INTRODUCTION
Osteosarcoma is the most common bone cancer in children and adolescents [1-3]. Osteosarcoma usually arises in the distal femur and proximal tibia and proximal humerus.
Development of neo-adjuvant chemotherapy has increased the rate of overall survival and has enabled surgery, but the failure rate is still high, causing death in children and adolescents [4-7].
Novel more effective targets for osteosarcoma therapy are therefore needed. Toward this goal, αvβ3 integrin-over-expressing high-metastatic variants of the human osteosarcoma cell line 143B were previously isolated and termed 143B-LM4.
Over-expression of αvβ3 integrin in the variant selected for high-metastasis suggested that integrin maybe a promising target for osteosarcoma. The 143B-LM4 cells expressed green fluorescent protein (GFP) in the nucleus and red fluorescent protein (RFP) in the cytoplasm, and therefore could be imaged down to the subcellular level in vitro and in vivo [8].
Lung seeding by 143B-LM4 cells was directly imaged and found to be greatly inhibited by the anti-β1 integrin monoclonal antibody, AIIB2. AIIB2 also significantly inhibited spontaneous lung metastasis and increased survival of mice with orthotopically-growing 143B-RFP [9].
In the present study, we tested echistatin, a cyclic RGD peptide antagonist of αvβ3 integrin (disintegrin) [10], as a molecular-targeting drug in human metastatic osteosarcoma in vitro on the highly metastatic 143B-LM4 cell line which over-expresses αvβ3 integrin described above.
RESULTS AND DISCUSSION
Dual-color-labeled GFP- and RFP-expressing 143B-LM4 cells
The high-metastatic integrin-over-expressing 143B-LM4 cells have a strikingly bright GFP in the nucleus and RFP in the cytoplasm, in vitro (Figure 1).
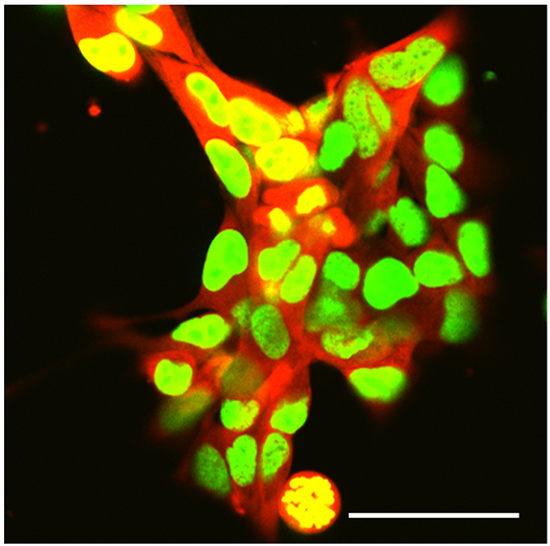
Figure 1: Dual-color selected 143B-LM4 human osteosarcoma cells expressing GFP in the nucleus and RFP in the cytoplasm in vitro. Images were obtained with a Fluoview FV1000 laser-scanning confocal microscope (Olympus Corp., Tokyo, Japan). GFP was excited at 488 nm, RFP at 543nm. Magnification 60x. Scale bar: 50 µm.
Echistatin inhibits cell proliferation, migration, invasion, and adhesion of 143B-LM4 cells
To determine whether echistatin inhibited 143B-LM4 cell proliferation, 143B-LM4 cells were treated with echistatin at various concentrations. 143B-LM4 cell proliferation decreased after treatment with echistatin in a time-dependent and a dose-dependent manner (P <0.01) (Figure 2A). After 24 hr treatment, 143B-LM4 cell proliferation was decreased to 44.0% at 0.5 μg/mL; 34.8% at 1.0 μg/mL echistatin; and 28.1% at 5.0 μg/mL echistatin, compared to control (P <0.01, respectively). At 72 hr after treatment, cell proliferation decreased to 74.2% at 0.1 μg/mL; 35.1% at 0.5 μg/mL echistatin, 19.1% at 1.0 μg/mL; and to 4.2% at 5.0 μg/mL echistatin, compared to control (P <0.01, respectively). Fluorescence microscopy showed that cell number decreased in a dose-dependent manner and the cancer cells appeared more shrunken at a high concentration of echistatin (Figure 2B).
