INTRODUCTION
Allogeneic hematopoietic stem cell transplantation (HSCT) is an important curative treatment option for patients with many malignant and non-malignant hematologic diseases [1, 2]. However, substantial risks remain for morbidity and mortality, which are largely attributable to disease relapse [3], graft-versus-host disease [4-6] and infection [7]. Allo-reactivity caused by immunologic reactions between two individuals has great therapeutic benefit through graft-versus-leukemia (GVL) reactions, but is also responsible for development of graft-versus-host disease (GVHD) that can be potentially fatal [8]. Because of treatment related mortality, GVHD, both in its acute and chronic forms, continues to be a major concern following allo-HSCT despite its association with a lower relapse rate [9, 10].
Current approaches for the prevention and treatment of GVHD involve direct blockade of T-cell function [11]. Cyclosporine (CsA), a calcineurin inhibitor (CNI), is one of the most commonly used pharmacologic agents for the prevention of GVHD [12]. CsA is thought to bind to its specific immunophilin, cyclophilin, of immunocompetent lymphocytes, especially T-lymphocytes [13]. This CNI-immunophilin complex inhibits calcineurin, which leads to a reduced function of effector T cells through inhibiting lymphokine production and interleukin release [14]. Despite its widespread use in clinical practice, the dose, target blood level of CsA, and schedule of administration vary among protocols [15]. In addition, although close surveillance, it is not always easy to keep the CsA levels within the desired range. Based on these findings, there have been interesting researches conferring impact of cyclosporine levels on the development of acute GVHD after allo-HSCT, in which higher CsA levels during early post-transplant period contributed to lower risk of acute GVHD [12, 16, 17]. However, the effect of blood level of CsA on the occurrence of chronic GVHD has rarely been studied.
The immune mechanisms eliciting chronic GVHD is differ from those of acute GVHD [18]. Acute GVHD is considered to be the consequent tissue damage initiated from the interaction between donor T cells and host antigen presenting cells, which is amplified by cytokine from damaged tissues and the lipopolysaccharide that leaks through the injured intestinal mucosa [19, 20]. The pathophysiology of chronic GVHD is much more complex and still remains to be determined. Among the possible explanations on how chronic GVHD develops, regulatory T cells (Treg) numbers have been reported to be diminished in chronic GVHD from a series of studies [21-23]. Of note, Tregs are known to be influenced by immunosuppressive therapy [24]. In particular, previous studies demonstrated that calcineurin inhibitors seem to reduce Treg function and T cell proliferation in vitro [25-29]. Owing to the fact that CsA has an impact on the number and activity of Tregs [30] and the possible role of T regs on suppression of chronic GVHD, we reasoned that there might be a relationship between blood concentration of CsA and the occurrence of chronic GVHD.
RESULTS
Patients’ characteristics
The analysis included 183 patients; their baseline characteristics are listed in Table 1. The median age was 45 years (range, 18-68), and more men than women were included. Acute leukemia was the most common disease and comprised 67.8% (n=124) of all cases. Myelodysplastic syndrome (MDS) and chronic myelomonocytic leukemia (CMML) came next (13.7%, n=25), and severe aplastic anemia, myeloproliferative neoplasm and lymphoma etc. accounted for the rest. Almost all patients received myeloablative conditioning (n=176, 96.2%). Total-body irradiation (TBI)-based conditioning was performed in 52 patients (28.4%), and 53 patients (29.0%) received anti-thymocyte globulin (ATG) during allo-HSCT. Peripheral blood was used as the source of stem cells in all patients. Acute GVHD was occurred in 51 patients (27.9%), 21 of whom experienced acute GVHD of grade 2 or more.
Table 1: Baseline characteristics
Characteristics (total n=183) |
Number (%) |
|
|---|---|---|
Age |
Median (range) |
45 years (18–68) |
Sex |
Male |
102 (55.7%) |
Female |
81 (44.3%) |
|
Disease |
Acute leukemia |
124 (67.8%) |
MDS* |
25 (13.7 %) |
|
MPN** |
13 (7.1%) |
|
Severe AA |
11 (6.0%) |
|
Others*** |
10 (5.5%) |
|
Conditioning |
Myeloablative |
176 (96.2%) |
TBI (+) |
52 (28.4%) |
|
ATG (+) |
53 (29.0%) |
|
Donor |
Matched sibling |
179 (97.8%) |
Haplo-identical family |
4 (2.2%) |
|
Graft source |
Peripheral blood |
183 (100.0%) |
Bone marrow |
0 (0.0%) |
|
Acute GVHD |
Any grade |
51 (27.9%) |
Grade 2 or more |
21 (11.5%) |
Abbreviations: MDS = myelodysplastic syndrome; MPN = myeloproliferative neoplasm; AA = aplastic anemia; TBI = total-body irradiation; ATG = anti-thymocyte globulin; GVHD
*Three chronic myelomonocytic leukemia (CMML) patients were included.
