INTRODUCTION
BMPs are important cytokines belonging to the Transforming Growth Factor (TGF)-β superfamily, which also includes TGF-βs, activins, inhibins, nodal, and myostatin [1, 2]. They were first described by Marshall R. Urist in the 1960s, when he suggested the presence of osteoinductive molecules in demineralized bone matrix extracts, but further gene identification only occurred in the late 1980s [3]. Thus far, more than 20 kinds of BMP ligands have been identified in humans. Based on the disparity of their sequences and functions, these ligands have been divided into at least four subgroups: BMP-2/-4 group; BMP-5/-6/-7/-8 group (osteogenic protein-1 [OP-1] group); BMP-9/-10 group; and BMP-3, -13, -11, -12, -14, and -15 group [4]. Among these ligands, the first three groups were profoundly studied in tumors and have been reported to participate in tumorigenesis and dissemination.
Several reviews have illustrated the rough backbone of the BMP signaling pathways [1, 5]. Briefly, BMP ligands bind to two receptor types (type I and type II) to form a heterotetrameric complex, which then binds to and phosphorylate the receptor-activated (R)-SMADs. The activated R-SMADs bind to the common SMAD (Co-SMAD) to form a complex, which translocates to the nucleus along with a number of transcription factors to drive the target genes expression [6-8]. In addition to the canonical SMAD pathway, BMPs activate the non-canonical SMAD pathways, such as phosphatidylinositol 3-kinase (PI3K)/AKT, mitogen-activated protein kinase (MAPK), nuclear factor kappa B (NF-κB), and Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling pathways, which form a complex network of molecular signals regulating a multitude of processes throughout the body [5, 9, 10].
BMPs were originally reported to induce bone and cartilage formation, which exhibit a wide range of biological effects on various cell types. BMPs play important roles in embryonic and postnatal development by regulating cell differentiation, proliferation, motility, and survival, thus maintaining homeostasis in different organs and tissues [6, 11]. Recently, more evidence demonstrated that BMPs participate in cancer development and progression.
PARADOXICAL EFFECTS OF BMP SIGNALING ON TUMORIGENESIS AND DISSEMINATION
Studies have shown that BMPs display significantly higher expression in tumors, which have been used as new biomarkers for the prognosis of cancer patients. In hepatocellular carcinoma (HCC), several BMPs (BMP-4, -6, -7, -8, -9, -10, -11, -13, and -15) revealed enhanced expression levels [12]. In advanced non-small cell lung cancer, serum increased the BMP-2 level, and the advanced clinical stages were significantly correlated with poor prognosis, which can be regarded as an independent negative predictor for the prognosis of patients [13]. BMP-4 upregulation is closely associated with shorter patients’ overall and disease-free survival, which serves as a novel marker for predicting the recurrence and prognosis of HCC patients after surgery [14]. In addition, high BMP-7 expression could be a useful predictive marker of poor prognosis in patients with lung cancer [15, 16], esophageal squamous cell carcinoma [17], colorectal cancer [18], and clear cell renal carcinoma [19].
The aberrant expression of BMPs is correlated with the proliferation, differentiation, and apoptosis of cancer cells and thus may be regarded as an oncogene. BMP-9 promotes the cell proliferation of ovarian cancer [20]. In addition, BMP-9 triggered the phosphorylation of SMAD1, 5, and 8 and the overexpression of inhibitors of DNA binding 1 (Id1), thereby promoting a proliferative response and exerting a remarkable anti-apoptotic function in HCC cells [21]. BMP-9 also led to an observable alteration in cell cycle regulator expression, including cyclinD1 protein upregulation and the downregulation of CDK-interacting protein p27 expression. Both events are involved in the progression from the G0/G1 phases toward the S-phase of the cell cycle [22, 23]. The BMP-downstream signaling pathway, such as SMAD, has a role in tumor development and metastasis. Reportedly, SMAD1, 5, and 8 promote tumors. For instance, SMAD5 expression is inversely correlated with the prognosis of serious ovarian cancer patients, and BMP-2 stimulated cellular proliferation by inducing phosphorylated SMAD5 (pSMAD5) translocation into the nucleus in ovarian cancer cells [24].
Aside from these impacts on tumorigenesis, BMP signaling is involved in the invasion and migration processes, which are prerequisites to metastatic spread. BMPs significantly promoted tumor migration by affecting the extracellular matrix (ECM) environment, such as integrin and matrix metalloproteinases (MMPs), which is a crucial factor in tumor migration. BMP-7 upregulates integrin avb3 expression, thereby inducing the migration activity in human chondrosarcoma cells [25]. BMPs accelerate pancreatic cancer cell invasiveness, which involves MMP-2 upregulation [26]. Furthermore, BMP-2-induced phosphorylation of SMAD2/3 promotes epithelial-mesenchymal transition (EMT) and induced cell invasion and migration in breast and pancreatic cancer cells [27].
However, some data revealed an opposite role of BMP signaling in tumors. BMP-10 was downregulated in gastric cancer samples [28]. BMP-6 expression was also absent in breast cancer tissues and might suppress breast cancer metastasis [29]. The inhibitory role of BMPs in tumorigenesis and dissemination has been widely reported in previous studies. For instance, BMP-2 and BMP-7 function as potent tumor suppressors in gastric carcinoma, renal cell carcinoma, lung and colorectal cancer, and osteosarcoma, in which BMPs suppress tumor growth by reducing the gene expression of tumorigenic factors and inducing the differentiation of cancer stem cells (CSCs) [30-33]. In HCC, BMP-2 induces apoptosis and plays an inhibitory role by virtue of their ability to increase the expression of the pro-apoptotic proteins caspase-3 and cleaved caspase-3 [34]. Correspondingly, BMP-4 and BMP-9 were also found to be potential anticancer agents in breast cancer [35]. BMP-4 causes a decline in granulocyte colony-stimulating factor (G-CSF) secretion, thereby reducing the number and activities of myeloid-derived suppressor cells (MDSCs) [36]. BMP-9 inhibits the bone metastasis of breast cancer cells by downregulating connective tissue growth factor expression [37]. BMP-9 also prevents the growth of prostate cancer cells by inducing tumor apoptosis, which is related to the upregulation of prostate apoptosis response-4 [38]. In other solid tumors, BMP-4 paraclinically inhibits tumor angiogenesis via the induction of thrombospondin-1 (TSP-1) [39]. At times, SMADs in the BMP signaling pathway could prevent tumor progression. The knockout of SMAD1 and SMAD5 in somatic cells of male and female gonads promotes metastatic granulosa cell tumorigenesis in mice, which implicated SMAD1 and SMAD5 as critical tumor suppressors [40]. In HCC, the inhibition of the BMP-4/SMAD1 signaling has been reported to suppress tumor migration, invasion, and EMT [41].
In conclusion, BMPs are described as both stimulator and inhibitor in different cancers; thus, we cannot simply define BMPs as oncogenes or anti-oncogenes. Collectively, the aforementioned evidence indicated that the effects of BMP signaling on tumor progress depends on the cell types and the tumor microenvironment. Therefore, in the current study, we reviewed recent studies focusing on BMP bilateral effects in tumorigenesis and the underlying signaling pathways regulating the paradoxical dilemmas.
BILATERAL ROLES OF BMP SIGNALING IN CELLULAR EVENTS OF CARCINOGENESIS
Considering that BMPs simultaneously displayed both tumor-promoting and tumor-inhibiting effects, we must emphasize on the disparity of biological behavior and molecular events along the BMP signaling to disclose the underlying mechanisms involved in such paradoxical biological behaviors. Therefore, we focus on the different aspects during tumorigenesis and metastasis, including EMT, CSCs, and angiogenesis.
BMP signaling and EMT
EMT is primarily defined as a phenotypic conversion that facilitates embryonic development and wound healing in physiological processes. Moreover, the acquisition of the EMT phenotype is related to fibrosis and tumor progression in certain pathological processes [42, 43]. Undergoing EMT, the epithelial cancer cells go through multiple changes, which mainly include the suppression of epithelial characteristics and the acquisition of a mesenchymal phenotype at the invasive front [44-46]. These hallmarks of EMT in cancers include the loss of E-cadherin expression, reduction of tight junction proteins [such as zona occludens-1 (ZO-1)] and cytokeratin, and increase of mesenchymal markers, such as vimentin, fibronectin, and N-cadherin [47, 48].
Multiple transcriptional factors and molecules take part in the tumor EMT procedure. Among them, the Snail superfamily (particularly Snail 1 and Snail 2), the basic helix-loop-helix family (such as Twist), and two Zeb factors (Zeb1 and Zeb2) regulate the expression of various epithelial and mesenchymal genes and thus affect the biological processes of cytoskeletal reorganization, extracellular matrix remodeling, and cell movements during EMT [49, 50]. For instance, Zeb1 and Zeb2 lead to E-cadherin repression, cause dramatic morphological transition of cells, and enhance migration and invasion during cancer progression [51]. Overexpression of Twist causes E-cadherin downregulation and vimentin upregulation to induce cellular morphological changes, expands the stem cell population, and promotes cell migration and invasion [52]. β-catenin and ZO-1 induced cytoplasmic/nuclear relocalization is a common process for EMT associated with tumor invasion [53].
