INTRODUCTION
Incontrovertibly and unfortunately, lung cancer is the most frequent reason of cancer-related deaths all over the world [1]. It is roughly estimated that there are 1.83 million new lung cancer cases and 1.59 million deaths annually around the world [2]. Approximately 83% of lung cancer patients are non-small cell lung cancer (NSCLC) patients, which 21% of those are alive at five years [3]. More powerful methods of diagnosis and treatment are indispensable to need for lung cancer patients.
Cancer stem cells (CSCs) could divide to produce heterogeneous lineages of cancer cells and new stem cells [4], which are making up a minority portion of the solid tumors, resisting to chemotherapy and radiation, correlating with targeted drug resistance and organ metastasis [5, 6]. This notion that tumors are maintained by their own stem cells has brought about novel directions to reveal the mechanisms of occurrence, progression, drug resistance, and metastasis of tumors and further seek for effective treatments of tumors. CD133 antigen, also known as prominin-1, is a member of pentaspan transmembrane glycoproteins specifically locating to cellular protrusions [7, 8]. It has been used extensively as a biomarker of CSCs in different types of cancers, such as hepatic cancer, gallbladder cancer, breast cancer, gastric cancer, pancreatic cancer and lung cancer [9–14].
Racial difference strongly affects the molecular characteristics of lung cancer [15]. Epidermal growth factor receptor mutations (mEGFR) and kirsten rat sarcoma viral oncogene mutations (mKRAS) are the most common mutations in lung cancer [16]. Alternatively, mEGFR and mKRAS usually do not occur in the same individual and have a significant association with race. For instance, Asian population have more frequently mEGFR but Caucasian population have more frequently mKRAS [17, 18]. Furthermore, it has been demonstrated that CD133 overexpressed in gefitinib-resistant tumors (GRTs) of EGFR-mutant NSCLC [19]. Therefore, we speculate that the prognostic value of CD133 in NSCLC patients might depend on given race because of various molecular characteristics.
Previous several studies about the prognostic value of CD133 in NSCLC patients suggested that NSCLC patients with higher CD133 expression have shorter overall survival (OS) time [9, 20–26] and disease free survival (DFS) [27, 28] time. On the contrary, several studies indicated that the expression level of CD133 was no association with OS and DFS [20, 29–35]. Additional, the relationship between CD133 and clinicopathological features was also in dispute [9, 20–22, 24–27, 29, 31, 33, 35–48], such as age, gender, smoking history, T stage, lymph node metastasis, distant metastasis, TNM stage, differentiation grade, and histological type. Wu. H et al. [49] and Wang. W et al. [50] have performed a meta-analysis on the prognostic value of CD133 expression in NSCLC patients, respectively. However, only 23 studies and 13 studies were included in their meta-analysis published in 2014, respectively. Additionally, several reduplicative articles (Okudela. K [28] and Woo. T [51]; Wei. YP [24] and Zhang. HZ [52]) in their meta-analysis which may limit the reliability of conclusion. Furthermore, their studies did not clarify the source of significant heterogeneity with sufficient subgroup analysis and sensitive analysis.
We performed this meta-analysis comprehensively to obtain further evidence that the biomarker of CSCs CD133 expression level may be associated with the prognosis of NSCLC patients and try to demonstrate our speculation that the prognostic value of CD133 in NSCLC patients might depend on given race for various molecular characteristics. Further, it may provide supportive evidence for the association between the cancer stem cells and the drive gene mutations of lung cancer in clinical trials and broaden new therapeutic strategy of NSCLC.
RESULTS
Eligible studies
We used the PRISMA 2009 flow diagram to screen the literature in Figure 1 [53]. A total of 1091 literature was identified through original searching from PubMed, Embase, and Web of Science. In total, 1009 Irrelevant and duplicate records were excluded through title review by two author independently (Engeng Chen and Zhiru Zeng). After that, we sorted the left literature through abstract review with double check and excluded 47 literature of meeting reports and reviews. Then we assessed the full text in the left thirty-five articles, and abandoned three articles that the sample data were reduplicate or insufficient. At last, 32 studies with 3595 participants were eligible in this meta-analysis.
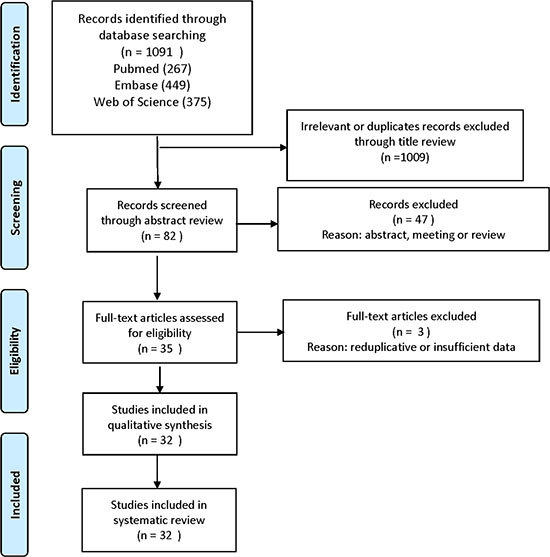
Figure 1: Flow diagram of study selection.
Study characteristics and quality assessment
The main characteristics of eligible studies were summarized in Table 1. The publication year was from 2008 to 2015. The race of study population was determined by its country. 18 Chinese studies and 4 Japanese studies composed East Asian ethnicity, and the Caucasian were from 3 Italian studies, 3 German studies, 1 Swiss study, 1 Czech study, 1 Australian study, 1 American study. Approximately 2412 male and 1183 female composed 3595 NSCLC patients in this meta-analysis, with the mean/median age range from 59 to 74.2. We defined overall survival (OS) and disease free survival (DFS) as primary endpoints. 15 studies [9, 20–26, 30–32, 42, 44, 54, 55] and 10 studies [20, 27–35] contained OS and DFS, respectively. Most of studies (29/32) used immunological histological chemistry (IHC) as experimental method for detecting CD133, and the left studies (3/32) chose quantitative real-time polymerase chain reaction (Q-PCR).
