Introduction
Prostate cancer (PCa) is the most commonly diagnosed cancer among men [1, 2]. It is the second leading cause of male cancer-related deaths in US with an estimated 26,120 deaths in 2016 [3]. Prostate tumors are often indolent [4] but a significant fraction of tumors are very aggressive, often metastasizing to bone and other organs, causing significant morbidity and mortality [5]. PSA (prostate-specific antigen) screening has revolutionized the clinical management of prostate cancer [6]. However, owing to the drawbacks of PSA including low specificity and inability to differentiate between indolent and aggressive prostate tumors, prostate cancer clinical management is often challenging. Treatment options for advanced metastatic disease are limited and cause marginal increases in survival [7, 8] Also, PCa is associated with high rates of disease recurrence, with ~40% of localized PCa cases having relapse after initial therapy [9] and tumor progression to a hormone refractory/castration resistant stage [1, 10] that is essentially untreatable [1, 11]. A major challenge is the elucidation of underlying molecular pathways of PCa progression, recurrence and metastasis that will open new avenues for the design of effective diagnostic, prognostic and therapeutic strategies for better clinical management of the disease.
Epithelial cancer cells convert to motile, mesenchymal cells by undergoing an epithelial-mesenchymal transition (EMT) [12-14] that plays a critical role in cancer metastasis and tumor invasion. EMT is characterized by changes in gene expression profile, including decreased expression of epithelial genes, such as E-cadherin (also known as cadherin 1 [CDH1], and increased expression of mesenchymal genes, such as N-cadherin and vimentin. The EMT process is a complex genetic program that involves complex interactions of diverse inducers [e.g. Transforming Growth Factor β (TGFβ), Fibroblast Growth Factor (FGF), multiple signaling pathways (e.g. Wnt signaling) and is coordinated in a large part by several EMT-transcription factors (TF). EMT and its reverse process, mesenchymal-epithelial transition (MET), play a critical role during organogenesis and tissue differentiation during normal embryonic development [14-17]. These embryonic processes are reactivated by primary tumor cells to acquire motility, invasiveness and form secondary tumors at distant sites. Following EMT, cells detach from the primary tumor, circulate through blood or lymphatic routes, and colonize a distant site wherein the cells undergo MET to revert back to their immobile epithelial states, leading to development of secondary tumors after metastasis [16]. It has been proven that the loss of the epithelial marker, E-cadherin (E-cad/CDH1), is a causal factor in cancer progression [18]. EMT promotes stem cell properties endowing cancer cells with the capabilities of continued self-renewal [19].
Several EMT-transcription factors (EMT-TFs) repress the expression of E-cad and other epithelial markers and induce the expression of mesenchymal markers [16]. There are three major families of transcription factors that control EMT - Zeb (Zeb1/Zeb2); zinc finger Snail (Snail/Slug); and basic helix-loop-helix families (e.g. Twist1) [16]. The Snail and Zeb family of transcription factors are highly conserved zinc finger transcription repressors that bind to E-box like promoter elements in DNA, thereby influencing gene expression and tumorigenesis. Zeb1 and Zeb2 have been shown to regulate the expression of various EMT- and tumor related genes and thereby have been implicated in EMT, tumorigenesis and metastasis [20-24]. It is now well established that EMT-TFs play important roles in tumorigenesis. Snai1, Snai2, Zeb1, Zeb2 and Twist expression have been correlated with increased metastasis and poor prognosis in human tumors [25]. In addition, epigenetic mechanisms also play a role in EMT and epigenetic regulators like Bmi1 promote EMT [26]. Several signaling pathways regulate EMT and include key molecules such as TGFβ (transforming growth factor beta), fibroblast growth factor receptors (FGFRs) and platelet-derived growth factor (PDGF) [18]. TGFβ is a well-known inducer of EMT [15, 16, 18] that works by targeting the EMT-TFs through Smad-dependent and -independent transcriptional pathways [15]. Other soluble growth factors that have been shown to induce EMT include EGF, IGF1, and β2-microglobulin [15]. In addition, TGFβ cross-talks with signaling pathways such as Wnt, Notch, NF-κB and Receptor Tyrosine Kinases (RTKs) to induce EMT. Signaling via RTKs and their downstream effectors such as PI3K, MAPK is crucial to regulate EMT [18].
MicroRNAs recently emerged as important regulators of EMT and MET in various cancers [16, 18]. MicroRNAs (miRNAs) are small, noncoding RNAs that suppress gene expression post transcriptionally via sequence-specific interactions with the 3’- untranslated regions (UTRs) of cognate mRNA targets [27]. miRNAs suppresses their cognate target genes through the destabilization or cleavage of the mRNA and/or translational inhibition [14, 16]. A single miRNA can target many mRNAs, and conversely, many miRNAs can target a single mRNA [16]. MicroRNAs regulate EMT through their ability to post-transcriptionally regulate various components of the process such as EMT-TFs, epithelial and mesenchymal genes or by regulating key signaling pathways [14, 16, 18]. A role for miRNAs that affects EMT and progression and metastasis of human cancers is increasingly emerging [25, 28, 29]. In prostate cancer, the role of miRNAs in EMT is being explored and there are few reports. Here we review and summarize the role of miRNAs in prostate cancer EMT that play a role in prostate cancer progression, metastasis and tumor recurrence.
Prominent microRNAs regulating EMT in various cancers
miRNAs have been estimated to target over one-third of the human genome [14, 18]. In view of the enormous regulatory potential of miRNAs, deregulated miRNA expression has been revealed in various disease states, including cancer [30, 31]. miRNAs can be oncogenic or tumor-suppressive [32]. In addition, several miRNAs called ‘metastamirs’ influence tumor metastasis [14]. miRNAs affect EMT and MET and thereby regulate progression and metastasis of various human cancers [25, 28, 29]. Prominent examples are the miR-200 family and miR-205 that regulates EMT through direct targeting of Zeb1 and Zeb2 [25, 33, 34]. miR-9, MYC/MYCN-induced miRNA, directly targets E-cadherin and breast cancer metastasis [35]. miR-103/107 induces EMT by downregulating miR-200 levels, and promotes metastatic dissemination of otherwise non-aggressive breast cancer cells in vivo [36]. miR-30a inhibits EMT by directly targeting Snai1 in non-small cell lung cancer cells [37]. Treatment of mammary epithelial cells with TGFβ induced miR-155, whose knockdown suppressed TGFβ-induced EMT, migration, and invasion through direct targeting of RhoA [38]. miR-204 plays a critical role in maintaining epithelial barrier function and cell physiology by directly targeting TGFβR2 and Snail2 in retinal pigment epithelium [39]. The miR-221/222 miRNA cluster has been found to induce EMT in breast cancer cells [40]. miR-27 is upregulated in gastric cancer metastasis and enhances EMT through regulation of Zeb1, Zeb2 and Slug [41]. These studies support a crucial role of microRNAs in controlling EMT and metastasis.
EMT in prostate cancer
In prostate cancer, EMT has been implicated particularly in the context of metastatic disease [26, 42-44]. E-cadherin expression is commonly lost or reduced in PCa [26, 42]. Loss of E-cadherin and gain of mesenchymal marker, N-cadherin has been associated with multiple end points of progression and cancer related mortality [20-24]. Also, PB-Cre4Ptenfl/flTP53fl/fl mouse model of prostate cancer demonstrate the role of EMT in development of aggressiveness. These mice represent prostatic intraepithelial neoplasia (PIN) at 8 weeks, adenocarcinoma formation by 4 months and the progressive development of EMT and sarcomatoid carcinoma with mesenchymal stem cell characteristics, leading to the death of most animals at about 7 months of age [45, 46]. Also, sarcomatoid carcinomas with a metastatic propensity have been described in humans with prostatic disease [47] supporting the existence of EMT like states in prostate cancer. Benign prostatic hyperplasia (BPH) of the prostate exhibits manifestations of EMT state characterized by accumulation of mesenchymal-like cells derived from the prostatic epithelium and endothelium [48]. TGFβ acts as a tumor suppressor in non-tumorigenic prostate epithelial cells, but promotes EMT in tumorigenic prostate cells [49]. In prostate cancer, TGFβ levels have been reported to increase with tumor burden and PCa metastases, also increasing in circulation [50, 51] and are negatively correlated with prognosis. ZEB1 is also a direct repressor of E cadherin in prostate cancer cell lines, and its level of expression correlates with Gleason score [44,45]. The Wnt signaling pathway has also been implicated in EMT-like states in prostate cancer [26].
