Introduction
In many aspects of development and disease, critical genetic and epigenetic alterations of genes encoding glycosyltansferases can cause pathologic changes [1-4]. N-linked and O-linked sugars are the two major glycosylation forms observed in secreted and cell-surface proteins. N-linked sugars contribute to protein folding, secretion and stability. Endoplasmic reticulum stress responses and cellular apoptosis are activated when N-glycosylation is disrupted. O-glycosylated proteins are also found in cell surface, serum and in the extracellular matrix (ECM). Therefore, altered cell-surface O-glycoproteins are often implicated in uncontrolled proliferation, invasion and metastasis [5], and similarly, the O-glycosylated ECM proteins are often involved in a variety of developmental pathologies. The intracellular localization of N-acetylgalactosaminyltransferases may become altered in cancer cells by redistributing to the endoplasmic reticulum, rather than being restricted to the Golgi [6, 7]. Alterations in protein O-glycosylation by a family of over 20 polypeptide GalNAc-transferases (GalNAcTs or GALNTs) catalyzing the transfer of N-acetylgalactosamine (GalNAc) from UDP-GalNAc to the hydroxyl group of a serine or threonine residue have been linked with a wide variety of epithelial developmental defects and neoplasms [4-6, 8-10]. The unusually large number of GalNAcTs is unique to O-glycosylation and the multiplicity of conserved isoforms in metazoan evolution suggests a need for cell or tissue-specific isoforms [11].
The GalNAcTs generate Tn antigens by transferring αGalNAc from UDP-GalNAc to Ser and Thr residues [12]. This Tn antigen may become sialylated on C6 to form sialyl-Tn antigen, or galactosylated on C3 to form the T antigen (or TF: Thomsen-Friedenreich, antigen) [12].
Studies on domain organizations of the different GalNAcTs have provided essential information on their enzymatic functions [13]. The type II transmembrane GalNAcTs insert into the Golgi membranes via a non-cleaved membrane-spanning domains and are connected with a luminal catalytic domain by a stem region. The catalytic domain of GalNAcTs is linked to a C-terminal, ricin-like lectin domain, a unique feature of eukaryotic GalNAcTs’. The linker sequence between catalytic and lectin domains is also important for the lectin domain to assist in the catalytic activity of the enzyme, but distinct GalNAc glycopeptide binding properties of different GalNAcTs lectin domains suggest additional functions beyond the regulation of catalytic domains [11, 13, 14].
GalNAc-type O-glycosylation may also correlate with proprotein convertase processing of >3000 protein substrates [15]. For instance, O-glycosylation in the vicinity of preprocessing sites have been shown to be altered in the rare disease familial tumoral calcinosis [16]. Using synthetic glycopeptides, Schjoldlager et al. 2011 have shown that O-glycans located +/- 3 residues from the RXXR furin cleavage site exert a co-regulatory role in preprotein processing [15].
Expression of GALNTs and their encoded GalNAcT proteins occurs in a wide range of tissues [6, 7, 17-19]. The distinct functions of their domains in the glycosylation process itself and their preferences for peptides or glycopeptides may contribute to a wide variety of disease-risks [4, 11, 13, 18, 20-24]. This review intends to highlight the neoplastic contexts where GALNTs are expressed and show their potential clinical usefulness for diagnostic and prognostic purposes.
N-Acetylgalactosaminyltransferase 1
Aberrant glycosylation resulting from mutations in GALNT1 was found to be involved in cardiac defects, and in melanoma, ovarian and bladder cancers [9, 11, 21, 23, 25, 26].
Expression of GALNT1 is required for O-glycosylation of many proteins of the extracellular matrix and basement membrane that regulate the normal heart valve development and submandibular gland development. Consequently, the loss of GALNT1 increases the bone morphogenetic protein (BMP) kinase and mitogen-activated protein kinase (MAPK) signaling, which are major contributing factors to the multiplication of endocardial cushion cells and integrin signaling in submandibular epithelial cells [9]. This phenotype suggests that transmembrane serine/threonine kinases are functionally altered in the absence GalNAcT1 and points to the incapacity of the TGFβ-family receptors to link the message of the agonist (BMP) to intracellular downstream signaling pathways (MAPK) [9].
In hepatocellular carcinoma (HCC), GalNAcT1 is frequently upregulated, which facilitates HCC cell migration and invasion by increasing the O-glycan addition to EGFR. Reciprocally, downregulation of GalNAcT1 decreases EGFR O-glycosylation and reduces the malignant behavior of HCC by decreasing EGF-stimulated EGFR phosphorylation. Hypophosphorylated EGFR is then internalized, which terminates EGF-induced signaling. In addition to EGF, downregulation of GalNAcT1 also decreases the PDGF- and VEGF-induced invasion of HCC [27].
In A375 human melanoma cells exposed to kojic acid (a tyrosinase inhibitor and skin whitener), seven likely tumor-suppressor genes— GALNT1, APOBEC1, ARHGEF16, CD22, FGFR3, UNC5C and ZNF146— were shown to be down-regulated, resulting in the loss of suppressor gene function [26]. The single-nucleotide polymorphism rs17647532 in GALNT1 was genotyped in fourteen studies to suggest association between GALNT1 alterations and ovarian cancer, but could not be confirmed in large populations [21, 25, 28]. In bladder cancer cells, expression of GALNT1-mRNA was higher by eleven-fold compared to normal cells, suggesting that hyperexpressed GALNT1 could be a novel marker for human bladder cancer [22]. In another report, a direct linkage between miR-129 and its putative targets SOX4 and GALNT1 have opened the possibility of a differential regulation of such genes in bladder cancer [23].
N-Acetylgalactosaminyltransferase 2
Different genome-wide association studies (GWAS) have documented the association of common variants of GALNT2 with both high-density lipoprotein cholesterol (HDLc) and triglyceride levels [24, 29]. A GWAS study identified the functional expression of GALNT2, TRIB1 and SORT1 in association with changes in lipid levels and degrees of heart disease [30]. Of the six newly identified chromosomal regions, one was associated with HDL cholesterol, two with low-density lipoprotein (LDL) cholesterol and five with triglycerides have been correlated with risk factors for cardiovascular disease [10]. Additionally, in mouse models, knockdown and overexpression of GALNT2 were inversely related to HDLc levels. Owing to the high homology between mouse and human transferase genes, the linkage of three newly identified human genes (GALNT2, WWOX, and CDH13) were suggested in silico to be associated with HDL levels [31].