**Six primary myelofibrosis (PMF), one post-essential thrombocythemia myelofibrosis (post ET-MF), and six chronic myeloid leukemia (CML) patients were included.
***Five non-hodgkin lymphoma (NHL), two paroxysmal nocturnal hemoglobinuria (PNH), one thalassemia, one chronic active EBV infection, and one blastic plasmacytoid dendritic cell neoplasm (BPDCN) patients were included.
Blood levels of cyclosporine according to post-transplant period
The mean values of CsAavr were 229.9, 197.0, and 168.5 ng/ml during the first 0-1 month, 1-2 month, 2-3 month after allo-HSCT, respectively (Table 2, Figure 1A). When grouping patients by drug levels, majority of patients were assigned to group 2 (CsAavr, ≥ 200 ~ <250 ng/ml) during the 1st period (n=110, 60.1%). As it went to the 2nd and 3rd period, the average drug levels tended to be lower and the proportion of patients with drug levels corresponding to group 1 is increased: 54.2% during the 2nd period, 70.0% during the 3rd period (Table 2, Figure 1B).
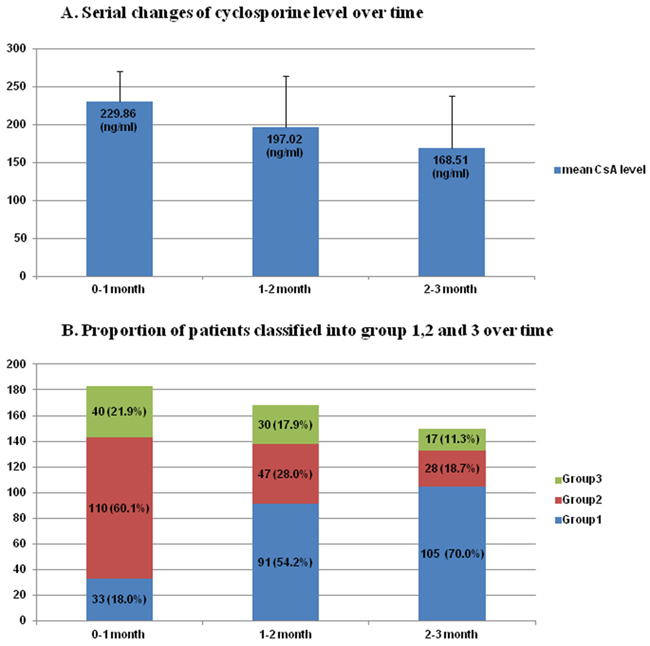
Figure 1: A. Serial changes of cyclosporine level over time B. Proportion of patients classified into group 1,2 and 3 over time
Table 2: Blood levels of cyclosporine according to post-transplant period
Post-transplant period |
Blood CsA level |
Patients, number |
|---|---|---|
0-1 month (available n=183) |
Mean, ng/ml (SE) |
229.9 (39.9) |
Group 1 |
33 (18.0%) |
|
Group 2 |
110 (60.1%) |
|
Group 3 |
40 (21.9%) |
|
1-2 month (available n=168) |
Mean, ng/ml (SE) |
197.0 (67.7) |
Group 1 |
91 (54.2%) |
|
Group 2 |
47 (28.0%) |
|
Group 3 |
30 (17.9%) |
|
2-3 month (available n=150) |
Mean, ng/ml (SE) |
168.5 (69.3) |
Group 1 |
105 (70.0%) |
|
Group 2 |
28 (18.7%) |
|
Group 3 |
17 (11.3%) |
Abbreviations: SE, standard error
The occurrence of moderate to severe chronic GVHD according to CsA blood concentration
Figure 2 shows the cumulative incidence of moderate to severe chronic GVHD (cGVHDmod-sev) according to the blood CsA levels during post-transplant period. Among the drug levels of different periods, the CsAavr level during the first month after transplantation showed significant association with the occurrence of cGVHDmod-sev, and the incidence was lowest in group 2 patients. In univariate analysis using Cox proportional hazards regression model, ATG usage was also associated with less occurrence of cGVHDmod-sev (p=0.007) (Table 3). In multivariate analysis adjusted for gender, age, total body irradiation, anti-thymocyte globulin, acute GVHD ≥ grade 2 and CsAavr levels of other periods, the CsAavr level during the first month were proven to significantly be associated with the occurrence of cGVHDmod-sev (p=0.003).