MicroRNAs and EMT in prostate cancer
Studies point to an important role of microRNAs in regulating EMT and MET in prostate cancer, that in turn regulates tumor progression and metastasis (Table 1). MicroRNAs control EMT and MET through efficiently targeting single or multiple components that impinge upon these regulatory pathways via multiple mechanisms- either by (i) directly repressing single or multiple EMT-TFs or regulating cytoskeletal components (epithelial and mesenchymal genes) (Figure 1) or (ii) by regulating key signaling pathways involved in EMT [14, 16, 18]. (Figure 2) Thus, miRNAs impact these pathways in various regulatory modes and their regulation is often controlled by double-negative feedback loops, leading to an efficient and flexible control of EMT. Dysregulation of miRNAs leads to induction of EMT states, leading to tumor progression, recurrence and metastasis. Tumor suppressive miRNAs act by inhibiting EMT and oncogenic microRNAs often act as EMT promoters. The following sections highlight the significant studies on suppressive/oncogenic microRNAs in PCa that act by regulating EMT. An understanding of the role of miRNA-mediated EMT program may help elucidate the mechanistic model of PCa progression and metastasis and identify novel targets for therapeutic intervention.
Table 1: MicroRNAs regulating EMT in prostate cancer
MicroRNA |
Effects on EMT |
Target(s) |
References |
miRNAs primarily targeting EMT transcription factors or cytoskeletal components |
|||
miR-1 |
Inhibit |
Slug, Twist1 |
|
miR-124 |
Inhibit |
Slug |
[70] |
miR-145 |
Inhibit |
ZEB2, HEF1 |
|
miR-200 |
Inhibit |
Zeb1, Zeb2, Slug |
[52] |
miR-203 |
Inhibit |
Zeb2, Bmi1, Survivin CKAP2, LASP1, WASF1, ASAP1 mRNAs |
|
miR-205 |
Inhibit |
Zeb2, Protein Kinase Cε |
[60] |
miR-186 |
Inhibit |
Twist1 |
[71] |
miR-23a-3p |
Promote |
E-cadherin |
[105] |
miRNAs targeting signaling pathways implicated in EMT |
|||
let 7 family |
Inhibit |
HMGA2 |
[82] |
miR-21 |
Promote |
BTG2, TGFβ |
|
miR-23b |
Inhibit |
Src kinase and Akt |
[91] |
miR-29b |
Inhibit |
MMP2 |
[95] |
miR-30 |
Inhibit |
ERG |
[92] |
miR-32 |
Promote |
BTG2 |
[108] |
miR-34a |
Inhibit |
LEF1 |
[89] |
miR-34b |
Inhibit |
Akt |
[90] |
miR-100 |
Inhibit |
AGO2 |
[93] |
miR-154 |
Inhibit |
HMGA2 |
[85] |
miR-154* |
Promote |
SMAD7 |
[112] |
miR-195 |
Inhibit |
FGF2, HMGA1, RPS6KB |
|
miR-223 |
Inhibit |
ITGA3, ITGB1 |
[100] |
miR-301a |
Promote |
p63 |
[104] |
miR-331-3p |
Promote |
NRP2, NACC1 |
[114] |
miR-379 |
Promote |
FOXF2 |
[112] |
miR-573 |
Inhibit |
FGFR1 |
[102] |
miRNAs affecting EMT and PCa stem cells |
|||
miR-200 |
Inhibit |
Notch1 |
[80] |
Let-7 |
Inhibit |
EZH2 |
|
miR-205 |
Inhibit |
Zeb2, Protein Kinase Cε |
[60] |
miR-145 |
Inhibit |
Zeb2 |
[67] |
miR-21 |
Promote |
[124] |
Table summarizes the miRNAs that have been reported to inhibit/promote EMT in prostate cancer. Target genes of the respective miRNAs are indicated in column 3.
MicroRNAs that inhibit EMT
Several tumor suppressive miRNAs act to inhibit PCa EMT by directly repressing one or more EMT-TFs or inhibiting signaling pathway components influencing EMT. These miRNAs negatively regulate tumor progression and metastasis as EMT is considered as the initiating and most critical step in invasion and metastasis [13].
EMT-inhibiting miRNAs primarily regulating EMT-TFs
MicroRNAs such as miR-200 family, miR-205 and miR-203 promote the epithelial state and inhibit EMT by primarily repressing EMT-TFs among other targets (Figure 1 and Table 1). Loss of these miRNAs leads to induction of EMT states as highlighted below:
miR-200 family and miR-1
The miR-200 family includes miR-200a, miR-200b, miR-200c, miR-141, and miR-429, all of which are significantly downregulated in PCa. Each of these miRNAs has been reported to suppress PCa metastasis via EMT inhibition. miR-200 inhibits Platelet-Derived Growth Factor-D (PDGF-D) induced EMT in PC3 cells by targeting and repressing Zeb1, Zeb2, and Slug expression (Figure 1) [52]. Similarly, miR-200b significantly inhibited tumor growth and cell proliferation in PC-3 cells due to inhibition of EMT [53]. Slabakova et al showed that miR-200 can counteract TGFβ1-induced EMT in benign prostate hyperplasia (BPH) cells [54]. Liu et al identified that miR-200 along with miR-1 act as negative regulators of EMT in Pten and TP53 null murine models [55]. miR-1 and miR-200 repress EMT by targeting Slug, which plays an important regulatory role in mesenchymal differentiation. Further, Slug was found as a direct repressor of miR-1 and miR-200 transcription suggesting that Slug and miR-1/miR-200 form a self-reinforcing regulatory loop that leads to EMT amplification (Figure 1) [55]. Also, transcription factor OVOL has been reported to induce miR-200 expression [56] and has been predicted to modulate PCa EMT by interacting with the miR-200/ZEB axis [57]. miR-1 targets Twist1 in PCa [55]. EGFR has been shown to act as a transcriptional repressor of miR-1 leading to oncogenic activation of Twist1, contributing to accelerated PCa bone metastasis [58].
miR-205
miR-205 shows significantly lower expression levels in prostate cancer cells in comparison to normal prostate cells [59]. miR-205 overexpression in a prostate cancer cell line led to alterations consistent with MET and a reduction in cell invasion primarily through downregulation of protein kinase Cε [60]. In addition, miR-205 was shown to directly target Zeb2, N-chimaerin, ErbB3, E2F1 and E2F5 in prostate cancer cells [60]. Another study showed that miR-205 is essential for the inhibitory effects of p63 on PCa EMT markers, such as Zeb1, and metastasis (Figure 1) [61]. miR-205 expression in PCa was shown to be regulated by p63 (both TAp63 and ΔNp63 isoforms). ΔNp63 or miR-205 significantly inhibited the incidence of lung metastasis in vivo in a mouse tail vein model. Further, in human PCa specimens, partial or complete loss of the ∆Np63/miR-205 axis was found to be correlated with EMT expression patterns and a predictor of metastatic behavior and poor prognosis [61].
miR-203
miR-203 is a tumor suppressor miRNA that is downregulated in PCa [62, 63]. We provided first evidence that miR-203 represses EMT by directly targeting Zeb2 (Figure 1) and other EMT regulators, including Bmi1, that in turn regulate invasion and metastasis of prostate cancer [26, 64]. In addition, miR-203 regulates a cohort of pro-metastatic genes including survivin and bone-specific effectors including Runx2, a master regulator of bone metastasis [62, 63]. Upon overexpression in prostate cancer cell lines, miR-203 induces MET and inhibits cell invasion [62, 63]. Viticchie et al. demonstrated that miR-203 additionally targets a series of metastasis-promoting genes, including CKAP2, LASP1, WASF1 and ASAP1 mRNAs [63].
miR-143/miR-145
miR-143/miR-145 form a cluster and are both reported to be downregulated in metastatic PCa [65]. Peng et al identified that downregulation of miR-143 and miR-145 is associated with induction of EMT and PCa bone metastasis [66]. miR-145 is a strong EMT repressor that directly targets Zeb2 [67]. Further, Zeb2 directly targets miR-145, repressing its transcription, thereby forming a double-negative feedback loop that regulates EMT, stemness and PCa bone metastasis (Figure 1). Zeb2 expression was positively correlated with bone metastasis in PCa patients and negatively correlated with miR-145 expression in primary PCa samples [67]. Guo et al showed that a cytoplasmic scaffolding protein, human enhancer of filamentation 1 (HEF1; also known as NEDD9/Cas-L), is also a direct miR-145 target. miR-145 and HEF1 expression levels were negatively correlated in primary prostate cancer and bone metastatic specimens [68]. Further, miR-145 expression is controlled by DNA hypermethylation and tumor suppressor p53 in PCa [65, 69]. WT-p53 upregulates the expression of miR-145, thereby suppressing metastasis and EMT in PC3 prostate cancer cells and these effects could be reversed by anti-miR-145. Thus, miR-145 plays at least a partial role in the EMT inhibitory effects of WT-p53 [69].
miR-124
Qin et al found that transforming growth factor-α (TGF-α) promotes EMT through downregulation of miR-124 in prostate cancer cell line. Overexpression of miR-124 on the TGF-α treated PCa cells inhibited the EMT inducing effects of TGF-α and reduced cellular invasion. Further, the authors demonstrated that miR-124 directly targets Slug (Figure 1) [70].
miR-186
miR-186 has been reported to be downregulated in human PCa tissues, particularly in metastatic samples and metastatic prostate cancer cell line [71]. Low miR-186 expression was found to be correlated with poor patient survival. miR-186 was found to inhibit EMT and PCa metastasis by directly targeting and downregulating Twist1 [71].