GALNT2 overexpression altered the Tn antigen expression by neuroblastoma (NB) cells and suppressed malignant proliferation of NB cells, as a result of decreased dimerization of IGF-1R (Insulin-like growth factor receptor), a critical glycosylation target for GalNAcT2 [32]. The activation of IGF-1Rs following GALNT2 knockout suggests that non-glycosylated IGF-1R dimerizes and increases the IGF-l (Insulin-like growth factor)-induced signals for cancer cell growth, migration, and invasion. Reciprocally, over-expression of GALNT2 significantly inhibited IGF-l-stimulated growth, migration, and invasion of NB cells, suggesting that O-glycosylated IGF-1R did not dimerize and thus failed to support the malignant behavior of NB cells [32]. Quite differently, in oral squamous cell carcinoma (OSCC), GalNAcT2 enhanced the invasive properties of OSCC cells by O-glycosylation the EGFR, and was selectively demonstrable in cells of the invading front of the tumor. In gastric adenocarcinoma, downregulation of GALNT2 increased the cell proliferation, migration, invasion and tumor metastasis by increasing the phosphorylation of hepatocyte growth factor receptor (MET: a receptor tyrosine kinase like IGF-l) and decreasing the expression of the Tn antigen on MET [33]. Likewise, downregulation of GalNAcT2 in hepatocellular carcinoma (HCC) cells promoted cell growth, migration, and invasion in vitro and in vivo by modulating the O-glycan pattern on EGFR, resulting in EGFR activation by dimerization, and phosphorylation of downstream signaling proteins [34]. In breast cancer cells (MDA231), increased Tn antigen amounts were observed in the endoplasmic reticulum (ER) concomitantly with GalNAcT2, thereby suggesting a role for COPI-based translocation of GalNAcT2 to the ER in increasing Tn levels in the ER [7]. In tumors such as breast, OSCC, HCC, and gastric carcinoma, future development of specific inhibitors of GalNAcTs (and GalNAcT2 in particular) might have potential therapeutic use [35].
Expression of GALNT1, GALNT2, GALNT3, GALNT4, GALNT5, GALNT6, GALNT9 and GALNT14 in B-cells has highlighted the significant role of GALNTs in B-cell biology. In particular, GALNT2 activation was found necessary for the initiation of O- glycosylation of the IgA1 hinge region, but the lack of GALNT1 resulted in decreased immunoglobulin G production, increased germinal center B-cell apoptosis and reduced numbers of plasma cells [36]. In myeloid cells, the expression of GALNT2, GALNT4, GALNT5 and GALNT7 was observed in K562 cells (chronic myeloid leukemia line), and of GALNT1, GALNT2, GALNT3 and GALNT4 in SHI-1 (acute monocytic leukemia line) [36, 37].
N-acetylgalactosaminyltransferase 3
Owing to the high structural similarity between GALNT3 and GALNT6, and the similar enzymatic properties of their encoded products, Bennett et al. proposed a classification of the twenty GALNT genes into distinct gene families [11, 38]. In this classification, the structurally identical GALNT3 and GALNT6 with nine intron/exon boundaries have been grouped in the Ic subfamily [11]. Except for GALNT3 and GALNT6, no correlation was found between structure and co-expression of other transferases, either in the same cancer cell type, or in other specific clinicopathological contexts (Figure 1). Despite their structural similarities, GALNT3 and GALNT6 do not co-localize to a single locus, are differentially expressed (as in the WI38 fibroblast cell line) and glycosylate fibronectin differently [38, 39]. For instance, the GalNAcT6 enzyme, unlike GalNAcT3, O-glycosylates oncofetal fibronectin efficiently [38]. Likewise, GalNAcT6 was found to function as an independent prognostic marker in pancreatic cancer, although both GalNAcT3 and GalNAcT6 were co-expressed in a cohort of pancreatic cancer cases [40]. In another study of renal cell carcinomas (RCCs), GalNAcT3 was predicted as an independent prognostic factor for high-grade tumor and poor prognosis in RCC patients, whereas GalNAcT6 was not [41]. Finally, much more is needed to correlate GalNAcT3 and GalNAcT6 expression with their capacity to aberrantly O-glycosylate the Muc1, and how this could affect the morphology, invasiveness and migration properties of RCC [41, 42].
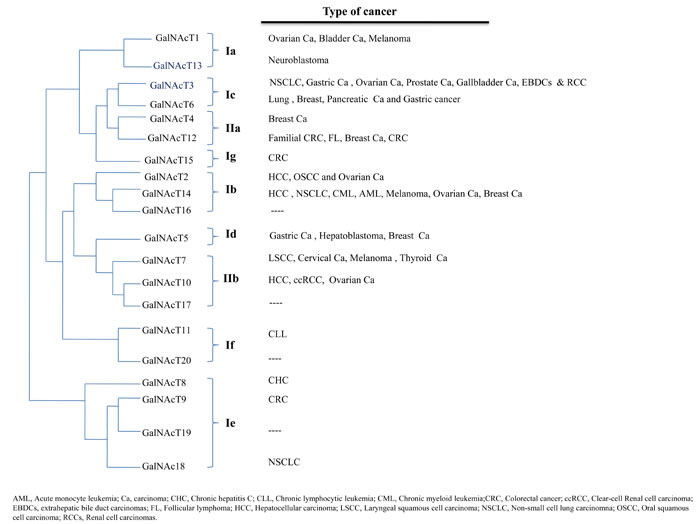
Figure 1: Phylogenetic tree showing the classification of GalNAcTs [11]. The implication of each transferase in individual cancers is shown in relationship to the phylogenetic tree.
