INTRODUCTION
Oral cancer is one of the major public health problems worldwide, as well as a major cause of cancer morbidity and mortality. In the United States, approximately 30,260 new cases are estimated and 5,990 cases may die in 2015 [1]. Oral squamous cell carcinoma (SCC) is the most common type of oral cancer, which usually develops from precancerous lesions such as oral leukoplakia (OLK) and erythroplakia, and histopathologically follows a step-wise pattern of hyperplasia, dysplasia and SCC [2]. Overall survival of these patients remained unchanged despite the advances in radiotherapy and chemotherapy. The five-year survival rate for patients with early and localized lesions is ~80%, whereas it is only 19% for patients with distant metastasis [3]. Thus it is important to develop preventive strategies for this deadly disease.
Oxidative stress has long been known to play an important role in the development of human oral cancer. 8-oxo-2’-deoxyguanosine (8OHdG), a marker of DNA oxidative damage, is known to increase in OLK and SCC as compared with normal mucosa [4–6]. Major risk factors of oral cancer (tobacco, alcohol drinking, and betel nut chewing) all generate oxidative stress in the epithelial cells of oral mucosa [7]. Although antioxidants have been proposed and tested for oral cancer prevention, randomized controlled clinical trials did not provide evidence that dietary antioxidant supplements are beneficial in primary cancer prevention. A systematic review conducted for the United States Preventive Services Task Force failed to find clear benefits of vitamin and mineral supplements for the prevention of chronic diseases including cancer [8]. Thus activation of the antioxidative stress response by chemicals (indirect antioxidants) has become a reasonable and promising approach.
As a major cellular defense pathway, nuclear factor erythroid 2-like 2 (NRF2) is known to regulate expression of genes involved in detoxification and anti-oxidative stress response. NRF2 forms heterodimers with small MAF proteins and binds to the antioxidant response elements of target genes when cells are exposed to oxidative stress or xenobiotics. Kelch-like ECH-associated protein 1 (KEAP1) inhibits the function of NRF2 by retaining NRF2 in the cytoplasm under normal physiological conditions, and by allowing nuclear translocation of NRF2 under stress conditions [9]. Paradoxically, NRF2 is known to be overexpressed in human oral cancer suggesting the involvement of NRF2 in oral carcinogenesis [10, 11].
In this study, we aimed to elucidate the chemopreventive effect and mechanisms of a chemical activator of NRF2 on oxidative stress-associated oral carcinogenesis. Sulforaphane (SFN) is a compound extracted from broccoli. Its isothiocyanate moiety (-N=C=S) is highly electrophilic and reacts directly but reversibly with the sulfhydryl groups of specific cysteine residues of Keap1, especially Cys151. Disruption of KEAP1-NRF2 interactions by SFN leads to nuclear accumulation of NRF2 and up-regulation of its target genes [12]. The 4NQO-induced oral carcinogenesis model in mice was used to test the chemopreventive efficacy and mechanisms of SFN in vivo [13, 14]. Cultured human cells and human tissue samples were also employed to understand chemopreventive mechanisms and establish clinical relevance.
RESULTS
Oxidative damage and NRF2 activation in human oral leukoplakia
In OLK samples, NRF2 was activated as evidenced by its nuclear accumulation (Figure 1A, 1B). The number of epithelial cells with nuclear NRF2 was significantly higher in OLK than in the normal oral mucosa (Figure 1C). As a marker of oxidative DNA damage, 8OHdG was increased in OLK (Figure 1D, 1E).
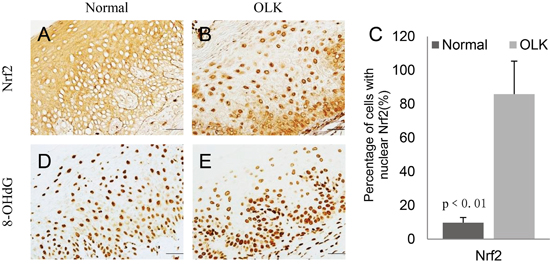
Figure 1: Increased expression of Nrf2 and 8OHdG in the nuclei of oral epithelial cells in OLK patients as compared with normal subjects. A-D. IHC staining of NRF2 and 8OHdG in normal and OLK samples; E. Quantitation showed a significant increase of the number of epithelial cells with nuclear NRF2 in OLK samples as compare with normal samples.
4NQO produces oxidative DNA damage and gene mutations, and activates the Nrf2 pathway in mouse tongue
With gene microarray analysis and a stringent data analysis method, we found 32 genes up-regulated and one gene down-regulated in mouse tongue due to 4NQO treatment (100μg/ml in drink for 8 weeks) as compared to control. Among these 33 genes, 9 are known Nrf2-regulated genes (Slc7a11, Akr1b8, Gsta1/a2, Gclc, Ptgr1, Gsta4, Gsta3, Gpx2, Ftl1/Ftl2). Several other genes (Brca1, Mcm5, Mcm3, Slc2a1, Clspn, Ucp3) are functionally associated with NRF2 or oxidative stress response [15–21] (Suppl. Excel 1). GSA_CP analysis showed that 58 canonical pathways were enriched in 4NQO-treated samples. These pathways mainly belonged to three categories: (1) 10 gene sets associated with DNA damage repair, such as nuclear excision repair, mismatch repair, DNA repair, p53 signaling pathway, ATM pathway; (2) 28 gene sets associated with cell cycle and DNA replication; (3) 6 gene sets associated with stress response and detoxification, such as glutathione metabolism, metabolism of xenobiotics, drug metabolism, biological oxidations, glutathione conjugation, Phase II conjugations (Suppl. Excel 2). In addition, an NRF2-regulated gene set, was also enriched in 4NQO-treated samples (Suppl. Excel 3).
IHC was performed to compare expression of 8OHdG, NRF2, and four classical NRF2-regulated genes (NQO1, HO1, MT1 and GCLC) in 4NQO-treated tongue and control tongue. All these markers were dramatically overexpressed in the 4NQO-treated tongue epithelial cells (Figure 2). We further analyzed DNA mutations in 4NQO-treated tongue using whole exome sequencing. Single nucleotide variants (SNVs) mainly took place at the G nucleotide. In sample 4NQO1, 28 out of 75 single SNVs took place at the G nucleotides. In sample 4NQO2, 54 out of 153 SNVs took place at the G nucleotides (Suppl. Excel 4). The preference for mutations at the G nucleotide is consistent with a recent whole genome sequencing study on the mutagenic spectrum of 4NQO in Aspergillus nidulans [22]. These data clearly demonstrated that 4NQO treatment results in oxidative damage, DNA mutations and Nrf2 activation in squamous epithelial cells of mouse tongue, and also suggested that the 4NQO-induced oral carcinogenesis model in mice is a relevant model system for studies on oxidative stress-associated oral cancer.
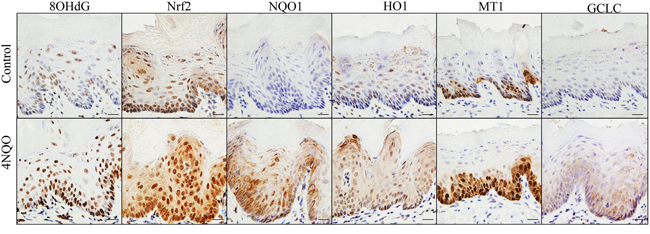
Figure 2: 4NQO treatment enhances oxidative DNA damage (8OHdG) and up-regulates expression of NRF2 and its target genes in mouse tongue (NQO1, HO1, MT1 and GCLC).