Let7 family
The let-7 family of miRNAs include let-7a, let-7b, let-7c, let-7d, let-7e, let-7f, let-7g, and let-7i [72], all of which are significantly downregulated in PCa, compared to benign tissues and act as tumor suppressors [32]. Lin28 is a highly conserved RNA-binding protein and an embryonic stem cell factor that blocks the processing of let-7 precursors into mature miRNAs [72-74]. Lin28 is frequently overexpressed in primary human tumors [75] and promotes oncogenesis and proliferation of cancer cells, by repression of the let-7 family [76]. Let-7 expression has been reported to be downregulated in localized PCa tissues relative to benign peripheral zone tissue [77, 78]. Loss of let-7 expression has been linked to the development of poorly differentiated and aggressive cancers [79]. The let-7 miRNAs are tumor suppressive and have been shown to target oncogenes such as Ras, Myc and genes involved in cell cycle regulation. These miRNAs thereby target oncogenes at various metastatic steps including EMT [17]. Kong et al. reported an important link between let-7 and miR-200 with EMT states in prostate cancer [52, 80]. One of the let-7 family’s target genes in EMT is high mobility group protein A (HMGA), a chromatin associated nonhistone protein [81] that has been implicated in EMT. HMGA2 is highly expressed in PCa, and upon let-7 inhibition, HMGA2 was de-repressed [82]. Thualt et al have demonstrated that HMGA2 cooperates with Smad to regulate Snail1 expression and to induce EMT in human hepatocarcinoma cells [83]. Therefore, these results suggest that let-7 could inhibit EMT in PCa by targeting HMGA2 [84] (Figure 1).
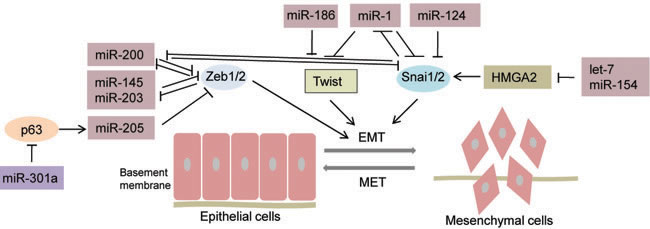
Figure 1: MicroRNAs regulating EMT-TFs in prostate cancer. Schematic illustration depicting microRNAs that have been reported to regulate major families of EMT- transcription factors (Zeb, Snail and Twist) in prostate cancer. MicroRNAs mediate PCa EMT by directly repressing single or multiple EMT-TFs. ![]() EMT-inhibiting miRNAs,
EMT-inhibiting miRNAs, ![]() EMT-promoting miRNAs, solid bars denote transcriptional repression, solid arrows denote transcriptional activation. As indicated, microRNAs inhibit EMT by directly or indirectly repressing Zeb1/2, Twist, and/or Snai1/2. miR-200, miR-145, miR-203, miR-205 repress ZEB1/2; miR-1 and miR-186 inhibit TWIST; miR-1 and miR-124 repress SNAI2; let-7 and miR-154 inhibit EMT by targeting HMGA2. Conversely, miR-301a represses p63 which downregulates miR-205 causing an upregulation of Zeb1/2. In addition, some of the depicted miRNAs participate in double-negative feedback loops with their respective targets as represented for miR-200, miR-145 and miR-1.
EMT-promoting miRNAs, solid bars denote transcriptional repression, solid arrows denote transcriptional activation. As indicated, microRNAs inhibit EMT by directly or indirectly repressing Zeb1/2, Twist, and/or Snai1/2. miR-200, miR-145, miR-203, miR-205 repress ZEB1/2; miR-1 and miR-186 inhibit TWIST; miR-1 and miR-124 repress SNAI2; let-7 and miR-154 inhibit EMT by targeting HMGA2. Conversely, miR-301a represses p63 which downregulates miR-205 causing an upregulation of Zeb1/2. In addition, some of the depicted miRNAs participate in double-negative feedback loops with their respective targets as represented for miR-200, miR-145 and miR-1.
miR-154
Similarly, miR-154 inhibits EMT in prostate cancer cells by directly targeting HMGA2 [85]. HMGA2 is mostly upregulated in PCa tissue samples as compared to normal. Overexpression of miR-154 inhibited EMT and reduced migratory and invasive capabilities of PCa cells in vitro concomitant with HMGA2 repression (Figure 1) [85].
MicroRNAs regulating key signaling pathways implicated in EMT
MicroRNAs also impinge upon key signaling pathways involved in EMT including TGFβ, Wnt, PI3K/Akt and Receptor Tyrosine Kinases (RTKs) to regulate EMT (Figure 2 and Table 1) [18] as summarized below:
miR-34a/b
The miR-34 family of tumor suppressive miRNAs include miR-34a/b/c that are encoded by two different genes. miR-34a precursor is transcribed from chromosome 1 while miR-34b and miR-34c precursors are co-transcribed from a region on chromosome 11 [86]. miR-34 is an important component of the p53 network and is directly transactivated by p53 [86, 87]. miR-34 has been reported to target key genes that function in cell cycle, apoptosis, senescence, cell migration, and invasion [86]. In mice, prostate epithelium-specific inactivation of miR-34 and p53 leads to early invasive adenocarcinomas and high-grade prostatic intraepithelial neoplasia with expansion of the prostate stem cell compartment [88]. miR-34a has been reported to be significantly downregulated in human PCa specimens in contrast to benign prostate tissues [89]. Further, it has been demonstrated that miR-34a regulates PCa EMT by directly repressing LEF1 (lymphoid enhancer-binding factor-1), a transcription factor in the Wnt signaling pathway that plays a role in cell proliferation and invasion (Figure 2) [89]. We reported that miR-34b is tumor suppressive in PCa and is silenced through CpG hypermethylation [90]. Reconstitution of miR-34b inhibited cell proliferation/invasion and EMT by directly targeting Akt and its downstream proliferative genes [90].
miR-23b
miR-23b is a methylation-silenced tumor suppressor in prostate cancer that works by directly targeting proto oncogene Src kinase and Akt (Figure 2) [91]. Overexpression of miR-23b in prostate cancer cells led to a decrease in mesenchymal markers and an increase in epithelial markers suggesting that miR-23b inhibits EMT and promotes MET [91].
miR-30
In PCa, Ets-related gene (ERG) is a frequently overexpressed oncogene that is activated by fusion events between promoter sequences of the TMPRSS2 and coding sequences of ERG. Kao et al showed that ERG is a direct target of miR-30 [92]. miR-30 is underexpressed in prostate cancer specimens and its overexpression in prostate cancer cells suppressed EMT and inhibited cell migration and invasion [92]. Further, Src inhibitors upregulated miR-30 levels suggesting that TMPRSS2-ERG gene fusions are regulated by Src via miR-30 [92].
miR-100
miR-100 negatively regulated EMT in PC3 and DU145 cells and its expression was negatively correlated with bone metastasis of prostate cancer patients [93]. Downregulation of miR-100 promoted prostate cancer metastasis by upregulating Argonaute 2 (AGO2), a core effector protein of the miRNA-induced silencing complex which facilitates EMT [93].
miR-29b
miR-29b is downregulated in PCa cells as compared to normal prostate epithelial cells, and its expression was further lowered in metastatic prostate cancer cells [94]. Upon miR-29b transfection in PC3 prostate cancer cells, E-cadherin was upregulated while N-cadherin, Twist, and Snail expression were downregulated [94]. Owing to its EMT-inhibiting effects, ectopic miR-29b expression suppressed PCa metastasis in a mouse model [94]. Matrix metallopeptidase-2 (MMP-2) has been identified as a miR-29b target in prostate cancer cells [95]. Further, miR-29b has been shown to enhance the chemotherapeutic effects of cisplatin through its direct targeting of DNMT3B (DNA (Cytosine-5-)-Methyltransferase 3 Beta) and Akt3 (v-Akt Murine Thymoma Viral Oncogene Homolog 3) [96].
miR-195
miR-195 is downregulated in prostate cancer tissues and low miR-195 is significantly associated with high Gleason score, biochemical recurrence and metastasis [97, 98]. Prostate cancer cases with low miR-195 expression had a shorter relapse-free survival (RFS) time [98]. miR-195 overexpression in DU-145 and PC3 PCa cell lines led to EMT inhibition and reduced invasion and migration by directly targeting fibroblast growth factor 2 (FGF2) [98]. Also, miR-195 has been reported to directly repress HMGA1 in prostate cancer [99]. Another study identified Ribosomal protein S6 kinase, RPS6KB, an important component of mTOR signaling pathway as a novel direct miR-195 target (Figure 2) [97]. The latter study suggests that miR-195 loss upregulates RPS6KB1, leading to a decreased expression of E-cadherin and increased expression of MMP-9 and VEGF proteins that culminates in effects on tumor invasion, angiogenesis, EMT, and survival [97].
miR-223
miR-223 is significantly downregulated in PCa tissues and restoration of miR-223 led to inhibited cell proliferation/invasion [100]. miR-223 was shown to directly target integrin alpha 3 (ITGA3) and integrin β1 (ITGβ1) [100]. These integrins are involved in focal adhesion and extracellular matrix (ECM) receptor interactions that play a role in EMT induction in prostate cancer cells [64, 101].
miR-573
miR-573 is downregulated in metastatic PCa in comparison to primary PCa [102]. miR-573 overexpression inhibited TGF-β1 induced EMT in vitro and lung metastasis in vivo by directly targeting the fibroblast growth factor receptor 1 (FGFR1) gene [102]. FGFR1 plays a critical role in prostate tumorigenesis, and miR-573 directly suppresses FGFR1 expression in PCa cells [102, 103]. Further, miR-573 was found to be directly regulated by transcription factor GATA3 suggesting the involvement of GATA3, miR-573 and FGFR1 in controlling PCa EMT [102].