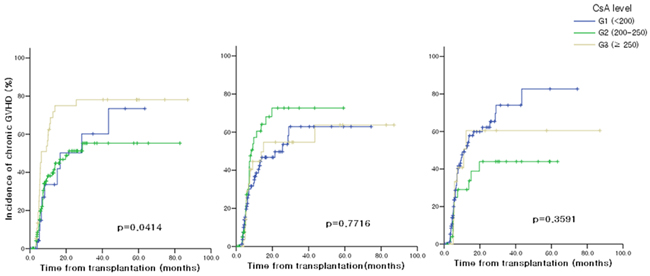
Figure 2: Moderate to severe chronic GVHD according to blood CsA levels A. 0-1 month after allo-HSCT B. 1-2 months after allo-HSCT C. 2-3 months after allo-HSCT
Table 3: Univariate and multivariate analyses of cGHVD, DFS, and OS
cGVHD |
DFS |
OS |
|||||||
|---|---|---|---|---|---|---|---|---|---|
univariate |
multivariate |
univariate |
multivariate |
univariate |
multivariate |
||||
P-value |
P-value |
HR (95% CI) |
P-value |
P-value |
HR (95% CI) |
P-value |
P-value |
HR (95% CI) |
|
Sex |
0.141 |
0.274 |
0.753 (0.453-1.252) |
0.634 |
0.360 |
0.719 (0.355-1.457) |
0.363 |
0.614 |
0.856 (0.469-1.563) |
Age* |
0.088 |
0.859 |
1.048 (0.621-1.796) |
0.877 |
0.763 |
1.112 (0.558-2.214) |
0.007 |
0.036 |
1.935 (1.045-3.585) |
TBI |
0.694 |
0.073 |
0.594 (0.337-1.050) |
0.337 |
0.452 |
1.342 (0.624-2.886) |
0.663 |
0.663 |
0.859 (0.434-1.701) |
ATG |
0.007 |
0.073 |
0.462 (0.199-1.076) |
0.032 |
0.476 |
0.683 (0.239-1.949) |
0.626 |
0.531 |
1.292 (0.579-2.882) |
acute GVHD ≥ grade 2 |
0.901 |
0.731 |
1.178 (0.463-2.998) |
0.489 |
0.130 |
2.308 (0.781-6.818) |
0.090 |
0.004 |
3.647 (1.504-8.843) |
CsAavr during 0-1 month** |
0.051 |
0.003 |
0.264 |
1.000 |
1.000 |
1.000 |
|||
2 vs 1 |
1 |
1 |
0.752 (0.235-2.403) |
0.396 |
1.000 |
2.128 (0.329-13.78) |
1.000 |
1.000 |
0.858 (0.242-3.042) |
3 vs 1 |
0.711 |
0.423 |
2.294 (0.710-7.414) |
1.000 |
1.000 |
1.371 (0.172-10.94) |
1.000 |
1.000 |
0.875 (0.207-3.698) |
2 vs 3 |
0.054 |
<0.001 |
0.328 (0.144-0.746) |
1.000 |
1.000 |
1.553 (0.445-5.422) |
1.000 |
1.000 |
0.981 (0.335-2.874) |
CsAavr during 1-2 month** |
0.795 |
0.156 |
0.282 |
0.264 |
0.018 |
0.030 |
|||
2 vs 1 |
1.000 |
0.153 |
2.114 (0.876-5.101) |
0.27 |
0.252 |
2.566 (0.771-8.538) |
0.234 |
0.09 |
2.544 (0.925-6.998) |
3 vs 1 |
1.000 |
1.000 |
1.198 (0.440-3.258) |
1.000 |
1.000 |
1.757 (0.445-6.933) |
1.000 |
1.000 |
0.742 (0.169-3.248) |
2 vs 3 |
1.000 |
1.000 |
1.765 (0.626-4.979) |
1.000 |
1.000 |
1.460 (0.382-5.589) |
0.036 |
0.162 |
3.430 (0.794-14.82) |
CsAavr during 2-3 month** |
0.393 |
0.105 |
1.000 |
1.000 |
1.000 |
1.000 |
|||
2 vs 1 |
0.405 |
0.090 |
0.387 (0.138-1.087) |
1.000 |
1.000 |
0.718 (0.181-2.845) |
1.000 |
1.000 |
0.790 (0.251-2.484) |
3 vs 1 |
1.000 |
1.000 |
0.922 (0.298-2.855) |
1.000 |
1.000 |
1.233 (0.289-5.259) |
1.000 |
1.000 |
0.995 (0.245-4.046) |
2 vs 3 |
1.000 |
0.756 |
0.419 (0.102-1.718) |
1.000 |
1.000 |
0.582 (0.093-3.657) |
1.000 |
1.000 |
0.794 (0.145-4.340) |
Abbreviations: TBI = total-body irradiation; ATG = anti-thymocyte globulin; OS = overall survival; DFS = disease-free survival; HR = hazard ratio; CI = confidence interval
* Age: <45 and ≥ 45 years of patients were compared.
