INTRODUCTION
Gastric cancer is one of the most common malignancies and displays variable incidence around the globe. About 90–95% of the cases are sporadic, it’s incidence is highest in East Asia, Central Eastern Europe, and approximately 75% of cases present in less developed countries [1]. In most developed regions, rates of stomach cancer have variably but uniformly declined over the past decades, fairly due to active surveillance methods in selected societies [2]. There are no solid biomarkers besides HER2 [3] and regular clinicopathological parameters predicting prognosis and response to therapy, and ultimately, there are no efficient therapeutic options available which prove to change the outcome of patients in a groundbreaking manner.
Following endoscopic examination and histologic confirmation of malignancy in the harvested biopsy, the basis of therapy is still removal of the tumor mass utilizing surgery. The 5-year survival rate for R0 surgical resection ranges from 30 to 50% for patients with stage II and from 10 to 25% for patients with stage III disease [4]. As these patients have a high likelihood of local and systemic relapse, most centers offer them systemic therapy forming the other cornerstone of the treatment. In addition, radiation therapy has proved to improve 5-year survival in resectable tumors [5].
Applied prognostic factors of gastric cancer are limited to the clinicopathological properties in the routine setting today, and classically include the WHO histopathological type, Lauren-Järvi classification, size of the tumor, grade, invasion through the gastric wall (pT), vascular invasion, lymph node involvement (pN), etiological background (EBV or Helicobacter pylori) and HER2 overexpression [2, 5–7]. Biomarkers for diagnosis and prognosis of gastric cancer that have previously been identified are mostly non tumor tissue based, and include carcinoembryonic antigen (CEA), CA 125, CA 19–9, CA 72–4 and alpha-fetoprotein [6, 8], serum pepsinogen I, and proteases (pepsinogen C, plasminogen activator, matrix metalloproteinases and their inhibitors) [9]. Cadherins, mucins and CD44 splicing variants are related to invasion/metastasis and extracellular matrix adhesion and degradation [7].
Among tissue based markers, overexpression of human epidermal growth factor receptor 2 (HER2) has been identified as a negative prognostic factor [7]. Trastuzumab with chemotherapy in HER2-positive advanced gastric cancer was investigated in the ToGA study. In this phase 3 trial, 22% of advanced stage cancers overexpressed HER2 and overall survival with trastuzumab was 2.7 months longer (hazard ratio, HR = 0.74, P = 0.0046) [10]. In addition, trastuzumab improved all of the secondary end points as well.
In a search for robust cancer tissue related biomarkers, first we intended to perform a literature review and identify previously described markers for gastric cancer outcome. We merged transcriptomic data of multiple independent datasets to enable a cross-validation of these in a uniform independent cohort. We used uni- and multivariate analyses to assess the prognostic potential for each of the candidate markers. Finally, we compared expression in normal and gastric cancer samples to evaluate the change of the gene expression during tumor formation.
RESULTS
Database setup
The entire gastric cancer database includes 1,065 samples from seven independent datasets. Of these, 652 samples were measured with the Affymetrix Human Genome U133 Plus 2.0 Array, 145 with the Human Genome U133A 2.0 Array and 268 with the Human Genome U133A Array. Five arrays did not pass quality control and were excluded from the cross-validation analysis (all five arrays originated in the Bethesda dataset).
Gender and stage were available for most patients –70% of samples were male and stage III was most common (Figure 1A). Additional clinical parameters including TNM stages, histology and systemic treatment were available for about half the patients – the aggregate clinical characteristics are summarized in Table 1. The median time to first progression (FP) was 18.3 months and the median overall survival (OS) was 28.9 months. Even with these numerically significant differences, the survival curves comparing FP and OS display minor difference (Figure 1B) indicating a short post-progression survival – in the 503 patients with a first event and a known OS, the median post progression survival was 9.4 months.
