INTRODUCTION
HCC is a common malignant tumor causing high cancer related mortality in East Asia, especially in China [1]. Hepatic resection and liver transplantation have been commonly applied in patients with early stage of the disease; and these approaches have largely improved the disease free survival (DFS) and overall survival (OS) in some HCC patients. However, intrahepatic recurrence and distant metastasis remain to be main factors accounting for the failure of treatments [2]. Therefore, it is urgent to delineate factors that promote the recurrence and metastasis of the disease and its underlying mechanisms.
Emerging evidences suggest that PVI occurs in early stage of HCC is associated with the metastasis and predicts poor prognosis [3–5]. HCC patients complicated with PVI will have a median survival time of less than 4 months if no effective interventions were taken promptly [5]. Recently, studies have demonstrated several risk or predicting factors for PVI, such as tumor size, grade of differentiation, capsule formation, serum level of alpha fetoprotein (AFP), alkaline phosphatase (ALP) and platelet counts, by using univariable or multivariable analysis [6]. However, few of them give a distinct definition for the relationship between the factors and PVI. Currently, knowledge on the risk factors and the corresponding underlying mechanisms that account for early presence of PVI are still lacking.
Vasculogenic mimicry (VM) was identified in melanoma at the end of last century and is accepted presently as a predictor for poor prognosis in numerous solid malignant tumors including HCC [7–12]. During tumor development, when the endothelia dependent angiogenesis from host is incapable of matching the need of nutrition and oxygen supplements for tumor growth, VM occurs and is in charge of the blood supply [13]. A classic VM channel is lined internally with high aggressive tumor cells and a layer of basal lamina rich in extracellular matrix (ECM) [14]. With the progression of cancer, VM gradually develops into another mosaic vessel, a vessel constructed with tumor cells combining with endothelial cells together, ultimately connects and merges with endothelial dependent vessels (EDV) [15]. Cumulating discoveries show that presence of VM could be considered as a metastatic route as well as a potential pro-metastasis factor [13].
Notch1 is a predominant receptor of Delta-like ligand and Notch (DLL-Notch) receptor family and contributes to cell fate decision, proliferation and differentiation. As an angiogenesis promoting molecule, Notch1 together with vascular endothelial growth factor A (VEGFA) manipulate sprouting angiogenesis and guarantee the vessels functions [16]. In addition, Notch1 also functions as an oncogene in a number of malignancies including HCC [17–19]. There are accumulating evidences indicating the association between Notch and VM formation in HCC [20, 21]. Our previous results (manuscript in press) proved that increased Notch1 was associated with VM development in HCC, and overexpression of Notch1 in HepG2 cells could endow the cells with the VM forming ability. Underlying mechanism was that overexpression of Notch1 probably induced epithelial to mesenchymal transition (EMT).
Because of the marked vascularity inside HCC, regional portal vein and its branches turn into the main countercurrent vessel for tumor mass [22]. Tumor related angiogenesis thus potentially contributes to PVI. A study has demonstrated that microvascular density and VEGF have close association with PVI [23]. However, the relationship between VM and PVI is still unclear. We speculated that VM may contribute to the incidence of PVI. The present study was conducted to investigate the relationship between VM and PVI in patients with HCC.
RESULTS
Demographics and clinical characteristics of HCC patients with and without PVI
A total of 44 HCC patients receiving anatomical resection in our hospital was enrolled in the present study and among them, 18 (40.91%) cases were identified with PVI by postoperation pathological examination. To determinate whether demographics and clinical characteristics contributed to PVI, all the patients were divided into two groups, one with PVI and the other without PVI (PVI vs. non-PVI). The demographics and clinical characteristics of these two groups were examined and evaluated using t-test or Chi-square test (Table 1). The majority of these baseline variables showed no significant differences between the two groups except AFP. Statistical result showed a significant increase of AFP in PVI group compared to non-PVI group (P<0.05). As for the etiology, a total of 30 patients (68.18%) were found to be HBV carriers and HBV-DNA copies increased in PVI group, but there was no significant difference between the PVI and non-PVI group (P>0.05). This finding was not surprising because HBV is still the main cause of hepatitis and HCC in China. Among patients with and without PVI, there were significant differences in tumor diameters and Edmonson Steiner grade (P<0.05 and 0.05 respectively), while there were no statistical significances in the tumor marginal state and liver cirrhosis complication (P>0.05 and 0.05 respectively).