** Blood drug levels (CsAavr) corresponding to Group 1 (CsAavr, <200 ng/ml) vs 2 (CsAavr, ≥ 200 ~ <250 ng/ml) vs 3 (CsAavr, ≥ 250 ng/ml) were compared.
DFS and OS according to CsA blood concentration
In the univariate and multivariate analysis for DFS, none of the factors including sex, age, TBI, ATG, acute GVHD ≥ Gr.2 and the CsAavr levels of each period, influenced DFS (Table 3) (Supplementary Figure 1). The univariate analysis for OS revealed that age < 45 years and the CsAavr level ≥ 250 ng/ml during 1-2 month after HSCT showed a significant association with better OS. The result of multivariate analysis for OS was similar to that of univariate analysis showing a significance of age (p=0.036, HR=1.935) and the CsAavr level during 1-2 month (p=0.030), but acute GVHD ≥ Gr.2 was also determined as an independent prognostic factors for worse OS (p=0.004, HR=3.647). Figure 3 demonstrates OS according to the blood CsA levels during post-transplant period.
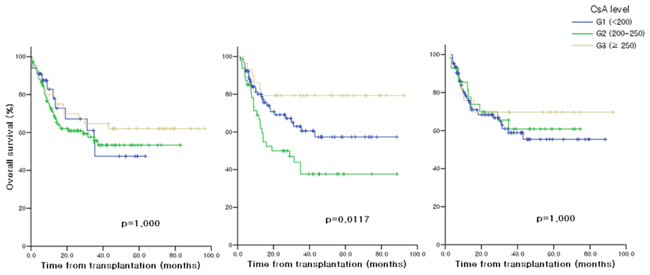
Figure 3: Overall survival according to CsA levels A. 0-1 month after allo-HSCT B. 1-2 months after allo-HSCT C. 2-3 months after allo-HSCT
DISCUSSION
This study began with clinical question of whether or not there would be an association between post transplant CsA concentration and chronic GVHD. There has already been substantial investigation on how CsA levels affects acute GVHD occurrence, however, relatively little attention has been paid to this question. In this study, we found that average drug levels of first month after transplantation was particularly relevant to the occurrence of chronic GVHD. Of note, chronic GVHD occurred less in group 2 patients, whose average drug levels corresponded to the range of 200-250ng/ml, compared to group 1 or 3 patients. This means it can be disadvantageous for preventing chronic GVHD if the CsA levels are less (group 1) or greater (group 3) than some degree of range. In the analyses of previous studies on acute GVHD, it was observed that higher drug levels within 3 or 4 weeks after HSCT were more effective in prevention of acute GVHD [12, 16, 17], and this is consistent with common concept that prevention of acute GVHD can be achievable by pharmacologic suppression of effector T cell.