Table 1: Characteristics of eligible studies in the meta-analysis
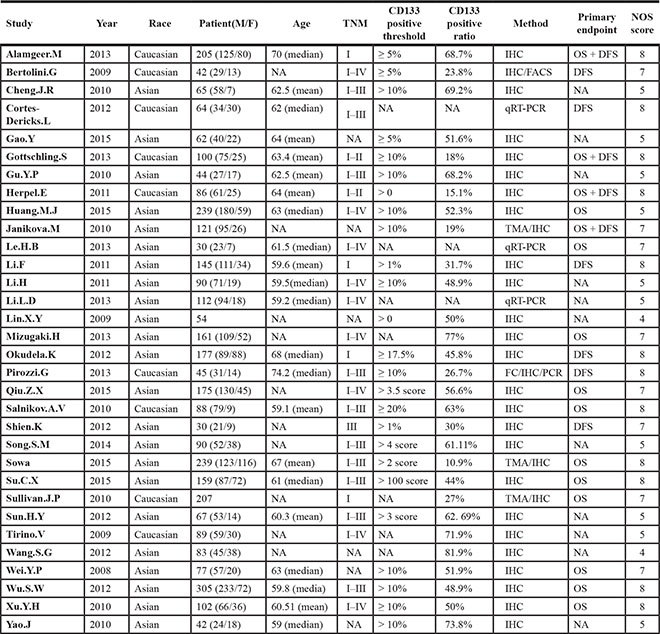
The quality of studies were assessed by Newcastle-Ottawa Quality Assessment Scale (NOS) [56]. 62.5% (20/32) of studies were more than 6 score which were deemed as high quality studies (see Supplementary Table S1 in Supplementary Material).
Association between CD133 and OS
Random-effects model was used to analyze the HRs of OS from 15 eligible studies because of significant heterogeneity (I2 = 83.7%, p < 0.001). NSCLC patients with higher CD133 expression showed a shorter OS time (HR = 1.98, 95% CI: 1.30–3.02, p = 0.002; I2 = 83.7%) (Figure 2A). Subgroup analysis indicated that both race and sample size were contributed to substantial heterogeneity. The subgroup of Caucasian from 6 studies was contributed to tiny heterogeneity (I2 = 0%, p = 0.426; HR = 1.15, 95% CI: 0.88–1.52, p = 0.307), while the subgroup of Asian from 9 studies was contributed to subtotal heterogeneity (I2 = 82.7%, p < 0.001; HR = 2.59, 95% CI: 1.58–4.25, p < 0.001). (Figure 3A). In consideration of significant heterogeneity in the subgroup of Asian, we continued to divide the 9 Asian studies into groups by sample size. The pooled HR of studies with large sample size (n > 100) was 2.83 (95% CI: 1.63–4.90, p < 0.001; I2 = 85.5%, p < 0.001) (Figure 3C). Sensitive analysis in Asian studies with large sample size showed that whatever study was removed, the result was stable as before (see Supplementary Figure S1 in Supplementary Material). Furthermore, the heterogeneity decreased (I2 = 32%, p = 0.196) after dropped out one study (Su. C.X 2015) (HR = 3.80, 95% CI: 3.12–4.04, p < 0.001) (Figure 3D). These results suggested that NSCLC patients with higher CD133 expression had poor prognosis only in Asian patients but not in Caucasian patients, which was quite different from the conclusion of Wang. W et al. [50]. Additionally, the subgroup analysis on OS by sample size showed studies with large sample size (n > 100) were associated with OS (HR = 2.45, 95% CI: 1.45–4.03, p = 0.001; I2 = 87.4%, p < 0.001) but not studies with small sample size (≤ 100) (HR = 1.28, 95% CI: 0.71–2.29, p = 0.415; I2 = 40.8%, p = 0.149) (Figure 3B), and neither Wu. H et al. [24] nor Wang. W et al. [25] analyzed this in their studies.
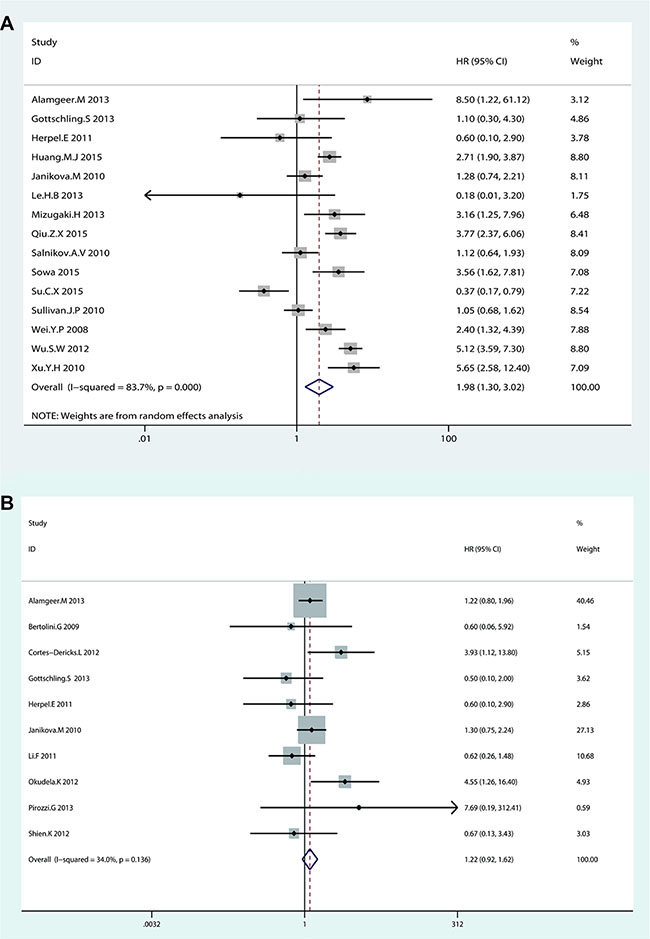
Figure 2: Forest plot of HRs for the association of CD133 expression in NSCLC patients with (A) OS and (B) DFS.