Table 1: Demographics and clinical characteristics of HCC patients with and without PVI
Characteristics |
All (n=44) |
PVI |
Non-PVI (n=26) |
P value |
|
Age |
58.52±8.13 |
58.67±8.76 |
58.42±7.83 |
0.924 |
|
Gender |
Male |
38 (86.36%) |
16 (88.89%) |
22 (84.62%) |
1.000 |
Female |
6 (13.64%) |
2 (11.11%) |
4 (15.38%) |
||
Alcoholism |
Yes |
9 (20.45%) |
3 (16.67%) |
6 (23.08%) |
0.716 |
No |
35 (79.55%) |
15 (83.33%) |
20 (76.92%) |
||
HBV-DNA |
≥1000cps/ml |
30 (68.18%) |
15 (83.33%) |
15 (57.69%) |
0.104 |
<1000cps/ml |
14 (31.82%) |
3 (16.67%) |
11(42.31%) |
||
HCV* |
N/A |
N/A |
N/A |
-# |
|
ALT (IU/L) |
48.89±6.28 |
56.16±45.32 |
43.85±44.08 |
0.373 |
|
TBIL (μmol/ml) |
15.73±6.28 |
16.51±5.11 |
15.20±7.02 |
0.504 |
|
Child-Pugh classification |
A |
37 (84.09%) |
14 (77.78%) |
23 (88.46%) |
0.419 |
B |
7 (15.91%) |
4 (22.22%) |
3 (11.54%) |
||
AFP |
≥400 ng/ml |
34 (77.27%) |
17 (94.44%) |
17 (65.38%) |
0.031* |
<400 ng/ml |
10 (22.73%) |
1 (5.56%) |
9 (34.62%) |
||
ECOG Scores |
0-1 |
34 (77.27%) |
12 (66.67%) |
22 (84.62%) |
0.273 |
2 |
10 (22.73%) |
6 (33.33%) |
4 (15.38%) |
||
Resection margin |
R0 |
40 (90.91%) |
17 (94.44%) |
23 (88.46%) |
0.634 |
R1 |
4 (9.09%) |
1 (5.56%) |
3 (11.54%) |
||
Cirrhosis |
Yes |
35 (79.55%) |
15 (83.33%) |
20 (76.92%) |
0.716 |
No |
9 (20.45%) |
3 (16.67%) |
6 (23.08%) |
||
Tumor diameters |
6.84±2.85 |
8.06±2.14 |
6.00±3.01 |
0.017* |
|
Edmonson-Steiner grade |
I-II |
37 (84.09%) |
12 (66.67%) |
25 (96.15%) |
0.013* |
III-IV |
7 (15.91%) |
6 (33.33%) |
1 (3.85%) |
SD, standard deviation; HBV, hepatitis B virus; HCV, hepatitis C virus; ALT, alanine aminotransferase; AFP, Alpha-fetoprotein; TBIL, total bilirubin; N/A, not available. # missing data. * P<0.05.
Prevalence of VM in HCC patients with and without PVI
Since VM has no endothelial cells and is consisted of tumor cells with basement membrane components, it is not stained by CD34 but stained positively by PAS. Therefore, net-like structures with PAS positive and CD34 negative staining were considered as positive VM formation in tumor tissues. In this study, 17 (33.64%, 17/44) cases with VM were found. Among the clinical characteristics summarized in Table 2, except for Edmonson-Steiner grade (r=0.421, P<0.01, Figure 1 and Table 2), none of them were found to be associated with VM formation. Positive VM structures were found both in PVI and non-PVI group. There were 13 (72.22%, 13/18) cases with VM in PVI group while only 4 (15.38%, 4/26) cases in non-PVI group (P<0.001, Figure 1). The linear regression analysis further confirmed that VM formation had a positive correlation with PVI (r=0.574, P<0.001, Figure 1).
Table 2: Correlations between VM and clinical characteristics
Variables |
Correlation coefficient |
P value |
Age |
0.044 |
0.776 |
Gender |
0.093 |
0.549 |
Alcoholism |
0.119 |
0.443 |
HBV-DNA |
0.141 |
0.361 |
HCV* |
- |
- |
ALT (IU/L) |
0.226 |
0.140 |
TBIL (μmol/ml) |
0.1242 |
0.359 |
Child-Pugh classification |
0.176 |
0.253 |
AFP |
0.010 |
0.948 |
ECOG Scores |
0.015 |
0.922 |
Resection margin |
0.021 |
0.873 |
Cirrhosis |
0.055 |
0.722 |
Tumor diameters |
0.204 |
0.184 |
Edmonson-Steiner grade |
0.421 |
0.004# |
* missing data, # significant difference.
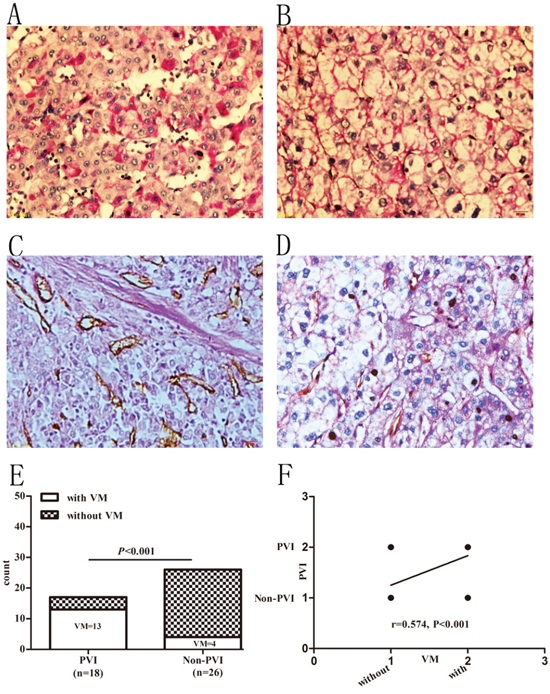
Figure 1: Prevalence of VM in HCC patients with and without PVI. A and B. PAS staining in HCC specimens. A: PAS staining in well to moderate differentiated samples. B: PAS staining in poorly differentiated samples. C and D. PAS-CD34 double-staining. C: specimen without VM. D: specimen with VM. Original magnification: 400×. E. Prevalence of VM in PVI and non-PVI groups. F. Presence of VM positively associated with PVI.