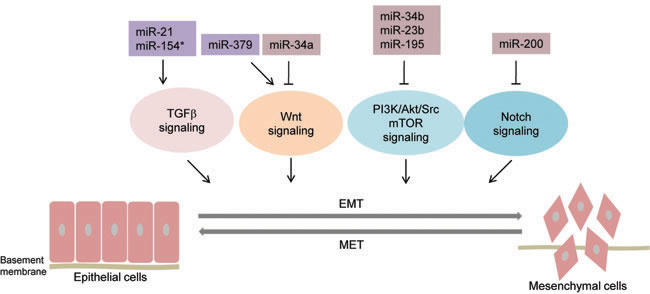
Figure 2: MicroRNAs regulating major signaling pathways involved in EMT in prostate cancer. Schematic illustration depicting microRNAs that have been reported to regulate major signaling pathways involved in EMT in prostate cancer. These pathways include TGFβ signaling, Wnt signaling, PI3K/Akt/Src mTOR signaling, and Notch signaling. The key microRNAs regulating these pathways are indicated. ![]() EMT-inhibiting miRNAs,
EMT-inhibiting miRNAs, ![]() EMT-promoting miRNAs, solid bars denote transcriptional repression, solid arrows denote transcriptional activation. miR-21, miR-154* impinge upon TGFβ pathway; miR-379 and miR-34a promote and inhibit Wnt signaling, respectively. miR-34b, miR-23b, miR-195 inhibit PI3K/Akt/Src/mTOR signaling and miR-200 inhibits Notch signaling.
EMT-promoting miRNAs, solid bars denote transcriptional repression, solid arrows denote transcriptional activation. miR-21, miR-154* impinge upon TGFβ pathway; miR-379 and miR-34a promote and inhibit Wnt signaling, respectively. miR-34b, miR-23b, miR-195 inhibit PI3K/Akt/Src/mTOR signaling and miR-200 inhibits Notch signaling.
MicroRNA that promote EMT
In prostate cancer, various oncogenic microRNAs often repress epithelial characteristics or promote EMT as summarized below.
EMT-promoting miRNAs regulating EMT-TFs or cytoskeletal components
Oncogenic miRNAs can promote EMT by augmenting the expression of EMT-TFs indirectly as exemplified by miR-301a or by directly targeting cytoskeletal components (epithelial genes).
miR-301a
In a recent study, Nam et al showed that miR-301a induces EMT in prostate cancer cells by directly targeting and inhibiting the tumor suppressor p63. The loss of p63 results in the downregulation of tumor suppressive miR-205, causing upregulation of ZEB1 and ZEB2 (Fig.1), which in turn, suppresses E-cadherin [104].
miR-23a-3p
Wen et al. demonstrated that miR-23a-3p regulates TGF-β-induced EMT in androgen independent prostate cancer (AIPC) cell lines PC3 and Du145 [105]. Treatment of PCa cell lines with TGF-β led to miR-23a-3p upregulation concomitant with E-cadherin downregulation. Further, miR-23a-3p was found to directly repress E-cadherin expression by binding to its 3′UTR. Also, a bioactive coumarin derivative, osthole, suppressed EMT in vitro and metastasis in vivo by inhibiting miR-23a-3p expression and Snail signaling in AIPC cells [105].
EMT-promoting miRNAs regulating signaling pathways implicated in EMT
In addition, oncogenic miRNAs may repress tumor suppressor genes or act by repressing inhibitors of signaling pathways involved in EMT as summarized below (Table 1).
miR-21
miR-21 is commonly upregulated in various cancers, including prostate cancer [106]. miR-21 has been reported to be Ras-induced, leading to tumor aggressiveness [107]. Overexpression of miR-21 in prostate cells promotes EMT leading to the transformation of normal prostate cells into prostate cancer cells [108] primarily from repression of B-cell translocation gene 2 (BTG2). B-cell translocation gene 2 (BTG2) is a basal protein that has been implicated in prostate cancer transformation and progression [108] and also possesses anti-proliferative activity [109]. A recent study shows that miR-21 overexpression concomitant with loss of the miR-15/16 cluster leads to aberrant TGFβ signaling, EMT, increased aggressiveness and PCa metastasis [110].
miR-32
Jalava et al showed that miR-32 also targets BTG2 and that their expression levels are inversely correlated in castration-resistant prostate cancer (CRPC) [111]. miR-32 is an androgen-regulated miRNA that is highly expressed in CPRC samples leading to reduced expression of BTG2 [111].
miR-154* and miR-379
miR-154* and miR-379 are members of the delta-like 1 homolog-deiodinase, iodothyroxine (DLK1-DIO3) cluster - a cluster that plays an important role in EMT and embryonic development. Gururajan et al demonstrated that both miR-154* and miR-379 have elevated expression in bone metastatic PCa cell lines and tissues and miR-379 expression correlated with progression-free survival of PCa [112]. Overexpression of these miRNAs promoted EMT in PCa [112] while their inhibition resulted in EMT reversal and decreased invasive capacities of PCa cells [112]. Analyses of the potential target pathways showed that these miRNAs regulate oncogenic pathways that activate EMT such as E2F signaling, Ras pathway, WNT and TGFβ pathways. [112]. miR-154* targets SMAD7, an inhibitor of the TGFβ pathway [113] while miR-379 was predicted to inhibit forkhead box F2 (FOXF2), a WNT pathway inhibitor (Figure 2) [112].
miR-331-3p
miR-331-3p expression promotes EMT as its overexpression in the PC3 cell line led to upregulation of mesenchymal markers such as vimentin and N-cadherin, and downregulation of epithelial markers such as E-cadherin and desmoplakin [114]. Further, the authors demonstrated that miR-331-3p- mediated EMT was due to direct targeting of Neuropilin 2 (NRP2) and Nucleus Accumbens-Associated Protein 1(NACC1) expression.
miR-331 expression is elevated in PCa cases with high Gleason score and was shown to be controlled by Syndecan-1 [114].
miR-129-3p
miR-129-3p has been reported to be upregulated in metastatic PCa cells [115]. miR-129-3p was found to directly repress Centriolar Coiled-Coil Protein 110kDa (CP110), a centrosomal protein that is downregulated in metastatic PCa. Lack of CP110 inhibition led to excessive centrosome amplification accompanied by increased E-cadherin expression, deregulated F-actin cytoskeleton and decreased invasion and metastasis [115]. Though a direct correlation was observed between CP110 expression, E-cadherin expression and PCa metastases, no significant correlation could be established between CP110 expression and the expression of EMT-related genes in clinical prostate cancer samples [115].
EMT promoting miRNAs with undefined mechanisms miR-409
miR-409-3p/-5p expression is elevated in bone metastatic PCa cell lines and in PCa tissues with higher Gleason scores [116]. High miR-409-3p expression was found to be correlated with progression-free survival of PCa patients. Further, orthotopic delivery of miR-409-3p/-5p in the murine prostate gland induced EMT and tumors expressing EMT and stemness markers. Inhibition of miR-409-5p in the bone metastatic ARCaPM prostate cancer cell line led to decreased bone metastasis and increased survival in an experimental mouse model [116].