Mutations in GALNT3 (c.484 C>T; p.R162X in exon1; c.1524+5G>A; splicing error in intron 7) impair the structure and function of GalNAcT3 and may cause modifications in growth regulation, immune recognition, and cell adhesion of cancer cells. Skipping of GALNT3 exon 7 caused an in-frame deletion of 44 amino acids, eliminating the linker region between the catalytic and lectin domains of GalNAcT3 [43]. Previous studies of GalNAcT3 glycosylating glycopeptide substrates have suggested that the lectin domain is involved in binding glycopeptide substrates [13]. The glycosylation of the endocrine FGF23 (fibroblast growth factor 23) by GalNAcT3 occurs via a lectin-dependent, selective O-glycosylation at Thr178. This site is adjacent to the proprotein convertase (PC) cleavage site of FGF23, such that O-glycosylation prevents FGF23 proteolytic degradation. The loss of active GalNAcT3 results in excessive FGF23 proteolytic processing and FGF23 deficit. However, prior to FGF23 Thr178 glycosylation, the first glycosylation occurs at the Thr171 in a lectin-independent manner and may serve as a target switching or enhancer for the second glycosylation at Thr178 [13, 16, 43].
The low expression of GALNT3 in non-small cell lung carcinoma (NSCLC) was shown to be an unfavorable prognostic factor for stage-I NSCLC and stage-I non-squamous cell carcinomas [44]. Altered GalNAcT3 expression may decrease protein O-glycosylation, resulting in altered biological activities of NSCLCs and shorter survival times [44-47]. Strong GalNAcT3 expression was characterized in both early-stage undifferentiated and differentiated gastric carcinomas whereas higher frequencies of metastatic lymph node involvement was seen in undifferentiated gastric carcinomas [48]. In addition, GalNAcT3 expression was correlated with the aggressive behavior of oesophageal squamous cell carcinomas [49]. In a recent report on papillary thyroid carcinoma (PTC), WNK1-B4GALNT3 fusion was correlated with the overexpression of B4GALNT3 [50]. In another study, GalNAcT3 expression in gastric carcinoma was found to positively correlate with differentiation. High expression of GalNAcT3 was more frequent in differentiated carcinomas and low expression of GalNAcT3 in less differentiated, more malignant carcinomas [11, 51].
In T2-stage gallbladder carcinomas (granular and diffuse-type), higher levels of mRNA and GalNAcT3 protein were detected than in adjacent non-cancerous tissues, and diffuse-type localization of GalNAcT3 deep in the subserosal layer correlated with postsurgical recurrence and poor survival [52]. Similarly, in prostate carcinoma, higher expression of GalNAcT3 was associated with increased numbers of malignant cells in the in situ epithelial lesions [53]. In a study of extrahepatic bile duct carcinomas (EBDCs), GalNAcT3 expression during tumor growth was found associated with lymph node metastasis [54]. Likewise, GalNAcT3 expression in RCC patients correlated with RCC invasion and metastasis, probably by decreasing O-glycosylation on cell-adhesion molecular markers β-catenin and E-cadherin and decreasing tumor cell adhesion to the stroma [41]. However, in other studies, weak GalNAcT3 expression was associated with poor differentiation and aggressiveness of ductal adenocarcinoma of the pancreas [41, 55].
N-acetylgalactosaminyltransferase 4
N-acetylgalactosaminyltransferase 4 (GALNT4)
encoding the fourth human N-acetylgalactosaminyltransferase (GalNAcT4) shares GalNAc-glycopeptide substrates with GalNAcT12, and was classified as group IIa [11].
Estrogens and estrogen receptors (ERs) are critical regulators of breast cell tumorigenesis. By quantitative, real-time PCR (qPCR), five genes (SYTL5, RAB27B, SNX24, GALNT4 and SLC12A2/NKCC1/BSC2) have been implicated in estrogen regulation [56]. How does the vesicle trafficking genes SYTL5, RAB27B, SNX24 and SLC12A2 cooperate with GALNT4 to mediate the 17β-estradiol (E2) signaling in breast cancer cells is another key question. Immunohistochemical detection of GalNAcT4 in renal tumor cells of relapse-free surviving patients with clear-cell renal cell carcinoma (ccRCC) strongly suggested that GALNT4 expression is a positive prognostic factor [57].
N-acetylgalactosaminyltransferase 5
The N-acetylgalactosaminyltransferase 5 gene with three unique intron positions did not show any significant relationship with other transferases and was classified as Id subfamily [11]. GalNAcT5 expression was found to be highly tissue-specific and catalyzing the glycosylation of a particular subset of peptides [14, 58]. Moreover, GalNAcT5 was localized in gastric epithelial cancer cells, and strong expression of GalNAcT5 correlated with well-differentiated gastric carcinoma whereas moderate or poor expression correlated with less differentiated carcinomas. In the same study, a closer follow-up and aggressive therapeutic treatment was advised for the gastric cancer patients with low intratumoral GalNAcT5 expression [59]. In breast cancers, presence of two somatic missense mutations (p.E507D and p.L692F in the catalytic domain) of GALNT5 appeared to reduce the transferase activity of GalNAcT5 [60]. However, the sequence analyses of GALNT5 in a group of thirty microsatellite-stable colon cancer cell lines failed to confirm the presence of these previously identified somatic mutations [60, 61].
Cytogenetic data of hepatoblastoma have revealed that up-regulation of GALNT5, DAPL1, ERMN, SCN1A and SCN3A plays a major role in the development and progression of the disease [62]. However, the post-translational modifications regulated by GalNAcT5 in hepatoblastoma cells remain undefined, as the mechanism of action of DAPL1, ERMN, GALNT5, SCN1A and SCN3A genes.
N-acetylgalactosaminyltransferase 6
The GALNT6 gene is structurally identical to GALNT3, and grouped in the Ic subfamily [11]. Numerous studies based on different experimental approaches have suggested a correlation between expression of GalNAcT6 and T3 with tumor differentiation [41]. However, only GalNAcT6 expression was predicted as a prognostic factor whereas GalNAcT3 was not [40, 63].