Notch1 associated with VM formation and PVI
Some researchers suggested that Notch1 played important roles in VM formation and correlated with poor prognosis in HCC [20]. Our previous results showed that Notch1 expression has a positive correlation with VM in HCC (r=0.590, P<0.001) and artificial overexpression of Notch1 in HCC cells could promote VM (manuscript in press). To evaluate whether Notch1 expression correlated with PVI, we compared it among HCC patients with and without PVI. As shown in Figure 2, both in PVI and non-PVI groups, IHC scores for Notch1 (116.81±25.57 and 72.22±35.53) were found increased compared to corresponding adjacent non-HCC tissues (49.99±21.63, P<0.001 and 0.001 respectively). In addition, PVI group had a higher Notch1 score than non-PVI group (P<0.01, Figure 2). Correlation analysis indicated that Notch1 expression had a close association with PVI (r=0.593, P<0.001, Figure 2). Combining with our previous findings, the results suggested that expression of Notch1 is associated with VM formation and PVI.
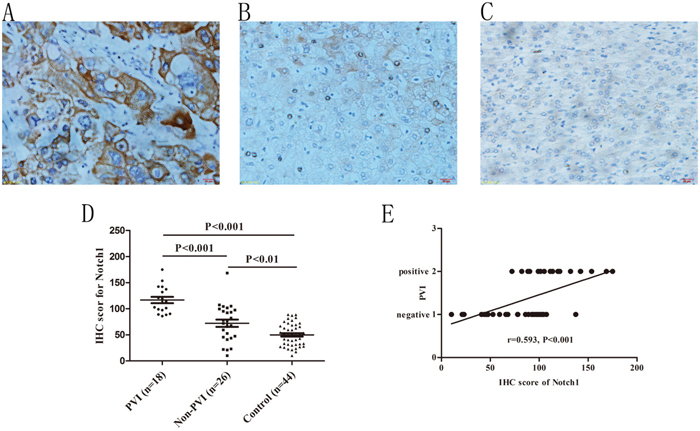
Figure 2: Immunohistochemical staining of Notch1 in HCC specimens and its association with PVI. A. Notch1 staining in HCC specimens with PVI, B. Notch1 staining in HCC specimens without PVI, C. Notch1 staining in adjacent non-tumor specimens. Original magnification: 400×. D. Different staining density and proportion of Notch1 in specimens of PVI, non-PVI and control (non-tumor) specimens. E. Spearman correlation analysis indicated a positive correlation between Notch1 expression and PVI.
Positive vimentin staining in HCC cells associated with PVI
Vimentin was normally expressed in the stroma and microvascular wall of HCC but not in HCC cells. A positive staining of Vimentin in HCC cells tended to reflect epithelial to mensenchymal transition (EMT) which was suggested to contribute to VM formation [24]. In this study we found 12 (27.27%, 12/44) specimens with positive Vimentin staining in the cytoplasm of HCC cells. As for its counterpart, E-cadherin was found decreased in the 12 cases compare to the rest 32 cases (26.09±12.70 vs. 48.51±31.15, P<0.01, Figure 3). Correlation analysis confirmed that Vimentin was positively correlated with VM formation (r=0.963, P<0.001) while E-cadherin was negatively correlated with VM (r=-0.527, P<0.001). In our previous study we found that overexpression or knockdown expression of Notch1 in HCC cells could increase or decrease Vimentin expression (manuscript in press). In accordance with this finding, we found in this study that increased Vimentin and decreased E-cadherin were positively and negatively correlated with Notch1 expression respectively (r=0.561, P<0.001; r=-0.524, P<0.001). In PVI group we found 10 (55.56%, 10/18) cases with positive Vimentin staining while only 2 (7.69%, 2/26) cases in non-PVI group (P<0.001, Figure 3). E-cadherin was found significantly declined in PVI group compared with non-PVI (24.30±12.83 vs. 54.92±30.64, P<0.001, Figure 3). Liner regression analysis confirmed that increasing Vimentin and decreasing E-cadherin were closely associated with PVI (r=0.582, P<0.001; r=-0.572, P<0.001, respectively. Figure 3).
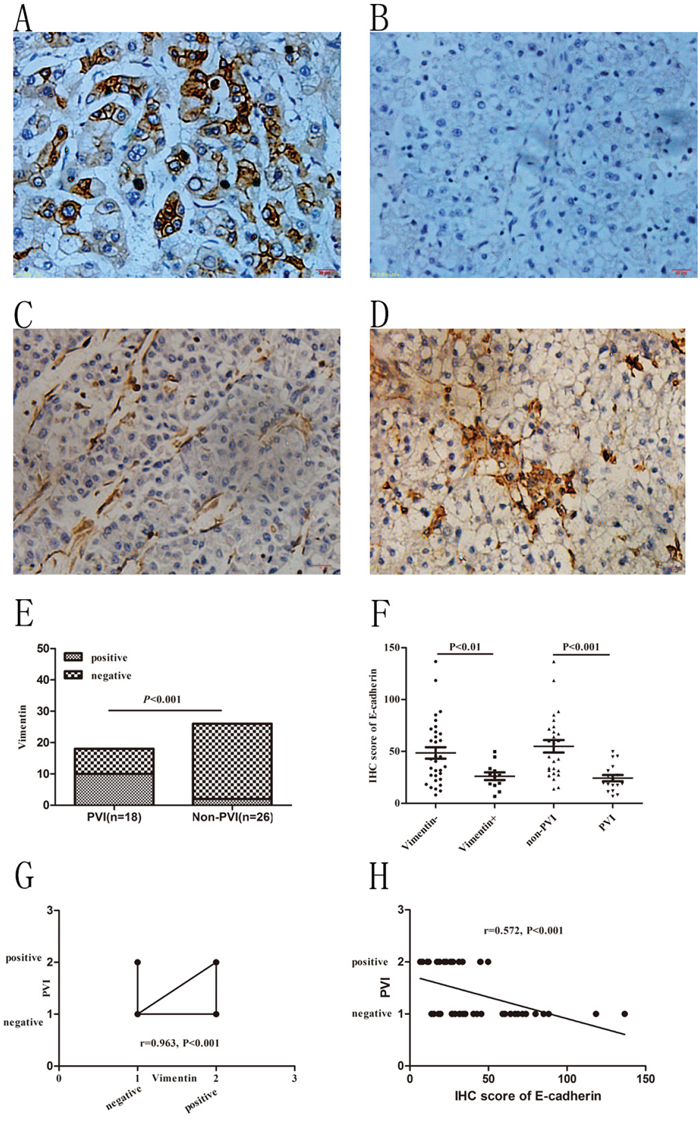
Figure 3: Immunohistochemical staining of Vimentin in HCC specimens and its association with VM and PVI. A and B. E-cadherin staining in non-PVI and PVI samples. C and D. Negative and positive Vimentin staining in non-PVI and PVI samples. Original magnification was 400×. E. Incidence of positive vimentin staining in PVI and non-PVI patients. Chi-Square test was used. F. E-cadherin staining in Vimentin positive and negative samples; E-cadherin staining in non-PVI and PVI samples. T-test was used. G. Increased Vimentin positively associated with PVI. Spearman correlation was used. H. Decreased E-cadherin negatively associated with PVI. Spearman Correlation analysis was used.