SFN activates Nrf2 in oral squamous epithelial cells and prevents 4NQO-induced oxidative damage in vitro and in vivo
SFN was examined for its activating effect on Nrf2 and inhibitory effect on oxidative DNA damage in vitro and in vivo. SFN treatment of DOK cells not only increased NRF2 expression, but also its nuclear accumulation (Figure 3A). Western blotting confirmed that SFN up-regulated the expression of NRF2 and its target genes (HO1 and NQO1) in a dose-dependent manner (Figure 3B). More importantly, treatment of DOK cells with SFN prior to 4NQO treatment prevented 4NQO-induced oxidative DNA damage in a dose-dependent manner. On the contrary, MSB, a SFN analogue without the isothiocyanate moiety, did not have any protective effect on oxidative DNA damage (Figure 3C).
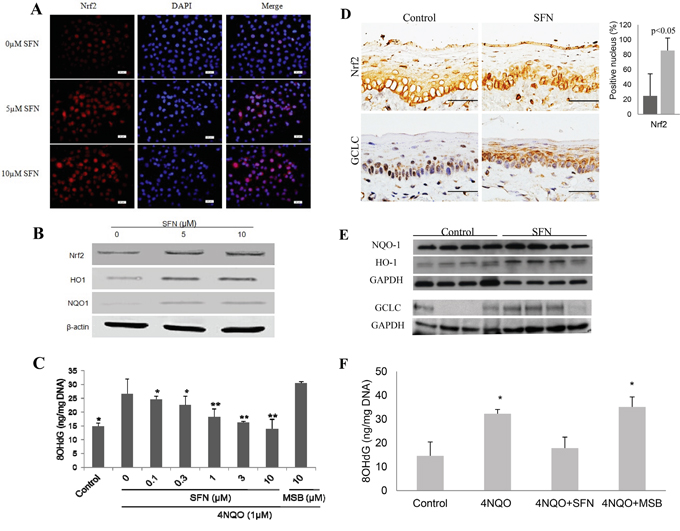
Figure 3: SFN treatment up-regulates NRF2 and its target genes, and suppresses 4NQO-induced oxidative DNA damage in vitro and in vivo. A. There is a dose-dependent increase of nuclear NRF2 in DOK cells after SFN treatment as determined by immunofluoresent staining with DAPI counterstaining. B. SFN treatment up-regulates expression of NRF2, HO1 and NQO1 in DOK cells as determined by Western blotting. C. Pre-treatment of SFN inhibits the level of 8OHdG after 4NQO treatment of DOK cells, whereas MSB does not have such an effect. D. IHC and quantitation shows a significant increase of the number of epithelial cells with nuclear Nrf2 and an increase of GCLC expression in mouse tongue after topical SFN treatment. E. Western blotting of NQO1, HO1 and GCLC shows a significant increase in HO1 (p<0.05) and slight increase of NQO1 and GCLC in SFN-treated mouse tongue. F. SFN treatment reduces the level of 8OHdG in mouse tongue due to 4NQO treatment. MSB does not have such an effect.
Topical application of SFN on mouse tongue for 2 weeks induced nuclear accumulation of NRF2 and up-regulation of GCLC in squamous epithelial cells of mouse tongue. Semi-quantitation of NRF2 staining showed a significant increase of epithelial cells with nuclear NRF2 in SFN-treated samples (Figure 3D). Western blotting also showed up-regulation of HO1, NQO1 and GCLC by SFN (Figure 3E). As expected, topical application of SFN for two weeks prior to 4NQO treatment inhibited oxidative DNA damage. In contrast, topical application of MSB did not have a significant effect on 4NQO-induced oxidative DNA damage (Figure 3F).
SFN prevents 4NQO-induced oral carcinogenesis in wild-type mice, but not in Nrf2-/- mice
In a long-term experiment, SFN were topically applied to tongues of wild-type and Nrf2-/- mice for 25 weeks (1 week prior to 4NQO treatment, 16 weeks during 4NQO treatment, and 8 weeks afterwards). SFN significantly reduced the number of visible tumors and the incidence of tongue SCC in wild-type mice. However the chemopreventive effect of SFN was completely lost in Nrf2-/- mice (Table 1).
Table 1: Chemopreventive effects of sulforaphane on 4NQO-induced oral carcinogenesis a
Group |
Genotype |
Treatment |
No |
Visible Tumor (%) |
No. of Tumor |
Tumor Volume (mm3) |
Dysplasia |
SCC |
|---|---|---|---|---|---|---|---|---|
A |
Wild-type |
- |
10 |
- |
- |
- |
- |
- |
B |
Nrf2-/- |
- |
10 |
- |
- |
- |
- |
- |
C |
Wild-type |
4NQO |
26 |
21 (80.8%) |
1.00±0.63 |
5.27±10.09 |
8(30.8%) |
18(69.2%) |
D |
Wild-type |
4NQO, SFN (-1~24w) |
28 |
14 (50%)b |
0.61±0.69 |
5.80±13.90 |
17(60.7%) |
11(39.3%)b |
E |
Nrf2-/- |
4NQO, SFN (-1~24w) |
24 |
17 (70.8%) |
1.42±1.21c |
5.35±9.01 |
8(33.3%) |
16(66.7%)d |
a 4NQO was given in drinking water (50μg/ml) for 16 weeks, and then replaced by water for 8 weeks. SFN was topically applied on the tongue at the concentration of 50 mM (50μl, 3 times per week).
b P<0.05 (as compared with Group C)
c P<0.01 (as compared with Group D)
d P<0.05 (as compared with Group D)
With IHC staining, we examined expression of Nrf2, HO1, NQO1 and 8OHdG in mouse tongue. Increased expression of NRF2 was observed in dysplasia but not in SCC as compared with normal samples (Figure 4A, 4B, 4C). Semi-quantitation of NRF2 expression confirmed this expression pattern (Figure 4D). Using IHC and Western, we also examined expression of HO1 and NQO1. Both NRF2-regulated genes shared the same expression pattern of NRF2 in normal, dysplasia and SCC tissues (Figure 4E-4J; semiquantiatation data of HO1 and NQO1 expression are not shown). Topical treatment with SFN up-regulated expression of HO1 and NQO1 in wild-type tongue (Group D), but not in Nrf2-/- tongue (Group E) (Figure 4K). Consistent with this observation, 8OHdG was at the highest level in dysplasia, and slightly reduced by SFN in wild-type tongue, but not in Nrf2-/- tongue (Figure 4O).