EMT is associated with cancer invasion and metastasis in various tumor types, and the acquisition of mesenchymal features is related to the enhancement of tumor invasive capacity during cancer progression [54-56]. In breast cancer, the downregulation of the epithelial marker E-cadherin and upregulation of the mesenchymal markers, N-cadherin and vimentin, were positively correlated with the high aggressiveness and rapid spread of cancer [57]. Consistently, EMT plays a crucial role in the early steps of metastasis in HCC where the low E-cadherin expression and high vimentin expression were closely associated with high-grade tumor vascular invasion [58, 59]. The mesenchymal to epithelial transition (MET) is the reverse procedure of EMT, during which cell motility dramatically decreases [46]. MET attenuates the malignancy of cancer cells in squamous cell carcinoma [60]. Similarly, the proliferation, migration, and invasion of gastric carcinoma cells are suppressed during MET [61]. Therefore, MET plays an inhibitory role in tumor metastasis.
Observable EMT features of cancer cells could be induced by BMPs via SMAD and non-SMAD signaling pathways, which promote tumor invasion and metastasis in vitro and in vivo [62-64]. For instance, in breast epithelial and ovarian cancer cells, BMP-4 could induce EMT by decreasing E-cadherin, increasing N-cadherin, disrupting the polarity of ZO-1, and inducing transcription factors, Slug and Snail, to facilitate tumor progression [65, 66]. The reduction of E-cadherin and ZO-1 and an induction of vimentin and Snail expression have also been observed after the BMP-9 treatment in HCC [67]. Similarly, BMP-2 and BMP-7 were reported to promote the characteristic morphologic conversion of EMT in gastric and prostate cancer cells [62, 63].
Aside from the positive effects on EMT, literature has shown the opposite roles of the BMP signaling in inhibiting EMT-related metastasis of cancer. In melanoma cells, BMP-7 induced MET, a process opposite to EMT at the primary tumor site, thereby leading to metastasis inhibition [68]. BMP-7 also inhibited cholangiocarcinoma cell migration by suppressing TGF-β-mediated Twist expression, which is an important EMT transcription factor [69]. Accordingly, the effects of BMPs reversing EMT have been determined in breast cancer cells where BMP signaling induced E-cadherin expression and limited cancer cell metastatic potential by repressing EMT-activator Zeb1genes [70].
BMP signaling and CSCs
CSCs were described as immortal, possessing self-renewal capacity [71, 72], highly tumorigenic [73], and resistant to conventional chemotherapies [74]. They were initially reported in acute myeloid leukemia [75]. Recent reports verified the CSCs’ presence in breast [76], colon [77], prostate [78], skin [79], liver [80], stomach [81], lung [82], and brain cancers [83]. BMP signaling participated in CSC-related tumor maintenance and progression by influencing the CSCs’ functional properties, such as self-renewal, chemo-resistance, and tumor-initiating capacities [84]. For instance, BMP signaling hyper-activated and accelerated the amplification of tumor stem cell populations during the initiation and progression of breast cancer and oral squamous cell carcinoma [85, 86]. BMP-2 enhanced the motility and invasiveness of colon cancer cells by inducing CSC proliferation [87]. In ovarian cancer, human carcinoma-associated mesenchymal stem cells could increase the number of CSCs and promote the chemotherapy resistance of ovarian cancer by activating the BMP-4/Hedgehog signaling pathway [88]. BMP signaling could crosstalk with several known stem cell pathways in cancer, such as the Notch pathway and the Wnt pathway. In colorectal cancer, Notch signaling interacts with the BMP signaling in CSC regulation, in which Notch-1 expression increased along with the upregulation of multiple EMT/stemness-associated molecules CD44, Slug, and SMAD3 that led to a more aggressive phenotype [89].
By contrast, other reports demonstrate that BMP signaling diminishes the CSC pool. In colorectal cancer and nervous system tumors, BMP-4 promotes the differentiation, apoptosis, and chemo-sensitization of CSCs and restricts the self-renewal capacity of CSCs, which plays an inhibitory role on tumor progression [90-92]. Furthermore, other known stem cell pathways, such as the Wnt/β-catenin signaling, have been found to crosstalk with the BMP signaling in CSC regulation. For example, intestinal adenoma cells produce BMP-4 to counteract the Wnt/β-catenin signaling-related CSC-like traits, such as losing self-renewal capacity and initiating irreversible cellular differentiation [93]. In human renal cancer, BMP-2 suppresses the growth of aldehyde dehydrogenase (ALDH)+ cells, downregulates the expression of embryonic stem cell markers, and inhibits renal CSC migration [94]. BMP-2/-7 heterodimer, the most efficient stimulator of BMP signaling, diminishes the ALDHhi/CD44hi/CD24low CSC pool and effectively reduces the activation of TGF-β-driven SMAD signaling pathway, thereby inhibiting tumor invasion in breast cancer [95]. In prostate cancer, BMP-7 increases the expression of cell cycle inhibitor p21 and metastasis suppressor gene NDRG1 (N-myc downstream-regulated gene1) to induce CSC senescence [96]. In glioblastoma, BMP-7 reduces cell growth, inhibits sphere formation, and decreases self-renewal capacity via canonical SMAD1, 5, and 8 signaling [97]. Similarly, a BMP-7 variant (BMP-7v) represses the proliferation of stem-like cells and the expression of stem cell markers and enhances the expression of differentiation marker in glioblastoma [98]. The BMP-mediated repression on CSCs was also found in head and neck squamous cell carcinoma (HNSCC), in which inhibiting BMP signaling potentiated the long-term survival of HNSCC CSCs [99].
BMP signaling and angiogenesis
Tumor lymphatic and vascular angiogenesis has a key role in cancer development and progression by providing a faster and easier route for cancer cells to spread to other body parts. The tumor-associated angiogenesis is affected by multiple factors, among which BMPs are considered as important modulators. BMPs participate in angiogenesis not only by directly regulating the functions of vascular endothelial cells but also by indirectly influencing the expression of multiple angiogenic factors [100, 101]. For instance, BMPs upregulate the expression of vascular endothelial growth factor (VEGF) in both prostate cancer cells and osteoblasts, thereby induce brain metastases [102]. BMP-9 and BMP-10 increase gene expression along the Notch signaling pathway in vascular endothelial cells, thereby coordinating postnatal vascular remodeling [103].
Recently, increasing evidence indicated that BMP signaling promotes tumor angiogenesis. BMP co-receptor repulsive guidance molecule b (RGMb) was upregulated in vascular endothelial cells after hepatocyte growth factor (HGF) stimulation, which was combined with BMP-7 to induce angiogenesis in breast and prostate cancers [104]. In human dermal microvascular endothelial cells, BMP-2 induces Id1 expression and cooperates with VEGF signaling to promote angiogenesis in murine breast cancer xenograft models [105]. Another case for the pro-angiogenic role of the BMP signaling during cancer development came from colorectal cancer studies in which miR-885-3p inhibited the growth of HT-29 colon cancer cell xenografts by disrupting angiogenesis via targeting BMPRIA and blocking the activation of the BMP/SMAD/Id1 signaling [106].
However, most data implied the paradoxical role of BMP signaling in tumor angiogenesis. For example, BMP-4 was downregulated in high endothelial venules of lymph nodes draining metastatic tumors [107]. In multiple myeloma, BMP-6 induces cell apoptosis, inhibits angiogenesis, and causes growth suppression [108]. BMP-9 inhibits basic fibroblast growth factor (bFGF)-induced proliferation and migration of bovine aortic endothelial cells and represses VEGF-stimulated angiogenesis in glioblastoma [109]. BMP-9/ALK1-induced Crossveinless 2 and matrix Gla protein inhibits angiogenesis by limiting proliferation, tube formation, and expression of VEGF of endothelial cells [110]. In addition, BMP-9 suppresses lymphatic vessel formation and restrains sprouting angiogenesis and blood circulation [111, 112].
Molecular pathways regulated by the BMP signaling pathway
As previously described, BMP signaling plays a paradoxical role in cancer by affecting various features of carcinogenesis, which include EMT, CSCs, and angiogenesis. Increasing evidence has focused on multiple molecular events regulating BMP-induced biological processes in cancer and demonstrated that BMP signaling influenced the tumorigenesis and dissemination by modulating either the canonical SMAD or the non-canonical SMAD signaling pathways [113].