Increase of MMPs associated with VM formation and indicated higher incidence of PVI
Emerging evidences have suggested that Notch1 induces increase of MMP-2 and MMP-9 expression that were important in cancer invasion and the extracellular matrix remolding during VM development [25, 26]. In this study, we found that expression of MMP-2 and MMP-9 were positively correlated with Notch1 expression (r=0.405, P<0.01; r=0.409, P<0.01 respectively, Figure 4). Furthermore, the increased expressions of MMP-2 and MMP-9 were positively associated with VM formation (r=0.425, P<0.001; r=0.399, P<0.01 respectively, Figure 4). In patients with PVI, IHC scores of MMP-2 and MMP-9 were 98.63±24.89 and 105.21±34.48 respectively, while in patients without PVI, the scores were 71.94±37.14 and 74.42±27.87 respectively. The expressions of MMP-2 and MMP-9 have significant difference between the PVI and non-PVI groups (P<0.01 and 0.01 respectively, Figure 4). Further correlation analysis indicated that MMP-2 and MMP-9 have close associations with PVI (r=0.0.382, P<0.05; r=0.422, P<0.01 respectively, Figure 4). These results suggested that overexpression of MMP-2 and MMP-9 is associated with VM formation and PVI.
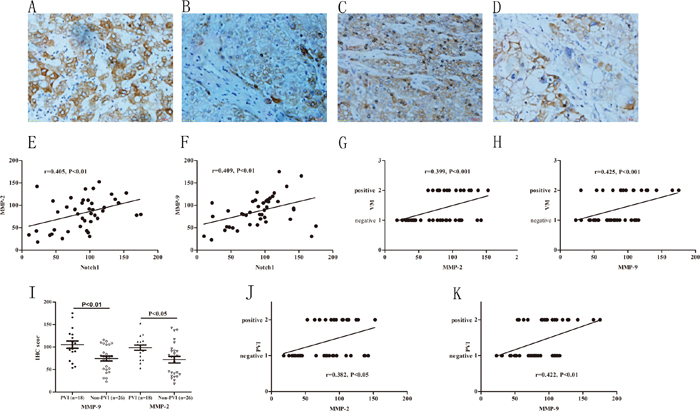
Figure 4: Increased expressions of MMPs associated with VM and higher incidence of PVI. A and B. MMP-9 immunohistochemical staining in HCC with and without PVI, original magnification: 400×. C and D. MMP-2 immunohistochemical staining in HCC with and without PVI, original magnification: 400×. E and F. MMP-2, MMP-9 IHC score were correlated with Notch1. G and H. MMP-2 and MMP-9 expression positively correlated with VM. I: MMP-2 and MMP-9 significantly increased in PVI group compared with non-PVI group. J and K: MMP-2, MMP-9 IHC score were positively correlated with PVI.
DISCUSSION
A number of studies have demonstrated that PVI exists in 44%-62.2% of early and mid-stage HCC patients who receive hepatic resection or liver transplantation [22]. Occurrence of PVI indicates a poor prognosis [4]. Cancer staging and postoperative treatment varied in patients with or without PVI. Currently, clinical trials have been conducted to validate various techniques for example surgical resection, transcatheter arterial chemoembolization (TACE), hepatic arterial infusion chemotherapy, radioembolization and sorafenib for the treatment of HCC [27–29]. Besides, attention has been paid to understand the promoting factors of PVI and corresponding mechanisms [22]. Our current study contributed to elucidate the role of angiogenesis related molecules such as Notch1, Vimentin, MMP-2 and MMP-9 in promoting VM formation and PVI in HCC. Evidence provided in this study supports the hypothesis that VM, as a pathological auto circulation in the tumor mass, may induce PVI in HCC.
The interaction between tumor internal circulation and the host blood supply are critical in malignancies that depend on hematogenous spread such as HCC. VM channel provides a pathway and functions as an important promoter in early tumor invasion and metastasis. VM normally exists in the early phase of tumor expansion and supplies the nutrition and oxygen together with mosaic vessel and endothelial dependent vessel and thus provides a chance for tumor cells to be exposed directly to blood flow [30]. On the other hand, cells lining VM channels normally exhibit poor pathological characteristics such as high invasiveness, plasticity, self-differentiation and potentiality of ECM [31]. These features have been suggested to be associated with tumor metastasis.