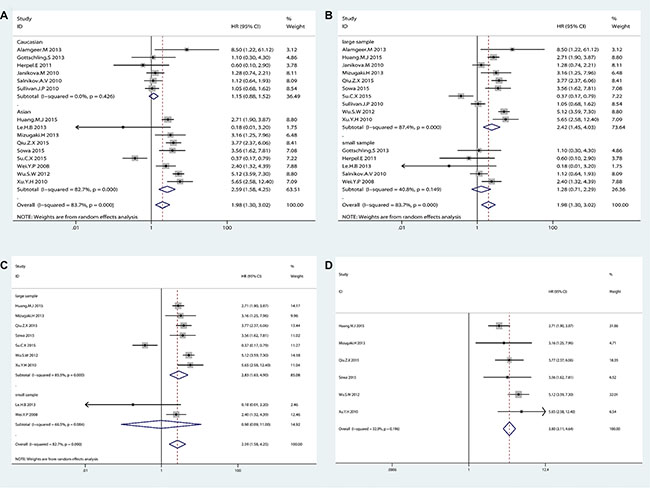
Figure 3: The subgroup analysis exploring the significant heterogeneity of CD133 expression with (A) OS by racial classification in NSCLC patients (B) OS by sample size classification in NSCLC patients (C) OS by sample size classification in Asian patients (D) OS after removed one study in Asian patients with large sample size.
Association between CD133 and DFS
Fixed-effects model was used to analyze the HRs of DFS from 10 eligible studies for tiny heterogeneity (I2 = 34%, p = 0.136). No significant association was found between CD133 expression level and DFS in NSCLC patients (HR = 1.22, 95% CI: 0.92–1.62, p = 0.173) (Figure 2B). Though the heterogeneity was no significant (I2 = 34%, p = 0.136), the subgroup analysis by race and sample size were still performed. The results showed that there was no significant association between CD133 expression level and DFS in NSCLC patients by dividing race and sample into groups (see Supplementary Figure S2 in Supplementary Material).
Association between CD133 and clinicopathological features
The frequency distributions of clinicopathological features in NSCLC patients with negative and positive expression of CD133 were summarized in Table 2. The pooled ORs of CD133 expression level and clinicopathological features were summarized in Figure 4. There were no associations between CD133 expression level and age, gender, smoking history, T stage, distant metastasis or TNM stage (detailed forest plot figures see Supplementary Figure S3–S8 in Supplementary Material). However, higher CD133 expression level was associated with poor/moderate differentiation (OR = 2.03, 95% CI: 1.32–3.14, p = 0.001; I2 = 54.8%, p = 0.007), lymph node metastasis (OR = 2.39, 95% CI: 1.62–3.52, p < 0.001; I2 = 61.5%, p = 0.001) and histological type (OR = 1.21, 95% CI: 1.01–1.46, p = 0.041; I2 = 21.8%, p = 0.18) (detailed forest plot figures see Supplementary Figure S9–S11 in Supplementary Material).
Table 2: The frequency distribution of clinicopathological features in NSCLC patients with negative and positive expression of CD133
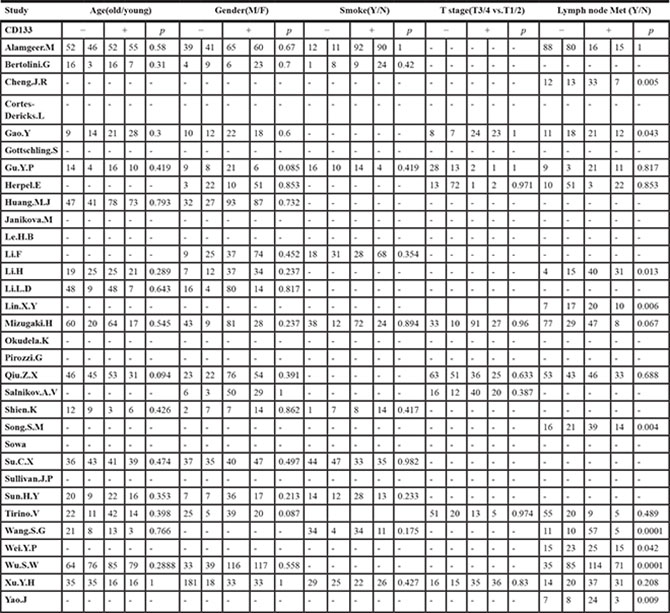
Table 2 (Continued): The frequency distribution of clinicopathological features in NSCLC patients with negative and positive expression of CD133
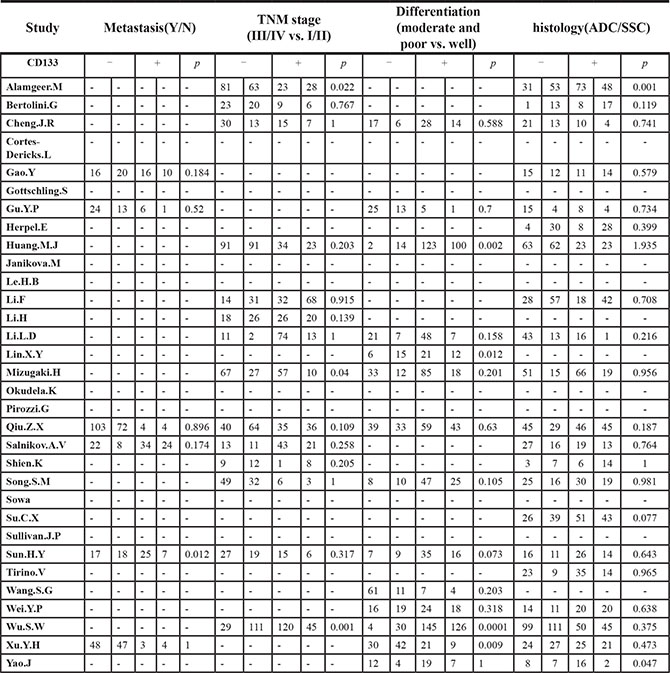
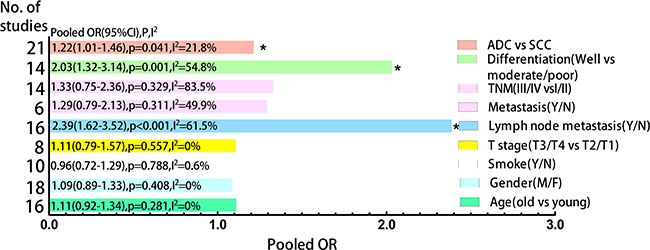
Figure 4: Clustered bars of pooled ORs for the association of CD133 expression with clinicopathological features in NSCLC patients.