Table 1: Summary of aggregate clinicopathological data for all patient samples included in the cross-validation
Parameter |
% |
N |
|---|---|---|
Gender |
||
Male |
53.1% |
566 |
Female |
22.9% |
244 |
No data |
23.9% |
255 |
Systemic treatment |
||
No Adjuvant |
36.9% |
393 |
Adjuvant |
22.3% |
238 |
No data |
40.8% |
434 |
Histology (Lauren class.) |
||
Diffuse |
23.3% |
248 |
Intestinal |
31.5% |
336 |
Mixed |
3.1% |
33 |
No data |
42.1% |
448 |
Differentiation |
||
Poor |
15.6% |
166 |
Moderate |
6.3% |
67 |
Well |
3.0% |
32 |
No data |
75.1% |
800 |
pT |
||
T1 |
1.3% |
14 |
T2 |
23.8% |
253 |
T3 |
19.5% |
208 |
T4 |
3.7% |
39 |
No data |
51.7% |
551 |
pN |
||
N0 |
7.1% |
76 |
N1 |
21.8% |
232 |
N2 |
12.1% |
129 |
N3 |
7.1% |
76 |
No data |
51.8% |
552 |
cM |
||
M0 |
43.1% |
459 |
M1 |
5.4% |
58 |
No data |
51.5% |
548 |
AJCC Stage |
||
I |
6.5% |
69 |
II |
13.6% |
145 |
III |
30.0% |
319 |
IV |
14.3% |
152 |
No data |
35.7% |
380 |
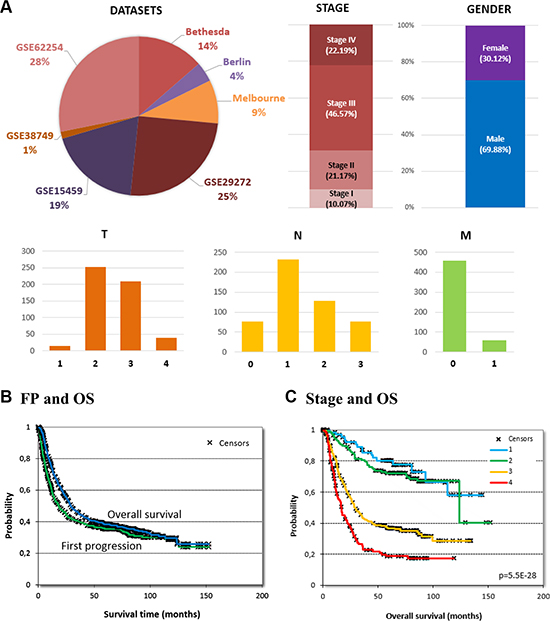
Figure 1: Database setup and clinical characteristics. List of datasets included in the database as well as basic clinical characteristics (A). Number of patients are given for TNM, because not all patients had these data available. Overall survival and time to first progression in all patients, (B) and effect of stage on overall survival (C).
Of the clinical parameters, gender, differentiation and histology were not significantly correlated to overall survival. Stage (p = 5.5E-28, see Figure 1C), T (p = 7.9E–15), and N (p = 1.1E-19) delivered high significance while there were not sufficient events to compute correlation to OS for M. Similar results were delivered for FP survival (stage: p = 1.7E-31, T: p = 9.2E-14, and N: p = 4.3E-20). In addition, M was also significant for FP (p = 1.3E-16).
Identification of biomarker candidates
The keyword search in PubMed resulted in 775 hits, of which 749 were in English language, and 398 were published between 2012–2015. Of these, 40 publications were categorized as review. Following careful and critical evaluation, a list of 29 markers emerged (Supplementary Table 1). Of these candidates, one gene was not present on the gene chips (AFAP1L2), and the remaining 28 were evaluated in the cross-validation.
Validation of previously identified prognostic markers
Out of the 28 biomarkers, 19 reached significance level with a FDR below 5% for FP and 20 for OS in the univariate analysis investigating gene expression only. Eighteen markers were significant for both FP and OS. Higher expression of BECN1, CASP3, COX2, CTGF, CTNNB1, MET, and SIRT1 correlated to better survival. Higher expression of BIRC5, CNTN1, EGFR, ERCC1, HER2, MMP2, PFKB4, SPHK, SP1, SPARC, TIMP1 and VEGF were predictive for poor outcome. For OS, the direction of correlation to survival was the same for all significant genes. The significant genes with hazard rates and p values are listed in Table 2. Supplementary Table 1. lists the results for all genes.
Table 2: List of significant gastric cancer genes evaluated in independent studies between 2012 and 2015
Symbol |
Affy ID |
Gene name |
Ref. |
First progression HR (95% CI), p |
Overall survival HR (95% CI), p |
|---|---|---|---|---|---|
BECN1 |
208946_s_at |
Beclin-1 |
[44] |
HR = 0.68 (0.55–0.84)p = 0.00042 |
HR = 0.68 (0.57–0.81) p= 1.5e-05 |
BIRC5 |
202094_at |
Survivin |
[25] |
HR = 1.52 (1.22–1.89)p = 0.00016 |
HR = 1.45 (1.2–1.75) p= 1e-04 |
CASP3 |
202763_at |
Caspase-3 |
[21] |
HR = 0.52 (0.42–0.64)p = 3e-10 |
HR = 0.5 (0.42–0.6) p = 6e-14 |
CNTN1 |
211203_s_at |
Contactin-1 |