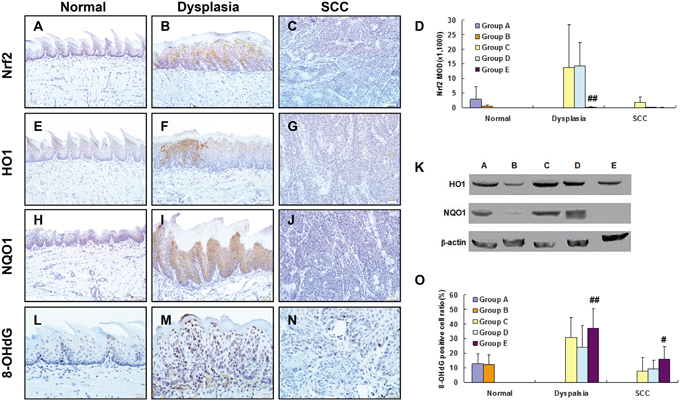
Figure 4: Expression of NRF2, HO1, NQO1 and 8OHdG in 4NQO/SFN-treated mouse tongue: Normal epithelium (A, E, H, L), dysplasia (B, F, I, M) and SCC (C, G, J, N). Expression of NRF2 in the epithelial cells oral tissues was semi-quantitated after (D). ## Statistically different from Group C (p<0.01). Expression of HO1 and NQO1 as determined by Western blotting (K). DNA oxidative damage level (the percentage of 8-OHdG positive cells) in the epithelial cells was semi-quantitated after IHC staining (O). # Statistically different from Group C (p<0.05); ## Statistically different from Group C (p<0.01).
DISCUSSION
In this study, SFN was shown to prevent 4NQO-induced oxidative damage and oral carcinogenesis in mouse tongue. Both pharmacological and genetic approaches demonstrated that SFN exerted its chemopreventive effects through NRF2 and the isothiocyanate moiety in vitro and in vivo. Using cell culture, mouse model, and human samples, we validated SFN as an effective chemopreventive agent for oxidative stress-associated oral carcinogenesis.
The 4NQO-induced oral carcinogenesis model with mice has been widely used for mechanistic and chemopreventive studies on oral cancer [13, 14]. It is known that 4NQO produces oxidative stress directly through generating reactive oxygen species (ROS) and indirectly through depleting GSH, and thus generates oxidative DNA damages [23, 24]. 4NQO also induced the formation of cellular topoisomerase I-DNA cleavage complexes, which may contribute to its mutagenesis and carcinogenesis [25]. Our microarray data of 4NQO-treated mouse tongue confirmed NRF2 activation and up-regulation of NRF2-regulated genes as oxidative stress response (Figure 2; Suppl. Excel 1, 2, 3). These data are consistent with a RNA-seq study comparing control mouse tongue with 4NQO-treated mouse tongue. In addition to up-regulation of 375 genes and down-regulation of 310 genes in that study, pathway analysis showed an enrichment of gene sets like “response to oxidative stress, glutathione metabolism, metabolism of xenobiotics by cytochrome P450” [26]. These data support the notion that 4NQO-induced oral carcinogenesis in mice (Figure 5) mimics oral carcinogenesis in humans at least in oxidative stress-associated mechanisms. It should be noted that exome sequencing data of 4NQO-treated mice showed some differences between mouse model and human cancer. In human oral cancer, C:G>A:T transversion and mutations at C nucleotides are more commonly seen [27, 28]. In this study, we also found that NRF2 activation was more prominent at the stage of dysplasia than the stage of cancer (Figure 4). These differences suggest multiple etiological factors for human oral cancer apart from oxidative stress.
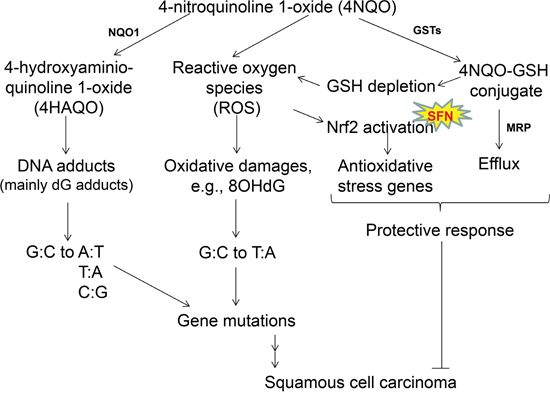
Figure 5: Mechanisms of 4NQO-induced oral carcinogenesis, and the chemopreventive mechanisms of SFN. When 4NQO is quickly metabolized in the squamous epithelial cells by NQO1, the ultimate carcinogen (4-hydroxyaminoquinoline 1-oxide, 4HAQO) causes gene mutations. Meanwhile, metabolism by glutathione transferases (GSTs) results in glutathione (GSH) depletion in these cells as well, and generates ROS which also produces oxidative damages and gene mutations. These gene mutations are presumably the driving force leading to SCC in mouse tongue. As a protective response, 4NQO-GSH conjugate is expected to be eliminated through multidrug resistant protein (MRP), and ROS activates the Nrf2 pathway which regulates antioxidative stress genes such as MT1, HO1, superoxide dismutase (SOD) and catalase. It is known that NQO1, GSTs, MRP, SOD, catalase are all transcriptionally regulated by NRF2. Based on this proposed mechanism, the 4NQO-induced oral carcinogenesis model in mice shares the same mechanism of oxidative damage as human oral carcinogenesis. SFN, which acts as a chemopreventive agent for 4NQO-induced oral carcinogenesis in mouse tongue through NRF2 and the isothiocyanate moiety, is therefore potentially effective for chemoprevention of human oral cancer.
The NRF2 pathway is a major cellular defense pathway through transcriptional regulation of its target genes involved in detoxification and anti-oxidative stress response. For example, NQO1 is a widely-distributed FAD-dependent flavoprotein that promotes obligatory 2-electron reductions of quinones, quinoneimines, nitroaromatics, and azo dyes. By reducing quinone levels, NQO1 minimizes reactive oxygen intermediates and prevents depletion of intracellular thiol pools [29]. HO1 is an oxidative stress-inducible protein which catabolizes heme into carbon monoxide, biliverdin/bilirubin and free iron, and thereby maintains intracellular redox homeostasis [30]. As a small, cysteine-rich and heavy metal-binding protein, MT1 protects cells from oxidative stress [31]. GCLC is the catalytic unit of glutamate cysteine ligase which catalyzes the rate-limiting step in the formation of the cellular antioxidant glutathione [32]. That’s why Nrf2-/- mice were more susceptible to 4NQO-induced tongue and esophageal carcinogenesis than wild-type mice, whereas Keap1-knockdown mice were resistant [33]. Thus, chemical activators of NRF2 are anticipated to be chemopreventive against oxidative stress-associated carcinogenesis. Such chemicals are classified into about ten chemically distinct classes based on their chemical structures and the nature of reaction with cysteine sulfhydryl groups [34]. Irrespective of their chemical differences, many of these compounds have shown chemopreventive effects against 4NQO-induced mutagenesis or carcinogenesis, such as green tea polyphenols [35], lycopene [36], selenium and vitamin E [37], butylated hydroxyanisole [38], naturally occurring plant phenolics [39], dietary flavonoids chalcone [40], naturally occurring xanthophylls [41], curcumin [42], and indole-3-carbinol [43].