Currently, the precise mechanisms related to PVI development are still contentious. Not only the aberrant vascular structures inside tumor mass but also the malignant phenotype of tumor cells and microenvironment in local portal vein are all contributors of PVI. In this study, we demonstrated that VM participates in tumor blood supply via connecting to endothelia-derived-vessels (EDV) originated from the host. This pathological vasoganglion not only promotes vascular abnormality, decreases blood supply resistance, subsequently leads to the hypertension of local vein; but also provides a shortcut for tumor cells to escape from the origin and migrate to the distance. In addition, with the development of VM, tumor cells obtain more malignant characteristics such as deattachment, migration, invasion and the ability to degrade and remold extracellular matrix (ECM), these factors are the co-promotors of PVI [22].
It has been documented that angiogenesis related molecules such as VEGFR1, Notch, Twist, ZEB1, Snail and Slug are involved in VM formation [30]. Among these molecules, Notch is extensively studied in various malignancies for its roles in proliferation, differentiation, invasion and metastasis. A previous report demonstrates that Notch1 is involved in VM formation in HCC and suppressing Notch1 inhibits the progression of VM [20]. A recent study confirmed that Notch participated in VM by manipulating NODAL expression [32]. Our previous study shows that increased Notch1 is associated with VM formation in HCC. Increased Notch1 induced by TGF-β1 or lentivirus mediated overexpression can promote HCC cell forming VM both in vitro and in vivo (manuscript in press). In the present study, Notch1 expression is higher in PVI group compared with non-PVI group. Increased Notch1 has a close association with PVI. Combining with our previous results, it is rational to consider that Notch1 promotes VM formation in HCC and subsequently induces the occurrence of PVI.
Existing data have shown a positive impact of EMT and ECM degradation on VM channel formation [33]. During EMT process, Vimentin is predominantly expressed while E-cadherin is suppressed. As a result, vimentin and E-cadherin were used as important markers for EMT in studies related to VM formation [34]. Alteratively, acquisition of mesenchymal phenotype, especially endothelial phenotype indicates the acquisition of potentiality for angiogenesis. In addition, decrease of E-cadherin and increase of Vimentin allow tumor cells easily to escape from the origin and metastasize. During degradation of ECM, MMPs especially MMP-2 and MMP-9 contribute to the proteolysis of the ECM whose degradation products together with tumor cells take part in the VM formation [35]. Although the roles of EMT and ECM degradation in VM formation have been well documented, the association between the two episodes and PVI was hardly proved. As shown in Figure 5, EMT promotes VM formation thus offers structure base for PVI. Simultaneously, changes of Vimentin and E-cadherin, increase of MMPs lead HCC cells easily to enter into portal vein and subsequently degrade matrix and seed on the vessel wall. Collectively, these findings support the hypothesis that VM formation is associated with PVI.
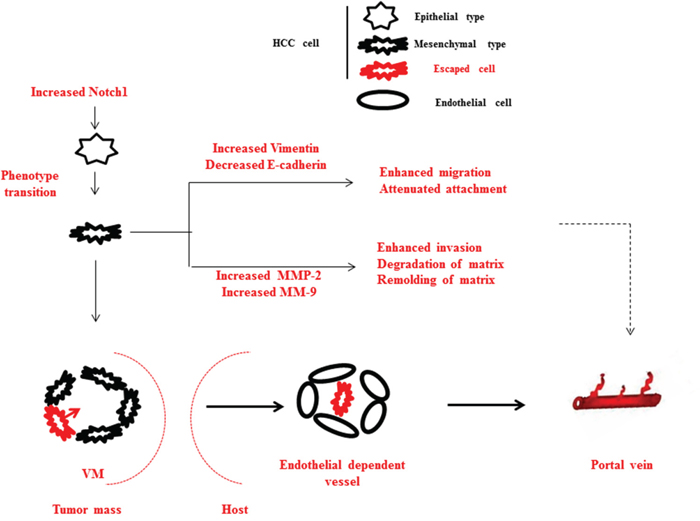
Figure 5: Schematic diagram of summary of this study. Elevated Notch1 induces a cohort of tumor cells translate from epithelial to mesenchymal phenotype in HCC. During the process, Vimentin increases and E-cadherin decreases in cells with malignant phenotype, therefore migration and deattachment are enhanced; MMP-2 and MMP-9 increases, therefore ECM degradation and remolding are enhanced. These support the PVI development at the cellular level. Additionally, EMT induced VM aggravates vasoganglion abnormality inside tumor mass, connects portal vein and internal circulation of tumor mass, therefore offers pathways for tumor cells to metastasize into portal vein. These support the PVI development at the circulation level.
This study has some limitations. It was reported that HBV infection can promote PVI via activating TGF-β signaling [22]. However we fail to find the significant differences of HBV infection and hepatic fibrosis between PVI and non-PVI group. Further analysis based on larger sample size and broader mechanism exploration depended on cellular and molecular biological experiments should be performed. Although we proved that VM connects to dismal branches of portal vein via EDV by pathological staining methods and statistics, corresponding ultrastructure and mechanisms are still unclear. In our previous study we have demonstrated that Notch1 is an upstream factor that regulate the expression of Vimentin and E-cadherin, MMP-9 and MMP-2. However, in the current study, we are still unable to explain how does Notch1 decide or impact the HCC cells to complete the terminal seeding and transplantation in portal vein. A larger cohort observation and some complex mechanism studies should be performed. AFP was found different between PVI and non-PVI group. We consider this may be associated with potentially subclinical tumor activation.