[19] |
HR = 1.41 (1.15–1.73)p = 0.0011 |
HR = 1.44 (1.21–1.7) p = 3.5e-05 |
COX2 |
204748_at |
Cyclooxygenase-2 |
[16] |
HR = 0.73 (0.59–0.91)p = 0.0056 |
HR = 0.72 (0.59–0.88) p= 0.0013 |
CTGF |
209101_at |
Connective tissue growth factor |
[22] |
HR = 0.71 (0.58–0.89p = 0.0022 |
HR = 0.72 (0.59–0.87) p= 0.00051 |
CTNNB1 |
201533_at |
Beta-catenin |
[18] |
HR = 0.52 (0.42–0.64)p = 3.2e-10 |
HR = 0.47 (0.38–0.57) p= 4.3e-15 |
EGFR |
201983_s_at |
Epidermal growth factor receptor |
[12] |
HR = 1.85 (1.49–2.29)p = 1.6e-08 |
HR = 1.86 (1.54–2.25) p= 8.5e-11 |
ERCC1 |
203720_s_at |
Excision repair complementation group 1 |
[45] |
HR = 1.38 (1.12–1.69) p = 0.002 |
HR = 1.36 (1.13–1.63) p= 0.0012 |
HER2 |
216836_s_at |
Human epidermal growth factor receptor 2 |
[46] |
HR = 1.38 (1.12–1.69)p = 0.0021 |
HR = 1.41 (1.18–1.68) p= 0.00011 |
HIF1a |
200989_at |
Hypoxia-inducible factors-1 alpha |
[14] |
n.s. |
HR = 0.73 (0.62–0.87) p= 0.00036 |
MET (HGFR) |
203510_at |
Hepatocyte growth factor receptor |
[47] |
HR = 0.69 (0.55–0.87)p = 0.0018 |
HR = 0.63 (0.51–0.77) p= 1.3e-05 |
MMP-2 |
201069_at |
Matrix metalloproteinase 2 |
[24] |
HR = 1.64 (1.33–2.02)p = 2.8e-06 |
HR = 1.78 (1.47–2.16) p= 2.6e-09 |
NOV |
200724_at |
Nephroblastoma Overexpressed |
[22] |
n.s. |
HR = 1.45 (1.22–1.72) p = 1.7e-05 |
PFKB4 |
206246_at |
6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-4 |
[48] |
HR = 1.7 (1.33–2.19) p = 2.5e-05 |
HR = 1.56 (1.32–1.86) p= 3.2e-07 |
SIRT1 |
218878_s_at |
Silent mating type information regulation 1 |
[49] |
HR = 0.56 (0.45–0.7) p = 1.1e-07 |
HR = 0.64 (0.54–0.76) p= 2.2e-07 |
SPHK1 |
219257_s_at |
Sphingosine kinase 1 |
[50] |
HR = 1.62 (1.31–1.99)p = 5.6e-06 |
HR = 1.61 (1.31–1.96) p= 3.1e-06 |
SP1 |
214732_at |
Specificity protein 1 |
[20] |
HR = 1.47 (1.19–1.82)p = 4e-04 |
HR = 1.45 (1.23–1.72) p= 1.6e-05 |
SPARC |
212667_at |
Secreted protein acidic and rich in cysteine |
[51] |
HR = 1.34 (1.08–1.66)p = 0.007 |
n.s. |
TIMP-1 |
201666_at |
Tissue inhibitor of metalloproteinase-1 |
[23] |
HR = 1.77 (1.42–2.22)p = 3.9e-07 |
HR = 1.92 (1.57–2.36) p= 2.2e-10 |
VEGF |
210512_s_at |
Vascular endothelial growth factor |
[15] |
HR = 1.75 (1.41–2.17)p = 2.9e-07 |
HR = 1.53 (1.27–1.85) p= 5.7e-06 |
Statistical test: Cox univariate regression analysis, HR: hazard rate, CI: confidence interval, n.s.: p value over the 5% FDR cutoff. Bold: see survival plots in Figure 2.
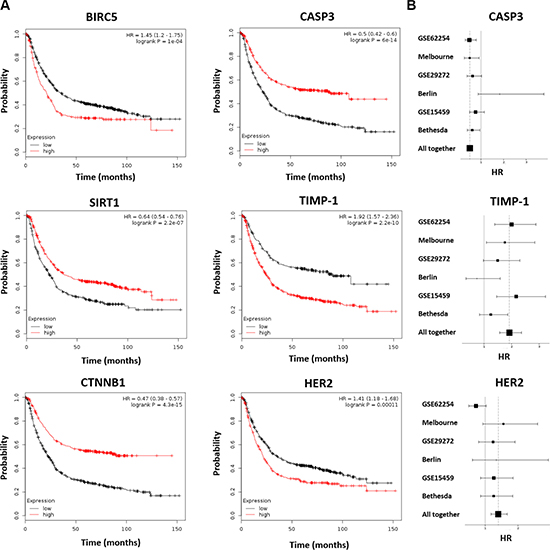
Figure 2: Survival for a selected set of the best performing markers. Kaplan-Meier survival plots show that higher expression of CASP3, CTNNB1 and SIRT1 results in a better OS, while higher expression of BIRC5, TIMP-1 and HER2 lead to worse survival (A). Forest plots for CASP3, TIMP-1, and HER2 (B).
Kaplan-Meier survival plots for three of the best performing genes with higher expression correlated to better survival (CASP3, SIRT1 and CTNNB1) and for three of the strongest genes with higher expression correlating to worse survival (BIRC5, HER2 and TIMP- 1) are presented in Figure 2.
Multivariate analysis
When running a multivariate analysis for OS using all genes, BIRC5 (p = 0.0018), CASP3 (p = 2.3E-04), and CTNNB1 (p = 0.0011) were significant at p < 0.05. Marginal significance was achieved by SP1 (p = 0.07) and VEGF (p = 0.07). When also including stage in the multivariate model, only BIRC5 (p = 0.05) and stage (p = 1.1E-06) were significant. When including stage and age in the multivariate model, only HIF1alpha (p = 0.02), SPARC (p = 0.03), stage (p = 6.8E-07), and age (p = 0.002) were significant.