We performed subgroup analysis regularly. Concerning differentiation, subgroup analysis by sample size but not race was performed due to all eligible studies were Asian. Large sample size group (n > 100) was contributed to the main heterogeneity (I2 = 66.0%, p = 0.012) with significant association (OR = 2.86, 95% CI: 1.46–5.58, p = 0.002) but not small sample size group (OR = 1.51, 95% CI: 0.85–2.68, p = 0.162; I2 = 42.9%, p = 0.092) (Figure 5A). As for lymph node metastasis, subgroup analysis by sample size could not explain the source of heterogeneity but race could (see Supplementary Figure S12 in Supplementary Material). The subgroup analysis by race showed that Asian group was contributed to a large proportion of heterogeneity (I2 = 52.7%, p = 0.013) with significant association (OR = 2.97, 95% CI: 2.03–4.34, p < 0.001) compared with Caucasian group (OR = 0.87, 95% CI: 0.48–1.56, p = 0.638; I2 = 0%, p = 0.768) (Figure 5B). These results suggested that NSCLC patients with higher CD133 expression had poor prognosis only in Asian patients but not in Caucasian patients.
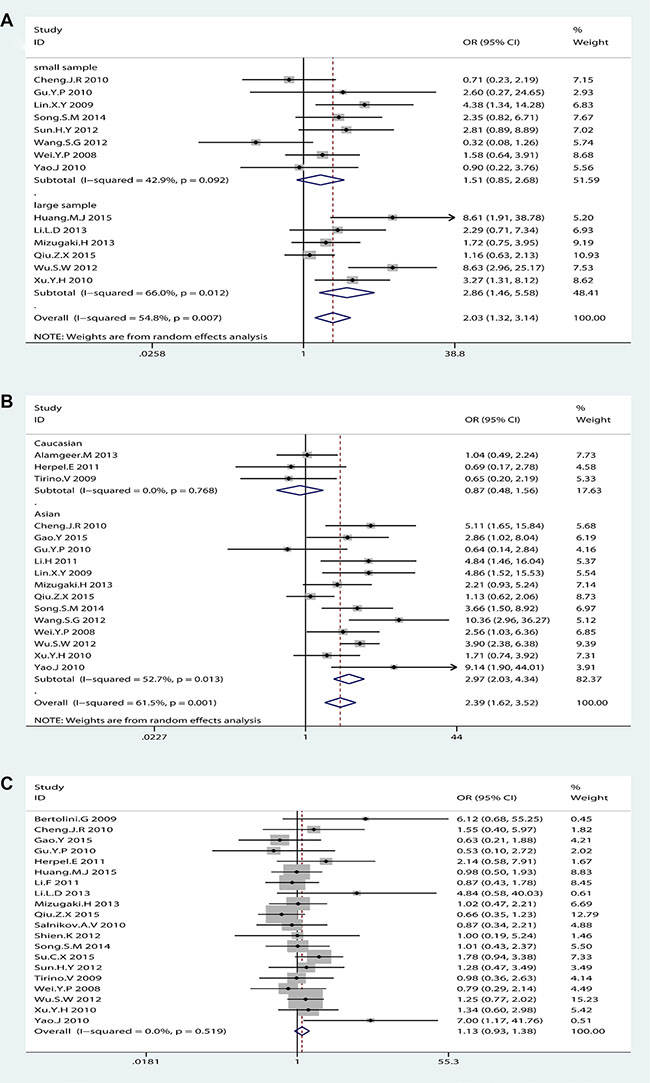
Figure 5: The subgroup analysis exploring the significant heterogeneity of CD133 expression with (A) differentiation by sample size (B) lymph node metastasis by race (C) histological type (adenocarcinoma vs. Squamous-cell carcinoma) after removed one study.
It seemed that the expression of CD133 in lung adenocarcinoma patients (ADC) was more than in lung squamous-cell carcinoma (SCC) patients, which was in agreement with Wang. W et al. [25]. However, sensitive analysis showed that the OR of the association between CD133 expression and histological type (ADC vs. SCC) in NSCLC patients was dramatically changed after removed one study (Alamgeer.M 2013) (OR = 1.13, 95% CI: 0.93–1.38, p = 0.3; I2 = 0%, p = 0.522) (Figure 5C). Thus, it could not come to a conclusion that there was significant difference of CD133 expression level between ADC and SCC in NSCLC patients, which was different from Wang. W et al. [25].
Sensitive analysis and publication bias
Sensitive analysis showed that regardless of which one study removed, pooled HRs of left studies on OS and DFS were remain robust and stable (see Supplementary Figure S13 in Supplementary Material). Begg’s funnel plot and Egger’s publication bias plot were used to evaluate to the publication bias on OS (Figure 6A) and DFS (Figure 6B), respectively. No publication bias evidence was found in OS (Begg’s test: p = 0.621; Egger’s test: p = 0.318) or DFS (Begg’s test: p = 0.858; Egger’s test: p = 0.926). Same as in clinicopathological features (see Supplementary Table S2 in Supplementary Material).
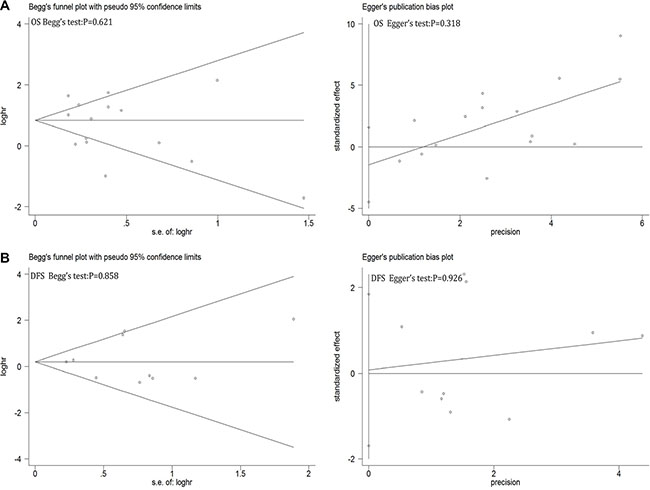
Figure 6: Begg’s funnel plot and Egger’s test to evaluate the publication bias for (A) OS and (B) DFS.