SFN was chosen as a candidate chemopreventive agent in this preclinical study due to its advantages in multiple aspects. First of all, SFN is quite potent in activating NRF2 when topically applied on the skin [44, 45]. Indeed, SFN is ranked at the top among the natural NRF2 activators [46]. Secondly, SFN is a small molecule compound (MW 177) and stable in the polyethylene glycol based formulation suitable for topical application [47]. Thirdly, as a compound within the isothiocyanate group of organosulfur compounds, SFN is particularly rich in young cruciferous vegetables such as broccoli, Brussels sprouts or cabbages. It is produced when the enzyme myrosinase transforms glucoraphanin upon damage to the plant. SFN can be administered to the oral mucosa not only through topical application of the pure compound, but also through chewing on SFN-rich broccoli sprouts. Both the natural source and synthetic form of SFN are inexpensive and thus would allow the long-term use for oral cancer prevention. Fourthly, SFN has been used in clinical trials for human diseases with very good safety profiles [48].
There are two major concerns of using SFN for oral cancer prevention. NRF2 is a double-edged sword and can be carcinogenic when overactive. Nrf2 was found to prevent inhibition but accelerate progression of lung carcinogenesis in vivo [49]. K-Ras(G12D), B-Raf(V619E) and Myc(ERT2) each increased the transcription of Nrf2 to confer a more reduced intracellular environment. Genetic targeting of the Nrf2 pathway impairs K-Ras(G12D)-induced proliferation and tumorigenesis in vivo [50]. These data suggest that careful timing of NRF2 activation is a critical issue in balancing the induction and prevention of oral carcinogenesis. Although there is a need to induce NRF2 before cancer initiation, further activation of NRF2 at certain late stage may promote carcinogenesis. To reconcile the good and bad sides of NRF2, Kensler and Wakabayashi proposed a concept of “inflection point” to explain why intermittent dosing with an NRF2 activator (e.g., one or several doses a day) is unlikely to promote carcinogenesis in contrast to constitutive NRF2 activation when Nrf2 or Keap1 is mutated [51]. In fact, SFN acts on KEAP1 in a reversible manner. As a result, systemic administration of SFN did not promote the growth of K-ras(G12D)-induced lung tumors and had no significant effect on the growth of established tumor xenografts in nude mice [52]. Therefore we do not expect topical application of SFN would promote oral carcinogenesis. It is also possible that SFN may exert its chemopreventive effect through NRF2-independent mechanisms, such as cell cycle regulation, induction of apoptosis, etc [53, 54]. Since these NRF2-independent effects usually take place at high concentrations and the chemopreventive effect of SFN was diminished in Nrf2-/- mice, it is therefore believed that SFN mainly acts through NRF2 in this current study.
While this manuscript was in preparation, one abstract was published to evaluate the chemopreventive efficacy and mechanisms of NRF2 for oral carcinogenesis in vitro and in vivo. It is very interesting that short-term treatment of healthy volunteers with topical or oral SFN-rich broccoli sprout extract resulted in up-regulation of NQO1 expression in oral mucosa of 70% subjects (7 out of 10) [55].
In summary, these data clearly demonstrate that SFN has chemopreventive effects on oxidative stress-associated oral carcinogenesis, and such chemopreventive effects depend on NRF2 and the isothiocyanate moiety. Clinical chemoprevention trials using topical SFN in humans are therefore warranted.
MATERIALS AND METHODS
Collection of human samples
A total of 15 samples of normal epithelium and 21 samples of OLK were collected from human patients in the Department of Oral Medicine, Beijing Stomatological Hospital, Capital Medical University, from 2012 to 2014. The informed consent was obtained from all patients. The OLK lesion and normal mucosa were biopsied, fixed in 10% buffered formalin and embedded in paraffin. OLK was defined as a white patch or plaque that cannot be characterized clinically or pathologically as any other disease [56].
Synthesis of sulforaphane (SFN) and 4-methylsufinylbutylamine (MSB)
SFN is known to activate NRF2 through a reaction between its isothiocyanate moiety with Keap1 cysteine sulfhydryl group [57]. SFN was synthesized by us (SZ and XC) using a previously published method of chemical synthesis [58]. MSB was synthesized as a SFN analogue without the isothiocyanate moiety (Figure 6). The detailed procedure of chemical synthesis and MRI data are provided in Supplementary File.
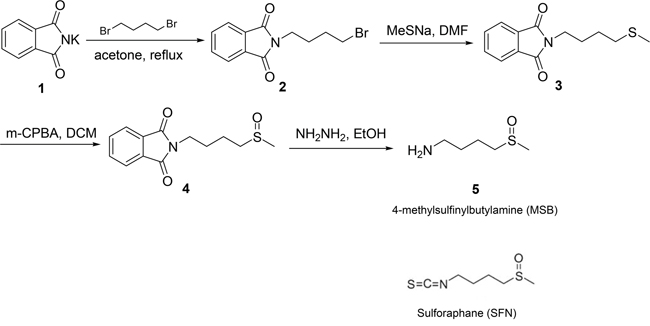
Figure 6: Chemical structures of SFN and MSB, and the synthetic process of MSB.
Cell culture and treatment
Human dysplastic oral keratinocytes (DOK) were obtained from Sigma (St Louis, MO). The DOK cells were cultured with DMEM/High glucose (Hyclone) medium supplemented with 15% FBS in culture incubator at 37C in 5% CO2. To determine whether SFN treatment may activate Nrf2 pathway, we determined Nrf2 localization with immunofluorescent staining. A total of 5×104 cells were cultured on a 24-well plate and treated with SFN (5 or 10μM) for 24 hours. Cells were fixed in cold 10% buffered neutral formalin for 10 minutes for immunofluorescent staining of NRF2.
To determine whether SFN treatment may inhibit 4NQO-induced oxidative damage, cells were treated by SFN (0.1, 0.3, 1, 3, or 10μM) or MSB (10μM) for 24 hr, and then 4NQO (1μM) for 12hr. These concentrations were determined in a preliminary experiment in which SFN and MSB (≤10μM) and 4NQO (≤1μM) did not significantly suppress cell growth as determined by a cell proliferation assay (data not shown). The cells were harvested for measurement of 8-OHdG by enzyme immunoassay (EIA).
Immunohistochemical staining (IHC) and immunofluorescent staining
Human samples of OLK lesion and normal mucosa were stained with a rabbit anti-NRF2 antibody (1:200) [59] or anti-8-oxo-2’-deoxyguanosine (8OHdG, 1:800; Abcam, Cambridge, MA). After deparaffinization, sections were submerged in methanol containing 0.3% hydrogen peroxide for 15 min to inhibit endogenous peroxidase activity. Antigen retrieval was done for incubating the sections in 0.01 mol/L citrate buffer (pH 6) in microwave. The antigen was detected with astreptavidin-peroxidase reaction kit (VECTASTAIN Elite ABC Kit; Vector Labs, Burlingame, CA). To ensure the specificity of the primary antibody, control tissue sections were incubated in the absence of primary antibody.
Immunofluorescent staining of NRF2 was conducted on cells with a rabbit polyclonal anti-NRF2 antibody (1:400), and then a goat anti-rabbit IgG secondary antibody labeled with Alexa Fluor 546 (Invitrogen, Carlsbad, CA). After counterstaining with DAPI, fluorescent signals were visualized under a fluorescent microscope.