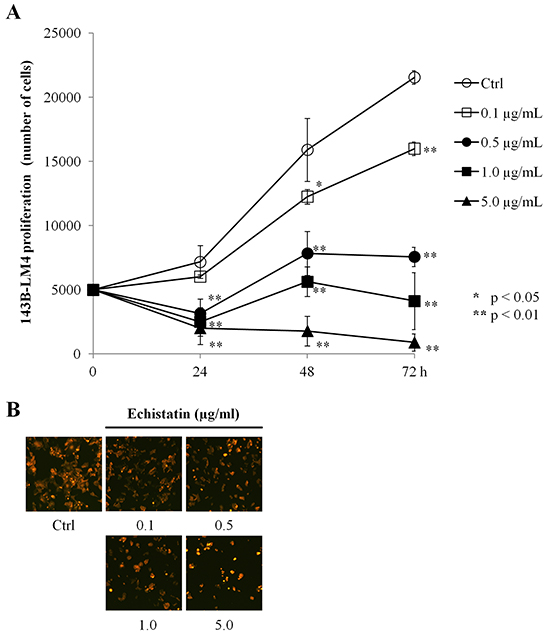
Figure 2: Echistatin decreased proliferation of 143B-LM4 cells in vitro. A. Efficacy of echistatin on 143B-LM4 cell proliferation. Proliferation of 143B-LM4 cells was inhibited by echistatin and decreased in a time-dependent and a dose-dependent manner (P <0.01). Error bars: SEM. B. visualization of the efficacy of echistatin at various concentrations on 143B-LM4 cell proliferation at 72 hours. Cell number decreased in a dose-dependent manner. Images were obtained with the Olympus IX71 fluorescence microscope. Magnification, 20×.
The effect of echistatin on the ability of 143B-LM4 cells to migrate was then tested. In vitro, migration of 143B-LM4 cells decreased to 59.4% at 1.0 μg/mL and to 8.5% at 5.0 μg/mL echistatin, compared to control (P <0.01 respectively) (Figure 3A).
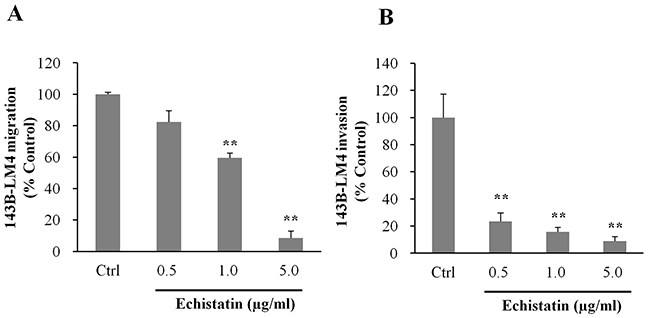
Figure 3: Echistatin decreased migration and invasion of 143B-LM4 cells in vitro. A. Efficacy of echistatin on 143B-LM4 cell migration in vitro. 143B-LM4 cells were seeded into the upper compartment of transwell chambers (Corning® HTS Transwell-96 uncoated plates, (Tewksbury, MA). Migration of 143B-LM4 cells decreased in a dose-dependent manner (P <0.01). Error bars: SEM. B. Efficacy of echistatin on 143B-LM4 invasion in vitro. 143B-LM4 cells were seeded into the upper compartment of transwell chambers with the surface coated with a basement membrane extract. Invasion of 143B-LM4 cells decreased in a dose-dependent manner (P <0.01). Error bars: SEM. Absorbance was evaluated with a plate reader after cells which migrated or invaded were treated with MTS.
Invasion of 143B-LM4 cells decreased to 23.3% at 0.5 μg/mL, 15.7% at 1.0 μg/mL and to 9.0% at 5.0 μg/mL echistatin, compared to control (P <0.01, respectively) (Figure 3B).
To determine whether echistatin could inhibit adhesion to vitronectin, which is a specific ligand of αvβ3 integrin, 143B-LM4 cells were seeded on vitronectin coated-dishes and treated with echistatin. Adhesion to vitronectin of 143B-LM4 cells decreased to 18.5% at 0.5 μg/mL, 14.6% at 1.0 μg/mL and to 6.5% at 5.0 μg/mL echistatin, compared to control (P <0.01, respectively) (Figure 4).