In skull-based chordomas, the BMP-4/SMAD signaling pathway upregulation was reported to be the dominant molecular mechanism of chordoma pathogenesis, which indicated poor clinical outcome [114]. Growth and Differentiation Factor (GDF)-9, a member of the BMP ligand family, promotes the adhesive and motile capacity of cancer cells by upregulating focal adhesion-associated proteins FAK and paxillin via the SMAD-dependent pathway, which implied pro-tumorigenic effects of the canonical BMP/SMAD signaling pathway in prostate cancer [115].
In addition to the canonical SMAD signaling pathways, the non-canonical SMAD signaling pathways participate in BMP-related cancer development and progression. BMPs activate the PI3K/AKT signaling pathways in gastric cancer, chondrosarcoma, and pancreatic cancer [116-118]. The PI3K/AKT pathway, a major cascade-promoting cell migration and invasion, could be activated by BMP-2 in gastric and pancreatic cancer cells, which can dramatically enhance the phosphorylation level of AKT protein. The blockage of the PI3K/AKT pathway using specific inhibitor LY294002 significantly reverts EMT and inhibits BMP-2-induced motility and invasiveness [62]. In addition, MAPK/ERK signaling pathway is another important regulator in cell migration and invasion, which was promoted by BMPs in which the inhibition of the RAF/MEK/ERK cascaded along the MAPK pathway reduced BMP7-induced motility and migration that subsequently led to cell apoptosis in prostate cancer cells [63]. Furthermore, BMP-2 enhances the phosphorylation of IκBα and the nuclear translocation of NF-κB in gastric and prostate cancer cells [119, 120]. Similarly, exogenous BMP-7 activates the c-Src/PI3K/AKT/IKK/NF-κB signaling pathway, thus resulting in the trans-activation of avβ3 integrin expression, which promotes tumor progression in human chondrosarcoma cells [25]. BMP-2 induces EMT and promotes colon cancer cell migration and invasion by increasing STAT3-mediated tumor stemness [87].
However, some studies pointed out that BMPs could exert an antitumor effect by blocking non-canonical signaling pathways in certain cancers. BMP-9 prevented the proliferation of HER2-positive breast cancer cells by inactivating ERK1/2 protein and repressing the PI3K/AKT signaling pathway [35]. In gastric cancer, PI3K/AKT pathway inhibition was involved in the tumor-suppressor effects of BMP-9 [121]. Similar to BMP-9, BMP-2 inhibits the growth and migration of HCC cells by attenuating the PI3K/AKT signaling pathway [34]. In addition, BMP-2 causes cell cycle arrest at the G1 phase and induces the apoptosis of myeloma cells by STAT3 inactivation [122]. Additionally, BMP-4 reduces the secretion of G-CSF and decreases the number and activities of MDSCs by counteracting NF-κB activity in tumor cells [36].
Collectively, BMPs play bidirectional and paradoxical effects on cancer development and invasion both at the molecular and cellular levels, in which the dysregulation of both the canonical and non-canonical SMAD pathways, including PI3K/AKT, MAPK/ERK, NF-κB, and STAT3 pathways, produces absolutely opposite influences on cell proliferation, apoptosis, migration, and invasion by affecting tumor EMT, generation and amplification of CSCs, and angiogenesis development.
THERAPEUTIC APPROACHES AGAINST BMP SIGNALING IN CANCER
Considering the extensive involvement of BMP signaling in carcinogenesis and dissemination, target therapy against BMPs and their receptors is a promising approach for cancer treatment. However, given that BMPs produce paradoxical effects in different types of cancer, the personalized treatment against BMPs should be discussed considering the characteristics of cancer cells, the disparity of components in tumor microenvironment, and the interaction among different signal pathways in each study model to achieve the best therapeutic efficiency.
Therapy against BMPs in cancer
BMPs are the hot topic of target therapy where some recombinant human BMPs, such as BMP-2 and BMP-7, have been used in orthopaedic and dental surgery [123-127]. However, clinical applications of BMPs in cancers are fewer because of their paradoxical effects on carcinogenesis and dissemination. Some studies have proposed potential applications for BMPs in cancer therapy.
In certain cancer types, BMPs that play pro-tumorigenic effects have been identified as novel prognostic biomarkers and potential therapy targets for cancer diagnosis and treatment. BMP-4, BMP-6, BMP-7, and BMP-9 are being proposed as biomarkers for HCC recurrence prediction and prognosis [14, 21, 128-131]. Some inhibitors that target BMPs have been proposed to be used in these cancers. Berberine, a natural alkaloid with important antitumor activities, has exerted inhibitory effects on the migratory and invasive abilities of highly metastatic prostate cancer cells by downregulating BMP-7 [132]. The active compounds tetramethylpyrazine, which was extracted from a Chinese medicinal plant, and heparan sulfate mimetic WSS25 respectively inhibited angiogenesis and tumor growth of lung and hepatocellular cancer by blocking BMP/SMAD/Id-1 signaling [133, 134]. Coleusin factor, an inhibitor targeting BMP-2, exerted its anticancer effects on osteosarcoma by inducing osteoblast differentiation [135]. Moreover, phosphoprotein Spp24 secreted by BMP binding protein diminished BMP-2-initated tumor growth and thus resulted in significant apoptosis of cancer cells, which would be developed into a new therapeutic agent for clinical applications [136].
By contrast, BMPs function as tumor suppressors in some cancer types. BMP-4, BMP-6, and BMP-9 have been reported to inhibit metastasis in breast cancer. BMP-2 plays a key inhibitory role in governing the proliferation and aggressive features of human cancer cells in HCC and colorectal carcinoma. Therefore, therapies based on BMP signaling activation may offer a novel treatment strategy for these cancer types [29, 33, 37]. Recombinant BMP ligand domains, which are being used as efficient agents for the repair of bony defects in preclinical and clinical studies of orthopaedic and maxillofacial surgery, are currently being tested for their therapeutic feasibilities in cancer. For instance, recombinant human (rh) BMP-7 exerted antineoplastic effects in HNSCC and rhBMP-2 was applied to treat osteosarcoma by increasing caspase-3 and Bax-mediated cell apoptosis in cancer [137, 138].
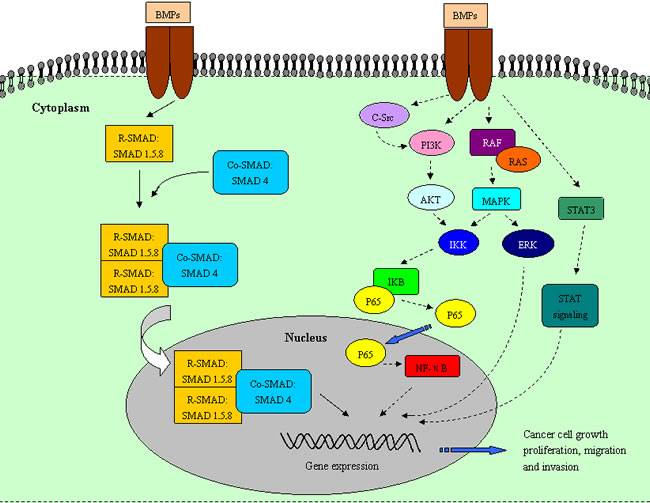
Figure 1: Relevant pathways regulate the paradoxical effects of BMP signaling in tumors. BMP signaling influences cancer cell progression through canonical SMAD and non-SMAD signaling pathways. The canonical SMAD-signaling pathway functions as a stimulator for cancer progression. However, non-SMAD signaling pathways, including PI3K/AKT, MAPK, NF-κB, and JAK/STAT signaling pathways, play bidirectional roles in various cancers. BMP signaling can cause the activation and inactivation of these non-SMAD signaling pathways to promote or suppress tumor progression.
Therapy against BMP receptors in cancer
BMP receptors have also been studied as target candidates for cancer therapy. Activin receptor-like kinases 1 (ALK1), a BMP receptor type I, has been confirmed as a target for anti-angiogenesis in cancer. Both ALK1 neutralizing antibodies and soluble ALK1 extracellular domain/Fc fusion protein (ALK1-Fc) attenuate the BMP signaling activity [139]. For example, Dalantercept, a fusion product composed of extracellular domain of ActRIIA and IgG-Fc fragment, was proposed as a novel anti-angiogenic therapy for treating a variety of cancers both in preclinical and clinical studies [140, 141].
Furthermore, the therapeutic effects of specific inhibitors against BMP receptor kinases have been investigated in various cancer types. Dorsomorphin and its analogue LDN-193189, which are the original inhibitors of ALK1, produce a block in cell migration and increase survival in human epithelial ovarian cancer [142]. Another small molecule inhibitor K02288, a 2-aminopyridine compound targeting ALK1, could inhibit BMP-9 signal transduction and thus repress tumor angiogenesis in diffused intrinsic pontine glioma and other tumors [143]. EW-7197, a novel ALK5 kinase inhibitor, represses the activation of the SMAD/TGF-β signaling pathway, thereby preventing lung metastasis in mouse 4T1 mammary cancer models and prolonging the survival of 4T1-bearing mice [144]. Recently, a selectively small molecule inhibitor DMH1, which specifically antagonizes the intracellular kinase domain of ALK2, significantly reduces cell proliferation, promotes cell death, and decreases cell invasion in NSCLC, thereby providing a promising development of target therapeutic strategy for clinical applications [145].