Mouse tissue sections were analyzed for 8OHdG and expression of NRF2, NAD(P)H dehydrogenase [quinone] 1 (NQO1), heme oxygenase 1 (HO1), metallothionein 1 (MT1), and glutamate-cysteine ligase, catalytic subunit (GCLC) by IHC with the following antibodies overnight at 4C: a rabbit polyclonal anti-8-OHdG antibody (1:8000; Abcam), a rabbit polyclonal anti-NRF2 (1:200) [59], a rabbit polyclonal anti-NQO1 antibody (1:8000; Abcam), a mouse monoclonal anti-MT1 (1:100; Accurate Chemical & Scientific, Westbury, NY), a rabbit polyclonal anti-HO1 (1:25; Abcam), and a rabbit polyclonal anti-GCLC (1:100; LifeSpan BioSciences, Seattle, WA).
Immunostaining was semiquantitated for comparison between groups in some experiments. Expression of NRF2 was semi-quantitated using a computerized image analysis system (ImagePro Plus; Media Cybernetics, Rockville, MD). The area of positive staining and the mean optical density were measured for calculation of integrated optical density (IOD) by multiplying density by area. The percentage of 8OHdG-positive cells was calculated as the number of positively stained cells divided by the total number of tongue epithelial cells. Three non-contiguous, randomly selected, high-power fields (x400) were photographed and counted per sample.
Western blotting
Cells were treated with 5 or 10μM SFN for 24 hours, lysed and fractionated to obtain cytoplasmic and nuclear fractions. The nuclear extract was used for Western blotting of NRF2, and the cytoplasmic extract for HO1 and NQO1. Western blotting was done with a standard protocol. In brief, 20μg of protein was resolved on a SDS-polyacrylamide gel and transferred to nitrocellulose membranes. NRF2, HO1, and NQO1 were detected with a rabbit polyclonal anti-NRF2 antibody (1:1000; Abcam), a rabbit polyclonal anti-HO1 antibody (1:2000; Abcam), and a mouse polyclonal anti-NQO1 antibody (1:1000; Cell Signaling, Danvers, MA), respectively. Membranes were developed with ECL chemiluminescence and exposed on X-ray film. Quantitation of the bands on the film was carried out by a densitometer. Mouse tongue samples were homogenized and lysed in RIPA buffer for Western blotting of NQO1, HO1 and GCLC in a similar way. GAPDH or β-actin was detected as a loading control.
Enzyme immunoassay (EIA) for 8-OHdG
The cells were lysed in 100μl buffer (25 mM NaOH, 0.2 mM EDTA) at 95C for 60 min. After DNA extraction, 8OHdG was measured using 8OHdG ELISA Kit (Rapidbio, West Hills, CA) according to the manufacturer’s instructions. The level of 8OHdG was expressed as ng per mg total DNA. Mouse tongue was weighed and homogenized, lysed in the buffer, DNA extracted, and measured for 8-OHdG in a similar way.
Affymetrix gene microarray and data analysis
All the experimental procedures involving the use of mice were approved by the Institutional Animal Use and Care Committee, Beijing Stomatological Hospital. C57BL/6J mice were given 4NQO (100μg/ml in drink) for 8 weeks. Non-treated control samples were used for comparison. Total RNA was extracted from individual mouse tongue tissue using TRIzol (Invitrogen, Carlsbad, CA). These RNA samples were checked for their quality using gel electrophoresis and their concentrations were measured using spectrophotometry. GeneChip mouse Genome Array (Affymetrix, Santa Clara, CA) containing 45,101 oligo probes were used to detect differential gene expression between tongue tissues of control and treated mice. One μg of total RNA was used for labeling and processing after quality validation. GeneChip hybridization and scanning were performed according to the Affymetrix protocols. Briefly, double-stranded cDNA containing T7 promoter (Genset, La Jolla, CA) was synthesized from total RNA using the SuperScript Choice System (Invitrogen). Biotinylated cRNAs were generated from cDNAs by in vitro transcription and amplified by using the BioArray T7 RNA polymerase labeling kit (Enzo Diagnostics). After purification of cRNAs by GeneChip Sample Cleanup Module (Affymetrix), 15 μg of cRNA was fragmented at 94°C for 35 min. Approximately 12.5 μg of fragmented cRNA was used in a 250-μl hybridization mixture containing herring-sperm DNA (0.1 mg/ml; Promega), plus bacterial and phage cRNA controls (1.5 pM BioB, 5 pM BioC, 25 pM BioD, and 100 pM Cre) to serve as internal controls for hybridization efficiency. Aliquots (200 μl) of the mixture were hybridized to arrays for 16 h at 45°C in a GeneChip Hybridization Oven 640 (Affymetrix). Each array was washed and stained with streptavidin–phycoerythrin (Invitrogen) and amplified with biotinylated anti-streptavidin antibody (Vector Laboratories) on the GeneChip Fluidics Station 450 (Affymetrix). Arrays were scanned with the GeneArray G7 scanner (Affymetrix) to obtain image and signal intensities. Data pre-processing was carried out in R with Bioconductor package biocLite for quality filtering and data normalization. All probe sequences were BLAT against Affymetrix mouse annotation (Mouse430_2.na32.annot.csv), and were annotated with Gene Symbol.
Significance analysis of microarrays (SAM) was used for identification of differentially expressed genes. Pre-processed data were used to construct a series of data matrix files for further analysis. For a given data matrix, the rows were excluded if more than 40% of missing values were observed. The rest of missing data was imputed with a K-nearest neighbor (k=9) approach. Differentially expressed genes were obtained from two-class SAM in Excel with the median number of false positive less than 1. Gene set analysis (GSA) was carried out as an add-in in Excel to identify differentially expressed gene sets. 1,000 permutations were applied to generate a null distribution for statistical testing, and significantly enriched gene sets were obtained at a false discovery rate cutoff of 0.5. Curated gene sets in canonical pathway (CP; 880 gene sets) were downloaded from the GSEA web portal and used in this study (http://www.broadinstitute.org/gsea/index.jsp). In addition, we generated an NRF2-regulated gene set based on our previous gene array study [60] (Suppl. Excel 3), and conducted GSA accordingly. The microarray data have been submitted to the GEO database (GSE39629).
Whole exome sequencing
DNA samples of 3 mouse tongues were analyzed by whole exome sequencing: one control, one 4NQO-treated (100μg/ml in drink for 8 weeks) and one 4NQO plus ethanol treated (100μg/ml 4NQO and 8% ethanol in drink for 8 weeks). Genomic DNA was extracted from mouse tongue epithelium, quality checked, and submitted to Otogenetics Corporation (Norcross, GA) for exome capture and sequencing. Briefly, genomic DNA was subjected agarose gel and OD ratio tests to confirm the purity and concentration prior to Covaris (Covaris, Inc., Woburn, MA) fragmentation. Fragmented DNAs were tested for size distribution and concentration using an Agilent Bioanalyzer 2100 and Nanodrop. Illumina libraries were made from qualified fragmented DNA using NEBNext reagents (New England Biolabs, Ipswich, MA) and the resulting libraries were subjected to exome enrichment using SureSelect XT Mouse All Exon Kit (Agilent Technologies, Wilmington, DE) following manufacturer’s instructions. Enriched libraries were tested for enrichment by qPCR and for size distribution and concentration by an Agilent Bioanalyzer 2100. The samples were then sequenced on an Illumina HiSeq2000 which generated paired-end reads of 100 nucleotides (nt). Data was analyzed for data quality, exome coverage, and exome-wide SNP/InDel using the platform provided by DNAnexus (DNAnexus, Inc, Mountain View, CA). This method targets 49.6 Mb of mouse exome. Exon definition is derived from Ensembl and RefSeq, designed against mm9 reference from UCSC. Gene mutations were analyzed by comparing data of the control tongue with that of 4NQO-treated tongue (one treated with 4NQO alone and another with 4NQO and ethanol) after noncoding and synchronymous SNVs were removed. The minimum depth of sequencing was set at 10.