Most studies on GalNAcT6 have shown its pivotal role in breast cancers [63-65]. During human breast carcinogenesis, strong expression of GalNAcT6 in ductal carcinoma in situ (DCIS) was considered an early event leading to aberrant mucin O-glycosylation [63]. Moreover, the selective expression of GalNAcT6 in myoepithelial cells in some breast cancer patients was associated with angiogenesis and invasiveness [63].
In breast cancer cells, GalNAcT6 may cause aberrant glycosylation of Muc1, thus mediating mammary carcinogenesis by up-regulating cell adhesion molecules β-catenin and E-cad [66]. The level of GalNAcT6 was found significantly higher in breast cancer cells as compared to normal or benign mammary cells [64]. Moreover, fibronectin, an important in vivo substrate of GalNAcT6/GalNAcT3, was stabilized by GalNAcT6-induced O-glycosylation and did not undergo degradation after endocytosis. Overexpression of GALNT6 and O-glycosylation of fibronectin increased the transformation of mammary epithelial cells and abrogated their proliferative behavior in vivo [65]. This GalNAcT6-fibronectin pathway may therefore matter in breast cancer development and progression, but tumor heterogeneity and the multiplicity of GalNAcT6 substrates in breast cancer cells are still to be accounted for in the evaluation of the GalNAcT6-fibronectin pathway.
In response to cigarette smoke, degradation of E-cadherin (E-cad) in lung cancer cells resulted from shedding of a 400 kDa Muc1-N isoform, leading to Muc1-C glycosylation and complex formation with p120ctn through bridging of Src/Muc1-C/galectin-3/EGFR signalosomes. This smoke-induced Muc1-C glycosylation and Muc1-C/p120ctn interaction was suppressed with GALNT6 shRNA, thereby inhibiting E-cad degradation— a major hallmark of epithelial-mesenchymal transition (EMT)— by blocking the smoke-induced Muc1-N shedding and maintaining cellular polarity [67].
N-acetylgalactosaminyltransferase 7
GalNAcT7 is a peptide-preferring transferase that was grouped with GALNT10 and GALNT17 in the IIb subfamily [11].
Overexpression of GALNT7 is associated with carcinogenesis and metastasis in laryngeal squamous cell carcinoma (LSCC) [1], hepatocellular carcinoma [68], cervical cancer [2], superficial spreading melanoma (SSM) and nodular melanoma (NM) [69]. Through reduced expression of GALNT7, miR-34a and miR-34c are tumor suppressors in laryngeal squamous carcinoma cell (LSCC), thereby serving as novel potential markers for LSCC therapy [1]. Likewise, down-regulation of miR-214 and up-regulation of its target gene GALNT7 have been documented in cervical cancer cells but not in adjacent normal tissues [2]. Superficial spreading melanoma (SSM) and nodular melanoma (NM characteristically express the eight genes GALNT7, DIS3, FGFR1OP, G3BP2, MTAP, SEC23IP, USO1, and ZNF668 [69].
Up-regulation of miR-30d or silencing of GALNT7 has significant effects on the O-glycosylation of melanomas. During melanoma progression, the expression of GALNT7 is decreased by miR-30b/30d, which promotes cell invasion and immunosuppression by altering the O-glycosylation patterns of membrane proteins interacting with the ECM and cells of the tumor environment [20]. The suppression of GALNT7 with upregulation of miR-30d increases the secretion of IL-10, providing a possible mechanistic explanation for the immunosuppressive behavior of melanomas [20].
Moreover, GALNT7, together with the PLA2 and SIAT8B genes were shown to encode and metabolize the ligands (e.g. acetylated galactosamine by GalNAcT7) to which cytomegalovirus (CMV) herpes simplex and influenza virus bind, and are further used by the epidermal growth factor receptor (EGFR). These genes were also shown to encode an interleukin (IL10)-like protein that binds IL10R [70].
In young-onset, post-Chernobyl papillary thyroid cancers (PTCs), eleven genes: ACVR2A, AJAP1, CA12, CDK12, FAM38A, GALNT7, LMO3, MTA1, SLC19A1, SLC43A3, ZNF493, were differentially expressed in an iodine-131 dose-dependent manner following ionizing radiation [71]. Remarkably, of the eleven, four genes, CA12, GALNT7, LMO3, and SLC43A3, were previously found deregulated in post-Chernobyl thyroid cancer [72].
N-acetylgalactosaminyltransferase 8
GALNT8 was classified in the Ie subfamily with GALNT9, GALNT18 and GALNT19 [11], however, in another classification system, the GALNT8, together with GALNT9, GALNT17 and GALNT18, were defined as the Y-subfamily [73].
Through large-scale association analysis including over a thousand Japanese patients with chronic hepatitis C (CHC), GALNT8 variants were found associated with the outcome of interferon therapy [74]. In another report on a Pakistani population, no such correlation was apparent for the GALNT8 variant rs10849138 [75]. However, further large scale population-based studies are needed to assess the role of rs10849138 in this Pakistani population.
N-acetylgalactosaminyltransferase 9
The N-acetylgalactosaminyltransferase 9 gene (GALNT9) belongs to both Y-subfamily [73] and Ie subfamily of GALNTs [11].
Among thirty-three oncogenes, fourteen genes including GALNT9 have been characterized with a novel hotspot mutation in colorectal cancer (CRC) [76]. Although the molecular mechanism involving GALNT9 and other factors in low-risk neuroblastoma patients remain largely unknown, the GALNT9 has been validated as a prognostic marker that may be helpful to guide therapy in low-risk neuroblastoma patients [77]. Lastly, a recent bioinformatic screen suggested that GALNT9, together with CCDC8 (involved in microtubule regulation) and BNC1 (transcription factor), may support brain metastatisation of breast carcinoma [78].
N-acetylgalactosaminyltransferase 10
GalNAcT10 is a glycopeptide-preferring transferase exhibiting unique sequence-recognition properties and stands out among other glycopeptide-preferring transferases [11, 13].