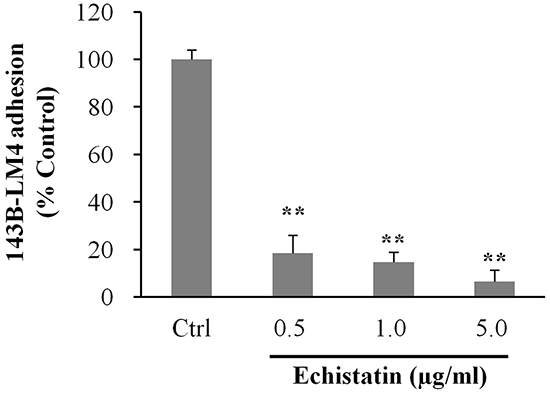
Figure 4: Echistatin decreased adhesion to vitronectin of 143B-LM4 cells in vitro. Efficacy of echistatin on 143B-LM4 cell adhesion to Vitronectin (Trevigen, Gaithersburg, MD) in vitro (Corning® HTS Transwell-96 plates (Tewksbury, MA) coated with Vitronectin. Adhesion of 143B-LM4 cells decreased in a dose-dependent manner (P <0.01). Error bars: SEM. Absorbance was evaluated with a plate reader after adherent cells were treated with MTS.
Aggressive chemotherapy of osteosarcoma in patients with metastatic or recurrent disease, most commonly in the lung [11-13], still results in poor prognosis with less than a 20% 5-year overall survival rate [14-16]. Therefore, novel targets are needed to overcome recurrence or metastasis and to improve the disease-free survival rate.
In the present study, we demonstrated that echistatin resulted in a significant decrease of cell proliferation, migration, invasion, and adhesion of 143B-LM4 cells in vitro. As 143B-LM4 is a high-metastatic variant that over-expresses αvβ3 integrin, these results suggest that αvβ3 integrin may be an effective target for osteosarcoma that has future clinical potential.
Previously-developed concepts and strategies of highly-selective tumor targeting can take advantage of molecular targeting of tumors, including tissue-selective therapy which focuses on unique differences between normal and tumor tissues [17-22].
MATERIALS AND METHODS
Cells
143B-LM4 cells [8] expressing GFP in the nucleus and RFP in the cytoplasm (Figure 1) were generated, as previously described, and maintained in RPMI 1640 medium (Irvine Scientific, Santa Ana, CA) containing 15% fetal bovine serum (FBS) (Omega Scientific, San Diego, CA) and 1% penicillin/streptomycin at 37°C in 5% CO2.
Drug
Echistatin [10] (Tocris Bioscience, Ellisville, MO) was diluted with distilled water and stored at -20°C.
Cell proliferation assay
143B-LM4 cells (5×103) were added to 96-well tissue culture plates and placed overnight at 37°C in a CO2 incubator. The cells were treated with various concentrations of echistatin. At various time intervals, 100 µl of fresh medium was replenished and 20 µl of 3-(4, 5-dimethylatiazol-2yl)-5-(3-carboxymethoxyphenyl)-2H-tetrazolium (MTS) (Promega, Madison, WI) was added to the cells. After incubation for 1 h, absorbance was measured using a microplate reader (TECAN Group Ltd., Männedorf, Switzerland) at 490 nm. The assay was performed in triplicate and at least twice. Cell images were observed with a fluorescence microscope (IX71, Olympus Corp., Tokyo, Japan).
Migration and invasion assay
An in vitro migratory/invasiveness assay was carried out with Corning® (Tewksbury, MA) HTS Transwell-96 plates uncoated or coated, respectively, with a basement membrane extract (Trevigen, Gaithersburg, MD) according to manufacturer’s instructions. 143B-LM4 cells (5×104) were added to the upper chamber and various concentrations of echistatin were added to the lower chamber (0.5 µg/mL, 1.0 µg/mL, 5.0 µg/m), for both the migration and invasion assays. The lower chamber had the same conditions for the migration and invasion assays. For the migration assay, an uncoated well was used for the upper chamber. For the invasion assay, a well coated with a basement membrane was used as the upper chamber. For both assays, cancer cells were seeded in the upper chamber. The plate was placed for 24 h at 37°C in a tissue culture incubator. After incubation for 24 h, 100 µl of fresh medium was gently replenished in the lower chamber and 20 µl MTS was added to the lower chamber to determine cell viability. After incubation for 1 h, the absorbance was measured using a microplate reader at 490 nm. The assays were performed in triplicate and at least twice, independently.