Striking finding from the current study is that drug level of 250ng/ml or more were more disadvantageous for preventing chronic GVHD than that of 200-250ng/ml. The result of this study implies that the degree of immune suppression during early post-transplantation period might also influence late allogeneic reactivity such as chronic GVHD. We don’t know exactly how this happened. One of the possible explanations is that the effects of CsA on T cells may vary among the different subsets of T cell population [29]. Actually, T cells consist of several subsets whose responses differ based on antigenic stimuli, activation thresholds, and effector functions [31]. Cyclosporine is the agent that inhibits IL-2 transcription, which leads to fail in T cell proliferation. The role of IL-2 as a growth factor for effector T cells has been well known. However, ironically, IL-2 functions as an indispensable factor for inducing activation-induced cell death (AICD) of activated effector T cells [31, 32], which means IL-2 is necessarily needed to reduce the mass of alloreactive cytopathic effector T cells. In addition, of note, presence of IL-2 is the prerequisite for survival and growth of regulator T cells [33-35]. And, there was a report showing that IL-2 exerts an anti-apoptotic effect on CD4+CD25+ T reg population [36]. Taken together, inhibition of T cell proliferation via IL-2 blockade can be achieved by calcineurin inhibitor such as CsA, and this would be advantageous for prevention of acute GVHD during early post-transplant period. But excessive IL-2 blockade potentially leave relatively large burden of alloreactive T cells through insufficient AICD, and may also hamper survival and growth of regulatory T cells as well as decrease the number of these cells. We cannot assure whether or not suppression of T regs and effector T cell mass over the limit during the early post-transplant period can really instigate a development of chronic GVHD, however, it might be worth considering an impact of excessive IL-2 suppression during early post-transplant period on the late immunologic reaction in stem cell transplanted patients. Other than chronic GVHD, the results in this study indicated that overall survival was influenced by CsAavr level during 1-2 month. Specifically, OS was longest in patients with the CsAavr level ≥ 250ng/ml (group 3), and worst in Group 2 patients (CsAavr, ≥ 200 ~ <250 ng/ml). We cannot assure the precise reason for this finding; but, there might be a causal relationship between the CsA level and the occurrence of acute GVHD. If one experienced acute GVHD, the CsA level would probably be targeted higher. On the contrary, one can had acute GVHD because CsA level was not sufficiently achieved. Given the acute GVHD ≥ Gr.2. was worse prognostic factor for OS, the inseparable connection between CsA level and acute GVHD can affect the results of the analysis.
We acknowledge that there are substantial limitations to the current study. First, institutional strategy for the ATG use has been changed since April 2013. Before then, ATG was not routinely used in matched sibling donor (MSD) transplantation, but only used in transplantation from unrelated donor. Whereas, ATG has actively incorporated into HSCT process even in transplantation from MSD since 2013 based on the recent observations that ATG use was associated with less occurrence of chronic GVHD, but not linked to increasing risk of relapse [37-39]. Although ATG showed significant association for preventing chronic GVHD only in an univariate analysis in this study, it may still be an important factor that can affect chronic GVHD development. In addition, the dose and duration of steroid treatment was not considered in an analysis. In some patients, for example, steroid could have been used for the treatment of acute GVHD, for hormonal replacement in case of adrenal insufficiency, or could have been introduced to patients who were not able to tolerate the use of calcineurin inhibitor. Consequently, heterogeneous distribution of treatment procedures, especially with regard to whether or not patients were treated with ATG and/or steroid that can affect T cell activity as well, might possibly confuse the interpretation of the results. And importantly, the study is limited in its retrospective nature and the patients’ composition of heterogeneous diseases. Lastly, our hypothesis that T regs and effector T cell mass during an early post-transplant period can influence development of chronic GVHD is limited by lack of concomitant experimental data involving T cell subsets, which has to be validated in a prospective manner.
In summary, we found that blood level of CsA during the first month after transplantation was relevant to the occurrence of chronic GVHD. The average drug level between 200 and 250 mg/ml (versus <200 or ≥ 250ng/ml) was significantly associated with the less occurrence of moderate to severe cGVHD.
MATERIALS AND METHODS
Patients
One hundred and eighty three patients who received allogeneic HSCT in Samsung Medical Center (Seoul, South Korea) between Sep 2006 and June 2014 were included. Patients who had received methotrexate and cyclosporine for GVHD prophylaxis were identified and retrospectively reviewed. According to institutional strategy, cyclosporine had been used for the prevention of GVHD in matched sibling donor transplantation or in transplantation from haploidentical family donor whereas tacrolimus was the agent used in transplantation from unrelated donor. Consequently, four patients who underwent allo-HSCT from family donors were only included in this study. Patients and donors were HLA typed using high-resolution molecular techniques. Genomic DNA isolated from peripheral blood samples were subjected to sequence-based typing at exons 2, 3 and 4 of HLA-A,B and C genes and at exon 2 of HLA-DRB1 with a commercial SBT kit (AlleleSEQR HLA sequencing kit, Atria Genetics, San Francisco, CA), nucleotide sequencing was performed using an ABI 3730 DNA Sequencer (Applied Biosystems, Foster City, CA) and sequences were analyzed with Assign version 3.5+ software (Conexio Genomics, Applecross, Australia).