In the liver, GALNT10 promotes carcinogenesis and was recognized as a bona fide target for miR-122, a liver-specific mammalian miRNA. Moreover, in hepatitis B virus (HBV)-associated hepatocellular carcinoma (HCC), hepatocyte nuclear factor 4α (Hnf4α) was found to activate miR-122 gene transcription. The reduced expression of miR-122 might facilitate GalNAcT10 expression to promote proliferation and apoptosis resistance of HCC in a glycosyltransferase-dependent manner [79]. By modifying the O-glycosylation of EGFR and subsequent phosphorylation of AKT, the GalNAcT10 increased EGFR signaling, the development of HBV-associated HCC proliferation and resistance to apoptosis. Actually, GalNAcT10 promoted HCC by aberrantly glycosylating Muc1 and several other glycoproteins and surface molecules, in addition to EGF. Activated GALNT10 therefore, facilitates HCC tumor growth in HBV-infected cells, but GALNT10 activity appears to be decreased under the influence of Hnf4α and miR-122 [79].
The involvement of PSG11 and GALNT10, together with SLC2A2, SLC17A, CD53, THBS2, LCT, and GYPA was suggested in O-glycoprotein synthesis [80]. The coactivation of these genes suggest early tumor growth and could provide useful information for risk assessment in ovarian cancer patients. The elevated GalNAcT10 expression was correlated with advanced TNM stages of clear-cell renal cell carcinoma (ccRCC) and evaluated as a potential independent adverse prognostic factor for recurrence and survival of patients with ccRCC after surgical resection [80]. Recent GWAS studies have shown association of different SNPs at or nearby GALNT10 with high body mass index (BMI), predominantly in African and European populations, suggesting a distinct role for GALNT10 in adiposity within different ethnic populations [81-83].
The biological consequences of organ-specific glycosylation by GalNAcT10 in the stomach, small intestine, liver, pancreas, ovary, spleen, and central nervous system (CNS) are still to be explored [84, 85]. The same holds true for the molecular mechanisms underlying the relationship between adiposity and GALNT10 expression.
N-acetylgalactosaminyltransferase 11
The expression of the Tn and sialyl-Tn antigens usually reflects incomplete/aberrant O-glycosylation and are a hallmark of different types of cancers, frequently associated with poor prognosis. In a recent report on chronic lymphocytic leukemia (CLL), a low density of Tn residues and higher expression of GALNT11 was observed in B-CLL cells and healthy T-cells suggesting that GalNAcT11 contributes to B- and T lymphocyte differentiation and transformation [36]. In another context, meta-analyses of GWAS for 63,558 individuals of European origin correlated GALNT11, CDH23 and UMOD with the rapid decline of kidney function [86].
N-acetylgalactosaminyltransferase 12
Unfolded protein response (UPR) pathways are activated by aberrant glycosylation that induces ER (endoplasmic reticulum) stress [87]. UPR restores ER homeostasis, but prolonged ER stress causes apoptosis. Deficiency in Mixed Lineage Leukemia 1 (MLL1: a mammalian histone H3K4 methyltransferase) enhanced UPR and apoptosis caused by the N-glycosylation inhibitor tunicamycin (TM), and the direct binding of MLL1 to the promoters of GALNT12, H6PD and UGP2 activated the expression of these genes. When all three genes were knocked-down, TM-induced apoptosis was enhanced. However, transfecting the GALNT12, H6PD and UGP2 enhanced glycosylation and maturation of Lamp2 (lysosome-associated membrane protein 2). Therefore, faulty glycosylation of Lamp2 by transferases (e.g. GalNAcT12) may lead to cancer and developmental defects [86, 87].
In two-hundred and forty-four follicular lymphoma (FL) cases recognized during a population-based case-control study of non-Hodgkin lymphoma (NHL), five genes (GALNT12, BMP7, DUSP2, GADD45B, and ADAM17) were found associated with the overall survival of FL and control of B-cell activity. Among these five genes, mutated GALNT12 was associated with aberrant glycosylation, but inherited differences in growth-regulatory pathways of immune cells may have influenced the phenotype and progression of FL [88]. In a colon cancer study, eight mutations inactivated the normal function of the GalNAcT12 to variable degrees (reduction in enzyme activity: p.R382H, 1%; p.T491M, 2%; p.R373H, 5%; and p.R279W, 7% and p.D303N, 37%), leading to variable phenotypes of colon and breast carcinoma [60]. Although breast cancer is commonly regulated by GalNAcT4 and GalNAcT12, more is needed to assess the underlying mechanism associating the two enzymes in the same cancer cell.
In the majority of hereditary colorectal cancer (CRC) families, GALNT12 variants were characterized as highly penetrant variants that influence the pathogenesis of CRC [89, 90], but little is known regarding the genotypic and allelic frequencies of GALNT12 in different racial/ethnic groups. Moreover, in addition to GALNT12, different other genes (ZNF367, HABP4, and GABBR2) are also associated with CRC susceptibility.
N-acetylgalactosaminyltransferase 13
Both GALNT1 and GALNT13 were found almost 90% identical in structure, substrate specificity and kinetic properties, and grouped in subclass Ia [11]. However, GalNAcT13 showed a higher specificity for Muc5Ac and Muc7 than GalNAcT1 [18].
GALNT13, like GALNT1, is highly expressed in all neuroblastoma (NB) cells, but not in glioblastoma cells [18]. As the bone marrow (BM) is the preferential site for NB dissemination, BM molecular analysis of human neuroblastoma patients has shown high expression of GALNT13, which was identified as a new indicator for disseminated neuroblasts in BM of NB patients. GalNAcT13, unlike GalNAcT1, may be a major glycosyltransferase in the biosynthesis of O-glycans in the brain and contribute to the formation of a triplet Tn epitope on peptides on cell-surface transmembrane heparan sulfate proteoglycans (syndecan-3) in neurons and Schwann cells [91]. However, the role of the triplet O-glycan formation on syndecan-3, if any in brain cancers, is not known.
In an immunohistochemical study, inactivation of GALNT13 showed a remarkable decrease in expression of the Tn antigen in cerebellum, suggesting the implication of the GalNAcT13 enzyme in neuronal function [18].