Table 1: The list for BMP family members
Known receptors |
||||
Ligand |
Gene locus |
Type I receptors |
Type II receptors |
Functions |
BMP-1 |
8p21.3 |
|
Extracellular matrix maintenance, chondrogenesis |
|
BMP-2 |
20p12 |
ALK-2, ALK-3, ALK-6 |
BMPR-II, ActR-IIA, ActR-IIB |
Osteoblast differentiation, bone and cartilage formation. Aretinoid mediator. Involved indorsoventral patterning, craniofacial and heart development |
BMP-3 |
14p21.21 |
ALK-4 |
ActR-IIA, ActR-IIB |
Bone formation |
BMP-4 |
14q22-q23 |
ALK-2, ALK-3, ALK-5, ALK-6 |
BMPR-II, ActR-IIA |
Fracture repair, Formation of teeth, limbs, lung, eye, and bone fromMesoderm, Dorsoventral patterning and craniofacial development |
BMP-5 |
6p12.1 |
ALK-3 |
BMPR-II, ActR-IIA, ActR-IIB |
Chondrogenesis |
BMP-6 |
6p24-p23 |
ALK-2, ALK-3, ALK-6 |
BMPR-II, ActR-IIA, ActR-IIB |
Involved in joint integrity, osteogenesis and chondrogenesis |
BMP-7 |
20q13 |
ALK-2, ALK-3, ALK-6 |
BMPR-II, AMHR-II |
Osteoblast differentiation, renal development/repair, eye and craniofacial development |
BMP-8a |
1p34.3 |
ALK-2, ALK-3, ALK-4, ALK-7 |
BMPR-II, AMHR-II |
Osteogenesis, chondrogenesis and craniofacial development |
BMP-8b |
1p35-p32 |
ALK-3, ALK-6 |
BMPR-II, ActR-IIA, ActR-IIB |
Osteogenesis, chondrogenesis and craniofacial development |
BMP-9 |
10q11.22 |
ALK-1, ALK-2 |
BMPR-II, ActR-IIA, ActR-IIB |
Chondrogenesis, nervous system, hepatogenesis and hepatic reticuloendothelial system development |
BMP-10 |
2p13.3 |
ALK-1, ALK-3, ALK-6 |
ActR-IIA, ActR-IIB |
Trabeculation of embryonic heart |
BMP-11 |
12q13.2 |
ALK-3, ALK-4, ALK-5, ALK-7 |
BMPR-II, ActR-IIA, ActR-IIB |
Mesodermal patterning and nervous system development |
BMP-12 |
2p24.1 |
ALK-3, ALK-6 |
BMPR-II, ActR-IIA |
Joint morphogenesis, Facilitates growth of ligament and tendon |
BMP-13 |
8q22.1 |
ALK-3, ALK-6 |
BMPR-II, ActR-IIA, ActR-IIB |
Joint morphogenesis, Facilitates growth of ligament and tendon |
BMP-14 |
20q11.2 |
ALK-3, ALK-6 |
BMPR-II, ActR-IIA |
Chondrogenesis, limb development, fracture healing and facilitates growth of tendon |
BMP-15 |
Xp11.2 |
ALK-6 |
Oocyte and follicular development |
This table shows the members of BMP family, gene locus, relative receptors and functions (if known).
CONCLUSIONS AND PERSPECTIVES
In this review, we summarized studies regarding the paradoxical roles of BMP signaling in tumor generation and progression. The effects of BMP signaling on cancer are closely related to the pathological type, the tumor origin, the activation status of downstream signaling pathways, and the various factors in tumor microenvironment. BMP signaling plays a paradoxical effects on cancer development and progression by serving as either tumor promoters or tumor suppressors, which dramatically affects tumor EMT, stemness, and angiogenesis. Targeting BMPs and BMPRs were successful in preventing tumor growth and invasion in some preclinical and clinical studies, which implies a promising future on BMPs target therapy in cancer treatment.
ACKNOWLEDGMENTS
This study was supported by the National Key Scientific and Technological Project of China (No. 2015BAI12B12), National Key Scientific and Technological Project of China (No. 2015BAI12B15), National Natural Science Foundation of China (81272360), National Natural Science Foundation of China (81472473), and Scientific and Technological Project of Tianjin, China (No.13ZCZCSY20300).
ConflictS of Interest
There is no conflict of interest.
References
1. Blanco Calvo M, Bolós Fernández V, Medina Villaamil V, Aparicio Gallego G, Díaz Prado S, Grande Pulido E. Biology of BMP signalling and cancer. Clinical and Translational Oncology. 2009; 11: 126-137.
2. Miyazono K, Kamiya Y, Morikawa M. Bone morphogenetic protein receptors and signal transduction. J Biochem. 2010; 147: 35-51.
3. Carreira AC, Lojudice FH, Halcsik E, Navarro RD, Sogayar MC, Granjeiro JM. Bone morphogenetic proteins: facts, challenges, and future perspectives. J Dent Res. 2014; 93: 335-345.
4. Lissenberg-Thunnissen SN, De Gorter DJJ, Sier CFM, Schipper IB. Use and efficacy of bone morphogenetic proteins in fracture healing. International Orthopaedics. 2011; 35: 1271-1280.
5. Sanchez-Duffhues G, Hiepen C, Knaus P, Ten Dijke P. Bone morphogenetic protein signaling in bone homeostasis. Bone. 2015; 80: 43-59.
6. Bragdon B, Moseychuk O, Saldanha S, King D, Julian J, Nohe A. Bone morphogenetic proteins: a critical review. Cell Signal. 2011; 23: 609-620.
7. Lin RL, Zhao LJ. Mechanistic basis and clinical relevance of the role of transforming growth factor-beta in cancer. Cancer Biol Med. 2015; 12: 385-393.
8. Thawani JP, Wang AC, Than KD, Lin CY, La Marca F, Park P. Bone morphogenetic proteins and cancer: review of the literature. Neurosurgery. 2010; 66: 233-246; discussion 246.
9. Ortuno MJ, Ruiz-Gaspa S, Rodriguez-Carballo E, Susperregui AR, Bartrons R, Rosa JL, Ventura F. p38 regulates expression of osteoblast-specific genes by phosphorylation of osterix. J Biol Chem. 2010; 285: 31985-31994.
10. Tang Y, Xie H, Chen J, Geng L, Chen H, Li X, Hou Y, Lu L, Shi S, Zeng X, Sun L. Activated NF-kappaB in bone marrow mesenchymal stem cells from systemic lupus erythematosus patients inhibits osteogenic differentiation through downregulating Smad signaling. Stem Cells Dev. 2013; 22: 668-678.
11. Holien T, Sundan A. The role of bone morphogenetic proteins in myeloma cell survival. Cytokine Growth Factor Rev. 2014; 25: 343-350.
12. Maegdefrau U, Bosserhoff AK. BMP activated Smad signaling strongly promotes migration and invasion of hepatocellular carcinoma cells. Exp Mol Pathol. 2012; 92: 74-81.
13. Fei Z-H, Yao C-Y, Yang X-L, Huang X-E, Ma S-L. Serum BMP-2 Up-regulation as an Indicator of Poor Survival in Advanced Non-small Cell Lung Cancer Patients. Asian Pacific Journal of Cancer Prevention. 2013; 14: 5293-5299.
14. Guo X, Xiong L, Zou L, Zhao J. Upregulation of bone morphogenetic protein 4 is associated with poor prognosis in patients with hepatocellular carcinoma. Pathol Oncol Res. 2012; 18: 635-640.
15. Chen J, Ye L, Xie F, Yang Y, Zhang L, Jiang WG. Expression of bone morphogenetic protein 7 in lung cancer and its biological impact on lung cancer cells. Anticancer Res. 2010; 30: 1113-1120.
16. Liu Y, Chen J, Yang Y, Zhang L, Jiang WG. Muolecular impact of bone morphogenetic protein 7, on lung cancer cells and its clinical significance. Int J Mol Med. 2012; 29: 1016-1024.
17. Megumi K, Ishigami S, Uchikado Y, Kita Y, Okumura H, Matsumoto M, Uenosono Y, Arigami T, Kijima Y, Kitazono M, Shinchi H, Ueno S, Natsugoe S. Clinicopathological significance of BMP7 expression in esophageal squamous cell carcinoma. Ann Surg Oncol. 2012; 19: 2066-2071.