In case of FP, when including all genes in a multivariate Cox regression, BIRC5 (p = 0.0017), CASP3 (p = 9.7E-05), CTNNB1 (p = 0.01), MMP-2 (p = 0.0092), SIRT1 (p = 0.035), SPARC (p = 0.0024), and VEGF (p = 0.027) were significant at p < 0.05. However, when including stage or stage and age, only VEGF (p = 0.02), stage (p = 9.3E-07), and age (p = 0.01) remained significant. We have to note that the multivariate analysis used only a fraction of patients included in the univariate analysis, as not all patients had complete clinical annotation (n = 316 for OS and n = 240 for FP).
Correlation to proliferation and HER2 expression
We used the expression of MKI67 as a surrogate of proliferation and run a Spearman correlation analysis for all genes. MKI67 itself had a strong prognostic value when examined in a univariate analysis for both overall survival (p = 0.0017, HR = 1.32) and relapse-free survival (p = 0.0015 and HR = 1.39).
Positive correlation to MKI67 expression was delivered by BIRC5 (coeff = 0.57, p < 1E-20), uPAR (coeff = 0.27, p = 3.9E-19), mTOR (coeff = 0.26, p = 1.6E- 18), SPHK1 (coeff = 0.21, p = 2.1E-12), and HER2 (coeff = 0.21, p = 2.8E-12). Negative correlation was observed for CTGF (coeff = –0.34, p = 9.6E- 31), SPARC (coeff = -0.31, p = 3E-25), PECAM-1 (coeff = –0.30, p = 9.2E-24), and SIRT1 (coeff = –0.23, p = 1.6E-14). As higher expression of multiple genes with negative correlation resulted in better survival (e.g. CTGF, SIRT1), and higher expression of genes with positive correlation delivered worse survival (e.g. BIRC5, HER2, SP1), we computed the correlation between the achieved hazard rate and the correlation coefficient against MKI67 expression. This analysis delivered a borderline significance (coeff = 0.32, p = 0.04). The same analysis performed for HER2 identified SP1 (coeff = 0.26, p = 5.1E-18), BIRC5 (coeff = 0.26, p = 5.2E-18), and EGFR (coeff = 0.20, p = 4.9E-17) having the highest correlation between gene expression and HER2 expression.
Expression in non-tumor gastric tissues
The keywords “gastric” and “normal” GEO delivered 266 datasets. When reducing the search to individual platforms, nine datasets were generated with the GPL96, 35 datasets with the GPL570 and two datasets with the GPL571 platform. Of these, five datasets (GSE44740, GSE51725, GSE13911, GSE43346, and GSE3526) contained expression data for a total of 57 normal gastric tissue specimens.
When comparing gastric normal and tumor samples, of all 28 genes, 6 were significant below p < 0.01 and had a fold change increase over 1.5 (BIRC5, CTNNB1, HER2, MET, PECAM-1 and uPAR) while only one gene had a 1.5-fold change reduction at the same significance (MMP- 2). The means with 95% confidence intervals for these genes are presented in Figure 3. Supplementary Table 2 contains all the expression values with the Mann-Whitney p value for each gene.
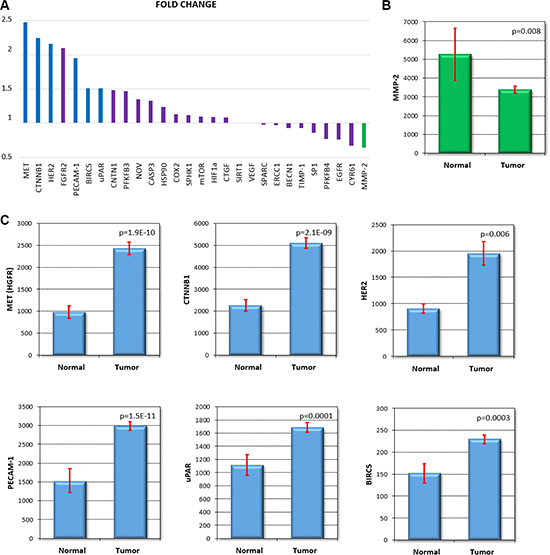
Figure 3: Expression change comparing normal and cancer tissue. All markers ranked by the fold change (A), MMP-2 was the only gene down regulated at p < 0.01 and FC < 0.66 (B). Six genes had an expression increase over 1.5 fold with a p < 0.01 (C). The normalized expression values are shown for each gene. p: Mann-Whitney p value comparing normal and tumor samples. Red bar: 95% confidence interval.
DISCUSSION
In this project, we performed a validation for previously identified biomarkers for gastric cancer outcome and prognosis prediction. To assemble a sizeable patient number, we collected data from three cancer centers around the globe (Europe, USA, Australia), and integrated with additional publicly available datasets. Then, in a literature search we selected 28 relevant markers from the past few years (2012–2015), which were disclosed to be related to survival of gastric adenocarcinoma. Analysis was restricted to the most relevant genes by assessing only markers which had been previously published in review manuscripts.