Adhesion assay
The adhesion assay was carried out with CultureCoat® Vitronectin 96-well dishes (Trevigen) according to the manufacturer’s instructions. 143B-LM4 cells were labeled with 2 µM calcein AM (Invitrogen, Carlsbad, CA), harvested and then re-suspended in medium to a final concentration of 1.5×105 cells/ml. Only live cells can absorb this agent. 143B-LM4 cells (1.5×104/100 µl) were added to each well and were either left untreated or treated with echistatin (0.5 µg/mL, 1.0 µg/mL, 5.0 µg/mL), and placed for 1 h at 37°C in a CO2 incubator. The medium was gently removed. The wells were washed twice gently with PBS. The fluorescence intensity of the cells remaining on the bottom of the well was measured using a SPECTRAmax GEMINI Dual-Scanning Microplate Spectrofluorometer, (Molecular Devices, Sunnyvale, CA) at an excitation/emission wavelength of 485/520 nm. The assay was performed in triplicate and at least twice independently.
Microscopy
In vitro 143B-LM4 cells were imaged with a Fluoview FV1000 laser-scanning confocal microscope (Olympus Corp., Tokyo, Japan) with a XLUMPLFL 60xW (0.90 NA) water-immersion objective [23]. GFP was excited at 488 nm, RFP at 543 nm. In the proliferation assay, cells were imaged with a fluorescence microscope (IX71, Olympus Corp., Tokyo, Japan).
Statistical analysis
The data are presented as mean ± SD or mean ± SEM. Statistical analysis was with ANOVA for multiple data sets. P-values of less than 0.05 were considered significant.
DEDICATION
This paper is dedicated to the memory of A. R. Moossa, M.D. and Sun Lee, M.D.
ACKNOWLEDGMENTS
This work was supported in part by a grant from the Nakatomi Foundation.
CONFLICTS OF INTEREST
No potential conflicts of interest were disclosed.
REFERENCES
1. Hong WK, Bast RC Jr, Hait WN, Kufe DW, Pollock RE, Weichselbaum RR, Holland JF, Frei E III. Holland-Frei Cancer Medicine, 8th edition, People’s Medical Publishing House, Shelton, CT, 2010.
2. Huvos AG. Osteogenic sarcoma. In: Bone Tumors: Diagnosis, Treatment and Prognosis. Second edition. Huvos AG (eds.). Philadelphia: WB Saunders; pp. 85-155, 1991.
3. Unni KK. Osteosarcoma. In: Dahlin’s Bone Tumors: General Aspects and Data on 11,087 Cases. Fifth edition. Unni KK and Inwards CY (eds.). New York: Lippincott-Raven, pp. 143-183, 1996.
4. Tsuchiya H, Tomita K, Mori Y, Asada N, Morinaga T, Kitano S, Yamamoto N. Caffeine-assisted chemotherapy and minimized tumor excision for nonmetastatic osteosarcoma. Anticancer Res 1998;18:657-666.
5. Bacci G, Picci P, Ferrari S, Ruggieri P, Casadei R, Tienghi A, Brach del Prever A, Gherlinzoni F, Mercuri M, Monti C. Primary chemotherapy and delayed surgery for nonmetastatic osteosarcoma of the extremities. Results in 164 patients preoperatively treated with high doses of methotrexate followed by cisplatin and doxorubicin. Cancer 1993;72:3227-3238.
6. Tsuchiya H, Wan SL, Sakayama K, Yamamoto N, Nishida H, Tomita K. Reconstruction using an autograft containing tumour treated by liquid nitrogen. J Bone Joint Surg Br 2005;87:218-225.