18. Motoyama K, Tanaka F, Kosaka Y, Mimori K, Uetake H, Inoue H, Sugihara K, Mori M. Clinical significance of BMP7 in human colorectal cancer. Ann Surg Oncol. 2008; 15: 1530-1537.
19. Basic-Jukic N, Hudolin T, Radic-Antolic M, Coric M, Zadro R, Kastelan Z, Pasini J, Bandic-Pavlovic D, Kes P. Bone morphogenetic protein-7 expression is down-regulated in human clear cell renal carcinoma. Journal of Nephrology. 2011; 24: 91-97.
20. Herrera B, Van Dinther M, Ten Dijke P, Inman GJ. Autocrine bone morphogenetic protein-9 signals through activin receptor-like kinase-2/Smad1/Smad4 to promote ovarian cancer cell proliferation. Cancer Res. 2009; 69: 9254-9262.
21. Herrera B, Garcia-Alvaro M, Cruz S, Walsh P, Fernandez M, Roncero C, Fabregat I, Sanchez A, Inman GJ. BMP9 is a proliferative and survival factor for human hepatocellular carcinoma cells. PLoS One. 2013; 8: e69535.
22. Besson A, Dowdy SF, Roberts JM. CDK inhibitors: cell cycle regulators and beyond. Dev Cell. 2008; 14: 159-169.
23. Garcia-Alvaro M, Addante A, Roncero C, Fernandez M, Fabregat I, Sanchez A, Herrera B. BMP9-Induced Survival Effect in Liver Tumor Cells Requires p38MAPK Activation. Int J Mol Sci. 2015; 16: 20431-20448.
24. Peng J, Yoshioka Y, Mandai M, Matsumura N, Baba T, Yamaguchi K, Hamanishi J, Kharma B, Murakami R, Abiko K, Murphy SK, Konishi I. The BMP signaling pathway leads to enhanced proliferation in serous ovarian cancer-a potential therapeutic target. Mol Carcinog. 2015.
25. Chen JC, Yang ST, Lin CY, Hsu CJ, Tsai CH, Su JL, Tang CH. BMP-7 enhances cell migration and alphavbeta3 integrin expression via a c-Src-dependent pathway in human chondrosarcoma cells. PLoS One. 2014; 9: e112636.
26. Gordon KJ, Kirkbride KC, How T, Blobe GC. Bone morphogenetic proteins induce pancreatic cancer cell invasiveness through a Smad1-dependent mechanism that involves matrix metalloproteinase-2. Carcinogenesis. 2009; 30: 238-248.
27. Holtzhausen A, Golzio C, How T, Lee YH, Schiemann WP, Katsanis N, Blobe GC. Novel bone morphogenetic protein signaling through Smad2 and Smad3 to regulate cancer progression and development. FASEB J. 2014; 28: 1248-1267.
28. Lei H, Wang J, Lu P, Si X, Han K, Ruan T, Lu J. BMP10 inhibited the growth and migration of gastric cancer cells. Tumour Biol. 2015.
29. Hu F, Zhang Y, Li M, Zhao L, Chen J, Yang S, Zhang X. BMP-6 inhibits the metastasis of MDA-MB-231 breast cancer cells by regulating MMP-1 expression. Oncol Rep. 2016; 35: 1823-1830.
30. Shi Y, Massagué J. Mechanisms of TGF-β Signaling from Cell Membrane to the Nucleus. Cell. 2003; 113: 685-700.
31. Wang L, Park P, Zhang H, La Marca F, Claeson A, Valdivia J, Lin C-Y. BMP-2 inhibits the tumorigenicity of cancer stem cells in human osteosarcoma OS99-1 cell line. Cancer Biology & Therapy. 2014; 11: 457-463.
32. Yeh LC. In vitro and in vivo studies on the effects of bone morphogenetic protein-7 on human kidney and lung tumor cells. Int J Biomed Sci. 2010; 6: 176-181.
33. Zhang Y, Chen X, Qiao M, Zhang BQ, Wang N, Zhang Z, Liao Z, Zeng L, Deng Y, Deng F, Zhang J, Yin L, Liu W, et al. Bone morphogenetic protein 2 inhibits the proliferation and growth of human colorectal cancer cells. Oncol Rep. 2014; 32: 1013-1020.
34. Zheng Y, Wang X, Wang H, Yan W, Zhang Q, Chang X. Bone morphogenetic protein 2 inhibits hepatocellular carcinoma growth and migration through downregulation of the PI3K/AKT pathway. Tumour Biol. 2014; 35: 5189-5198.
35. Ren W, Liu Y, Wan S, Fei C, Wang W, Chen Y, Zhang Z, Wang T, Wang J, Zhou L, Weng Y, He T, Zhang Y. BMP9 inhibits proliferation and metastasis of HER2-positive SK-BR-3 breast cancer cells through ERK1/2 and PI3K/AKT pathways. PLoS One. 2014; 9: e96816.
36. Cao Y, Slaney CY, Bidwell BN, Parker BS, Johnstone CN, Rautela J, Eckhardt BL, Anderson RL. BMP4 inhibits breast cancer metastasis by blocking myeloid-derived suppressor cell activity. Cancer Res. 2014; 74: 5091-5102.
37. Ren W, Sun X, Wang K, Feng H, Liu Y, Fei C, Wan S, Wang W, Luo J, Shi Q, Tang M, Zuo G, Weng Y, et al. BMP9 inhibits the bone metastasis of breast cancer cells by downregulating CCN2 (connective tissue growth factor, CTGF) expression. Mol Biol Rep. 2014; 41: 1373-1383.
38. Ye L, Kynaston H, Jiang WG. Bone morphogenetic protein-9 induces apoptosis in prostate cancer cells, the role of prostate apoptosis response-4. Mol Cancer Res. 2008; 6: 1594-1606.
39. Tsuchida R, Osawa T, Wang F, Nishii R, Das B, Tsuchida S, Muramatsu M, Takahashi T, Inoue T, Wada Y, Minami T, Yuasa Y, Shibuya M. BMP4/Thrombospondin-1 loop paracrinically inhibits tumor angiogenesis and suppresses the growth of solid tumors. Oncogene. 2014; 33: 3803-3811.
40. Pangas SA, Li X, Umans L, Zwijsen A, Huylebroeck D, Gutierrez C, Wang D, Martin JF, Jamin SP, Behringer RR, Robertson EJ, Matzuk MM. Conditional deletion of Smad1 and Smad5 in somatic cells of male and female gonads leads to metastatic tumor development in mice. Mol Cell Biol. 2008; 28: 248-257.
41. Wang Y, Sun B, Zhao X, Zhao N, Sun R, Zhu D, Zhang Y, Li Y, Gu Q, Dong X, Wang M, An J. Twist1-related miR-26b-5p suppresses epithelial-mesenchymal transition, migration and invasion by targeting SMAD1 in hepatocellular carcinoma. Oncotarget. 2016; 7:24383-401. doi: 10.18632/oncotarget.8328.
42. Hollier BG, Evans K, Mani SA. The epithelial-to-mesenchymal transition and cancer stem cells: a coalition against cancer therapies. J Mammary Gland Biol Neoplasia. 2009; 14: 29-43.
43. Lee JM, Dedhar S, Kalluri R, Thompson EW. The epithelial-mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol. 2006; 172: 973-981.
44. Demirkan B. The Roles of Epithelial-to-Mesenchymal Transition (EMT) and Mesenchymal-to-Epithelial Transition (MET) in Breast Cancer Bone Metastasis: Potential Targets for Prevention and Treatment. Journal of Clinical Medicine. 2013; 2: 264-282.
45. Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. Journal of Clinical Investigation. 2009; 119: 1420-1428.
46. Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009; 139: 871-890.
47. Gonzalez DM, Medici D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal. 2014; 7: re8.
48. Micalizzi DS, Farabaugh SM, Ford HL. Epithelial-mesenchymal transition in cancer: parallels between normal development and tumor progression. J Mammary Gland Biol Neoplasia. 2010; 15: 117-134.
49. Moreno-Bueno G, Portillo F, Cano A. Transcriptional regulation of cell polarity in EMT and cancer. Oncogene. 2008; 27: 6958-6969.
50. Peinado H, Olmeda D, Cano A. Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? Nat Rev Cancer. 2007; 7: 415-428.
51. Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem. 2008; 283: 14910-14914.
52. Wang Y, Liu J, Ying X, Lin PC, Zhou BP. Twist-mediated Epithelial-mesenchymal Transition Promotes Breast Tumor Cell Invasion via Inhibition of Hippo Pathway. Sci Rep. 2016; 6: 24606.
53. Polette M, Mestdagt M, Bindels S, Nawrocki-Raby B, Hunziker W, Foidart JM, Birembaut P, Gilles C. Beta-catenin and ZO-1: shuttle molecules involved in tumor invasion-associated epithelial-mesenchymal transition processes. Cells Tissues Organs. 2007; 185: 61-65.