N-acetylgalactosaminyltransferase 14
Overexpression of GALNT14, which initiates the O-glycosylation of mucin substrates Muc2, Muc5Ac, Muc7 and Muc13, activates the invasion and migration of breast cancer cells by up-regulating MMP-2, VEGF, TGF-β, N-cadherin, vimentin and down-regulating E-cad [92]. Of the two single nucleotide polymorphisms rs9679162 and rs6752303 flanking GALNT14, the TT genotype of rs9679162 was strongly associated with the non-viral etiology of HCC [93]. In another similar study on advanced HCC patients treated with 5-fluorouracil, mitoxantrone and cisplatin chemotherapy, the GALN14 genotype rs9679162 was found to be a reliable marker for a positive therapeutic outcome [86]. The GALNT14 genotype and α-fetoprotein (AFP) levels were identified as pre-therapeutic markers for a favorable response in HCC [86]. In another study on ovarian cancer, suppression of cell migration and altered cellular morphology were found to result from the knockdown of GALNT14 by small interfering RNA. In the same report, interleukin-8 (IL-8) had no significant effect on the function of GalNAcT14 and the tumor-associated Tn antigen [94]. However, co-expression of GalNAcT14 and transmembrane mucin 13 (Muc13) in ovarian cancer tissues, but not in normal tissues, suggested a contribution of GalNAcT14 to ovarian carcinogenesis through aberrant glycosylation of Muc13. The exact molecular mechanism underlying the Muc13-GalNAcT14-ERK1/2 pathway remains however undefined [94].
In pancreatic carcinoma, non-small-cell lung carcinoma and a melanoma cell line, expression of GalNAcT14 was correlated with Apo2L/TRAIL sensitivity [95]. Following O-glycosylation of proapoptotic death receptors (DR4 and DR5) by GalNAcT14, clustering of DR4/DR5 by TRAIL (tumor necrosis factor-related apoptosis-inducing ligand) mediates cancer cell death [95]. Although the over-expression of GALNT14 mRNA in tumor cell lines increased the responsiveness to TRAIL, no significant interaction was observed between tumor GALNT14 expression and the outcome of NSCLC patients treated with dulanermin, a recombinant apoptosis ligand. A trend toward increased progression-free survival and overall survival was seen in GALNT14-positive patients under dulanermin treatment, and the presence of DR4 and/or DR5 was essential but not sufficient for stimulation of apoptosis by dulanermin [96].
In CRC cell lines, dulanermin sensitivity was correlated with GalNAcT14 activity on Muc1 substrate and the fucosyltransferase enzymes FUT3 and FUT6 [95, 97]. In patients with advanced hepatocellular carcinoma, linkage of the germline SNP marker rs9679162 with GALNT14 could be correlated with the objective response to the first course of 5-fluorouracil, mitoxantrone and cisplatin (FMP) chemotherapy [98]. However, the role of rs9679162 in the FMP response and its influence on the alternative splicing of GALNT14 and other GALNTs [98] remain unknown.
N-acetylgalactosaminyltransferase 15 (GALNT15) to N-acetylgalactosaminyltransferase 20
Similar to GALNT5, GALNT15 did not show any significant relationship with other GALNT family members and was grouped in the Ig subfamily [11]. After genotyping of CRC cases and controls in the EPICOLON consortium, four genetic variants, rs2102302 in GALNTL2, rs3803185 in ARL11, rs698 in ADH1C and rs1800795 in IL6, were found with potential association to CRC risk factor [99].
GALNT16 shares the same intron numbers (with minor variations in introns positions) with GALNT2 and GALNT14 and the three genes were grouped together as Ib subfamily [11]. The GalNAcT18 (encoded by GALNT18/GALNTL4) was classified as Ie subfamily with GALNT8, GALNT9 and GALNT19 [11, 100, 101], but in another classification system, the GALNT8, GALNT9, GALNT17 and GALNT18 were defined as the Y-subfamily. All Y-subfamily member enzymes fail to glycosylate classical substrates because of an altered UDP-GalNAc-binding pocket in LDX(5)YGGENXE. Nonetheless, the expression profile of GalNAcT18 in the endoplasmic reticulum of lung carcinoma cells and the observed enhancement of catalytic activity of both GalNAcT2 and GalNAcT10 in the presence of GalNAcT18 suggests that GalNAcT18 could be a chaperone-like protein [73].
N-acetylgalactosaminyltransferase-like protein 5 (GALNTL5) is the only GalNAcT family member of which lacks the lectin domain, and has been called GalNAcT19 or GalNAcT20 in the literature (Refseq accession no.: NP_660335.2) [102]. However, no clinical evidence has been reported for any involvement of GALNTL5 in cancer.
Conclusions and perspectives
The GalNAcTs family is involved to variable degrees in carcinomas, leukemias, lymphomas and central nervous system cancers, and several non-neoplastic diseases and developmental defects. Developmental pathologies involving cardiac valves and submandibular gland generation also depend on the timely and efficient participation of N-acetylgalactosaminyltransferases during ontogeny. This critical role of the sugar transferases catalyzing O-glycosylation of cell-surface proteins (mucins and surface receptors like DR4/DR5, IGF-lR, MET, and EGFR) and ECM proteins (fibronectin) points to the fundamental involvement of glycoproteins in interactions of epithelial and hemopoietic cells with the ECM and small proteins like cytokines, growth factors and hormones. Therefore, proliferative activities and metastatic behaviors may all depend upon O-glycosylated cell-surface receptors and cell-cell interaction proteins. Lastly, metabolic diseases such as diabetes and calcinosis were shown to display genetic alterations of GALNT2 and GALNT3, respectively, alterations reflected by inappropriate insulin responses and calcium deposition in the case of calcinosis [103-106].
Site-specific GalNAc-type O-glycosylation is also a major regulator of proprotein convertase processing, which generates a variety of active extracellular agonists like the endocrine FGF23 [107]. It has been estimated that the number of proteins generated in this manner well exceeds 3000, including cytokines and hormones, but also proteases and receptors. The range of effects generated by deficient preprocessed proteins is expected to be very large, and will certainly be part of developmental defects and neoplastic phenotypes.