MicroRNAs, EMT and prostate cancer stem cells
Prostate cancer recurrence and progression has been associated with the existence of prostate cancer stem cells (CSC) or tumor-initiating cells (TIC). These cells are refractory to current therapies [117] and are characterized by various cell surface markers [118-122] such as CD44, CD133, integrin α2β1 and stem-cell signature genes such as Sox2, Nanog and Oct4. EMT endows cancer cells with stem cell properties [19, 123]. It has been reported that EMT is mechanistically linked to stem cell signatures in prostate cancer [80]. Several miRNAs implicated in EMT have been reported to influence prostate cancer stem cells indirectly by regulating EMT-TFs or by influencing the signaling pathways that impinge upon both EMT and stemness such as TGFβ, MAP kinase and Wnt signaling pathways [19, 123]. Decreased expression of miR-200 and/or let-7 family has been reported in PCa cells that have undergone EMT and display stem cell- like features [80]. miR-200 reexpression led to reversal of EMT and inhibition of stem cell properties such as decreased prostasphere-forming ability and clonogenicity concomitant with reduced expression of Notch1 and Lin28B [80]. Similarly, miR-205 reverses EMT progression and inhibits stem-cell properties of PCa cells through repression of its targets Zeb2 and protein kinase Cε [60]. miR-145 inhibits EMT and stem cell properties in PCa [67] via its regulation of Zeb2. miR-21 is involved in EMT, maintenance of CSCs and therapeutic resistance [124]. Downregulation of miR-21 led to repression of EpCAM, CD44 and VEGF [124]. Let-7 directly targets Enhancer of Zeste homolog 2 (EZH2) and thereby regulates PCa CSCs. Loss of let-7 during PCa progression leads to increased expression of EZH2, contributing to PCa aggressiveness [125]. In addition to influencing CSCs indirectly through EMT, miRNAs can also regulate CSCs via their ability to directly regulate CSC markers [123]. As an example, miR-34a directly represses CD44 thereby inhibiting PCa stem cells and metastasis [126-128]. We showed that miR-708 is a key negative regulator of the CD44(+) subpopulation of prostate cancer cells by directly regulating CD44 [129]. Also, miR-708 directly targets Akt2, a component of the PI3K/Akt pathway, with roles in tumor progression and CSCs [129].
Conclusions and future perspectives
In conclusion, the role of miRNAs in PCa EMT is emerging and warrants further investigation. Studies till date suggest that microRNAs mediate efficient and reversible post-transcriptional control of prostate cancer EMT via multiple mechanisms including direct targeting of EMT-TFs, affecting signaling pathway components controlling EMT or by directly modulating cytoskeletal components. Oncogenic microRNAs often act as EMT promoters by repressing epithelial characteristics and tumor suppressive miRNAs act by inhibiting mesenchymal progression. Loss of EMT-inhibiting miRNAs and/or gain of EMT promoting miRNAs lead to induction of PCa EMT, leading to tumor progression, metastasis and recurrence. Restoring expression of tumor suppressive miRNAs and inhibiting oncogenic miRNAs represent therapeutic opportunities to prevent PCa metastasis and recurrence.
ACKNOWLEDGMENTS
We thank Dr. Roger Erickson for his support and assistance with preparation of the manuscript.
CONFLICTS OF INTEREST
The author declares that there are no conflicts of interest.
GRANT SUPPORT
The authors are supported by the National Cancer Institute at the National Institutes of Health (Grant Number RO1CA177984 to SS, RO1CA138642 to RD).
References
1. Barlow LJ, Shen MM. SnapShot: Prostate cancer. Cancer cell. 2013; 24: 400 e401.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA: a cancer journal for clinicians. 2016; 66: 7-30.
3. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA: a cancer journal for clinicians. 2015; 65: 5-29.
4. Jaiswal S, Sarmad R, Arora S, Dasaraju R, Sarmad K. Prostate Cancer for the Internist. N Am J Med Sci. 2015; 7: 429-435.
5. Loberg RD, Logothetis CJ, Keller ET, Pienta KJ. Pathogenesis and treatment of prostate cancer bone metastases: targeting the lethal phenotype. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2005; 23: 8232-8241.
6. Cary KC, Cooperberg MR. Biomarkers in prostate cancer surveillance and screening: past, present, and future. Therapeutic advances in urology. 2013; 5: 318-329.
7. Fong MK, Hare R, Jarkowski A. A new era for castrate resistant prostate cancer: a treatment review and update. Journal of oncology pharmacy practice : official publication of the International Society of Oncology Pharmacy Practitioners. 2012; 18: 343-354.
8. Rodrigues DN, Butler LM, Estelles DL, de Bono JS. Molecular Pathology and Prostate Cancer Therapeutics: From Biology to Bedside. The Journal of pathology. 2013.
9. Grubb RL, 3rd, Kibel AS. Prostate cancer: screening, diagnosis and management in 2007. Mo Med. 2007; 104: 408-413; quiz 413-404.
10. Kasper S, Cookson MS. Mechanisms leading to the development of hormone-resistant prostate cancer. Urol Clin North Am. 2006; 33: 201-210, vii.
11. Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, Oudard S, Theodore C, James ND, Turesson I, Rosenthal MA, Eisenberger MA. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004; 351: 1502-1512.
12. Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. The Journal of clinical investigation. 2009; 119: 1420-1428.
13. Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nature reviews Cancer. 2002; 2: 442-454.
14. Zhang J, Ma L. MicroRNA control of epithelial-mesenchymal transition and metastasis. Cancer Metastasis Rev. 2012; 31: 653-662.
15. Kyprianou N. ASK-ing EMT not to spread cancer. Proceedings of the National Academy of Sciences of the United States of America. 2010; 107: 2731-2732.
16. Lamouille S, Subramanyam D, Blelloch R, Derynck R. Regulation of epithelial-mesenchymal and mesenchymal-epithelial transitions by microRNAs. Curr Opin Cell Biol. 2013; 25: 200-207.
17. Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A, Labourier E, Reinert KL, Brown D, Slack FJ. RAS is regulated by the let-7 microRNA family. Cell. 2005; 120: 635-647.
18. Zaravinos A. The Regulatory Role of MicroRNAs in EMT and Cancer. J Oncol. 2015; 2015: 865816.
19. Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, Campbell LL, Polyak K, Brisken C, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008; 133: 704-715.
20. Imamichi Y, Konig A, Gress T, Menke A. Collagen type I-induced Smad-interacting protein 1 expression downregulates E-cadherin in pancreatic cancer. Oncogene. 2007; 26: 2381-2385.
21. Maeda G, Chiba T, Okazaki M, Satoh T, Taya Y, Aoba T, Kato K, Kawashiri S, Imai K. Expression of SIP1 in oral squamous cell carcinomas: implications for E-cadherin expression and tumor progression. International journal of oncology. 2005; 27: 1535-1541.
22. Spaderna S, Schmalhofer O, Hlubek F, Berx G, Eger A, Merkel S, Jung A, Kirchner T, Brabletz T. A transient, EMT-linked loss of basement membranes indicates metastasis and poor survival in colorectal cancer. Gastroenterology. 2006; 131: 830-840.
23. Spaderna S, Schmalhofer O, Wahlbuhl M, Dimmler A, Bauer K, Sultan A, Hlubek F, Jung A, Strand D, Eger A, Kirchner T, Behrens J, Brabletz T. The transcriptional repressor ZEB1 promotes metastasis and loss of cell polarity in cancer. Cancer research. 2008; 68: 537-544.
24. Spoelstra NS, Manning NG, Higashi Y, Darling D, Singh M, Shroyer KR, Broaddus RR, Horwitz KB, Richer JK. The transcription factor ZEB1 is aberrantly expressed in aggressive uterine cancers. Cancer research. 2006; 66: 3893-3902.
25. Bracken CP, Gregory PA, Khew-Goodall Y, Goodall GJ. The role of microRNAs in metastasis and epithelial-mesenchymal transition. Cellular and molecular life sciences : CMLS. 2009; 66: 1682-1699.
26. Nauseef JT, Henry MD. Epithelial-to-mesenchymal transition in prostate cancer: paradigm or puzzle? Nature reviews Urology. 2011; 8: 428-439.
27. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009; 136: 215-233.
28. Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004; 118: 277-279.
29. Zhang J, Ma L. MicroRNA control of epithelial-mesenchymal transition and metastasis. Cancer metastasis reviews. 2012.
30. Calin GA. MicroRNAs and cancer: what we know and what we still have to learn. Genome medicine. 2009; 1: 78.
31. Garzon R, Calin GA, Croce CM. MicroRNAs in Cancer. Annual review of medicine. 2009; 60: 167-179.
32. Nadiminty N, Tummala R, Lou W, Zhu Y, Shi XB, Zou JX, Chen H, Zhang J, Chen X, Luo J, deVere White RW, Kung HJ, Evans CP, et al. MicroRNA let-7c is downregulated in prostate cancer and suppresses prostate cancer growth. PloS one. 2012; 7: e32832.
33. Gregory PA, Bracken CP, Bert AG, Goodall GJ. MicroRNAs as regulators of epithelial-mesenchymal transition. Cell Cycle. 2008; 7: 3112-3118.
34. Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y, Goodall GJ. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008; 10: 593-601.
35. Ma L, Young J, Prabhala H, Pan E, Mestdagh P, Muth D, Teruya-Feldstein J, Reinhardt F, Onder TT, Valastyan S, Westermann F, Speleman F, Vandesompele J, et al. miR-9, a MYC/MYCN-activated microRNA, regulates E-cadherin and cancer metastasis. Nature cell biology. 2010; 12: 247-256.