Transplantation procedure and administration of cyclosporine
The conditioning regimen was mainly a combination of fludarabine (30mg/m2/day, 6 days) and busulfan (3.2mg/kg/day, 2 or 4 days) although cyclophosphamide (60mg/kg/day, 2 days) and total body irradiation (TBI) (999cGy, total) combination was primarily used in patients with acute lymphoblastic leukemia. Cyclosporine and methotrexate was used for GVHD prevention, and methotrexate was administered intravenously at 15mg/m2 on day 1 and 10mg/m2 on day 3, 6, and 11. Cyclosporine was started at 5mg/kg/day as a continuous infusion on day -1, and the dose was reduced to 3mg/kg/day on day 0. Since then, the whole blood cyclosporine concentration was measured every day, and the dose was titrated with a target blood trough level between 150 and 250 ng/ml. The route of CsA administration was switched to oral at a ratio of 1:3 when patients showed acceptable oral intake. After discharge from the hospital at least 3 weeks after transplantation, patients visited outpatient clinic at least once a week for 4 weeks, and at least every 2 week for next 8 weeks with monitoring of CsA blood level at every visit. Blood samples were performed 12 hours after the prior dose, just before the morning dose.
Blood concentration of cyclosporine and statistical considerations
The blood concentration of CsA was measured by high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS), and the sensitivity of the assay was 5 ng/ml. In the analyses of previous studies on acute GVHD, CsA levels during early post-transplant period, especially within 3 or 4 weeks after HSCT, was a major interest and drug levels were considered usually on a weekly basis [12, 16, 17]. However, because our primary interest was the occurrence of chronic GVHD instead of acute GVHD, we examined drug levels for a longer period of time (for 3 months after HSCT), and the time periods required for the analysis were categorized on a monthly basis. The average CsA blood concentration (CsAavr, ng/ml) during a post-transplant period, between 0-1 month, 1-2 months, and 2-3 months after allo-HSCT, was calculated in each patient. The median value of CsAavr was examined in each period, and its correlation with the occurrence of chronic GVHD, overall survival (OS) and disease free survival (DFS) were assessed. Patients were grouped as group 1, 2 and 3 according to average CsA level. When the CsAavr was below 200 ng/ml, they were classified as group 1; group 2 when the level was between 200 and 250 ng/ml, and group 3 if the level was 250 ng/ml or more. Chronic GVHD was determined and scored as mild, moderate or severe according to the National Institutes of Health (NIH) criteria [40]. Chronic GVHD of moderate to severe global severity, which needs systemic immune suppressive therapy, were only accounted for the analysis. The cumulative incidence of moderate to severe chronic GVHD, disease free survival (DFS) and overall survival (OS) according to CsA blood levels were estimated by the Kaplan-Meier method and compared by the log-rank test. The association of moderate to severe chronic GVHD, DFS and OS with clinical characteristics as well as blood CsA level was evaluated using the Cox proportional hazards regression model. P-value and 95% confidence interval (CI) were corrected by Bonferroni’s method due to multiple testing. A two-sided P value of <0.05 was considered significant. Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) v. 17.0 (SPSS Inc., Chicago, IL, USA).
CONFLICTS OF INTEREST
The authors declare that there is no conflict of interest regarding the publication of this paper.
REFERENCES
1. Lim Z, Brand R, Martino R, van Biezen A, Finke J, Bacigalupo A, Beelen D, Devergie A, Alessandrino E, Willemze R, Ruutu T, Boogaerts M, Falda M, et al. Allogeneic hematopoietic stem-cell transplantation for patients 50 years or older with myelodysplastic syndromes or secondary acute myeloid leukemia. J Clin Oncol. 2010; 28: 405-411.
2. Gupta V, Tallman MS and Weisdorf DJ. Allogeneic hematopoietic cell transplantation for adults with acute myeloid leukemia: myths, controversies, and unknowns. Blood. 2011; 117: 2307-2318.
3. Barrett AJ and Battiwalla M. Relapse after allogeneic stem cell transplantation. Expert Rev Hematol. 2010; 3: 429-441.
4. Lee SJ, Vogelsang G and Flowers ME. Chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2003; 9: 215-233.
5. Nash RA, Pepe MS, Storb R, Longton G, Pettinger M, Anasetti C, Appelbaum FR, Bowden RA, Deeg HJ, Doney K. Acute graft-versus-host disease: analysis of risk factors after allogeneic marrow transplantation and prophylaxis with cyclosporine and methotrexate. Blood. 1992; 80: 1838-1845.
6. Atkinson K, Horowitz MM, Gale RP, van Bekkum DW, Gluckman E, Good RA, Jacobsen N, Kolb HJ, Rimm AA, Ringden O. Risk factors for chronic graft-versus-host disease after HLA-identical sibling bone marrow transplantation. Blood. 1990; 75: 2459-2464.
7. Hiemenz JW. Management of infections complicating allogeneic hematopoietic stem cell transplantation. Semin Hematol. 2009; 46: 289-312.