The wide spectrum of biological consequences that could result from altered N-acetylgalactosaminyltransferase biological activities (see Figure 1) mostly fits the contexts of 1) cellular interactions with the ECM, as exemplified by neoplastic cells interacting with the peri-tumoral context during invasion, or epithelial cells contacting their basement membrane, 2) abnormal responses of abnormally O-glycosylated cell-surface receptors to ECM ligands, insulin and cytokines, as expected to happen in epithelial cells during oncogenesis, insulin resistance, inappropriate responses to growth factors and resistance to apoptosis-inducing ligands, 3) alteration of vesicle trafficking genes, as it has been suggested in 17β-estradiol-resistance in breast cancer cells, 4) altered mucin-type O-glycosylation of the cellular glycocalyx, as described in regulation of various carcinomas, and 5) COPI-based translocation of GalNAcTs to the endoplasmic reticulum, as it has been demonstrated in driving motility and invasiveness of tumor cells (Figure 2).
In particular, the frequent occurrence of modulation by GalNAcTs of transmembrane receptors points to the selective influence of O-glycosylation on receptor dimerization and activation of their intracytoplasmic tyrosine kinases (Figure 2). This applies to insulin and insulin-like receptors, apoptosis receptors (DR4/5), MET and TGF-β receptors, in normal and neoplastic cells, and most selectively to growth factor receptors (EGFRs & IGF-lR) in neoplastic cells. Mechanistically, inappropriate O-glycosylation of tyrosine kinase receptors in cancer cells may inactivate them by preventing their dimerization [27] or abnormally favor their dimerization [34, 108] and make them constitutively active (Figure 2). Both outcomes may take place in normal cells and lead to insufficient or excessive proliferation during embryogenesis and result in growth defects. In cancer cells, however, constitutive proliferation is a hallmark of neoplastic behavior [109]. It follows that carcinoma cells may express a wide variety of tyrosine kinases transmembrane proteins which may [27, 33, 34, 108] or may not be appropriately O-glycosylated by the available transferases. Given this unpredictability in oncogenic driver expression, as for instance in NSCLC [110], gastric adenocarcinoma and HCC, altered O-glycosylation of the oncogenic driver may sustain hyperproliferation in many different ways [27, 33].
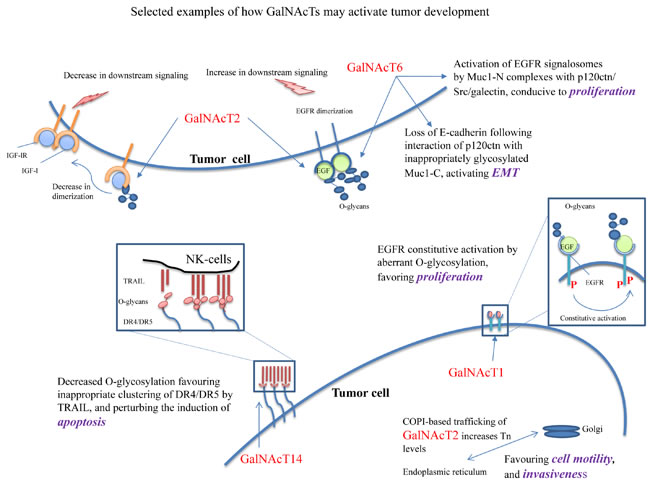
Figure 2: Selected examples of putative mechanisms whereby GalNAcTs could support the neoplastic phenotype. 1) Constitutive activation of IGFR-1 dimers, under O-glycosylated by GalNAcT2, 2) constitutive activation of EGFR dimers, under O-glycosylation by GalNAcT2 and T6), 3) loss of E-cadherin following interaction with p120-catenin with inappropriately glycosylated Muc1-C, by GalNAcT6, activating EMT, 4) activation of EGFR signalosomes of MuC-1 complexes with p120ctn/Src/galectin, by GalNAcT6, leading to proliferation, 5) COPI-based trafficking of GalNAcT2 increases the amounts of Tn antigen in the ER, leading to increased cell motility and invasiveness, and 6) decreased O-glycosylation of DR4/DR5 suppresses the clustering of TRAIL, perturbing the induction of apoptosis.
As highlighted in Figure 1, genetic alterations in GALNTs are almost never isolated events, but always associated with other genetic alterations, as shown by GWAS and non-GWAS studies. To find out how the expression of GALNTs products and other altered genes concurrently contribute to the pathologies ascribed to GALNTs’ alterations is a most interesting challenge.
It is expected that further GWAS and other linkage studies, and especially the availability of specific antibodies to individual transferases and biochemical characterization of the transferases themselves, will provide not only the means of ascribing individual transferase to specific clinical contexts, but also to identify those transferases that could be targeted therapeutically.
Acknowledgments
This work was supported by National Natural Science Foundation of China (Grant No. 31370877, 31322020), M.F. is supported by an award from the National Thousand Talent Plan of China (2012). Muhammad Ramzan Manwar (ID: 2014A8011307026) is supported by the CAS-TWAS President’s Fellowship. The authors acknowledge the helpful suggestions from Hussein Sheikh Ali Mohamoud (Human Genetics Research Centre, Division of Biomedical Sciences (BMS), St. George’s University of London (SGUL), London, UK) and Xuefeng Duan, Wenming Zhao, Kai Zhou, Wei Jiang, Dongfang Wang and Jun Fang (CAS-Institute of Microbiology, Beijing, China).
CONFLICTS OF INTEREST
The authors declared that they have no competing interests.
References
1. Li W, Ma H and Sun J. MicroRNA34a/c function as tumor suppressors in Hep2 laryngeal carcinoma cells and may reduce GALNT7 expression. Molecular medicine reports. 2014; 9:1293-1298.
2. Peng RQ, Wan HY, Li HF, Liu M, Li X and Tang H. MicroRNA-214 suppresses growth and invasiveness of cervical cancer cells by targeting UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferase 7. The Journal of biological chemistry. 2012; 287:14301-14309.