In summary, our results demonstrated that VM formation is positively correlated with PVI in HCC. Overexpression of Notch1, followed by the changes of expression of Vimentin, E-cadherin and MMPs are closely associated with PVI in HCC. These findings will help prognostic estimation and provide treatment target for HCC patients with PVI.
MATERIALS AND METHODS
Patients and HCC specimens
Previous study suggested that incidence of PVI in HCC patients receiving resection is approximately 46.8% [36]. According to the reported incidence, we estimated the sample size with the 80% statistical power and the result indicated the sample size should be more than 30 cases. As a result, a total of 44 patients who were diagnosed with HCC and received anatomic liver resection in the Second People’s Hospital of Taizhou (Jiangsu, China) between 2009 and 2015 were enrolled in the study. The post-operative histopathologic diagnosis for HCC and PVI were independently confirmed by two pathologists through routine histopathological examination based on H&E staining. Individuals with mixed hepatocellular carcinoma and cholangiocarcinoma were excluded. None of the 44 patients received any preoperative anti-cancer treatments. The demographic and clinical characteristics were collected from the electronic medical record system. All specimens were fixed in formalin and embedded in paraffin. This study was reviewed and approved by the institutional ethic committee of the Second People’s Hospital of Taizhou. All patients were requested to sign the informed consent for participation. The characteristics of patients were summarized in Table 1.
Immunohistochemical staining
Immunohistochemical staining was performed as previously described [37]. Briefly, 4μm thick sections were routinely dewaxed and rehydrated. Sections were then heated at 100°C in citrate buffer (0.01 M, pH 6.0) for 10 min using a microwave oven for antigen retrieval. After that, sections were treated with 3% H2O2 for 20 min to inactivate the endogenous peroxidase and incubated with 10% goat serum for 30 min to block the nonspecific binding. Next, sections were incubated with primary antibodies at 4°C overnight. The list of antibodies used was summarized in Table 3. After rinsing, sections were incubated with biotin labeled secondary antibody against responding primary antibodies for 30 min at 37°C. Then sections were rinsed and incubated with 3,3’-diaminobenzidinetetrahydrochloride (DAB) for 10 min at room temperature. Slides were then counterstained with hematoxylin. Negative control was achieved by replacing the primary antibodies with PBS.
Table 3: Primary antibodies used in immunohistochemical staining
Primary antibodies |
Serial Number |
Dilution |
Source |
Company |
Notch1 |
4380 |
1:100 |
Rabbit |
CST, Danforth, USA |
MMP-2 |
Ab7033 |
1:200 |
Mouse |
Abcam, Camb, UK |
MMP-9 |
Ab73734 |
1:200 |
Rabbit |
Abcam, Camb, UK |
E-cadherin |
3195 |
1:150 |
Rabbit |
CST, Danforth, USA |
Vimentin |
5741 |
1:100 |
Rabbit |
CST, Danforth, USA |
CD34 |
3569 |
1:50 |
Mouse |
CST, Danforth, USA |
CST, cell signaling technology.
PAS-CD34 dual staining
The staining procedure was performed as previous description [38]. Briefly, immunohistochemical staining was applied to perform CD34 staining. The procedure was the same as the description above (CD34 primary antibody dilution was 1:200). After immunihistochemical staining, sections were treated with 0.5% periodic acid solution for 10 min and rinsed with distilled water for 5 min. In a dark chamber, sections were treated with Schiff solution for 15-30 min. After rinsing with distilled water, sections were counterstained with hematoxylin.
Staining evaluation
All the slides were evaluated by two independent pathologists who were blinded with the study background and outcomes. For immunohistochemical staining of Notch1, E-cadherin, MMP-9 and MMP-2, the slides were imaged digitally with the same light exposure and evaluated by Image Pro Plus (IPP), a digitalized immunohistochemistry scoring program (Media Cybernetics, San Diego, CA). For immunohistochemical staining of Vimentin, because the target staining location was in cytoplasm or cell membrane other than microvascular wall, the staining located in HCC cells agreed by two pathologists was considered positive staining; conversely, it was considered negative staining if the staining was in microvascular wall.
Statistical analysis
Comparisons between PVI and non-PVI groups were performed using the Student’s t-test for continuous variables according to normal distribution and Chi-square test for categorical variables. Pearson and Spearman correlation coefficient were used to determine the relationship between two variables. P values less than 0.05 were considered statistically significant. All analysis was performed with SPSS software version 22.0 for Windows (SPSS, Chicago, IL).
ACKNOWLEDGMENTS
We thank the staffs in the Department of Clinical Pathology, the Second People’s Hospital of Taizhou City, Yangzhou University for help collecting specimens. We thank Prof. Shen Yi (Nan Tong University, Jiangsu, China) for her help in statistical work.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
GRANT SUPPORT
This work was supported by the National Natural Science Foundation (Grant No. 81274141, 81450051, 81403232 and 81573656).
REFERENCES
1. Chen BB, Murakami T, Shih TT, Sakamoto M, Matsui O, Choi BI, Kim MJ, Lee JM, Yang RJ, Zeng MS, Chen RC, Liang JD: Novel Imaging Diagnosis for Hepatocellular Carcinoma: Consensus from the 5th Asia-Pacific Primary Liver Cancer Expert Meeting (APPLE 2014), Liver cancer 2015; 4:215-227.
2. Yamamoto Y, Ikoma H, Morimura R, Konishi H, Murayama Y, Komatsu S, Shiozaki A, Kuriu Y, Kubota T, Nakanishi M, Ichikawa D, Fujiwara H, Okamoto K, Sakakura C, Ochiai T, Otsuji E: Changing trends in long-term outcomes after hepatic resection for hepatocellular carcinoma: A 30-year, single-center experience, Anticancer research 2013; 33:5097-5105.
