Reports. 2012; 2:362
29. Li H, Chen X, Yu Y, Wang Z, Zuo Y, Li S, Yang D, Hu S, Xiang M, Xu Z, Yu Z. Metformin inhibits the growth of nasopharyngeal carcinoma cells and sensitizes the cells to radiation via inhibition of the DNA damage repair pathway. Oncol Rep. 2014; 32:2596–2604.
30. Zhuang Y, Miskimins WK. Metformin induces both caspase-dependent and poly(ADP-ribose) polymerase-dependent cell death in breast cancer cells. Mol Cancer Res. 2011; 9:603–615.
31. Queiroz EA, Puukila S, Eichler R, Sampaio SC, Forsyth HL, Lees SJ, Barbosa AM, Dekker RF, Fortes ZB, Khaper N. Metformin induces apoptosis and cell cycle arrest mediated by oxidative stress, AMPK and FOXO3a in MCF-7 breast cancer cells. PLoS One. 2014; 9:e98207.
32. Cheng J, Huang T, Li Y, Guo Y, Zhu Y, Wang Q, Tan X, Chen W, Zhang Y, Cheng W, et al. AMP-activated protein kinase suppresses the in vitro and in vivo proliferation of hepatocellular carcinoma. PLoS One. 2014; 9:e93256.
33. Han G, Gong H, Wang Y, Guo S, Liu K. AMPK/mTOR-mediated inhibition of survivin partly contributes to metformin-induced apoptosis in human gastric cancer cell. Cancer Biol Ther. 2015; 16:77–87.
34. Feng T, Li L, Ling S, Fan N, Fang M, Zhang H, Fang X, Lan W, Hou Z, Meng Q, et al. Metformin enhances radiation response of ECa109 cells through activation of ATM and AMPK. Biomed Pharmacother. 2015; 69:260–266.
35. Honjo S, Ajani JA, Scott AW, Chen Q, Skinner HD, Stroehlein J, Johnson RL, Song S. Metformin sensitizes chemotherapy by targeting cancer stem cells and the mTOR pathway in esophageal cancer. Int J Oncol. 2014; 45:567–574.
36. Zhang Y, Storr SJ, Johnson K, Green AR, Rakha EA, Ellis IO, Morgan DA, Martin SG. Involvement of metformin and AMPK in the radioresponse and prognosis of luminal versus basal-like breast cancer treated with radiotherapy. Oncotarget. 2014; 5:12936–12949. doi: 10.18632/oncotarget.2683.
37. Cerezo M, Tichet M, Abbe P, Ohanna M, Lehraiki A, Rouaud F, Allegra M, Giacchero D, Bahadoran P, Bertolotto C, et al. Metformin blocks melanoma invasion and metastasis development in AMPK/p53-dependent manner. Mol Cancer Ther. 2013; 12:1605–1615.
38. Hsieh SC, Tsai JP, Yang SF, Tang MJ, Hsieh YH. Metformin inhibits the invasion of human hepatocellular carcinoma cells and enhances the chemosensitivity to sorafenib through a downregulation of the ERK/JNK-mediated NF-kappaB-dependent pathway that reduces uPA and MMP-9 expression. Amino Acids. 2014; 46:2809–2822.
39. Janzer A, German NJ, Gonzalez-Herrera KN, Asara JM, Haigis MC, Struhl K. Metformin and phenformin deplete tricarboxylic acid cycle and glycolytic intermediates during cell transformation and NTPs in cancer stem cells. Proc Natl Acad Sci USA. 2014; 111:10574–10579.
40. Lonardo E, Cioffi M, Sancho P, Sanchez-Ripoll Y, Trabulo SM, Dorado J, Balic A, Hidalgo M, Heeschen C. Metformin targets the metabolic achilles heel of human pancreatic cancer stem cells. PLoS One. 2013; 8:e76518.
41. Saito T, Chiba T, Yuki K, Zen Y, Oshima M, Koide S, Motoyama T, Ogasawara S, Suzuki E, Ooka Y, et al. Metformin, a diabetes drug, eliminates tumor-initiating hepatocellular carcinoma cells. PLoS One. 2013; 8:e70010.
42. Zhang Y, Guan M, Zheng Z, Zhang Q, Gao F, Xue Y. Effects of metformin on CD133+ colorectal cancer cells in diabetic patients. PLoS One. 2013; 8:e81264.
43. Xiao Z, Sperl B, Ullrich A, Knyazev P. Metformin and salinomycin as the best combination for the eradication of NSCLC monolayer cells and their alveospheres (cancer stem cells) irrespective of EGFR, KRAS, EML4/ALK and LKB1 status. Oncotarget. 2014; 5:12877–12890. doi: 10.18632/oncotarget.2657.
44. Gritti M, Wurth R, Angelini M, Barbieri F, Peretti M, Pizzi E, Pattarozzi A, Carra E, Sirito R, Daga A, Curmi PM, Mazzanti M, Florio T. Metformin repositioning as antitumoral agent: selective antiproliferative effects in human glioblastoma stem cells, via inhibition of CLIC1-mediated ion current. Oncotarget. 2014; 5:11252–11268. doi: 10.18632/oncotarget.2617.
45. Cui X, Oonishi K, Tsujii H, Yasuda T, Matsumoto Y, Furusawa Y, Akashi M, Kamada T, Okayasu R. Effects of carbon ion beam on putative colon cancer stem cells and its comparison with X-rays. Cancer Res. 2011; 71:3676–3687.
46. Oonishi K, Cui X, Hirakawa H, Fujimori A, Kamijo T, Yamada S, Yokosuka O, Kamada T. Different effects of carbon ion beams and X-rays on clonogenic survival and DNA repair in human pancreatic cancer stem-like cells. Radiother Oncol. 2012; 105:258–265.
47. Kanai T, et al. Examination of gye system for himac carbon therapy. Int J Radiat Oncol Biol Phys. 2006;64:650–656.