Most of the molecules were related to worse outcome, being negative prognostic factors. The significant genes include members of the epidermal growth factor family and related genes (EGFR, HER2, SP1) [11–13] , hypoxia-induced and angiogenic genes (HIF1A, VEGF, COX-2) [14–16], members of the MET and NOTCH signaling pathway (MET, CTNNB1, CNTN1) [17–19], regulators of survival and proliferation (SP1, CASP3, CTGF) [20–22], and genes involved in cellular motility (TIMP, MMP2) [23, 24]. All these genes are members of important pathways that contribute to progression of the neoplastic process through proliferation and survival by reprogramming the cell’s metabolism, mitochondrial functions, protein and lipid synthesis, cytoskeletal organization and signaling.
The strongest candidate gene was BIRC5 (baculoviral inhibitor of apoptosis repeat containing 5, also known as survivin) – although numerically other genes (like TIMP-1) reached a higher hazard rate, but survivin remained significant in a multivariate analysis as well. BIRC5 has multiple effects including inhibition of apoptosis, enhancing cell proliferation, and promotion of angiogenesis [25]. The correlation between survivin and gastric cancer survival was described in multiple studies [25, 26]. In addition to effect on survival, we observed the highest correlation between BIRC5 and MKI67 expression which supports the link between survivin expression and progression through the cell cycle. However, MKI67 expression per se was not correlated to survival in our Cox regression analysis suggesting that the main effect of BIRC5 on survival is independent of cell proliferation. Theoretically, targeted therapy could be pursued in gastric cancer with siRNA, e.g. LY2181308, is investigated against survivin in multiple types of cancer, however outcome data is pending. Furthermore, immunotherapy may hold promise for these patients as survivin is a strong antigenic peptide for the T cells [27].
Among the most promising markers is SIRT1 (Silent mating type information regulation 1), a key gene in the progression of gastric cancer. Earlier, it’s expression was associated with histological type, stage, lymph node status, and p53 expression [28], and proliferation as measured by Ki-67 index [29]. It was also identified as being down-regulated in gastric cancer and a key regulator of NFκB/Cyclin D1 signaling and G1 phase arrest [30], offering a possible therapeutic intervention in biological models. Here, our results confirm both the association between high SIRT1 and better survival and the correlation between SIRT1 and MKI67 expression. In theory, targeting SIRT1 can be utilized in cancer therapy, mostly cell cycle arrest in G1 phase showed promise in prostate cancer with DU145 [31].
Previously, receptor tyrosine kinase genomic alterations were detected in 20.6% of cases, affecting ERBB2, FGFR2, and MET, suggesting potential benefit from targeted therapy including MET-amplified gastric tumors and ERBB2 base substitutions [32]. Temporary but durable response to anti-MET agents have already been described [33]. Here, we observed a significant correlation between HER2 and MET and survival, but FGFR2 was not significant. Nevertheless, currently only one of the investigated genes, HER2 expression and amplification is utilized in the routine for prediction of response to anti-HER2 therapy [10].
Interestingly, the targetable genes (by administering e.g. cobimetinib, trastuzumab, and ponatinib, respectively, against) MET, HER2, and FGFR2 also displayed the highest difference with a fold change of the mean expressions over 2 when comparing gastric normal and cancer samples. However, again, FGFR2 was not significant – the reason for this is the uneven distribution of FGFR2 expression resulting in almost overlapping median expression at the same time.
Although our database represents a wide range of clinical cases, the patients are still not sufficiently characterized and this leads to a limitation of our study. While HER2 is an important marker also utilized clinically, our patient samples were collected before the introduction of anti-HER2 therapy. Thus, it was not possible to evaluate the effect of anti-HER2 therapy in the dataset. In the recent years, The Cancer Genome Atlas (TCGA) project proposed a molecular classification dividing gastric cancer into four subtypes [34]. We were also not able to validate the markers in these subtypes as the classification was not available for the patients included in the investigated datasets.
An alternative approach for survival prediction would be avoiding the utilization of a pre-defined gene to assign patients into prognostic cohorts. Rather, a whole transcriptome gene expression signature could be utilized to select molecularly similar patients and then determine prognostic expectations by evaluating the clinical outcome for these similar patients as has been demonstrated recently [35]. However, no similar methodology has been proposed for gastric cancer, thus we have not included such a model in our meta-analysis.
In summary, we collected gene expression data sets from three institutions and merged these with public datasets. Then, we performed a literature review and validated previously described markers for gastric cancer outcome. The major advantage of our analysis is that we evaluated all genes in the same set of patients thereby making direct comparison of the markers feasible. The best performing genes include BIRC5, CASP3, CTNNB1, TIMP-1, MMP-2, SIRT, SPARC, and VEGF. The importance of pathological parameters is supported by the fact that only a few genes remained significant when also including stage and age in a multivariate analysis.
MATERIALS AND METHODS
Identification of previously described biomarker candidates
We performed a literature search in PubMed (http://www.ncbi.nlm.nih.gov/pubmed) restricted to the timeframe of 2012 to 2015 utilizing the keywords “gastric”, “cancer”, “survival”, “gene expression” and “biomarker”. To limit the analysis to the most promising markers we selected only the English language manuscripts that were categorized as reviews. We performed the search in November/2015, and then manually continued with review of the publications one by one. Helicobacter pylori infection as a predisposing factor was not analyzed as a prognostic factor of outcome as there was no gene expression marker directly correlated to the infection. We assigned unique gene identifiers for each gene using the HUGO Gene Nomenclature Database (http://www.genenames.org/).