3. de la Torre MA, Buades-Mateu J, de la Rosa PA, Lue A, Bustamante FJ, Serrano MT, Testillano M, Lorente S, Arenas JI, Gil C, Inarrairaegui M, Sangro B: A comparison of survival in patients with hepatocellular carcinoma and portal vein invasion treated by radioembolization or sorafenib, Liver international: official journal of the International Association for the Study of the Liver 2016; 36:1206-1212.
4. Sinn DH, Cho JY, Gwak GY, Paik YH, Choi MS, Lee JH, Koh KC, Paik SW, Yoo BC: Different survival of Barcelona clinic liver cancer stage C hepatocellular carcinoma patients by the extent of portal vein invasion and the type of extrahepatic spread, PloS one 2015; 10:e0124434.
5. Shirabe K, Kajiyama K, Harimoto N, Masumoto H, Fukuya T, Ooya M, Maehara Y: Prognosis of hepatocellular carcinoma accompanied by microscopic portal vein invasion, World journal of gastroenterology 2009; 15:2632-2637.
6. Shirabe K, Kajiyama K, Abe T, Sakamoto S, Fukuya T, Akazawa K, Morita K, Maehara Y: Predictors of microscopic portal vein invasion by hepatocellular carcinoma: measurement of portal perfusion defect area ratio, Journal of gastroenterology and hepatology 2009; 24:1431-1436.
7. Folberg R, Hendrix MJ, Maniotis AJ: Vasculogenic mimicry and tumor angiogenesis, The American journal of pathology 2000; 156:361-381.
8. Wang L, Lin L, Chen X, Sun L, Liao Y, Huang N, Liao W: Metastasis-associated in colon cancer-1 promotes vasculogenic mimicry in gastric cancer by upregulating TWIST1/2, Oncotarget 2015; 6:11492-11506. doi: 10.18632/oncotarget.3416.
9. Zhou L, Yu L, Feng ZZ, Gong XM, Cheng ZN, Yao N, Wang DN, Wu SW: Aberrant Expression of Markers of Cancer Stem Cells in Gastric Adenocarcinoma and their Relationship to Vasculogenic Mimicry, Asian Pacific journal of cancer prevention: APJCP 2015; 16:4177-4183.
10. Wang H, Lin H, Pan J, Mo C, Zhang F, Huang B, Wang Z, Chen X, Zhuang J, Wang D, Qiu S: Vasculogenic Mimicry in Prostate Cancer: The Roles of EphA2 and PI3K, Journal of Cancer 2016; 7:1114-1124.
11. Chen YS, Chen ZP: Vasculogenic mimicry: a novel target for glioma therapy, Chinese journal of cancer 2014; 33:74-79.
12. Liu WB, Xu GL, Jia WD, Li JS, Ma JL, Chen K, Wang ZH, Ge YS, Ren WH, Yu JH, Wang W, Wang XJ: Prognostic significance and mechanisms of patterned matrix vasculogenic mimicry in hepatocellular carcinoma, Med Oncol 2011; 28:S228-238.
13. Seftor RE, Hess AR, Seftor EA, Kirschmann DA, Hardy KM, Margaryan NV, Hendrix MJ: Tumor cell vasculogenic mimicry: from controversy to therapeutic promise, The American journal of pathology 2012; 181:1115-1125.
14. Maniotis AJ, Folberg R, Hess A, Seftor EA, Gardner LM, Pe'er J, Trent JM, Meltzer PS, Hendrix MJ: Vascular channel formation by human melanoma cells in vivo and in vitro: vasculogenic mimicry, The American journal of pathology 1999; 155:739-752.
15. Sun B, Zhang D, Zhao N, Zhao X: Epithelial-to-endothelial transition and cancer stem cells: two cornerstones of vasculogenic mimicry in malignant tumors, Oncotarget 2016 Mar 29. doi: 10.18632/oncotarget.8461. [Epub ahead of print].
16. Li JL, Harris AL: Crosstalk of VEGF and Notch pathways in tumour angiogenesis: therapeutic implications, Front Biosci (Landmark Ed) 2009; 14:3094-3110.
17. Upadhyay P, Nair S, Kaur E, Aich J, Dani P, Sethunath V, Gardi N, Chandrani P, Godbole M, Sonawane K, Prasad R, Kannan S, Agarwal B, Kane S, Gupta S, Dutt S, Dutt A: Notch pathway activation is essential for maintenance of stem-like cells in early tongue cancer, Oncotarget 2016; 7:50437-50449. doi: 10.18632/oncotarget.10419.
18. Mirone G, Perna S, Shukla A, Marfe G: Involvement of Notch-1 in Resistance to Regorafenib in Colon Cancer Cells, Journal of cellular physiology 2016; 231:1097-1105.
19. Gao J, Song Z, Chen Y, Xia L, Wang J, Fan R, Du R, Zhang F, Hong L, Song J, Zou X, Xu H, Zheng G, Liu J, Fan D: Deregulated expression of Notch receptors in human hepatocellular carcinoma, Digestive and liver disease: official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver 2008; 40:114-121.
20. Vartanian A, Gatsina G, Grigorieva I, Solomko E, Dombrovsky V, Baryshnikov A, Stepanova E: The involvement of Notch signaling in melanoma vasculogenic mimicry, Clinical and experimental medicine 2013; 13:201-209.