8. Petersen SL. Alloreactivity as therapeutic principle in the treatment of hematologic malignancies. Studies of clinical and immunologic aspects of allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. Dan Med Bull. 2007; 54: 112-139.
9. Socie G, Stone JV, Wingard JR, Weisdorf D, Henslee-Downey PJ, Bredeson C, Cahn JY, Passweg JR, Rowlings PA, Schouten HC, Kolb HJ and Klein JP. Long-term survival and late deaths after allogeneic bone marrow transplantation. Late Effects Working Committee of the International Bone Marrow Transplant Registry. N Engl J Med. 1999; 341: 14-21.
10. Lee SJ, Klein JP, Barrett AJ, Ringden O, Antin JH, Cahn JY, Carabasi MH, Gale RP, Giralt S, Hale GA, Ilhan O, McCarthy PL, Socie G, et al. Severity of chronic graft-versus-host disease: association with treatment-related mortality and relapse. Blood. 2002; 100: 406-414.
11. Ho VT and Soiffer RJ. The history and future of T-cell depletion as graft-versus-host disease prophylaxis for allogeneic hematopoietic stem cell transplantation. Blood. 2001; 98: 3192-3204.
12. Rogosheske JR, Fargen AD, DeFor TE, Warlick E, Arora M, Blazar BR, Weisdorf DJ and Brunstein CG. Higher therapeutic CsA levels early post transplantation reduce risk of acute GVHD and improves survival. Bone Marrow Transplant. 2014; 49: 122-125.
13. Mukherjee S and Mukherjee U. A comprehensive review of immunosuppression used for liver transplantation. J Transplant. 2009; 2009: 701464.
14. Borel JF, Feurer C, Gubler HU and Stahelin H. Biological effects of cyclosporin A: a new antilymphocytic agent. Agents Actions. 1976; 6: 468-475.
15. Ruutu T, Niederwieser D, Gratwohl A and Apperley JF. A survey of the prophylaxis and treatment of acute GVHD in Europe: a report of the European Group for Blood and Marrow, Transplantation (EBMT). Chronic Leukaemia Working Party of the EBMT. Bone Marrow Transplant. 1997; 19: 759-764.
16. Kanda Y, Hyo R, Yamashita T, Fujimaki K, Oshima K, Onoda M, Mori T, Sakura T, Tanaka M, Sakai M, Taguchi J, Kurakawa M, Maruta A, et al. Effect of blood cyclosporine concentration on the outcome of hematopoietic stem cell transplantation from an HLA-matched sibling donor. Am J Hematol. 2006; 81: 838-844.
17. Garcia Cadenas I, Valcarcel D, Martino R, Pinana JL, Barba P, Novelli S, Esquirol A, Garrido A, Saavedra S, Granell M, Moreno C, Briones J, Brunet S, et al. Impact of cyclosporine levels on the development of acute graft versus host disease after reduced intensity conditioning allogeneic stem cell transplantation. Mediators Inflamm. 2014; 2014: 620682.
18. Min CK. The pathophysiology of chronic graft-versus-host disease: the unveiling of an enigma. Korean J Hematol. 2011; 46: 80-87.
19. Reddy P. Pathophysiology of acute graft-versus-host disease. Hematol Oncol. 2003; 21: 149-161.
20. Ferrara JL, Cooke KR and Teshima T. The pathophysiology of acute graft-versus-host disease. Int J Hematol. 2003; 78: 181-187.
21. Rieger K, Loddenkemper C, Maul J, Fietz T, Wolff D, Terpe H, Steiner B, Berg E, Miehlke S, Bornhauser M, Schneider T, Zeitz M, Stein H, et al. Mucosal FOXP3+ regulatory T cells are numerically deficient in acute and chronic GvHD. Blood. 2006; 107: 1717-1723.
22. Anderson BE, McNiff JM, Matte C, Athanasiadis I, Shlomchik WD and Shlomchik MJ. Recipient CD4+ T cells that survive irradiation regulate chronic graft-versus-host disease. Blood. 2004; 104: 1565-1573.
23. Zorn E, Kim HT, Lee SJ, Floyd BH, Litsa D, Arumugarajah S, Bellucci R, Alyea EP, Antin JH, Soiffer RJ and Ritz J. Reduced frequency of FOXP3+ CD4+CD25+ regulatory T cells in patients with chronic graft-versus-host disease. Blood. 2005; 106: 2903-2911.
24. Miroux C, Morales O, Carpentier A, Dharancy S, Conti F, Boleslowski E, Podevin P, Auriault C, Pancre V and Delhem N. Inhibitory effects of cyclosporine on human regulatory T cells in vitro. Transplant Proc. 2009; 41: 3371-3374.