Database setup
We assembled a gastric cancer database using samples measured in three different sources including previously partly published data at the Max Delbrück Center for Molecular Medicine, Berlin, Germany (“Berlin dataset”, published in GEO as GSE22377) [36]; at the Transgenic Oncogenesis and Genomics Section, Laboratory of Cancer Biology and Genetics, National Cancer Institute, Bethesda, Maryland, USA (“Bethesda dataset”, published in GEO as GSE14210) [37]; and at the Peter MacCallum Cancer Centre, Melbourne, Australia (“Melbourne dataset”, published in GEO as GSE51105) [38]. Sample collection, hybridization, and gene expression measurements were described previously. The clinical data was updated for each dataset at the end of 2014 and we utilized in the analysis the aggregate database containing all samples with available follow-up data.
Publicly available datasets
We further extended the database using gene expression data downloaded from GEO. For this, we utilized the keywords “gastric”, “cancer”, “GPL96”, and “GPL570” to search GEO (http://www.ncbi.nlm.nih.gov/geo/). Only publications with available raw data, clinical survival information, and at least 15 patients were included. Affymetrix HG-U133A (GPL96) and HG-U133 Plus 2.0 (GPL570) microarrays were considered because of their overlapping set of 22,277 probe sets and because of our datasets were also derived using these gene chips.
Database of normal gastric samples
To discriminate genes related to carcinogenesis, we assembled a database of normal tissues. For this, we used the keywords “gastric” and “normal” in GEO without any limitation regarding publication time or sample number within the study. We included only the GPL96, GPL570, and GPL571 platforms in the search. Samples with premalignant conditions such as intestinal metaplasia were not included as “normal”.
Statistical analyses
The raw CEL files were MAS5 normalized in the R statistical environment (http://www.r-project.org) using the Affy Bioconductor library. Quality control for gene chips and control for duplicate samples were performed as described previously [39]. Only arrays passing the quality criteria were utilized. After normalization, only probes measured on both GPL96 and GPL570 were retained (n = 22,277). We subsequently performed a second scaling normalization to set the average expression on each chip to 1000 to reduce batch effects [40]. Kaplan–Meier survival plot and the hazard ratio with 95% confidence intervals and log-rank P values were calculated and plotted in R using Bioconductor packages. False discovery rate (FDR) was computed to correct for multiple testing using the brainwaver library in R as described previously [41] – the FDR cutoff was set at 5%. Expression in cancerous and normal samples was compared using a Mann-Whitney U-test.
Multivariate analysis
We performed a multivariate analysis using Cox proportional hazards regression including the gene expression markers and clinical variables including stage, age, Lauren classification, differentiation, and gender. In addition to the clinical data, we also determined the HER2 and MKI67 expression using data provided on the gene chips. We computed HER2 status by using the probe set 216836_s_at and setting the cutoff for positivity at 4800 [42]. To assess correlation to proliferation, Spearman correlation to MKI67 expression (probes set 212021_s_at) was computed for each of the genes separately [43]. In addition, Spearman correlation was also run for HER2 without using the dichotomization.
ACKNOWLEDGMENTS AND FUNDING
BG was supported by the OTKA K108655 grant. AMSz was supported by a János Bolyai Research Scholarship of the Hungarian Academy of Sciences.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. International journal of cancer. 2015; 136:E359–386.
2. Hamashima C, Shibuya D, Yamazaki H, Inoue K, Fukao A, Saito H, Sobue T. The Japanese guidelines for gastric cancer screening. Jpn J Clin Oncol. 2008; 38:259–267.
3. Boku N. HER2-positive gastric cancer. Gastric Cancer. 2014; 17:1–12.
4. Climent M, Hidalgo N, Vidal O, Puig S, Iglesias M, Cuatrecasas M, Ramon JM, Garcia-Albeniz X, Grande L, Pera M. Postoperative complications do not impact on recurrence and survival after curative resection of gastric cancer. Eur J Surg Oncol. 2015.
5. Valentini V, Cellini F, Minsky BD, Mattiucci GC, Balducci M, D’Agostino G, D’Angelo E, Dinapoli N, Nicolotti N, Valentini C, La Torre G. Survival after radiotherapy in gastric cancer: systematic review and meta-analysis. Radiotherapy and oncology. 2009; 92:176–183.
6. Webb A, Scott-Mackie P, Cunningham D, Norman A, Andreyev J, O’Brien M, Bensted J. The prognostic value of serum and immunohistochemical tumour markers in advanced gastric cancer. Eur J Cancer. 1996; 32a:63–68.
7. Yasui W, Sentani K, Sakamoto N, Anami K, Naito Y, Oue N. Molecular pathology of gastric cancer: research and practice. Pathol Res Pract. 2011; 207:608–612.
8. He CZ, Zhang KH, Li Q, Liu XH, Hong Y, Lv NH. Combined use of AFP, CEA, CA125 and CAl9–9 improves the sensitivity for the diagnosis of gastric cancer. BMC Gastroenterol. 2013; 13:87.