54. Gravdal K, Halvorsen OJ, Haukaas SA, Akslen LA. A switch from E-cadherin to N-cadherin expression indicates epithelial to mesenchymal transition and is of strong and independent importance for the progress of prostate cancer. Clin Cancer Res. 2007; 13: 7003-7011.
55. Sawada K, Mitra AK, Radjabi AR, Bhaskar V, Kistner EO, Tretiakova M, Jagadeeswaran S, Montag A, Becker A, Kenny HA, Peter ME, Ramakrishnan V, Yamada SD, et al. Loss of E-cadherin promotes ovarian cancer metastasis via alpha 5-integrin, which is a therapeutic target. Cancer Res. 2008; 68: 2329-2339.
56. Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002; 2: 442-454.
57. Sarrio D, Rodriguez-Pinilla SM, Hardisson D, Cano A, Moreno-Bueno G, Palacios J. Epithelial-mesenchymal transition in breast cancer relates to the basal-like phenotype. Cancer Res. 2008; 68: 989-997.
58. Giannelli G, Koudelkova P, Dituri F, Mikulits W. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol. 2016.
59. Mima K, Hayashi H, Kuroki H, Nakagawa S, Okabe H, Chikamoto A, Watanabe M, Beppu T, Baba H. Epithelial-mesenchymal transition expression profiles as a prognostic factor for disease-free survival in hepatocellular carcinoma: Clinical significance of transforming growth factor-beta signaling. Oncol Lett. 2013; 5: 149-154.
60. Takaishi M, Tarutani M, Takeda J, Sano S. Mesenchymal to Epithelial Transition Induced by Reprogramming Factors Attenuates the Malignancy of Cancer Cells. PLoS One. 2016; 11: e0156904.
61. Chang Q, Zhang L, He C, Zhang B, Zhang J, Liu B, Zeng N, Zhu Z. HOXB9 induction of mesenchymal-to-epithelial transition in gastric carcinoma is negatively regulated by its hexapeptide motif. Oncotarget. 2015; 6: 42838-42853. doi:10.18632/oncotarget.5814.
62. Liao A, Wang W, Sun D, Jiang Y, Tian S, Li J, Yang X, Shi R. Bone morphogenetic protein 2 mediates epithelial-mesenchymal transition via AKT and ERK signaling pathways in gastric cancer. Tumour Biol. 2015; 36: 2773-2778.
63. Lim M, Chuong CM, Roy-Burman P. PI3K, Erk signaling in BMP7-induced epithelial-mesenchymal transition (EMT) of PC-3 prostate cancer cells in 2- and 3-dimensional cultures. Horm Cancer. 2011; 2: 298-309.
64. Tan XY, Zheng FL, Zhou QG, Duan L, Li Y. [Effect of bone morphogenetic protein-7 on monocyte chemoattractant protein-1 induced epithelial-myofibroblast transition and TGF-beta1-Smad 3 signaling pathway of HKC cells]. Zhonghua Yi Xue Za Zhi. 2005; 85: 2607-2612.
65. Park KS, Dubon MJ, Gumbiner BM. N-cadherin mediates the migration of MCF-10A cells undergoing bone morphogenetic protein 4-mediated epithelial mesenchymal transition. Tumour Biol. 2015; 36: 3549-3556.
66. Richter A, Valdimarsdottir L, Hrafnkelsdottir HE, Runarsson JF, Omarsdottir AR, Ward-Van Oostwaard D, Mummery C, Valdimarsdottir G. BMP4 promotes EMT and mesodermal commitment in human embryonic stem cells via SLUG and MSX2. Stem Cells. 2014; 32: 636-648.
67. Li Q, Gu X, Weng H, Ghafoory S, Liu Y, Feng T, Dzieran J, Li L, Ilkavets I, Kruithof-De Julio M, Munker S, Marx A, Piiper A, et al. Bone morphogenetic protein-9 induces epithelial to mesenchymal transition in hepatocellular carcinoma cells. Cancer Sci. 2013; 104: 398-408.
68. Na YR, Seok SH, Kim DJ, Han JH, Kim TH, Jung H, Lee BH, Park JH. Bone morphogenetic protein 7 induces mesenchymal-to-epithelial transition in melanoma cells, leading to inhibition of metastasis. Cancer Sci. 2009; 100: 2218-2225.
69. Duangkumpha K, Techasen A, Loilome W, Namwat N, Thanan R, Khuntikeo N, Yongvanit P. BMP-7 blocks the effects of TGF-β-induced EMT in cholangiocarcinoma. Tumor Biology. 2014; 35: 9667-9676.
70. Mock K, Preca BT, Brummer T, Brabletz S, Stemmler MP, Brabletz T. The EMT-activator ZEB1 induces bone metastasis associated genes including BMP-inhibitors. Oncotarget. 2015; 6: 14399-14412. doi:10.18632/oncotarget.3882.
71. Dalerba P, Clarke MF. Cancer stem cells and tumor metastasis: first steps into uncharted territory. Cell Stem Cell. 2007; 1: 241-242.
72. Liu C, Liu L, Chen X, Cheng J, Zhang H, Shen J, Shan J, Xu Y, Yang Z, Lai M, Qian C. Sox9 regulates self-renewal and tumorigenicity by promoting symmetrical cell division of cancer stem cells in hepatocellular carcinoma. Hepatology. 2016; 64: 117-129.
73. Li B, Lu Y, Wang H, Han X, Mao J, Li J, Yu L, Wang B, Fan S, Yu X, Song B. miR-221/222 enhance the tumorigenicity of human breast cancer stem cells via modulation of PTEN/Akt pathway. Biomed Pharmacother. 2016; 79: 93-101.
74. Liu H, Wang YJ, Bian L, Fang ZH, Zhang QY, Cheng JX. CD44+/CD24+ cervical cancer cells resist radiotherapy and exhibit properties of cancer stem cells. Eur Rev Med Pharmacol Sci. 2016; 20: 1745-1754.
75. Minami Y. Overview: Cancer Stem Cell and Tumor Environment. Oncology. 2015; 89 Suppl 1: 22-24.
76. Bahena-Ocampo I, Espinosa M, Ceballos-Cancino G, Lizarraga F, Campos-Arroyo D, Schwarz A, Maldonado V, Melendez-Zajgla J. miR-10b expression in breast cancer stem cells supports self-renewal through negative PTEN regulation and sustained AKT activation. EMBO Rep. 2016; 17: 648-658.
77. Shaheen S, Ahmed M, Lorenzi F, Nateri AS. Spheroid-Formation (Colonosphere) Assay for in Vitro Assessment and Expansion of Stem Cells in Colon Cancer. Stem Cell Rev. 2016; 12: 492-499.
78. Jaworska D, Krol W, Szliszka E. Prostate Cancer Stem Cells: Research Advances. Int J Mol Sci. 2015; 16: 27433-27449.
79. Adhikary G, Grun D, Balasubramanian S, Kerr C, Huang JM, Eckert RL. Survival of skin cancer stem cells requires the Ezh2 polycomb group protein. Carcinogenesis. 2015; 36: 800-810.
80. Ochiya T. Novel therapeutic strategies targeting liver cancer stem cells. Chin Clin Oncol. 2016.
81. Brungs D, Aghmesheh M, Vine KL, Becker TM, Carolan MG, Ranson M. Gastric cancer stem cells: evidence, potential markers, and clinical implications. J Gastroenterol. 2016; 51: 313-326.
82. Pore M, Meijer C, De Bock GH, Boersma-Van Ek W, Terstappen LW, Groen HJ, Timens W, Kruyt FA, Hiltermann TJ. Cancer Stem Cells, Epithelial to Mesenchymal Markers, and Circulating Tumor Cells in Small Cell Lung Cancer. Clin Lung Cancer. 2016.
83. Corsaro A, Bajetto A, Thellung S, Begani G, Villa V, Nizzari M, Pattarozzi A, Solari A, Gatti M, Pagano A, Wurth R, Daga A, Barbieri F, et al. Cellular prion protein controls stem cell-like properties of human glioblastoma tumor-initiating cells. Oncotarget. 2016; doi:10.18632/oncotarget.9575.
84. Baccelli I, Trumpp A. The evolving concept of cancer and metastasis stem cells. The Journal of Cell Biology. 2012; 198: 281-293.
85. Balboni AL, Hutchinson JA, Decastro AJ, Cherukuri P, Liby K, Sporn MB, Schwartz GN, Wells WA, Sempere LF, Yu PB, Direnzo J. DeltaNp63alpha-mediated activation of bone morphogenetic protein signaling governs stem cell activity and plasticity in normal and malignant mammary epithelial cells. Cancer Res. 2013; 73: 1020-1030.