25. Zeiser R, Nguyen VH, Beilhack A, Buess M, Schulz S, Baker J, Contag CH and Negrin RS. Inhibition of CD4+CD25+ regulatory T-cell function by calcineurin-dependent interleukin-2 production. Blood. 2006; 108: 390-399.
26. Kawai M, Kitade H, Mathieu C, Waer M and Pirenne J. Inhibitory and stimulatory effects of cyclosporine A on the development of regulatory T cells in vivo. Transplantation. 2005; 79: 1073-1077.
27. Wang H, Zhao L, Sun Z, Sun L, Zhang B and Zhao Y. A potential side effect of cyclosporin A: inhibition of CD4(+)CD25(+) regulatory T cells in mice. Transplantation. 2006; 82: 1484-1492.
28. San Segundo D, Ruiz JC, Fernandez-Fresnedo G, Izquierdo M, Gomez-Alamillo C, Cacho E, Benito MJ, Rodrigo E, Palomar R, Lopez-Hoyos M and Arias M. Calcineurin inhibitors affect circulating regulatory T cells in stable renal transplant recipients. Transplant Proc. 2006; 38: 2391-2393.
29. Kang HG, Zhang D, Degauque N, Mariat C, Alexopoulos S and Zheng XX. Effects of cyclosporine on transplant tolerance: the role of IL-2. Am J Transplant. 2007; 7: 1907-1916.
30. Miroux C, Morales O, Ghazal K, Othman SB, de Launoit Y, Pancre V, Conti F and Delhem N. In vitro effects of cyclosporine A and tacrolimus on regulatory T-cell proliferation and function. Transplantation. 2012; 94: 123-131.
31. Dai Z, Konieczny BT, Baddoura FK and Lakkis FG. Impaired alloantigen-mediated T cell apoptosis and failure to induce long-term allograft survival in IL-2-deficient mice. J Immunol. 1998; 161: 1659-1663.
32. Lai KN, Leung JC and Lai FM. Cyclosporine A inhibits lymphocyte activation at more than one site in vivo. Transplant Proc. 1989; 21: 878-880.
33. Furtado GC, Curotto de Lafaille MA, Kutchukhidze N and Lafaille JJ. Interleukin 2 signaling is required for CD4(+) regulatory T cell function. J Exp Med. 2002; 196: 851-857.
34. Malek TR, Yu A, Vincek V, Scibelli P and Kong L. CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rbeta-deficient mice. Implications for the nonredundant function of IL-2. Immunity. 2002; 17: 167-178.
35. Klebb G, Autenrieth IB, Haber H, Gillert E, Sadlack B, Smith KA and Horak I. Interleukin-2 is indispensable for development of immunological self-tolerance. Clin Immunol Immunopathol. 1996; 81: 282-286.
36. Bensinger SJ, Walsh PT, Zhang J, Carroll M, Parsons R, Rathmell JC, Thompson CB, Burchill MA, Farrar MA and Turka LA. Distinct IL-2 receptor signaling pattern in CD4+CD25+ regulatory T cells. J Immunol. 2004; 172: 5287-5296.
37. Ratanatharathorn V, Deol A, Ayash L, Cronin S, Bhutani D, Lum LG, Abidi M, Ventimiglia M, Mellert K and Uberti JP. Low-dose antithymocyte globulin enhanced the efficacy of tacrolimus and mycophenolate for GVHD prophylaxis in recipients of unrelated SCT. Bone Marrow Transplant. 2015; 50: 106-112.
38. Bacigalupo A, Lamparelli T, Barisione G, Bruzzi P, Guidi S, Alessandrino PE, di Bartolomeo P, Oneto R, Bruno B, Sacchi N, van Lint MT and Bosi A. Thymoglobulin prevents chronic graft-versus-host disease, chronic lung dysfunction, and late transplant-related mortality: long-term follow-up of a randomized trial in patients undergoing unrelated donor transplantation. Biol Blood Marrow Transplant. 2006; 12: 560-565.
39. Bonifazi F, Bandini G, Rondelli D, Falcioni S, Stanzani M, Bontadini A, Tazzari PL, Arpinati M, Giannini B, Conte R and Baccarani M. Reduced incidence of GVHD without increase in relapse with low-dose rabbit ATG in the preparative regimen for unrelated bone marrow transplants in CML. Bone Marrow Transplant. 2003; 32: 237-242.
40. Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, Martin P, Chien J, Przepiorka D, Couriel D, Cowen EW, Dinndorf P, Farrell A, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005; 11: 945-956.