9. Peyre S, Di Napoli A, Pelissero A, Sategna-Guidetti C. Diagnostic usefulness of serum group I pepsinogen determination. Gastroenterologie clinique et biologique. 1983; 7:793–798.
10. Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T, Aprile G, Kulikov E, Hill J, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010; 376:687–697.
11. Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008; 19:1523–1529.
12. Chen C, Yang JM, Hu TT, Xu TJ, Yan G, Hu SL, Wei W, Xu WP. Prognostic role of human epidermal growth factor receptor in gastric cancer: a systematic review and meta-analysis. Archives of medical research. 2013; 44:380–389.
13. Jiang W, Jin Z, Zhou F, Cui J, Wang L. High co-expression of Sp1 and HER-2 is correlated with poor prognosis of gastric cancer patients. Surg Oncol. 2015; 24:220–225.
14. Zhu CL, Huang Q, Liu CH, Lin XS, Xie F. Prognostic value of HIF-1alpha expression in patients with gastric cancer. Molecular biology reports. 2013; 40:6055–6062.
15. Liu L, Ma XL, Xiao ZL, Li M, Cheng SH, Wei YQ. Prognostic value of vascular endothelial growth factor expression in resected gastric cancer. Asian Pac J Cancer Prev. 2012; 13:3089–3097.
16. Cheng J, Fan XM. Role of cyclooxygenase-2 in gastric cancer development and progression. World J Gastroenterol. 2013; 19:7361–7368.
17. Peng Z, Zhu Y, Wang Q, Gao J, Li Y, Li Y, Ge S, Shen L. Prognostic significance of MET amplification and expression in gastric cancer: a systematic review with meta-analysis. PLoS One. 2014; 9:e84502.
18. Li LF, Wei ZJ, Sun H, Jiang B. Abnormal beta-catenin immunohistochemical expression as a prognostic factor in gastric cancer: a meta-analysis. World J Gastroenterol. 2014; 20:12313–12321.
19. Chen DH, Yu JW, Jiang BJ. Contactin 1: A potential therapeutic target and biomarker in gastric cancer. World J Gastroenterol. 2015; 21:9707–9716.
20. Jiang W, Jin Z, Zhou F, Cui J, Wang L, Wang L. High co-expression of Sp1 and HER-2 is correlated with poor prognosis of gastric cancer patients. Surg Oncol. 2015; 24:220–225.
21. Chen H, Yang X, Feng Z, Tang R, Ren F, Wei K, Chen G. Prognostic value of Caspase-3 expression in cancers of digestive tract: a meta-analysis and systematic review. Int J Clin Exp Med. 2015; 8:10225–10234.
22. Cheng TY, Wu MS, Hua KT, Kuo ML, Lin MT. Cyr61/CTGF/Nov family proteins in gastric carcinogenesis. World J Gastroenterol. 2014; 20:1694–1700.
23. Grunnet M, Mau-Sorensen M, Brunner N. Tissue inhibitor of metalloproteinase 1 (TIMP-1) as a biomarker in gastric cancer: a review. Scand J Gastroenterol. 2013; 48:899–905.
24. Shen W, Xi H, Wei B, Chen L. The prognostic role of matrix metalloproteinase 2 in gastric cancer: a systematic review with meta-analysis. J Cancer Res Clin Oncol. 2014; 140:1003–1009.
25. Liu JL, Gao W, Kang QM, Zhang XJ, Yang SG. Prognostic value of survivin in patients with gastric cancer: a systematic review with meta-analysis. PLoS One. 2013; 8:e71930.
26. Wang TT, Qian XP, Liu BR. Survivin: potential role in diagnosis, prognosis and targeted therapy of gastric cancer. World J Gastroenterol. 2007; 13:2784–2790.
27. Friedrichs B, Siegel S, Andersen MH, Schmitz N, Zeis M. Survivin-derived peptide epitopes and their role for induction of antitumor immunity in hematological malignancies. Leukemia & lymphoma. 2006; 47:978–985.
28. Cha EJ, Noh SJ, Kwon KS, Kim CY, Park BH, Park HS, Lee H, Chung MJ, Kang MJ, Lee DG, Moon WS, Jang KY. Expression of DBC1 and SIRT1 is associated with poor prognosis of gastric carcinoma. Clin Cancer Res. 2009; 15:4453–4459.
29. Feng AN, Zhang LH, Fan XS, Huang Q, Ye Q, Wu HY, Yang J. Expression of SIRT1 in gastric cardiac cancer and its clinicopathologic significance. Int J Surg Pathol. 2011; 19:743–750.
30. Yang Q, Wang B, Gao W, Huang S, Liu Z, Li W, Jia J. SIRT1 is downregulated in gastric cancer and leads to G1-phase arrest via NF-kappaB/Cyclin D1 signaling. Mol Cancer Res. 2013; 11:1497–1507.
31. Shukla S, Gupta S. Molecular mechanisms for apigenin-induced cell-cycle arrest and apoptosis of hormone refractory human prostate carcinoma DU145 cells. Molecular carcinogenesis. 2004; 39:114–126.