21. Qiu XX, Chen L, Wang CH, Lin ZX, Chen BJ, You N, Chen Y, Wang XF: The Vascular Notch Ligands Delta-Like Ligand 4 (DLL4) and Jagged1 (JAG1) Have Opposing Correlations with Microvascularization but a Uniform Prognostic Effect in Primary Glioblastoma: A Preliminary Study, World neurosurgery 2016; 88:447-458.
22. Cheng S, Yang J, Shen F, Zhou W, Wang Y, Cong W, Yang GS, Cheng H, Hu H, Gao C, Guo J, Li A, Meng Y et al. Multidisciplinary management of hepatocellular carcinoma with portal vein tumor thrombus - Eastern Hepatobiliary Surgical Hospital consensus statement, Oncotarget 2016; 7:40816-40829. doi: 10.18632/oncotarget.8386.
23. Zhou J, Tang Z, Fan J: [Expression rates of three angiogenic factors in hepatocellular carcinoma and their relation with microvessel density and portal vein tumor thrombosis], Zhonghua yi xue za zhi 2001; 81:462-464.
24. Meng J, Sun B, Zhao X, Zhang D, Gu Q, Dong X, Zhao N, Liu P, Liu Y: Doxycycline as an inhibitor of the epithelial-to-mesenchymal transition and vasculogenic mimicry in hepatocellular carcinoma, Molecular cancer therapeutics 2014; 13:3107-3122.
25. Zhao Y, Qiao X, Tan TK, Zhao H, Zhang Y, Liu L, Zhang J, Wang L, Cao Q, Wang Y, Wang YM, Lee VW, Alexander SI, Harris DC, Zheng G: Matrix metalloproteinase 9-dependent Notch signaling contributes to kidney fibrosis through peritubular endothelial-mesenchymal transition, Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 2016.
26. Yu B, Wei J, Qian X, Lei D, Ma Q, Liu Y: Notch1 signaling pathway participates in cancer invasion by regulating MMPs in lingual squamous cell carcinoma, Oncology reports 2012; 27:547-552.
27. Li J, Hou Y, Cai XB, Liu B: Sorafenib after resection improves the outcome of BCLC stage C hepatocellular carcinoma, World journal of gastroenterology 2016; 22:4034-4040.
28. Cho YY, Lee M, Kim HC, Chung JW, Kim YH, Gwak GY, Bae SH, Kim do Y, Heo J, Kim YJ: Radioembolization Is a Safe and Effective Treatment for Hepatocellular Carcinoma with Portal Vein Thrombosis: A Propensity Score Analysis, PloS one 2016; 11:e0154986.
29. Matsumoto T, Kubota K, Aoki T, Iso Y, Kato M, Shimoda M: Clinical Impact of Anatomical Liver Resection for Hepatocellular Carcinoma with Pathologically Proven Portal Vein Invasion, World journal of surgery 2016; 40:402-411.
30. Paulis YW, Soetekouw PM, Verheul HM, Tjan-Heijnen VC, Griffioen AW: Signalling pathways in vasculogenic mimicry, Biochimica et biophysica acta 2010; 1806:18-28.
31. Seftor RE, Seftor EA, Koshikawa N, Meltzer PS, Gardner LM, Bilban M, Stetler-Stevenson WG, Quaranta V, Hendrix MJ: Cooperative interactions of laminin 5 gamma2 chain, matrix metalloproteinase-2, and membrane type-1-matrix/metalloproteinase are required for mimicry of embryonic vasculogenesis by aggressive melanoma, Cancer research 2001; 61:6322-6327.
32. Hardy KM, Kirschmann DA, Seftor EA, Margaryan NV, Postovit LM, Strizzi L, Hendrix MJ: Regulation of the embryonic morphogen Nodal by Notch4 facilitates manifestation of the aggressive melanoma phenotype, Cancer research 2010; 70:10340-10350.
33. Hendrix MJ, Seftor EA, Seftor RE, Chao JT, Chien DS, Chu YW: Tumor cell vascular mimicry: Novel targeting opportunity in melanoma, Pharmacology & therapeutics 2016; 159:83-92.
34. Liu CY, Lin HH, Tang MJ, Wang YK: Vimentin contributes to epithelial-mesenchymal transition cancer cell mechanics by mediating cytoskeletal organization and focal adhesion maturation, Oncotarget 2015; 6:15966-15983. doi: 10.18632/oncotarget.3862.
35. Liu Y, Sun X, Feng J, Deng LL, Li B, Zhu M, Lu C, Zhou L: MT2-MMP induces proteolysis and leads to EMT in carcinomas, Oncotarget 2016; 7:48193-48205. doi: 10.18632/oncotarget.10194.
36. Miyata R, Tanimoto A, Wakabayashi G, Shimazu M, Nakatsuka S, Mukai M, Kitajima M: Accuracy of preoperative prediction of microinvasion of portal vein in hepatocellular carcinoma using superparamagnetic iron oxide-enhanced magnetic resonance imaging and computed tomography during hepatic angiography, Journal of gastroenterology 2006; 41:987-995.
37. Dai K, Qin F, Zhang H, Liu X, Guo C, Zhang M, Gu F, Fu L, Ma Y: Low expression of BMPRIB indicates poor prognosis of breast cancer and is insensitive to taxane-anthracycline chemotherapy, Oncotarget 2016; 7:4770-4784. doi: 10.18632/oncotarget.6613.
38. Zhu MS, Xu LB, Zeng H, Shi XD, Wu WR, Liu C: Association of Notch1 with vasculogenic mimicry in human hepatocellular carcinoma cell lines, International journal of clinical and experimental pathology 2014; 7:5782-5791.