DISCUSSION
Mainly benefit from tobacco control and improvements in early detection and treatment, mortality rates decreased for lung cancer by 45% and 8% since from 1990 to 2015 in men and women, respectively [57]. However, only a small proportion of lung cancers are currently detected early [57], and more effective methods are needed to reduce the morbidity and mortality of lung cancer.
The CSCs hypothesis elucidates that a small proportion of tumor cells drive the cancer growth, progression and recurrence [58], which is different from the classical stochastic hypothesis [59]. In a landmark experiment, Singh SK and his colleagues showed that injection of as few as 100 CD133+ tumor cells were tumorigenic but injection of 105 CD133− tumor cells were not, giving stable foundation for CSCs hypothesis in many solid tumors [60]. Recent studies showed that CD133 was a biomarker of putative CSCs in many solid tumors from brain [60] [61], lung [62, 63], liver [64], pancreas [65] [66] and colon [67–70]. However, controversies remain exist when referring to the prognostic value of CD133 in solid tumors [9, 20–48, 54, 55, 71].
In this meta-analysis, we tried to elucidate the potential prognostic and clinical value of CD133 by systematically reviewing and analyzing 32 eligible literature. Interestingly and notably particularly, we found that NSCLC patients with higher CD133 expression have shorter overall survival time only in Asian patients but not in Caucasian patients. It remains unknown why racial difference causes this significant difference. Recent studies showed that EGFR and EGFRvIII signaling are concerned with maintaining a CSCs phenotype [72]. The EGFR positive CSCs represented enhanced tumorigenic potential and highly invasive behavior whereas EGFR negative CSCs reduced their tumorigenic ability [73]. Furthermore, Mitsudomi et al. reported that the EGFR mutation rate was 32% in patients of East Asian compared with 7% in patients of non-Asian [74]. Probably as a consequence, we speculated that difference of mEGFR of CD133+ CSCs in different racial NSCLC patients might be the potential mechanism causing the significant difference on OS. Here to yonder, we speculated that only the intrinsic EGFR gene status of CSCs could predict the efficacy of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) in NSCLC patients, which are effective target drugs for NSCLC patients with EGFR mutations. But so far, general method for detecting EGFR mutations in lung cancer is direct sequencing with a low sensitivity, which could not uncover the EGFR gene status of tumor factually [75]. Therefore, detecting EGFR gene status after identification and isolation of CSCs using CD133 in NSCLC patients might be preferable strategy for choosing EGFR-TKIs.
Certain limitations in our study might influence the results. Firstly, these eligible studies were incorporated with varying TNM stage. Secondly, detection methods and threshold value of CD133 expression level were not consistent. Thirdly, though we performed subgroup analysis to explore the significant heterogeneity and further stabilized and consolidated our results that NSCLC patients with higher CD133 expression had poor overall survival time only in Asian patients but not in Caucasian patients, we could not explain fully the potential heterogeneity on differentiated degree and lymph node metastasis. Fourthly, relevant data in several eligible studies were too limited to pool all studies for evaluating the association between CD133 expression level and these parameters, which might overrate the clinical value of CD133.
Therefore, added large-scale sample, high-quality, and interethnic studies will be required to confirm the prognostic and clinical value of CD133. Far more than, the association between CD133+ CSCs and EGFR mutation in NSCLC patients is further deserving of attention and exploration, which may provide a new therapeutic perspective on the treatment of NSCLC patients according to the expression of CD133 and the intrinsic EGFR gene status of CD133+ CSCs.
MATERIALS AND METHODS
Search strategy
We searched PubMed, Embase, and Web of science to confirm relevant studies on CD133 expression level in NSCLC patients from each database since its inception up to May 4, 2016 without language restriction by using the keywords of CD133 and lung cancer (detail search strategy see Supplementary Material).
Inclusion and exclusion criteria
A study was selected when met the following criteria: (1) the study population were mainly NSCLC patients; (2) it investigated the prognostic role of CD133 with the survival outcomes and/or clinicopathological characteristics in NSCLC patients. The exclusion criteria: (1) meeting report, review, comment, or letter; (2) it was a reduplicative study whose data had been published in another study, and then left the complete one in this meta-analysis. Independently evaluations were performed by two authors (Engeng Chen and Zhiru Zeng) according to the inclusion and exclusion criteria.
Data extraction
A double abstraction process was performed for data extraction (Engeng Chen and Zhiru Zeng). Disagreements were resolved by consulting the third author (Bingjun Bai). The following data were collected from eligible studies: the first author, publication year, race, number of NSCLC patients with CD133 measured, gender distribution, age, TNM stage, CD133 positive threshold, CD133 positive ratio, experimental method, primary outcomes (reported HR with its 95% CI on OS and DFS), and essential clinicopathological characteristics (T stage, N stage, M stage, TNM stage, smoking history, differentiation grade, and histological type).
Quality assessment of eligible studies
The Newcastle-Ottawa Quality Assessment Scale (NOS) [56] was used to evaluate the quality of each eligible study by two authors (Engeng Chen and Zhiru Zeng) independently. This scale ranges from 0 to 9 score, and we consider the study as a high quality study if the score is not less than 6.
Statistical analysis
The main purpose of this meta-analysis was to estimate the pooled HRs of OS and DFS, then to validate the hypotheses: that NSCLC patients with higher CD133 expression would have a shorter OS and DFS time. The secondary purpose was to estimate the pooled ORs to analyze the correlation between CD133 expression level and clinicopathological features, with the doubts: that is there any cause-and-effect relationship between CD133 and these features.
We analyzed each eligible study to obtain HR and DFS with corresponding 95% CI from the results of multivariate Cox’s proportional hazards regression model reported in the study. Also we reconstructed and calculated the data from Kaplan-Meier survival curve using Engauge-Digitizer version 7.2 if there was no direct data in the study [76]. The ORs with corresponding 95% CIs were calculated according to the relevant parameters using chi-square test by SPSS version 21 (SPSS Inc. Chicago, USA) in eligible studies.