86. Qiao B, Johnson NW, Chen X, Li R, Tao Q, Gao J. Disclosure of a stem cell phenotype in an oral squamous cell carcinoma cell line induced by BMP-4 via an epithelial-mesenchymal transition. Oncol Rep. 2011; 26: 455-461.
87. Kim BR, Oh SC, Lee DH, Kim JL, Lee SY, Kang MH, Lee SI, Kang S, Joung SY, Min BW. BMP-2 induces motility and invasiveness by promoting colon cancer stemness through STAT3 activation. Tumour Biol. 2015; 36: 9475-9486.
88. Coffman LG, Choi YJ, Mclean K, Allen BL, Di Magliano MP, Buckanovich RJ. Human carcinoma-associated mesenchymal stem cells promote ovarian cancer chemotherapy resistance via a BMP4/HH signaling loop. Oncotarget. 2016; 7: 6916-6932. doi:10.18632/oncotarget.6870.
89. Fender AW, Nutter JM, Fitzgerald TL, Bertrand FE, Sigounas G. Notch-1 promotes stemness and epithelial to mesenchymal transition in colorectal cancer. J Cell Biochem. 2015; 116: 2517-2527.
90. Lombardo Y, Scopelliti A, Cammareri P, Todaro M, Iovino F, Ricci-Vitiani L, Gulotta G, Dieli F, De Maria R, Stassi G. Bone morphogenetic protein 4 induces differentiation of colorectal cancer stem cells and increases their response to chemotherapy in mice. Gastroenterology. 2011; 140: 297-309.
91. Piccirillo SG, Reynolds BA, Zanetti N, Lamorte G, Binda E, Broggi G, Brem H, Olivi A, Dimeco F, Vescovi AL. Bone morphogenetic proteins inhibit the tumorigenic potential of human brain tumour-initiating cells. Nature. 2006; 444: 761-765.
92. Whissell G, Montagni E, Martinelli P, Hernando-Momblona X, Sevillano M, Jung P, Cortina C, Calon A, Abuli A, Castells A, Castellvi-Bel S, Nacht AS, Sancho E, et al. The transcription factor GATA6 enables self-renewal of colon adenoma stem cells by repressing BMP gene expression. Nat Cell Biol. 2014; 16: 695-707.
93. Farrall AL, Riemer P, Leushacke M, Sreekumar A, Grimm C, Herrmann BG, Morkel M. Wnt and BMP signals control intestinal adenoma cell fates. Int J Cancer. 2012; 131: 2242-2252.
94. Wang L, Park P, La Marca F, Than KD, Lin CY. BMP-2 inhibits tumor-initiating ability in human renal cancer stem cells and induces bone formation. J Cancer Res Clin Oncol. 2015; 141: 1013-1024.
95. Buijs JT, Van Der Horst G, Van Den Hoogen C, Cheung H, De Rooij B, Kroon J, Petersen M, Van Overveld PG, Pelger RC, Van Der Pluijm G. The BMP2/7 heterodimer inhibits the human breast cancer stem cell subpopulation and bone metastases formation. Oncogene. 2012; 31: 2164-2174.
96. Kobayashi A, Okuda H, Xing F, Pandey PR, Watabe M, Hirota S, Pai SK, Liu W, Fukuda K, Chambers C, Wilber A, Watabe K. Bone morphogenetic protein 7 in dormancy and metastasis of prostate cancer stem-like cells in bone. J Exp Med. 2011; 208: 2641-2655.
97. Gonzalez-Gomez P, Crecente-Campo J, Zahonero C, De La Fuente M, Hernandez-Lain A, Mira H, Sanchez-Gomez P, Garcia-Fuentes M. Controlled release microspheres loaded with BMP7 suppress primary tumors from human glioblastoma. Oncotarget. 2015; 6: 10950-10963. doi:10.18632/oncotarget.3459.
98. Tate CM, Pallini R, Ricci-Vitiani L, Dowless M, Shiyanova T, D’alessandris GQ, Morgante L, Giannetti S, Larocca LM, Di Martino S, Rowlinson SW, De Maria R, Stancato L. A BMP7 variant inhibits the tumorigenic potential of glioblastoma stem-like cells. Cell Death Differ. 2012; 19: 1644-1654.
99. Khammanivong A, Gopalakrishnan R, Dickerson EB. SMURF1 silencing diminishes a CD44-high cancer stem cell-like population in head and neck squamous cell carcinoma. Mol Cancer. 2014; 13: 260.
100. David L, Feige JJ, Bailly S. Emerging role of bone morphogenetic proteins in angiogenesis. Cytokine Growth Factor Rev. 2009; 20: 203-212.
101. Ye L, Mason MD, Jiang WG. Bone morphogenetic protein and bone metastasis, implication and therapeutic potential. Front Biosci (Landmark Ed). 2011; 16: 865-897.
102. <Vascular Endothelial Growth Factor Contributes to the Prostate Cancer-Induced.pdf>.
103. Ricard N, Ciais D, Levet S, Subileau M, Mallet C, Zimmers TA, Lee SJ, Bidart M, Feige JJ, Bailly S. BMP9 and BMP10 are critical for postnatal retinal vascular remodeling. Blood. 2012; 119: 6162-6171.
104. Sanders AJ, Ye L, Li J, Mason MD, Jiang WG. Tumour angiogenesis and repulsive guidance molecule b: a role in HGF- and BMP-7-mediated angiogenesis. Int J Oncol. 2014; 45: 1304-1312.
105. Raida M, Clement JH, Leek RD, Ameri K, Bicknell R, Niederwieser D, Harris AL. Bone morphogenetic protein 2 (BMP-2) and induction of tumor angiogenesis. J Cancer Res Clin Oncol. 2005; 131: 741-750.
106. Xiao F, Qiu H, Cui H, Ni X, Li J, Liao W, Lu L, Ding K. MicroRNA-885-3p inhibits the growth of HT-29 colon cancer cell xenografts by disrupting angiogenesis via targeting BMPR1A and blocking BMP/Smad/Id1 signaling. Oncogene. 2015; 34: 1968-1978.
107. Farnsworth RH, Karnezis T, Shayan R, Matsumoto M, Nowell CJ, Achen MG, Stacker SA. A role for bone morphogenetic protein-4 in lymph node vascular remodeling and primary tumor growth. Cancer Res. 2011; 71: 6547-6557.
108. Seckinger A, Meissner T, Moreaux J, Goldschmidt H, Fuhler GM, Benner A, Hundemer M, Reme T, Shaughnessy JD, Jr., Barlogie B, Bertsch U, Hillengass J, Ho AD, et al. Bone morphogenic protein 6: a member of a novel class of prognostic factors expressed by normal and malignant plasma cells inhibiting proliferation and angiogenesis. Oncogene. 2009; 28: 3866-3879.
109. Scharpfenecker M, Van Dinther M, Liu Z, Van Bezooijen RL, Zhao Q, Pukac L, Lowik CW, Ten Dijke P. BMP-9 signals via ALK1 and inhibits bFGF-induced endothelial cell proliferation and VEGF-stimulated angiogenesis. J Cell Sci. 2007; 120: 964-972.
110. Yao Y, Jumabay M, Ly A, Radparvar M, Wang AH, Abdmaulen R, Bostrom KI. Crossveinless 2 regulates bone morphogenetic protein 9 in human and mouse vascular endothelium. Blood. 2012; 119: 5037-5047.
111. David L, Mallet C, Keramidas M, Lamande N, Gasc JM, Dupuis-Girod S, Plauchu H, Feige JJ, Bailly S. Bone morphogenetic protein-9 is a circulating vascular quiescence factor. Circ Res. 2008; 102: 914-922.
112. Yoshimatsu Y, Lee YG, Akatsu Y, Taguchi L, Suzuki HI, Cunha SI, Maruyama K, Suzuki Y, Yamazaki T, Katsura A, Oh SP, Zimmers TA, Lee SJ, et al. Bone morphogenetic protein-9 inhibits lymphatic vessel formation via activin receptor-like kinase 1 during development and cancer progression. Proceedings of the National Academy of Sciences of the United States of America. 2013; 110: 18940-18945.
113. Ehata S, Yokoyama Y, Takahashi K, Miyazono K. Bi-directional roles of bone morphogenetic proteins in cancer: another molecular Jekyll and Hyde? Pathol Int. 2013; 63: 287-296.
114. Feng Y, Zhang Q, Wang Z, Yan B, Wei W, Li P. Overexpression of the BMP4/SMAD signaling pathway in skull base chordomas is associated with poor prognosis. Int J Clin Exp Pathol. 2015; 8: 8268-8275.
115. Bokobza. Growth and differentiation factor-9 promotes adhesive and motile capacity of prostate cancer cells by up-regulating FAK and Paxillin via Smad dependent pathway. Oncology Reports. 2010; 24.