36. Martello G, Rosato A, Ferrari F, Manfrin A, Cordenonsi M, Dupont S, Enzo E, Guzzardo V, Rondina M, Spruce T, Parenti AR, Daidone MG, Bicciato S, et al. A MicroRNA targeting dicer for metastasis control. Cell. 2010; 141: 1195-1207.
37. Kumarswamy R, Mudduluru G, Ceppi P, Muppala S, Kozlowski M, Niklinski J, Papotti M, Allgayer H. MicroRNA-30a inhibits epithelial-to-mesenchymal transition by targeting Snai1 and is downregulated in non-small cell lung cancer. International journal of cancer Journal international du cancer. 2012; 130: 2044-2053.
38. Kong W, Yang H, He L, Zhao JJ, Coppola D, Dalton WS, Cheng JQ. MicroRNA-155 is regulated by the transforming growth factor beta/Smad pathway and contributes to epithelial cell plasticity by targeting RhoA. Molecular and cellular biology. 2008; 28: 6773-6784.
39. Wang FE, Zhang C, Maminishkis A, Dong L, Zhi C, Li R, Zhao J, Majerciak V, Gaur AB, Chen S, Miller SS. MicroRNA-204/211 alters epithelial physiology. FASEB journal : official publication of the Federation of American Societies for Experimental Biology. 2010; 24: 1552-1571.
40. Stinson S, Lackner MR, Adai AT, Yu N, Kim HJ, O’Brien C, Spoerke J, Jhunjhunwala S, Boyd Z, Januario T, Newman RJ, Yue P, Bourgon R, et al. miR-221/222 targeting of trichorhinophalangeal 1 (TRPS1) promotes epithelial-to-mesenchymal transition in breast cancer. Science signaling. 2011; 4: pt5.
41. Zhang Z, Liu S, Shi R, Zhao G. miR-27 promotes human gastric cancer cell metastasis by inducing epithelial-to-mesenchymal transition. Cancer genetics. 2011; 204: 486-491.
42. Cheng L, Nagabhushan M, Pretlow TP, Amini SB, Pretlow TG. Expression of E-cadherin in primary and metastatic prostate cancer. The American journal of pathology. 1996; 148: 1375-1380.
43. Sethi S, Macoska J, Chen W, Sarkar FH. Molecular signature of epithelial-mesenchymal transition (EMT) in human prostate cancer bone metastasis. American journal of translational research. 2010; 3: 90-99.
44. Wallerand H, Robert G, Pasticier G, Ravaud A, Ballanger P, Reiter RE, Ferriere JM. The epithelial-mesenchymal transition-inducing factor TWIST is an attractive target in advanced and/or metastatic bladder and prostate cancers. Urologic oncology. 2010; 28: 473-479.
45. Liu YN, Yin JJ, Abou-Kheir W, Hynes PG, Casey OM, Fang L, Yi M, Stephens RM, Seng V, Sheppard-Tillman H, Martin P, Kelly K. MiR-1 and miR-200 inhibit EMT via Slug-dependent and tumorigenesis via Slug-independent mechanisms. Oncogene. 2012.
46. Martin P, Liu YN, Pierce R, Abou-Kheir W, Casey O, Seng V, Camacho D, Simpson RM, Kelly K. Prostate epithelial Pten/TP53 loss leads to transformation of multipotential progenitors and epithelial to mesenchymal transition. The American journal of pathology. 2011; 179: 422-435.
47. Hansel DE, Epstein JI. Sarcomatoid carcinoma of the prostate: a study of 42 cases. The American journal of surgical pathology. 2006; 30: 1316-1321.
48. Alonso-Magdalena P, Brossner C, Reiner A, Cheng G, Sugiyama N, Warner M, Gustafsson JA. A role for epithelial-mesenchymal transition in the etiology of benign prostatic hyperplasia. Proceedings of the National Academy of Sciences of the United States of America. 2009; 106: 2859-2863.
49. Ao M, Williams K, Bhowmick NA, Hayward SW. Transforming growth factor-beta promotes invasion in tumorigenic but not in nontumorigenic human prostatic epithelial cells. Cancer Res. 2006; 66: 8007-8016.
50. Adler HL, McCurdy MA, Kattan MW, Timme TL, Scardino PT, Thompson TC. Elevated levels of circulating interleukin-6 and transforming growth factor-beta1 in patients with metastatic prostatic carcinoma. The Journal of urology. 1999; 161: 182-187.
51. Wikstrom P, Stattin P, Franck-Lissbrant I, Damber JE, Bergh A. Transforming growth factor beta1 is associated with angiogenesis, metastasis, and poor clinical outcome in prostate cancer. The Prostate. 1998; 37: 19-29.
52. Kong D, Li Y, Wang Z, Banerjee S, Ahmad A, Kim HR, Sarkar FH. miR-200 regulates PDGF-D-mediated epithelial-mesenchymal transition, adhesion, and invasion of prostate cancer cells. Stem Cells. 2009; 27: 1712-1721.
53. Williams LV, Veliceasa D, Vinokour E, Volpert OV. miR-200b inhibits prostate cancer EMT, growth and metastasis. PloS one. 2013; 8: e83991.
54. Slabakova E, Pernicova Z, Slavickova E, Starsichova A, Kozubik A, Soucek K. TGF-beta1-induced EMT of non-transformed prostate hyperplasia cells is characterized by early induction of SNAI2/Slug. The Prostate. 2011; 71: 1332-1343.
55. Liu YN, Yin JJ, Abou-Kheir W, Hynes PG, Casey OM, Fang L, Yi M, Stephens RM, Seng V, Sheppard-Tillman H, Martin P, Kelly K. MiR-1 and miR-200 inhibit EMT via Slug-dependent and tumorigenesis via Slug-independent mechanisms. Oncogene. 2013; 32: 296-306.
56. Roca H, Hernandez J, Weidner S, McEachin RC, Fuller D, Sud S, Schumann T, Wilkinson JE, Zaslavsky A, Li H, Maher CA, Daignault-Newton S, Healy PN, et al. Transcription factors OVOL1 and OVOL2 induce the mesenchymal to epithelial transition in human cancer. PloS one. 2013; 8: e76773.
57. Jia D, Jolly MK, Boareto M, Parsana P, Mooney SM, Pienta KJ, Levine H, Ben-Jacob E. OVOL guides the epithelial-hybrid-mesenchymal transition. Oncotarget. 2015; 6: 15436-15448. doi: 10.18632/oncotarget.3623.
58. Chang YS, Chen WY, Yin JJ, Sheppard-Tillman H, Huang J, Liu YN. EGF Receptor Promotes Prostate Cancer Bone Metastasis by Downregulating miR-1 and Activating TWIST1. Cancer Res. 2015; 75: 3077-3086.
59. Majid S, Dar AA, Saini S, Yamamura S, Hirata H, Tanaka Y, Deng G, Dahiya R. MicroRNA-205-directed transcriptional activation of tumor suppressor genes in prostate cancer. Cancer. 2010; 116: 5637-5649.
60. Gandellini P, Folini M, Longoni N, Pennati M, Binda M, Colecchia M, Salvioni R, Supino R, Moretti R, Limonta P, Valdagni R, Daidone MG, Zaffaroni N. miR-205 Exerts tumor-suppressive functions in human prostate through down-regulation of protein kinase Cepsilon. Cancer Res. 2009; 69: 2287-2295.
61. Tucci P, Agostini M, Grespi F, Markert EK, Terrinoni A, Vousden KH, Muller PA, Dotsch V, Kehrloesser S, Sayan BS, Giaccone G, Lowe SW, Takahashi N, et al. Loss of p63 and its microRNA-205 target results in enhanced cell migration and metastasis in prostate cancer. Proceedings of the National Academy of Sciences of the United States of America. 2012; 109: 15312-15317.
62. Saini S, Majid S, Yamamura S, Tabatabai L, Suh SO, Shahryari V, Chen Y, Deng G, Tanaka Y, Dahiya R. Regulatory Role of mir-203 in Prostate Cancer Progression and Metastasis. Clinical cancer research : an official journal of the American Association for Cancer Research. 2011; 17: 5287-5298.
63. Viticchie G, Lena AM, Latina A, Formosa A, Gregersen LH, Lund AH, Bernardini S, Mauriello A, Miano R, Spagnoli LG, Knight RA, Candi E, Melino G. MiR-203 controls proliferation, migration and invasive potential of prostate cancer cell lines. Cell Cycle. 2011; 10: 1121-1131.
64. Shirakihara T, Kawasaki T, Fukagawa A, Semba K, Sakai R, Miyazono K, Miyazawa K, Saitoh M. Identification of integrin alpha3 as a molecular marker of cells undergoing epithelial-mesenchymal transition and of cancer cells with aggressive phenotypes. Cancer science. 2013; 104: 1189-1197.
65. Suh SO, Chen Y, Zaman MS, Hirata H, Yamamura S, Shahryari V, Liu J, Tabatabai ZL, Kakar S, Deng G, Tanaka Y, Dahiya R. MicroRNA-145 is regulated by DNA methylation and p53 gene mutation in prostate cancer. Carcinogenesis. 2011; 32: 772-778.