The following analyses were performed using Stata version 12 software (Stata Corporation, College Station, Texas, USA). Pooled HRs of OS and DFS and pooled ORs for the relationship between CD133 and clinicopathological features were calculated by using fixed-effects model if I-square < 50%. Additionally, we used the Cochran’s Q-test and I-square statistics to test for between-study heterogeneity [77–78]. Instead of fixed-effects, random-effects model was used if I-square > 50% or corresponding p value < 0.05. Furthermore, subgroup analysis and sensitive analysis were applied to assess the source of heterogeneity. The potential publication bias was tested by using Begg’s test and Egger’s test [79–80]. All statistics p-value < 0.05 at two-tailed was considered statistically significant.
CONCLUSIONS
In summary, this meta-analysis showed that high expression level of CSCs marker CD133 was strongly in correlation with poor OS but not DFS in NSCLC patients. Subgroup analysis by race showed that NSCLC patients with higher CD133 expression had shorter overall survival time only in Asian patients but not in Caucasian patients, suggesting that differential prognostic value of CD133 expression in distinct ethnic group. Additionally, higher expression of CD133 was associated with poor differentiation and lymph node metastasis but there was no significant difference of CD133 expression between ADC and SCC in NSCLC patients. Therefore, added large-scale, prospective and clinical studies are required to further validate the prognostic and clinical value of CSCs marker CD133.
ACKNOWLEDGMENTS AND FUNDING
This work is supported by grants from the National Natural Science Foundation of China (No.81370461).
CONFLICTS OF INTEREST
No conflicts of interest was declared.
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7–30.
2. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108.
3. American Cancer Society. Cancer Facts & Figures 2016. Atlanta, American Cancer Society. 2016.
4. Kaiser J. The cancer stem cell gamble. Science. 2015; 347:226–229.
5. Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, Visvader J, Weissman IL, Wahl GM. Cancer stem cells—perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res. 2006; 66:9339–9344.
6. Marx J. Molecular biology. Cancer's perpetual source? Science. 2007; 317:1029–1031.
7. Yin AH, Miraglia S, Zanjani ED, Almeida-Porada G, Ogawa M, Leary AG, Olweus J, Kearney J, Buck DW. AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood. 1997; 90:5002–5012.
8. Corbeil D, Fargeas CA, Huttner WB. Rat prominin, like its mouse and human orthologues, is a pentaspan membrane glycoprotein. Biochem Biophys Res Commun. 2001; 285:939–944.
9. Qiu ZX, Zhao S, Mo XM, Li WM. Overexpression of PROM1 (CD133) confers poor prognosis in non-small cell lung cancer. Int J Clin Exp Pathol. 2015; 8:6589.
10. Cheung PF, Cheung TT, Yip CW, Ng LW, Fung SW, Lo CM, Fan ST, Cheung ST. Hepatic cancer stem cell marker granulin-epithelin precursor and beta-catenin expression associate with recurrence in hepatocellular carcinoma. Oncotarget. 2016; 7:21644–57. doi: 10.18632/oncotarget.7803.
11. Li C, Wang C, Xing Y, Zhen J, Ai Z. CD133 promotes gallbladder carcinoma cell migration through activating Akt phosphorylation. Oncotarget. 2016; 7:17751–9. doi: 10.18632/oncotarget.7474.
12. Kim D, Jung J, You E, Ko P, Oh S, Rhee S. mDia1 regulates breast cancer invasion by controlling membrane type 1-matrix metalloproteinase localization. Oncotarget. 2016; 7:17829–43. doi: 10.18632/oncotarget.7429.
13. Zhang X, Hua R, Wang X, Huang M, Gan L, Wu Z, Zhang J, Wang H, Cheng Y, Li J, Guo W. Identification of stem-like cells and clinical significance of candidate stem cell markers in gastric cancer. Oncotarget. 2016; 7:9815–9831. doi: 10.18632/oncotarget.6890.
14. Nomura A, Banerjee S, Chugh R, Dudeja V, Yamamoto M, Vickers SM, Saluja AK. CD133 initiates tumors, induces epithelial-mesenchymal transition and increases metastasis in pancreatic cancer. Oncotarget. 2015; 6:8313–8322. doi: 10.18632/oncotarget.3228.
15. Soh J, Toyooka S, Matsuo K, Yamamoto H, Wistuba, II, Lam S, Fong KM, Gazdar AF, Miyoshi S. Ethnicity affects and gene alterations of lung adenocarcinoma. Oncol Lett. 2015; 10:1775–1782.
16. Pao W, Girard N. New driver mutations in non-small-cell lung cancer. Lancet Oncol. 2011; 12:175–180.
17. Shigematsu H, Gazdar AF. Somatic mutations of epidermal growth factor receptor signaling pathway in lung cancers. Int J Cancer. 2005; 118:257–262.
18. Suda K, Tomizawa K, Mitsudomi T. Biological and clinical significance of KRAS mutations in lung cancer: an oncogenic driver that contrasts with EGFR mutation. Cancer Metastasis Rev. 2010; 29:49–60.
19. Kobayashi I, Takahashi F, Nurwidya F, Nara T, Hashimoto M, Murakami A, Yagishita S, Tajima K, Hidayat M, Shimada N, Suina K, Yoshioka Y, Sasaki S, et al. Oct4 plays a crucial role in the maintenance of gefitinib-resistant lung cancer stem cells. Biochem Biophys Res Commun. 2016; 473:125–132.
20. Alamgeer M, Ganju V, Szczepny A, Russell PA, Prodanovic Z, Kumar B, Wainer Z, Brown T, Schneider-Kolsky M, Conron M, Wright G, Watkins DN. The prognostic significance of aldehyde dehydrogenase 1A1 (ALDH1A1) and CD133 expression in early stage non-small cell lung cancer. Thorax. 2013; 68:1095–1104.
21. Huang M, Zhu H, Feng J, Ni S, Huang J. High CD133 expression in the nucleus and cytoplasm predicts poor prognosis in non-small cell lung cancer. Dis Markers. 2015; 2015:986095.
22. Mizugaki H, Sakakibara-Konishi J, Kikuchi J, Moriya J, Hatanaka KC, Kikuchi E, Kinoshita I, Oizumi S, Dosaka-Akita H, Matsuno Y, Nishimura M. CD133 expression: a potential prognostic marker for non-small cell lung cancers. Int J Clin Oncol. 2014; 19:254–259.