Animal experiments with SFN
In order to determine whether topical SFN may activate NRF2 in mouse tongue, a two-week study was performed in which C57BL/6J mice (6-8 weeks old; n=5; Beijing Vital River Laboratory Animal Company, Beijing, China) were topically treated with SFN (100 mM, 50μl, 5 times per week). In the literature, 0.1-1 μmole per day have been used for topical application on mouse skin for preventing skin cancer and treating pachyonychia congenital [44, 61]. We chose 5 μmole per day because this study was a proof-of-concept experiment which will be followed by a dose-response experiment. Unlike topical application on mouse skin, topical application into mouse oral cavity may be less bioavailable because mice tended to spit excessive liquid right after application. A control group of mice (n=5) were treated in the same way with corn oil. At the end of the study, half of the tongue was collected and fixed in 10% buffered formalin and embedded in paraffin for IHC of NRF2 and GCLC. The second half of the tongue was snap-frozen in liquid nitrogen and stored at -80C for Western blotting of NQO1, HO1, and GCLC.
In order to determine whether topical SFN may protect mouse tongue from 4NQO-induced oxidative damage, wild-type C57BL/6J mice (6-8 weeks old) were treated with 4NQO in drinking water (100μg/ml) for 8 weeks. Two weeks before and during 4NQO treatment, the first group did not receive any other treatment (n=15); the second group was topically treated with SFN (n=15; 100 mM, 50μl, 5/week); and the third group with MSB (n=15; 100 mM, 50μl, 5/week). Mice not treated with 4NQO (n=10) served as the negative control. All the mice were sacrificed at the end of week 10. One half of the tongue was snap-frozen in liquid nitrogen for analysis of 8-OHdG and the other half was fixed in formalin and embedded in paraffin.
In a long-term chemoprevention study with SFN, wild-type C57BL/6J mice (6-8 weeks old) and Nrf2-/- mice (RIKEN BioResource Center, Japan) were bred in-house and PCR genotyped [62]. Mice were assigned to 5 groups: Group A (n=10) as the wild-type negative control; Group B (n=10) as the Nrf2-/- negative control; Group C (n=30) as the positive control group; Group D (n=30) as the wild-type experimental group; and Group E (n=30) as the Nrf2-/- experimental group. Group C, D and E were given 4NQO in drinking water (50μg/ml for 16 weeks). SFN treatment (50 mM in corn oil, 50μl, 3/week) were administered to Group D and E throughout the whole experiment (-1~24w) (Table 1). An earlier study has clearly demonstrated that Nrf2-/- mice were more susceptible to 4NQO-induced tongue carcinogenesis than wild-type mice [33]. Therefore, we did not include an “Nrf2-/-+4NQO” group and an “Nrf2-/-+4NQO+ SFN” group for the sake of saving the number of animals. All mice were monitored for their body weights weekly and sacrificed at Week 24. The tongue was harvested and examined for the presence of macroscopic alterations, then split longitudinally. One half of the tongue was snap frozen in liquid nitrogen for Western blotting (HO1 and NQO1), and the half was fixed overnight in 10% neutral-buffered formalin, processed, and embedded in paraffin for histopathology.
Histopathological analysis was performed blind by our research pathologist without prior knowledge of the experiment. Dysplasia and SCC were diagnosed with established criteria. Dysplasia was characterized by irregular epithelial stratification, increased number of mitotic figures, increased nuclear-to-cytoplastic ratio, and loss of polarity of basal cells. SCC was diagnosed when dysplastic cells invaded underlying tissues [56]. Tissue sections were used for analysis of Nrf2, HO1, NQO1, and 8-OHdG with IHC.
Statistical analysis
Fisher’s exact test was used for evaluation of tumor incidence, Student’s t test for two-group comparisons, and one-way ANOVA for multiple-group comparisons. Values were expressed as mean ± SD. P<0.05 was considered statistically significant.
Abbreviations
4HAQO, 4-hydroxyaminoquinoline 1-oxide; 4NQO, 4-nitroquinoline 1-oxide; 8OHdG, 8-oxo-2’-deoxyguanosine; CP, canonical pathway; EIA, enzyme immunoassay; GCLC, glutamate-cysteine ligase, catalytic subunit; GSA, gene set analysis; GSH, glutathione; GST, glutathione transferase; HO1, heme oxygenase 1; IHC, immunohistochemical staining; IOD, integrated optical density; MSB, 4-methyl sulfinylbutylamine; MRP, multidrug resistance protein; MT1, metallothionein 1; NQO1, NAD(P)H dehydrogenase [quinone] 1; NRF2, nuclear factor, erythroid 2-like 2; OLK, oral leukoplakia; ROS, reactive oxygen species; SAM, significance analysis of microarrays; SFN, sulforaphane; SCC, squamous cell carcinoma; SNV, single nucleotide variant; SOD, superoxide dismutase.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
GRANT SUPPORT
This work was supported by the National Natural Science Foundation of China (No. 30973325 and 81372891) and the National Institutes of Health (U54 AA019765, U54 CA156735).
REFERENCES
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5-29.
2. Stelow EB, Mills SE. Squamous cell carcinoma variants of the upper aerodigestive tract. Am J Clin Pathol. 2005; 124 Suppl:S96-109.
3. Liu Y, Chen H, Sun Z, Chen X. Molecular mechanisms of ethanol-associated oro-esophageal squamous cell carcinoma. Cancer Lett. 2015; 361:164-173.
4. Agha-Hosseini F, Mirzaii-Dizgah I, Farmanbar N, Abdollahi M. Oxidative stress status and DNA damage in saliva of human subjects with oral lichen planus and oral squamous cell carcinoma. J Oral Pathol Med. 2012; 41:736-740.
5. Nagini S, Letchoumy PV, A T, Cr R. Of humans and hamsters: a comparative evaluation of carcinogen activation, DNA damage, cell proliferation, apoptosis, invasion, and angiogenesis in oral cancer patients and hamster buccal pouch carcinomas. Oral oncology. 2009; 45:e31-37.
6. Ma N, Tagawa T, Hiraku Y, Murata M, Ding X, Kawanishi S. 8-Nitroguanine formation in oral leukoplakia, a premalignant lesion. Nitric Oxide. 2006; 14:137-143.
7. Choudhari SK, Chaudhary M, Gadbail AR, Sharma A, Tekade S. Oxidative and antioxidative mechanisms in oral cancer and precancer: a review. Oral oncology. 2014; 50:10-18.
8. Fortmann SP, Burda BU, Senger CA, Lin JS, Whitlock EP. Vitamin and mineral supplements in the primary prevention of cardiovascular disease and cancer: An updated systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2013; 159:824-834.
9. Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol. 2007; 47:89-116.
10. Huang CF, Zhang L, Ma SR, Zhao ZL, Wang WM, He KF, Zhao YF, Zhang WF, Liu B, Sun ZJ. Clinical significance of Keap1 and Nrf2 in oral squamous cell carcinoma. PloS one. 2013; 8:e83479.
11. Stacy DR, Ely K, Massion PP, Yarbrough WG, Hallahan DE, Sekhar KR, Freeman ML. Increased expression of nuclear factor E2 p45-related factor 2 (NRF2) in head and neck squamous cell carcinomas. Head & neck. 2006; 28:813-818.
12. Posner GH, Cho CG, Green JV, Zhang Y, Talalay P. Design and synthesis of bifunctional isothiocyanate analogs of sulforaphane: correlation between structure and potency as inducers of anticarcinogenic detoxication enzymes. J Med Chem. 1994; 37:170-176.
13. Tang XH, Albert M, Scognamiglio T, Gudas LJ. A DNA methyltransferase inhibitor and all-trans retinoic acid reduce oral cavity carcinogenesis induced by the carcinogen 4-nitroquinoline 1-oxide. Cancer Prev Res (Phila). 2009; 2:1100-1110.
14. Tang XH, Osei-Sarfo K, Urvalek AM, Zhang T, Scognamiglio T, Gudas LJ. Combination of bexarotene and the retinoid CD1530 reduces murine oral-cavity carcinogenesis induced by the carcinogen 4-nitroquinoline 1-oxide. Proc Natl Acad Sci U S A. 2014; 111:8907-8912.
15. Gorrini C, Baniasadi PS, Harris IS, Silvester J, Inoue S, Snow B, Joshi PA, Wakeham A, Molyneux SD, Martin B, Bouwman P, Cescon DW, Elia AJ, et al. BRCA1 interacts with Nrf2 to regulate antioxidant signaling and cell survival. J Exp Med. 2013; 210:1529-1544.
16. Wang Q, Li J, Yang X, Sun H, Gao S, Zhu H, Wu J, Jin W. Nrf2 is associated with the regulation of basal transcription activity of the BRCA1 gene. Acta Biochim Biophys Sin (Shanghai). 2013; 45:179-187.
17. Bae I, Fan S, Meng Q, Rih JK, Kim HJ, Kang HJ, Xu J, Goldberg ID, Jaiswal AK, Rosen EM. BRCA1 induces antioxidant gene expression and resistance to oxidative stress. Cancer Res. 2004; 64:7893-7909.
18. Hast BE, Goldfarb D, Mulvaney KM, Hast MA, Siesser PF, Yan F, Hayes DN, Major MB. Proteomic analysis of ubiquitin ligase KEAP1 reveals associated proteins that inhibit NRF2 ubiquitination. Cancer Res. 2013; 73:2199-2210.
19. Kozlovsky N, Rudich A, Potashnik R, Ebina Y, Murakami T, Bashan N. Transcriptional activation of the Glut1 gene in response to oxidative stress in L6 myotubes. J Biol Chem. 1997; 272:33367-33372.
20. Kim HL, Seo YR. Molecular and genomic approach for understanding the gene-environment interaction between Nrf2 deficiency and carcinogenic nickel-induced DNA damage. Oncol Rep. 2012; 28:1959-1967.
21. Anedda A, Lopez-Bernardo E, Acosta-Iborra B, Saadeh Suleiman M, Landazuri MO, Cadenas S. The transcription factor Nrf2 promotes survival by enhancing the expression of uncoupling protein 3 under conditions of oxidative stress. Free Radic Biol Med. 2013; 61:395-407.
22. Downes DJ, Chonofsky M, Tan K, Pfannenstiel BT, Reck-Peterson SL, Todd RB. Characterization of the mutagenic spectrum of 4-nitroquinoline 1-oxide (4-NQO) in Aspergillus nidulans by whole genome sequencing. G3 (Bethesda). 2014; 4:2483-2492.
23. Arima Y, Nishigori C, Takeuchi T, Oka S, Morimoto K, Utani A, Miyachi Y. 4-Nitroquinoline 1-oxide forms 8-hydroxydeoxyguanosine in human fibroblasts through reactive oxygen species. Toxicol Sci. 2006; 91:382-392.
24. Nunoshiba T, Demple B. Potent intracellular oxidative stress exerted by the carcinogen 4-nitroquinoline-N-oxide. Cancer Res. 1993; 53:3250-3252.
25. Miao ZH, Rao VA, Agama K, Antony S, Kohn KW, Pommier Y. 4-nitroquinoline-1-oxide induces the formation of cellular topoisomerase I-DNA cleavage complexes. Cancer Res. 2006; 66:6540-6545.
26. Tang XH, Urvalek AM, Osei-Sarfo K, Zhang T, Scognamiglio T, Gudas LJ. Gene expression profiling signatures for the diagnosis and prevention of oral cavity carcinogenesis-genome-wide analysis using RNA-seq technology. Oncotarget. 2015; 6:24424-24435. doi: 10.18632/oncotarget.4420.
27. Pickering CR, Zhang J, Neskey DM, Zhao M, Jasser SA, Wang J, Ward A, Tsai CJ, Ortega Alves MV, Zhou JH, Drummond J, El-Naggar AK, Gibbs R, et al. Squamous cell carcinoma of the oral tongue in young non-smokers is genomically similar to tumors in older smokers. Clin Cancer Res. 2014; 20:3842-3848.
28. Mutational landscape of gingivo-buccal oral squamous cell carcinoma reveals new recurrently-mutated genes and molecular subgroups. Nat Commun. 2013; 4:2873.
29. Dinkova-Kostova AT, Talalay P. NAD(P)H:quinone acceptor oxidoreductase 1 (NQO1), a multifunctional antioxidant enzyme and exceptionally versatile cytoprotector. Arch Biochem Biophys. 2010; 501:116-123.
30. Fan W, Huang F, Zhu X, Li D, Fu S, He H. The heme oxygenase system and oral diseases. Oral Dis. 2011; 17:252-257.
31. Ruttkay-Nedecky B, Nejdl L, Gumulec J, Zitka O, Masarik M, Eckschlager T, Stiborova M, Adam V, Kizek R. The role of metallothionein in oxidative stress. Int J Mol Sci. 2013; 14:6044-6066.
32. Krejsa CM, Franklin CC, White CC, Ledbetter JA, Schieven GL, Kavanagh TJ. Rapid activation of glutamate cysteine ligase following oxidative stress. J Biol Chem. 2010; 285:16116-16124.
33. Ohkoshi A, Suzuki T, Ono M, Kobayashi T, Yamamoto M. Roles of Keap1-Nrf2 system in upper aerodigestive tract carcinogenesis. Cancer Prev Res (Phila). 2013; 6:149-159.
34. Magesh S, Chen Y, Hu L. Small molecule modulators of Keap1-Nrf2-ARE pathway as potential preventive and therapeutic agents. Med Res Rev. 2012; 32:687-726.