66. Peng X, Guo W, Liu T, Wang X, Tu X, Xiong D, Chen S, Lai Y, Du H, Chen G, Liu G, Tang Y, Huang S, et al. Identification of miRs-143 and -145 that is associated with bone metastasis of prostate cancer and involved in the regulation of EMT. PloS one. 2011; 6: e20341.
67. Ren D, Wang M, Guo W, Huang S, Wang Z, Zhao X, Du H, Song L, Peng X. Double-negative feedback loop between ZEB2 and miR-145 regulates epithelial-mesenchymal transition and stem cell properties in prostate cancer cells. Cell and tissue research. 2014; 358: 763-778.
68. Guo W, Ren D, Chen X, Tu X, Huang S, Wang M, Song L, Zou X, Peng X. HEF1 promotes epithelial mesenchymal transition and bone invasion in prostate cancer under the regulation of microRNA-145. Journal of cellular biochemistry. 2013; 114: 1606-1615.
69. Ren D, Wang M, Guo W, Zhao X, Tu X, Huang S, Zou X, Peng X. Wild-type p53 suppresses the epithelial-mesenchymal transition and stemness in PC-3 prostate cancer cells by modulating miR145. International journal of oncology. 2013; 42: 1473-1481.
70. Qin W, Pan Y, Zheng X, Li D, Bu J, Xu C, Tang J, Cui R, Lin P, Yu X. MicroRNA-124 regulates TGF-alpha-induced epithelial-mesenchymal transition in human prostate cancer cells. International journal of oncology. 2014; 45: 1225-1231.
71. Zhao X, Wang Y, Deng R, Zhang H, Dou J, Yuan H, Hou G, Du Y, Chen Q, Yu J. miR186 suppresses prostate cancer progression by targeting Twist1. Oncotarget. 2016; 7:33136-51. doi: 10.18632/oncotarget.8887.
72. Roush S, Slack FJ. The let-7 family of microRNAs. Trends in cell biology. 2008; 18: 505-516.
73. Viswanathan SR, Daley GQ. Lin28: A microRNA regulator with a macro role. Cell. 2010; 140: 445-449.
74. Viswanathan SR, Daley GQ, Gregory RI. Selective blockade of microRNA processing by Lin28. Science. 2008; 320: 97-100.
75. Viswanathan SR, Powers JT, Einhorn W, Hoshida Y, Ng TL, Toffanin S, O’Sullivan M, Lu J, Phillips LA, Lockhart VL, Shah SP, Tanwar PS, Mermel CH, et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nature genetics. 2009; 41: 843-848.
76. Peng S, Maihle NJ, Huang Y. Pluripotency factors Lin28 and Oct4 identify a sub-population of stem cell-like cells in ovarian cancer. Oncogene. 2010; 29: 2153-2159.
77. Jiang J, Lee EJ, Gusev Y, Schmittgen TD. Real-time expression profiling of microRNA precursors in human cancer cell lines. Nucleic acids research. 2005; 33: 5394-5403.
78. Ozen M, Creighton CJ, Ozdemir M, Ittmann M. Widespread deregulation of microRNA expression in human prostate cancer. Oncogene. 2008; 27: 1788-1793.
79. Boyerinas B, Park SM, Hau A, Murmann AE, Peter ME. The role of let-7 in cell differentiation and cancer. Endocrine-related cancer. 2010; 17: F19-36.
80. Kong D, Banerjee S, Ahmad A, Li Y, Wang Z, Sethi S, Sarkar FH. Epithelial to mesenchymal transition is mechanistically linked with stem cell signatures in prostate cancer cells. PloS one. 2010; 5: e12445.
81. Motoyama K, Inoue H, Nakamura Y, Uetake H, Sugihara K, Mori M. Clinical significance of high mobility group A2 in human gastric cancer and its relationship to let-7 microRNA family. Clinical cancer research : an official journal of the American Association for Cancer Research. 2008; 14: 2334-2340.
82. Dong Q, Meng P, Wang T, Qin W, Wang F, Yuan J, Chen Z, Yang A, Wang H. MicroRNA let-7a inhibits proliferation of human prostate cancer cells in vitro and in vivo by targeting E2F2 and CCND2. PloS one. 2010; 5: e10147.
83. Thuault S, Tan EJ, Peinado H, Cano A, Heldin CH, Moustakas A. HMGA2 and Smads co-regulate SNAIL1 expression during induction of epithelial-to-mesenchymal transition. The Journal of biological chemistry. 2008; 283: 33437-33446.
84. Lo UG, Yang D, Hsieh JT. The role of microRNAs in prostate cancer progression. Translational andrology and urology. 2013; 2: 228-241.
85. Zhu C, Li J, Cheng G, Zhou H, Tao L, Cai H, Li P, Cao Q, Ju X, Meng X, Wang M, Zhang Z, Qin C, et al. miR-154 inhibits EMT by targeting HMGA2 in prostate cancer cells. Molecular and cellular biochemistry. 2013; 379: 69-75.
86. Hermeking H. The miR-34 family in cancer and apoptosis. Cell death and differentiation. 2010; 17: 193-199.
87. He L, He X, Lim LP, de Stanchina E, Xuan Z, Liang Y, Xue W, Zender L, Magnus J, Ridzon D, Jackson AL, Linsley PS, Chen C, et al. A microRNA component of the p53 tumour suppressor network. Nature. 2007; 447: 1130-1134.
88. Cheng CY, Hwang CI, Corney DC, Flesken-Nikitin A, Jiang L, Oner GM, Munroe RJ, Schimenti JC, Hermeking H, Nikitin AY. miR-34 cooperates with p53 in suppression of prostate cancer by joint regulation of stem cell compartment. Cell reports. 2014; 6: 1000-1007.
89. Liang J, Li Y, Daniels G, Sfanos K, De Marzo A, Wei J, Li X, Chen W, Wang J, Zhong X, Melamed J, Zhao J, Lee P. LEF1 Targeting EMT in Prostate Cancer Invasion Is Regulated by miR-34a. Molecular cancer research : MCR. 2015; 13: 681-688.
90. Majid S, Dar AA, Saini S, Shahryari V, Arora S, Zaman MS, Chang I, Yamamura S, Tanaka Y, Chiyomaru T, Deng G, Dahiya R. miRNA-34b inhibits prostate cancer through demethylation, active chromatin modifications, and AKT pathways. Clinical cancer research : an official journal of the American Association for Cancer Research. 2013; 19: 73-84.
91. Majid S, Dar AA, Saini S, Arora S, Shahryari V, Zaman MS, Chang I, Yamamura S, Tanaka Y, Deng G, Dahiya R. miR-23b represses proto-oncogene Src kinase and functions as methylation-silenced tumor suppressor with diagnostic and prognostic significance in prostate cancer. Cancer Res. 2012; 72: 6435-6446.
92. Kao CJ, Martiniez A, Shi XB, Yang J, Evans CP, Dobi A, deVere White RW, Kung HJ. miR-30 as a tumor suppressor connects EGF/Src signal to ERG and EMT. Oncogene. 2014; 33: 2495-2503.
93. Wang M, Ren D, Guo W, Wang Z, Huang S, Du H, Song L, Peng X. Loss of miR-100 enhances migration, invasion, epithelial-mesenchymal transition and stemness properties in prostate cancer cells through targeting Argonaute 2. International journal of oncology. 2014; 45: 362-372.
94. Ru P, Steele R, Newhall P, Phillips NJ, Toth K, Ray RB. miRNA-29b suppresses prostate cancer metastasis by regulating epithelial-mesenchymal transition signaling. Molecular cancer therapeutics. 2012; 11: 1166-1173.
95. Steele R, Mott JL, Ray RB. MBP-1 upregulates miR-29b that represses Mcl-1, collagens, and matrix-metalloproteinase-2 in prostate cancer cells. Genes & cancer. 2010; 1: 381-387.
96. Yan B, Guo Q, Nan XX, Wang Z, Yin Z, Yi L, Wei YB, Gao YL, Zhou KQ, Yang JR. Micro-ribonucleic acid 29b inhibits cell proliferation and invasion and enhances cell apoptosis and chemotherapy effects of cisplatin via targeting of DNMT3b and AKT3 in prostate cancer. OncoTargets and therapy. 2015; 8: 557-565.
97. Cai C, Chen QB, Han ZD, Zhang YQ, He HC, Chen JH, Chen YR, Yang SB, Wu YD, Zeng YR, Qin GQ, Liang YX, Dai QS, et al. miR-195 Inhibits Tumor Progression by Targeting RPS6KB1 in Human Prostate Cancer. Clinical cancer research : an official journal of the American Association for Cancer Research. 2015; 21: 4922-4934.