23. Sowa T, Menju T, Sonobe M, Nakanishi T, Shikuma K, Imamura N, Motoyama H, Hijiya K, Aoyama A, Chen F, Sato T, Kobayashi M, Yoshizawa A, et al. Association between epithelial-mesenchymal transition and cancer stemness and their effect on the prognosis of lung adenocarcinoma. Cancer Med. 2015; 4:1853–1862.
24. Wei Y, Wang W, Hua P. Expression of Tumor Stem Cell Marker CD133 in Non-small Cell Lung Carcinoma and Its Clinical Significance. J Sun Yat-Sen Univ Med Sci. 2008; 29:312–316.
25. Wu S, Yu L, Wang D, Zhou L, Cheng Z, Chai D, Ma L, Tao Y. Aberrant expression of CD133 in non-small cell lung cancer and its relationship to vasculogenic mimicry. BMC cancer. 2012; 12:535.
26. Xu YH, Zhang GB, Wang JM, Hu HC. B7-H3 and CD133 expression in non-small cell lung cancer and correlation with clinicopathologic factors and prognosis. Saudi Med J. 2010; 31:980–986.
27. Cortes-Dericks L, Galetta D, Spaggiari L, Schmid RA, Karoubi G. High expression of octamer-binding transcription factor 4A, prominin-1 and aldehyde dehydrogenase strongly indicates involvement in the initiation of lung adenocarcinoma resulting in shorter disease-free intervals. Eur J Cardiothorac Surg. 2012; 41:e173–181.
28. Okudela K, Woo T, Mitsui H, Tajiri M, Masuda M, Ohashi K. Expression of the potential cancer stem cell markers, CD133, CD44, ALDH1, and beta-catenin, in primary lung adenocarcinoma—their prognostic significance. Pathol Int. 2012; 62:792–801.
29. Bertolini G, Roz L, Perego P, Tortoreto M, Fontanella E, Gatti L, Pratesi G, Fabbri A, Andriani F, Tinelli S, Roz E, Caserini R, Lo Vullo S, et al. Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment. Proc Natl Acad Sci U S A. 2009; 106:16281–16286.
30. Gottschling S, Jensen K, Herth FJ, Thomas M, Schnabel PA, Herpel E. Lack of prognostic significance of neuroendocrine differentiation and stem cell antigen co-expression in resected early-stage non-small cell lung cancer. Anticancer Res. 2013; 33:981–990.
31. Herpel E, Jensen K, Muley T, Warth A, Schnabel PA, Meister M, Herth FJ, Dienemann H, Thomas M, Gottschling S. The cancer stem cell antigens CD133, BCRP1/ABCG2 and CD117/c-KIT are not associated with prognosis in resected early-stage non-small cell lung cancer. Anticancer Res. 2011; 31:4491–4500.
32. Janikova M, Skarda J, Dziechciarkova M, Radova L, Chmelova J, Krejci V, Sedlakova E, Zapletalova J, Langova K, Klein J. Identification of CD133+/nestin+ putative cancer stem cells in non-small cell lung cancer. Biomed Pap. 2010; 154:321–326.
33. Li F, Zeng H, Ying K. The combination of stem cell markers CD133 and ABCG2 predicts relapse in stage I non-small cell lung carcinomas. Med Oncol. 2011; 28:1458–1462.
34. Pirozzi G, Tirino V, Camerlingo R, La Rocca A, Martucci N, Scognamiglio G, Franco R, Cantile M, Normanno N, Rocco G. Prognostic value of cancer stem cells, epithelial-mesenchymal transition and circulating tumor cells in lung cancer. Oncol Rep. 2013; 29:1763–1768.
35. Shien K, Toyooka S, Ichimura K, Soh J, Furukawa M, Maki Y, Muraoka T, Tanaka N, Ueno T, Asano H, Tsukuda K, Yamane M, Oto T, et al. Prognostic impact of cancer stem cell-related markers in non-small cell lung cancer patients treated with induction chemoradiotherapy. Lung Cancer. 2012; 77:162–167.
36. Cheng J, Wang S, Zhou J. Expressions of CD133 and CD105 in lung cancer tissue and their clinical significance. Tumor. 2010; 30:334–337.
37. Gao Y, Feng J, Wu L, Zhan S, Sun J. Expression and pathological mechanism of MMP-9 and HIF-2α in CD133 + lung cancer stem cells. Natl Med J Chin. 2015; 95.
38. Gu Y, Sun M, Gu L, Zhang H, Xie F. Expression and Significance of Cancer Stem Cell Marker CD133, ABCG2 and p75 NTR in Non-small Cell Lung Carcinoma. Suzhou Univ J Med Sci. 2010; 30:513–516.
39. Li H, Wang Y, Yu L, Zhao B. Clinicopathological significance of expression of CD133 protein in neuroendocrine lung carcinoma tissues. Chin J Cancer Prev Treat. 2011; 18:29–31.
40. Li L, Yu H, Wang X, Zeng J, Li D, Lu J, Wang C, Wang J, Wei J, Jiang M, Mo B. Expression of seven stem-cell-associated markers in human airway biopsy specimens obtained via fiberoptic bronchoscopy. J Exp Clin Cancer Res. 2013; 32:28.
41. Lin X, Liu S, Liu N, Yang X, Xu H, Wang E. [Expression and Significance of Stem Cell Markers CK19, Notch3, CD133, P75NTR, STRO-1 and ABCG2 in Pulmonary Squamous Carcinomas.]. Zhongguo Fei Ai Za Zhi. 2009; 12:316–321.
42. Salnikov AV, Gladkich J, Moldenhauer G, Volm M, Mattern J, Herr I. CD133 is indicative for a resistance phenotype but does not represent a prognostic marker for survival of non-small cell lung cancer patients. Int J Cancer. 2010; 126:950–958.
43. Song S, He X. CD133 and CD44 expression in non-small-cell lung cancer(NSCLC)and its clinical significance. J Modern Oncol. 2014; :1318–1320.
44. Su C, Xu Y, Li X, Ren S, Zhao C, Hou L, Ye Z, Zhou C. Predictive and prognostic effect of CD133 and cancer-testis antigens in stage IB–IIIA non-small cell lung cancer. Int J Clin Exp Pathol. 2015; 8:5509.