7. Torbert TJ, Fox EJ, Hosalkar HS, Ogilvie CM, Lackman RD. Endoprostethic reconstructions: Results of long-term follow-up of 139 patients. Clin Orthop Relat Res 2005;438:51-59.
8. Tome Y, Kimura H, Maehara H, Sugimoto N, Bouvet M, Tsuchiya H, Kanaya F, Hoffman RM. High lung-metastatic variant of human osteosarcoma cells, selected by passage of lung metastasis in nude mice, is associated with increased expression of αvβ3 integrin. Anticancer Res 2013;33:3623-3628.
9. Kimura H, Tome Y, Momiyama M, Hayashi K, Tsuchiya H, Bouvet M, Hoffman RM. Imaging the inhibition by anti-β1 integrin antibody of lung seeding of single osteosarcoma cells in live mice. Int J Cancer 2012;131:2027-2033.
10. Del Gatto A, Zaccaro L, Grieco P, Novellino E, Zannetti A, Del Vecchio S, Iommelli F, Salvatore M, Pedone C, Saviano M. Novel and selective alpha(v)beta3 receptor peptide antagonist: design, synthesis, and biological behavior. J Med Chem 2006;49:3416-3420.
11. Chi SN, Conklin LS, Qin J, Meyers PA, Huvos AG, Healey JH, Gorlick R. The patterns of relapse in osteosarcoma: the Memorial Sloan-Kettering experience. Pediatr Blood Cancer 2004;42:46-51.
12. Crompton BD, Goldsby RE, Weinberg VK, Feren R, O’Donnell RJ, Ablin AR. Survival after recurrence of osteosarcoma: a 20-year experience at a single institution. Pediatr Blood Cancer 2006;47:255-259.
13. Gelderblom H, Jinks RC, Sydes M, Bramwell VH, van Glabbeke M, Grimer RJ, Hogendoorn PC, McTiernan A, Lewis IJ, Nooij MA, Taminiau AH, Whelan J, European Osteosarcoma Intergroup. Survival after recurrent osteosarcoma: data from 3 European Osteosarcoma Intergroup (EOI) randomized controlled trials. Eur J Cancer 2011;47:895-902.
14. Meyers PA, Heller G, Healey JH, Huvos A, Applewhite A, Sun M, LaQuaglia M. Osteogenic sarcoma with clinically detectable metastasis at initial presentation. J Clin Oncol 1993;11:449-453.
15. Bacci G, Briccoli A, Mercuri M, Ferrari S, Bertoni F, Gasbarrini A, Fabbri N, Cesari M, Forni C, Campanacci M. Osteosarcoma of the extremities with synchronous lung metastases: long-term results in 44 patients treated with neoadjuvant chemotherapy. J Chemother 1998;10:69-76.
16. Kimura H, Tsuchiya H, Shirai T, Nishida H, Hayashi K, Takeuchi A, Ohnari I, Tomita K. Caffeine-potentiated chemotherapy for metastatic osteosarcoma. J Orthop Sci 2009;14:556-565.
17. Blagosklonny MV. Matching targets for selective cancer therapy. Drug Discov Today 2003;8:1104-1107.
18. Blagosklonny MV. Teratogens as anti-cancer drugs. Cell Cycle 2005;4:1518-1521.
19. Blagosklonny MV. Treatment with inhibitors of caspases, that are substrates of drug transporters, selectively permits chemotherapy-induced apoptosis in multidrug-resistant cells but protects normal cells. Leukemia 2001;15:936-941.
20. Blagosklonny MV. Target for cancer therapy: proliferating cells or stem cells. Leukemia 2006;20:385-391.
21. Apontes P, Leontieva OV, Demidenko ZN, Li F, Blagosklonny MV. Exploring long-term protection of normal human fibroblasts and epithelial cells from chemotherapy in cell culture. Oncotarget 2011;2:222-233. doi: 10.18632/oncotarget.248.
22. Blagosklonny MV. Tissue-selective therapy of cancer. Br J Cancer 2003;89:1147-1151.
23. Uchugonova A, Duong J, Zhang N, König K, Hoffman RM. The bulge area is the origin of nestin-expressing pluripotent stem cells of the hair follicle. J Cell Biochem 2011;112:2046-2050.