32. Ali SM, Sanford EM, Klempner SJ, Rubinson DA, Wang K, Palma NA, Chmielecki J, Yelensky R, Palmer GA, Morosini D, Lipson D, Catenacci DV, Braiteh F, et al. Prospective comprehensive genomic profiling of advanced gastric carcinoma cases reveals frequent clinically relevant genomic alterations and new routes for targeted therapies. Oncologist. 2015; 20:499–507.
33. Catenacci DVT, Henderson L, Xiao S-Y, Patel P, Yauch RL, Hegde P, Zha J, Pandita A, Peterson A, Salgia R. Durable Complete Response of Metastatic Gastric Cancer with Anti-Met Therapy Followed by Resistance at Recurrence. Cancer Discovery. 2011; 1:573–579.
34. Network TCGAR. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–209.
35. Gyorffy B, Karn T, Sztupinszki Z, Weltz B, Muller V, Pusztai L. Dynamic classification using case-specific training cohorts outperforms static gene expression signatures in breast cancer. Int J Cancer. 2015; 136:2091–2098.
36. Forster S, Gretschel S, Jons T, Yashiro M, Kemmner W. THBS4, a novel stromal molecule of diffuse-type gastric adenocarcinomas, identified by transcriptome-wide expression profiling. Mod Pathol. 2011; 24:1390–1403.
37. Kim HK, Choi IJ, Kim CG, Kim HS, Oshima A, Yamada Y, Arao T, Nishio K, Michalowski A, Green JE. Three-gene predictor of clinical outcome for gastric cancer patients treated with chemotherapy. Pharmacogenomics J. 2012; 12:119–127.
38. Busuttil RA, George J, Tothill RW, Ioculano K, Kowalczyk A, Mitchell C, Lade S, Tan P, Haviv I, Boussioutas A. A signature predicting poor prognosis in gastric and ovarian cancer represents a coordinated macrophage and stromal response. Clin Cancer Res. 2014; 20:2761–2772.
39. Gyorffy B, Surowiak P, Budczies J, Lanczky A. Online survival analysis software to assess the prognostic value of biomarkers using transcriptomic data in non-small-cell lung cancer. PLoS One. 2013; 8:e82241.
40. Fekete T, Raso E, Pete I, Tegze B, Liko I, Munkacsy G, Sipos N, Rigo J Jr, Gyorffy B. Meta-analysis of gene expression profiles associated with histological classification and survival in 829 ovarian cancer samples. International journal of cancer. 2012; 131:95–105.
41. Gyorffy B, Gyorffy A, Tulassay Z. The problem of multiple testing and solutions for genome-wide studies [Article in Hungarian]. Orv Hetil. 2005; 146:559–563.
42. Gyorffy B, Benke Z, Lanczky A, Balazs B, Szallasi Z, Timar J, Schafer R. RecurrenceOnline: an online analysis tool to determine breast cancer recurrence and hormone receptor status using microarray data. Breast Cancer Res Treat. 2012; 132:1025–1034.
43. Mihaly Z, Kormos M, Lanczky A, Dank M, Budczies J, Szasz MA, Gyorffy B. A meta-analysis of gene expression-based biomarkers predicting outcome after tamoxifen treatment in breast cancer. Breast Cancer Res Treat. 2013; 140:219–232.
44. He Y, Zhao X, Subahan NR, Fan L, Gao J, Chen H. The prognostic value of autophagy-related markers beclin-1 and microtubule-associated protein light chain 3B in cancers: a systematic review and meta-analysis. Tumour Biol. 2014; 35:7317–7326.
45. Deng Q, Yang H, Lin Y, Qiu Y, Gu X, He P, Zhao M, Wang H, Xu Y, Lin Y, Jiang J, He J, Zhou JX. Prognostic value of ERCC1 mRNA expression in non-small cell lung cancer, breast cancer, and gastric cancer in patients from Southern China. Int J Clin Exp Pathol. 2014; 7:8312–8321.
46. Jorgensen JT. Role of human epidermal growth factor receptor 2 in gastric cancer: biological and pharmacological aspects. World J Gastroenterol. 2014; 20:4526–4535.
47. Marano L, Chiari R, Fabozzi A, De Vita F, Boccardi V, Roviello G, Petrioli R, Marrelli D, Roviello F, Patriti A. c-Met targeting in advanced gastric cancer: An open challenge. Cancer letters. 2015; 365:30–36.
48. Minchenko OH, Tsuchihara K, Minchenko DO, Bikfalvi A, Esumi H. Mechanisms of regulation of PFKFB expression in pancreatic and gastric cancer cells. World J Gastroenterol. 2014; 20:13705–13717.
49. Qiu G, Li X, Che X, Wei C, He S, Lu J, Jia Z, Pang K, Fan L. SIRT1 is a regulator of autophagy: Implications in gastric cancer progression and treatment. FEBS Lett. 2015; 589:2034–2042.
50. Zhang Y, Wang Y, Wan Z, Liu S, Cao Y, Zeng Z. Sphingosine kinase 1 and cancer: a systematic review and meta-analysis. PLoS One. 2014; 9:e90362.
51. Wang Z, Hao B, Yang Y, Wang R, Li Y, Wu Q. Prognostic role of SPARC expression in gastric cancer: a meta-analysis. Arch Med Sci. 2014; 10:863–869.