45. Sun H, Yang M, Zheng M, Ren Z, Liu H. Expression and clinical significance of CD133 and ALDH1 in non-small cell lung cancer. Chin J Clin Exp Patho. 2012; 28:813–815.
46. Tirino V, Camerlingo R, Franco R, Malanga D, La Rocca A, Viglietto G, Rocco G, Pirozzi G. The role of CD133 in the identification and characterisation of tumour-initiating cells in non-small-cell lung cancer. Eur J Cardiothorac Surg. 2009; 36:446–453.
47. Wang S, Zeng Z, Yang S, Lin J, Yuan Y. The study of CD133 expression in 83 cases of human lung adenocarcinoma cells. J Cardio Pulm Dis. 2012; 31:727–729.
48. Yao J, Wang Z, Tong W, Li Z, Yu Z. Expression of cancer stem cell markers CD133 and CD44 in primary lung cancer and metastatic lymph nodes. Med J Natl Defending Forces Southwest China. 2010; 20:1300–1303.
49. Wu H, Qi XW, Yan GN, Zhang QB, Xu C, Bian XW. Is CD133 expression a prognostic biomarker of non-small-cell lung cancer? A systematic review and meta-analysis. PloS one. 2014; 9:e100168.
50. Wang W, Chen Y, Deng J, Zhou J, Zhou Y, Wang S, Zhou J. The prognostic value of CD133 expression in non-small cell lung cancer: a meta-analysis. Tumour Biol. 2014; 35:9769–9775.
51. Woo T, Okudela K, Mitsui H, Yazawa T, Ogawa N, Tajiri M, Yamamoto T, Rino Y, Kitamura H, Masuda M. Prognostic value of CD133 expression in stage I lung adenocarcinomas. Int J Clin Exp Med. 2009; 4:32–42.
52. Zhang H, Wei Y, Wang M, Wu C, YANG Y, Chen J, Cao Y. Association of CD133 and endothelin-converting enzyme expressions with prognosis of non-small cell lung carcinoma. J South Med Univ. 2007; 27:696–699.
53. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010; 8:336–341.
54. Le H, Zeng F, Xu L, Liu X, Huang Y. The role of CD133 expression in the carcinogenesis and prognosis of patients with lung cancer. Mol Med Rep. 2013; 8:1511–1518.
55. Sullivan JP, Spinola M, Dodge M, Raso MG, Behrens C, Gao B, Schuster K, Shao C, Larsen JE, Sullivan LA, Honorio S, Xie Y, Scaglioni PP, et al. Aldehyde dehydrogenase activity selects for lung adenocarcinoma stem cells dependent on notch signaling. Cancer Res. 2010; 70:9937–9948.
56. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009; 151:W65–94.
57. Byers T, Wender RC, Jemal A, Baskies AM, Ward EE, Brawley OW. The American Cancer Society challenge goal to reduce US cancer mortality by 50% between 1990 and 2015: Results and reflections. CA Cancer J Clin. 2016.
58. Lobo NA, Shimono Y, Qian D, Clarke MF. The biology of cancer stem cells. Annu Rev Cell Dev Biol. 2007; 23: 675–699.
59. Pardal R, Clarke MF, Morrison SJ. Applying the principles of stem-cell biology to cancer. Nat Rev Cancer. 2003; 3:895–902.
60. Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004; 432:396–401.
61. Hemmati HD, Nakano I, Lazareff JA, Masterman-Smith M, Geschwind DH, Bronner-Fraser M, Kornblum HI. Cancerous stem cells can arise from pediatric brain tumors. P Natl Acad Sci USA. 2003; 100:15178–15183.
62. Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2007; 15:504–514.
63. Ho MM, Ng AV, Lam S, Hung JY. Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res. 2007; 67:4827–4833.
64. Ma S, Chan KW, Hu L, Lee TK, Wo JY, Ng IO, Zheng BJ, Guan XY. Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology. 2007; 132:2542–2556.
65. Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V, Wicha M, Clarke MF, Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res. 2007; 67: 1030–1037.
66. Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M, Bruns CJ, Heeschen C. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2008; 1:313–323.
67. Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW, Hoey T, Gurney A, Huang EH, Simeone DM, Shelton AA, Parmiani G, Castelli C, et al. Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci U S A. 2007; 104:10158–10163.
68. Huang EH, Hynes MJ, Zhang T, Ginestier C, Dontu G, Appelman H, Fields JZ, Wicha MS, Boman BM. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res. 2009; 69:3382–3389.
69. O'Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2006; 445:106–110.
70. Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R. Identification and expansion of human colon-cancer-initiating cells. Nature. 2006; 445:111–115.
71. Zhao Y, Peng J, Zhang E, Jiang N, Li J, Zhang Q, Zhang X, Niu Y. CD133 expression may be useful as a prognostic indicator in colorectal cancer, a tool for optimizing therapy and supportive evidence for the cancer stem cell hypothesis: a meta-analysis. Oncotarget. 2016; 7:10023–10036. doi: 10.18632/oncotarget.7054.
72. Jutten B, Rouschop KM. EGFR signaling and autophagy dependence for growth, survival, and therapy resistance. Cell Cycle. 2014; 13:42–51.
73. Mazzoleni S, Politi LS, Pala M, Cominelli M, Franzin A, Sergi Sergi L, Falini A, De Palma M, Bulfone A, Poliani PL, Galli R. Epidermal growth factor receptor expression identifies functionally and molecularly distinct tumor-initiating cells in human glioblastoma multiforme and is required for gliomagenesis. Cancer Res. 2010; 70:7500–7513.
74. Mitsudomi T, Kosaka T, Yatabe Y. Biological and clinical implications of EGFR mutations in lung cancer. Int J Clin Oncol. 2006; 11:190–198.
75. Pao W, Ladanyi M. Epidermal growth factor receptor mutation testing in lung cancer: searching for the ideal method. Clin Cancer Res. 2007; 13:4954–4955.
76. Guyot P, Ades AE, Ouwens MJ, Welton NJ. Enhanced secondary analysis of survival data: reconstructing the data from published Kaplan-Meier survival curves. BMC Med Res Methodol. 2012; 12:9.
77. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560.
78. Thompson SG, Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Statist Med. 1999; 18:2693–2708.
79. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–1101.
80. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–634.