116. Chen X, Liao J, Lu Y, Duan X, Sun W. Activation of the PI3K/Akt pathway mediates bone morphogenetic protein 2-induced invasion of pancreatic cancer cells Panc-1. Pathol Oncol Res. 2011; 17: 257-261.
117. Fong YC, Li TM, Wu CM, Hsu SF, Kao ST, Chen RJ, Lin CC, Liu SC, Wu CL, Tang CH. BMP-2 increases migration of human chondrosarcoma cells via PI3K/Akt pathway. J Cell Physiol. 2008; 217: 846-855.
118. Kang MH, Kim JS, Seo JE, Oh SC, Yoo YA. BMP2 accelerates the motility and invasiveness of gastric cancer cells via activation of the phosphatidylinositol 3-kinase (PI3K)/Akt pathway. Exp Cell Res. 2010; 316: 24-37.
119. Graham TR, Odero-Marah VA, Chung LW, Agrawal KC, Davis R, Abdel-Mageed AB. PI3K/Akt-dependent transcriptional regulation and activation of BMP-2-Smad signaling by NF-kappaB in metastatic prostate cancer cells. Prostate. 2009; 69: 168-180.
120. Kang MH, Oh SC, Lee HJ, Kang HN, Kim JL, Kim JS, Yoo YA. Metastatic function of BMP-2 in gastric cancer cells: the role of PI3K/AKT, MAPK, the NF-kappaB pathway, and MMP-9 expression. Exp Cell Res. 2011; 317: 1746-1762.
121. Duan L, Ye L, Wu R, Wang H, Li X, Li H, Yuan S, Zha H, Sun H, Zhang Y, Chen X, Zhang Y, Zhou L. Inactivation of the phosphatidylinositol 3-kinase/Akt pathway is involved in BMP9-mediated tumor-suppressive effects in gastric cancer cells. J Cell Biochem. 2015; 116: 1080-1089.
122. Kawamura C, Kizaki M, Yamato K, Uchida H, Fukuchi Y, Hattori Y, Koseki T, Nishihara T, Ikeda Y. Bone morphogenetic protein-2 induces apoptosis in human myeloma cells with modulation of STAT3. Blood. 2000; 96: 2005-2011.
123. Axelrad TW, Einhorn TA. Bone morphogenetic proteins in orthopaedic surgery. Cytokine Growth Factor Rev. 2009; 20: 481-488.
124. Ayoub A, Roshan CP, Gillgrass T, Naudi K, Ray A. The clinical application of rhBMP-7 for the reconstruction of alveolar cleft. J Plast Reconstr Aesthet Surg. 2016; 69: 101-107.
125. Gomes-Ferreira PH, Okamoto R, Ferreira S, De Oliveira D, Momesso GA, Faverani LP. Scientific evidence on the use of recombinant human bone morphogenetic protein-2 (rhBMP-2) in oral and maxillofacial surgery. Oral Maxillofac Surg. 2016.
126. Herford AS, Tandon R, Stevens TW, Stoffella E, Cicciu M. Immediate distraction osteogenesis: the sandwich technique in combination with rhBMP-2 for anterior maxillary and mandibular defects. J Craniofac Surg. 2013; 24: 1383-1387.
127. Li Z, Lang G, Karfeld-Sulzer LS, Mader KT, Richards RG, Weber FE, Sammon C, Sacks H, Yayon A, Alini M, Grad S. Heterodimeric BMP-2/7 for nucleus pulposus regeneration-In vitro and ex vivo studies. J Orthop Res. 2016.
128. Li W, Cai HX, Ge XM, Li K, Xu WD, Shi WH. Prognostic significance of BMP7 as an oncogene in hepatocellular carcinoma. Tumour Biol. 2013; 34: 669-674.
129. Lu JW, Hsia Y, Yang WY, Lin YI, Li CC, Tsai TF, Chang KW, Shieh GS, Tsai SF, Wang HD, Yuh CH. Identification of the common regulators for hepatocellular carcinoma induced by hepatitis B virus X antigen in a mouse model. Carcinogenesis. 2012; 33: 209-219.
130. Maegdefrau U, Amann T, Winklmeier A, Braig S, Schubert T, Weiss TS, Schardt K, Warnecke C, Hellerbrand C, Bosserhoff AK. Bone morphogenetic protein 4 is induced in hepatocellular carcinoma by hypoxia and promotes tumour progression. J Pathol. 2009; 218: 520-529.
131. Maegdefrau U, Arndt S, Kivorski G, Hellerbrand C, Bosserhoff AK. Downregulation of hemojuvelin prevents inhibitory effects of bone morphogenetic proteins on iron metabolism in hepatocellular carcinoma. Lab Invest. 2011; 91: 1615-1623.
132. Liu CH, Tang WC, Sia P, Huang CC, Yang PM, Wu MH, Lai IL, Lee KH. Berberine inhibits the metastatic ability of prostate cancer cells by suppressing epithelial-to-mesenchymal transition (EMT)-associated genes with predictive and prognostic relevance. Int J Med Sci. 2015; 12: 63-71.
133. Jia Y, Wang Z, Zang A, Jiao S, Chen S, Fu Y. Tetramethylpyrazine inhibits tumor growth of lung cancer through disrupting angiogenesis via BMP/Smad/Id-1 signaling. Int J Oncol. 2016; 48: 2079-2086.
134. Qiu H, Yang B, Pei ZC, Zhang Z, Ding K. WSS25 inhibits growth of xenografted hepatocellular cancer cells in nude mice by disrupting angiogenesis via blocking bone morphogenetic protein (BMP)/Smad/Id1 signaling. J Biol Chem. 2010; 285: 32638-32646.
135. Geng S, Sun B, Lu R, Wang J. Coleusin factor, a novel anticancer diterpenoid, inhibits osteosarcoma growth by inducing bone morphogenetic protein-2-dependent differentiation. Mol Cancer Ther. 2014; 13: 1431-1441.
136. Li CS, Tian H, Zou M, Zhao KW, Li Y, Lao L, Brochmann EJ, Duarte ME, Daubs MD, Zhou YH, Murray SS, Wang JC. Secreted phosphoprotein 24 kD (Spp24) inhibits growth of human pancreatic cancer cells caused by BMP-2. Biochem Biophys Res Commun. 2015; 466: 167-172.
137. Grassi Rici R, Alcântara D, Fratini P, Wenceslau C, Ambrósio C, Miglino M, Maria D. Mesenchymal stem cells with rhBMP-2 inhibits the growth of canine osteosarcoma cells. BMC Veterinary Research. 2012; 8: 17.
138. Lappin DF, Abu-Serriah M, Hunter KD. Effects of recombinant human bone morphogenetic protein 7 (rhBMP-7) on the behaviour of oral squamous cell carcinoma: a preliminary in vitro study. British Journal of Oral and Maxillofacial Surgery. 2015; 53: 158-163.
139. Hawinkels LJaC, De Vinuesa AG, Ten Dijke P. Activin receptor-like kinase 1 as a target for anti-angiogenesis therapy (vol 22, pg 1371, 2013). Expert Opinion on Investigational Drugs. 2013; 22: 1695-1695.
140. Abdulkadyrov KM, Salogub GN, Khuazheva NK, Sherman ML, Laadem A, Barger R, Knight R, Srinivasan S, Terpos E. Sotatercept in patients with osteolytic lesions of multiple myeloma. Br J Haematol. 2014; 165: 814-823.
141. Gupta S, Gill D, Pal SK, Agarwal N. Activin receptor inhibitors—dalantercept. Curr Oncol Rep. 2015; 17: 14.
142. Ali JL, Lagasse BJ, Minuk AJ, Love AJ, Moraya AI, Lam L, Arthur G, Gibson SB, Morrison LC, Werbowetski-Ogilvie TE, Fu Y, Nachtigal MW. Differential cellular responses induced by dorsomorphin and LDN-193189 in chemotherapy-sensitive and chemotherapy-resistant human epithelial ovarian cancer cells. Int J Cancer. 2015; 136: E455-469.
143. Kerr G, Sheldon H, Chaikuad A, Alfano I, Von Delft F, Bullock AN, Harris AL. A small molecule targeting ALK1 prevents Notch cooperativity and inhibits functional angiogenesis. Angiogenesis. 2015; 18: 209-217.
144. Son JY, Park SY, Kim SJ, Lee SJ, Park SA, Kim MJ, Kim SW, Kim DK, Nam JS, Sheen YY. EW-7197, a novel ALK-5 kinase inhibitor, potently inhibits breast to lung metastasis. Mol Cancer Ther. 2014; 13: 1704-1716.
145. Hao J, Lee R, Chang A, Fan J, Labib C, Parsa C, Orlando R, Andresen B, Huang Y. DMH1, a small molecule inhibitor of BMP type i receptors, suppresses growth and invasion of lung cancer. PLoS One. 2014; 9: e90748.