35. Srinivasan P, Suchalatha S, Babu PV, Devi RS, Narayan S, Sabitha KE, Shyamala Devi CS. Chemopreventive and therapeutic modulation of green tea polyphenols on drug metabolizing enzymes in 4-Nitroquinoline 1-oxide induced oral cancer. Chem Biol Interact. 2008; 172:224-234.
36. El-Rouby DH. Histological and immunohistochemical evaluation of the chemopreventive role of lycopene in tongue carcinogenesis induced by 4-nitroquinoline-1-oxide. Arch Oral Biol. 2011; 56:664-671.
37. Guttenplan JB, Kosinska W, von Pressentin M, Rosa J, El-Bayoumy K. Effects of 1,4-phenylenebis(methylene)selenocyanate (p-XSC) and vitamin E on 4-nitroquinoline-N-oxide (4-NQO)-induced mutagenesis in lacZ mouse upper aerodigestive tissue. Mutat Res. 2002; 518:85-93.
38. Stanley JS, York JL, Benson AM. Nitroreductases and glutathione transferases that act on 4-nitroquinoline 1-oxide and their differential induction by butylated hydroxyanisole in mice. Cancer Res. 1992; 52:58-63.
39. Tanaka T, Kojima T, Kawamori T, Wang A, Suzui M, Okamoto K, Mori H. Inhibition of 4-nitroquinoline-1-oxide-induced rat tongue carcinogenesis by the naturally occurring plant phenolics caffeic, ellagic, chlorogenic and ferulic acids. Carcinogenesis. 1993; 14:1321-1325.
40. Makita H, Tanaka T, Fujitsuka H, Tatematsu N, Satoh K, Hara A, Mori H. Chemoprevention of 4-nitroquinoline 1-oxide-induced rat oral carcinogenesis by the dietary flavonoids chalcone, 2-hydroxychalcone, and quercetin. Cancer Res. 1996; 56:4904-4909.
41. Tanaka T, Makita H, Ohnishi M, Mori H, Satoh K, Hara A. Chemoprevention of rat oral carcinogenesis by naturally occurring xanthophylls, astaxanthin and canthaxanthin. Cancer Res. 1995; 55:4059-4064.
42. Tanaka T, Makita H, Ohnishi M, Hirose Y, Wang A, Mori H, Satoh K, Hara A, Ogawa H. Chemoprevention of 4-nitroquinoline 1-oxide-induced oral carcinogenesis by dietary curcumin and hesperidin: comparison with the protective effect of beta-carotene. Cancer Res. 1994; 54:4653-4659.
43. Tanaka T, Kojima T, Morishita Y, Mori H. Inhibitory effects of the natural products indole-3-carbinol and sinigrin during initiation and promotion phases of 4-nitroquinoline 1-oxide-induced rat tongue carcinogenesis. Jpn J Cancer Res. 1992; 83:835-842.
44. Xu C, Huang MT, Shen G, Yuan X, Lin W, Khor TO, Conney AH, Kong AN. Inhibition of 7,12-dimethylbenz(a)anthracene-induced skin tumorigenesis in C57BL/6 mice by sulforaphane is mediated by nuclear factor E2-related factor 2. Cancer Res. 2006; 66:8293-8296.
45. Dinkova-Kostova AT, Jenkins SN, Fahey JW, Ye L, Wehage SL, Liby KT, Stephenson KK, Wade KL, Talalay P. Protection against UV-light-induced skin carcinogenesis in SKH-1 high-risk mice by sulforaphane-containing broccoli sprout extracts. Cancer Lett. 2006; 240:243-252.
46. Wu KC, McDonald PR, Liu J, Klaassen CD. Screening of natural compounds as activators of the keap1-nrf2 pathway. Planta Med. 2014; 80:97-104.
47. Franklin SJ, Dickinson SE, Karlage KL, Bowden GT, Myrdal PB. Stability of sulforaphane for topical formulation. Drug Dev Ind Pharm. 2014; 40:494-502.
48. Houghton CA, Fassett RG, Coombes JS. Sulforaphane: translational research from laboratory bench to clinic. Nutr Rev. 2013; 71:709-726.
49. Satoh H, Moriguchi T, Takai J, Ebina M, Yamamoto M. Nrf2 prevents initiation but accelerates progression through the Kras signaling pathway during lung carcinogenesis. Cancer Res. 2013; 73:4158-4168.
50. DeNicola GM, Karreth FA, Humpton TJ, Gopinathan A, Wei C, Frese K, Mangal D, Yu KH, Yeo CJ, Calhoun ES, Scrimieri F, Winter JM, Hruban RH, et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature. 2011; 475:106-109.
51. Kensler TW, Wakabayashi N. Nrf2: friend or foe for chemoprevention? Carcinogenesis. 2010; 31:90-99.
52. Kombairaju P, Ma J, Thimmulappa RK, Yan SG, Gabrielson E, Singh A, Biswal S. Prolonged sulforaphane treatment does not enhance tumorigenesis in oncogenic K-ras and xenograft mouse models of lung cancer. J Carcinog. 2012; 11:8.
53. Clarke JD, Dashwood RH, Ho E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008; 269:291-304.
54. Sestili P, Fimognari C. Cytotoxic and Antitumor Activity of Sulforaphane: The Role of Reactive Oxygen Species. Biomed Res Int. 2015; 2015:402386.
55. Bauman JE, Zang Y, Sen M, Normolle DP, Kensler TW, Trivedi S, Egner PA, Sheth SH, Grandis JR, Johnson DE. (2015). Sulforaphane as a chemopreventive agent against oral carcinogenesis. Annual Meeting of AACR. (Philadelphia: AACR), pp. A894.
56. Barnes L, Eveson JW, Reichart P, Sidransky D. (2005). Pathology and Genetics of Head and Neck Tumours. Oxford University Press), pp. 177-178.
57. Hu C, Eggler AL, Mesecar AD, van Breemen RB. Modification of keap1 cysteine residues by sulforaphane. Chem Res Toxicol. 2011; 24:515-521.
58. Chen X, Li Z, Sun X, Ma H, Chen X, Ren J, Hu K. New method for the synthesis of sulforaphane and related isothiocyanates. Synthesis. 2011; 24:3991-3996.
59. Suvorova ES, Lucas O, Weisend CM, Rollins MF, Merrill GF, Capecchi MR, Schmidt EE. Cytoprotective Nrf2 Pathway Is Induced In Chronically Txnrd 1-Deficient Hepatocytes. PloS one. 2009; 4.
60. Chen H, Li J, Li H, Hu Y, Tevebaugh W, Yamamoto M, Que J, Chen X. Transcript profiling identifies dynamic gene expression patterns and an important role for Nrf2/Keap1 pathway in the developing mouse esophagus. PloS one. 2012; 7:e36504.
61. Kerns ML, Hakim JM, Lu RG, Guo Y, Berroth A, Kaspar RL, Coulombe PA. Oxidative stress and dysfunctional NRF2 underlie pachyonychia congenita phenotypes. J Clin Invest. 2016.
62. Wakabayashi N, Itoh K, Wakabayashi J, Motohashi H, Noda S, Takahashi S, Imakado S, Kotsuji T, Otsuka F, Roop DR, Harada T, Engel JD, Yamamoto M. Keap1-null mutation leads to postnatal lethality due to constitutive Nrf2 activation. Nat Genet. 2003; 35:238-245.