98. Liu C, Guan H, Wang Y, Chen M, Xu B, Zhang L, Lu K, Tao T, Zhang X, Huang Y. miR-195 Inhibits EMT by Targeting FGF2 in Prostate Cancer Cells. PloS one. 2015; 10: e0144073.
99. Zhang X, Tao T, Liu C, Guan H, Huang Y, Xu B, Chen M. Downregulation of miR-195 promotes prostate cancer progression by targeting HMGA1. Oncology reports. 2016.
100. Kurozumi A, Goto Y, Matsushita R, Fukumoto I, Kato M, Nishikawa R, Sakamoto S, Enokida H, Nakagawa M, Ichikawa T, Seki N. Tumor-suppressive microRNA-223 inhibits cancer cell migration and invasion by targeting ITGA3/ITGB1 signaling in prostate cancer. Cancer science. 2016; 107: 84-94.
101. Yang J, Hou Y, Zhou M, Wen S, Zhou J, Xu L, Tang X, Du YE, Hu P, Liu M. Twist induces epithelial-mesenchymal transition and cell motility in breast cancer via ITGB1-FAK/ILK signaling axis and its associated downstream network. The international journal of biochemistry & cell biology. 2016; 71: 62-71.
102. Wang L, Song G, Tan W, Qi M, Zhang L, Chan J, Yu J, Han J, Han B. MiR-573 inhibits prostate cancer metastasis by regulating epithelial-mesenchymal transition. Oncotarget. 2015; 6:35978-90. doi: 10.18632/oncotarget.5427.
103. Yang F, Zhang Y, Ressler SJ, Ittmann MM, Ayala GE, Dang TD, Wang F, Rowley DR. FGFR1 is essential for prostate cancer progression and metastasis. Cancer Res. 2013; 73: 3716-3724.
104. Nam RK, Benatar T, Wallis CJ, Amemiya Y, Yang W, Garbens A, Naeim M, Sherman C, Sugar L, Seth A. MiR-301a regulates E-cadherin expression and is predictive of prostate cancer recurrence. The Prostate. 2016.
105. Wen YC, Lee WJ, Tan P, Yang SF, Hsiao M, Lee LM, Chien MH. By inhibiting snail signaling and miR-23a-3p, osthole suppresses the EMT-mediated metastatic ability in prostate cancer. Oncotarget. 2015; 6: 21120-21136. doi: 10.18632/oncotarget.4229.
106. Volinia S, Calin GA, Liu CG, Ambs S, Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M, Prueitt RL, Yanaihara N, Lanza G, et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proceedings of the National Academy of Sciences of the United States of America. 2006; 103: 2257-2261.
107. Hatley ME, Patrick DM, Garcia MR, Richardson JA, Bassel-Duby R, van Rooij E, Olson EN. Modulation of K-Ras-dependent lung tumorigenesis by MicroRNA-21. Cancer cell. 2010; 18: 282-293.
108. Coppola V, Musumeci M, Patrizii M, Cannistraci A, Addario A, Maugeri-Sacca M, Biffoni M, Francescangeli F, Cordenonsi M, Piccolo S, Memeo L, Pagliuca A, Muto G, et al. BTG2 loss and miR-21 upregulation contribute to prostate cell transformation by inducing luminal markers expression and epithelial-mesenchymal transition. Oncogene. 2013; 32: 1843-1853.
109. Winkler GS. The mammalian anti-proliferative BTG/Tob protein family. Journal of cellular physiology. 2010; 222: 66-72.
110. Bonci D, Coppola V, Patrizii M, Addario A, Cannistraci A, Francescangeli F, Pecci R, Muto G, Collura D, Bedini R, Zeuner A, Valtieri M, Sentinelli S, et al. A microRNA code for prostate cancer metastasis. Oncogene. 2016; 35: 1180-1192.
111. Jalava SE, Urbanucci A, Latonen L, Waltering KK, Sahu B, Janne OA, Seppala J, Lahdesmaki H, Tammela TL, Visakorpi T. Androgen-regulated miR-32 targets BTG2 and is overexpressed in castration-resistant prostate cancer. Oncogene. 2012; 31: 4460-4471.
112. Gururajan M, Josson S, Chu GC, Lu CL, Lu YT, Haga CL, Zhau HE, Liu C, Lichterman J, Duan P, Posadas EM, Chung LW. miR-154* and miR-379 in the DLK1-DIO3 microRNA mega-cluster regulate epithelial to mesenchymal transition and bone metastasis of prostate cancer. Clinical cancer research : an official journal of the American Association for Cancer Research. 2014; 20: 6559-6569.
113. Edlund S, Bu S, Schuster N, Aspenstrom P, Heuchel R, Heldin NE, ten Dijke P, Heldin CH, Landstrom M. Transforming growth factor-beta1 (TGF-beta)-induced apoptosis of prostate cancer cells involves Smad7-dependent activation of p38 by TGF-beta-activated kinase 1 and mitogen-activated protein kinase kinase 3. Molecular biology of the cell. 2003; 14: 529-544.
114. Fujii T, Shimada K, Tatsumi Y, Tanaka N, Fujimoto K, Konishi N. Syndecan-1 up-regulates microRNA-331-3p and mediates epithelial-to-mesenchymal transition in prostate cancer. Molecular carcinogenesis. 2015.
115. Bijnsdorp IV, Hodzic J, Lagerweij T, Westerman B, Krijgsman O, Broeke J, Verweij F, Nilsson RJ, Rozendaal L, van Beusechem VW, van Moorselaar JA, Wurdinger T, Geldof AA. miR-129-3p controls centrosome number in metastatic prostate cancer cells by repressing CP110. Oncotarget. 2016; 7: 16676-16687. doi: 10.18632/oncotarget.7572.
116. Josson S, Gururajan M, Hu P, Shao C, Chu GY, Zhau HE, Liu C, Lao K, Lu CL, Lu YT, Lichterman J, Nandana S, Li Q, et al. miR-409-3p/-5p promotes tumorigenesis, epithelial-to-mesenchymal transition, and bone metastasis of human prostate cancer. Clinical cancer research : an official journal of the American Association for Cancer Research. 2014; 20: 4636-4646.
117. Tang DG, Patrawala L, Calhoun T, Bhatia B, Choy G, Schneider-Broussard R, Jeter C. Prostate cancer stem/progenitor cells: identification, characterization, and implications. Mol Carcinog. 2007; 46: 1-14.
118. Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res. 2005; 65: 10946-10951.
119. Li H, Chen X, Calhoun-Davis T, Claypool K, Tang DG. PC3 human prostate carcinoma cell holoclones contain self-renewing tumor-initiating cells. Cancer Res. 2008; 68: 1820-1825.
120. Patrawala L, Calhoun T, Schneider-Broussard R, Li H, Bhatia B, Tang S, Reilly JG, Chandra D, Zhou J, Claypool K, Coghlan L, Tang DG. Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells. Oncogene. 2006; 25: 1696-1708.
121. Patrawala L, Calhoun T, Schneider-Broussard R, Zhou J, Claypool K, Tang DG. Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic. Cancer Res. 2005; 65: 6207-6219.
122. Patrawala L, Calhoun-Davis T, Schneider-Broussard R, Tang DG. Hierarchical organization of prostate cancer cells in xenograft tumors: the CD44+alpha2beta1+ cell population is enriched in tumor-initiating cells. Cancer Res. 2007; 67: 6796-6805.
123. Fang YX, Chang YL, Gao WQ. MicroRNAs targeting prostate cancer stem cells. Exp Biol Med (Maywood). 2015; 240: 1071-1078.
124. Bao B, Ahmad A, Kong D, Ali S, Azmi AS, Li Y, Banerjee S, Padhye S, Sarkar FH. Hypoxia induced aggressiveness of prostate cancer cells is linked with deregulated expression of VEGF, IL-6 and miRNAs that are attenuated by CDF. PloS one. 2012; 7: e43726.
125. Kong D, Heath E, Chen W, Cher ML, Powell I, Heilbrun L, Li Y, Ali S, Sethi S, Hassan O, Hwang C, Gupta N, Chitale D, et al. Loss of let-7 up-regulates EZH2 in prostate cancer consistent with the acquisition of cancer stem cell signatures that are attenuated by BR-DIM. PloS one. 2012; 7: e33729.
126. Liu C, Kelnar K, Liu B, Chen X, Calhoun-Davis T, Li H, Patrawala L, Yan H, Jeter C, Honorio S, Wiggins JF, Bader AG, Fagin R, et al. The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat Med. 2011; 17: 211-215.
127. Liu C, Tang DG. MicroRNA regulation of cancer stem cells. Cancer Res. 2011; 71: 5950-5954.
128. Wicha MS. Stemming a tumor with a little miR. Nat Med. 2011; 17: 162-164.
129. Saini S, Majid S, Shahryari V, Arora S, Yamamura S, Chang I, Zaman MS, Deng G, Tanaka Y, Dahiya R. miRNA-708 control of CD44(+) prostate cancer-initiating cells. Cancer Res. 2012; 72: 3618-3630.