3. Kahai S, Lee SC, Lee DY, Yang J, Li M, Wang CH, Jiang Z, Zhang Y, Peng C and Yang BB. MicroRNA miR-378 regulates nephronectin expression modulating osteoblast differentiation by targeting GalNT-7. PloS one. 2009; 4:e7535.
4. Hussain MR, Nasir J and Al-Aama JY. Clinically significant missense variants in human GALNT3, GALNT8, GALNT12, and GALNT13 genes: intriguing in silico findings. Journal of cellular biochemistry. 2014; 115:313-327.
5. Hollingsworth MA and Swanson BJ. Mucins in cancer: protection and control of the cell surface. Nat Rev Cancer. 2004; 4:45-60.
6. Brockhausen I. Pathways of O-glycan biosynthesis in cancer cells. Biochim Biophys Acta. 1999; 1473:67-95.
7. Gill DJ, Tham KM, Chia J, Wang SC, Steentoft C, Clausen H, Bard-Chapeau EA and Bard FA. Initiation of GalNAc-type O-glycosylation in the endoplasmic reticulum promotes cancer cell invasiveness. Proceedings of the National Academy of Sciences of the United States of America. 2013; 110:E3152-3161.
8. Mohamoud HS, Hussain MR, El-Harouni AA, Shaik NA, Qasmi ZU, Merican AF, Baig M, Anwar Y, Asfour H, Bondagji N and Al-Aama JY. First comprehensive in silico analysis of the functional and structural consequences of SNPs in human GalNAc-T1 gene. Computational and mathematical methods in medicine. 2014; 2014:904052.
9. Tian E, Stevens SR, Guan Y, Springer DA, Anderson SA, Starost MF, Patel V, Ten Hagen KG and Tabak LA. Galnt1 is required for normal heart valve development and cardiac function. PloS one. 2015; 10:e0115861.
10. Kathiresan S, Melander O, Guiducci C, Surti A, Burtt NP, Rieder MJ, Cooper GM, Roos C, Voight BF, Havulinna AS, Wahlstrand B, Hedner T, Corella D, et al. Six new loci associated with blood low-density lipoprotein cholesterol, high-density lipoprotein cholesterol or triglycerides in humans. Nature genetics. 2008; 40:189-197.
11. Bennett EP, Mandel U, Clausen H, Gerken TA, Fritz TA and Tabak LA. Control of mucin-type O-glycosylation: a classification of the polypeptide GalNAc-transferase gene family. Glycobiology. 2012; 22:736-756.
12. Ju T, Otto VI and Cummings RD. The Tn antigen-structural simplicity and biological complexity. Angew Chem Int Ed Engl. 2011; 50:1770-1791.
13. Gerken TA, Revoredo L, Thome JJ, Tabak LA, Vester-Christensen MB, Clausen H, Gahlay GK, Jarvis DL, Johnson RW, Moniz HA and Moremen K. The lectin domain of the polypeptide GalNAc transferase family of glycosyltransferases (ppGalNAc Ts) acts as a switch directing glycopeptide substrate glycosylation in an N- or C-terminal direction, further controlling mucin type O-glycosylation. The Journal of biological chemistry. 2013; 288:19900-19914.
14. Ten Hagen KG, Fritz TA and Tabak LA. All in the family: the UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferases. Glycobiology. 2003; 13:1R-16R.
15. Schjoldager KT, Vester-Christensen MB, Goth CK, Petersen TN, Brunak S, Bennett EP, Levery SB and Clausen H. A systematic study of site-specific GalNAc-type O-glycosylation modulating proprotein convertase processing. The Journal of biological chemistry. 2011; 286:40122-40132.
16. Kato K, Jeanneau C, Tarp MA, Benet-Pages A, Lorenz-Depiereux B, Bennett EP, Mandel U, Strom TM and Clausen H. Polypeptide GalNAc-transferase T3 and familial tumoral calcinosis. Secretion of fibroblast growth factor 23 requires O-glycosylation. The Journal of biological chemistry. 2006; 281:18370-18377.
17. Potapenko IO, Haakensen VD, Luders T, Helland A, Bukholm I, Sorlie T, Kristensen VN, Lingjaerde OC and Borresen-Dale AL. Glycan gene expression signatures in normal and malignant breast tissue; possible role in diagnosis and progression. Mol Oncol. 2010; 4:98-118.
18. Zhang Y, Iwasaki H, Wang H, Kudo T, Kalka TB, Hennet T, Kubota T, Cheng L, Inaba N, Gotoh M, Togayachi A, Guo J, Hisatomi H, et al. Cloning and characterization of a new human UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferase, designated pp-GalNAc-T13, that is specifically expressed in neurons and synthesizes GalNAc alpha-serine/threonine antigen. The Journal of biological chemistry. 2003; 278:573-584.
19. Bennett EP, Hassan H and Clausen H. cDNA cloning and expression of a novel human UDP-N-acetyl-alpha-D-galactosamine. Polypeptide N-acetylgalactosaminyltransferase, GalNAc-t3. The Journal of biological chemistry. 1996; 271:17006-17012.
20. Gaziel-Sovran A, Segura MF, Di Micco R, Collins MK, Hanniford D, Vega-Saenz de Miera E, Rakus JF, Dankert JF, Shang S, Kerbel RS, Bhardwaj N, Shao Y, Darvishian F, et al. miR-30b/30d regulation of GalNAc transferases enhances invasion and immunosuppression during metastasis. Cancer cell. 2011; 20:104-118.
21. Sellers TA, Huang Y, Cunningham J, Goode EL, Sutphen R, Vierkant RA, Kelemen LE, Fredericksen ZS, Liebow M, Pankratz VS, Hartmann LC, Myer J, Iversen ES, Jr., et al. Association of single nucleotide polymorphisms in glycosylation genes with risk of epithelial ovarian cancer. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2008; 17:397-404.
22. Ding MX, Wang HF, Wang JS, Zhan H, Zuo YG, Yang DL, Liu JY, Wang W, Ke CX and Yan RP. ppGalNAc T1 as a potential novel marker for human bladder cancer. Asian Pacific journal of cancer prevention : APJCP. 2012; 13:5653-5657.
23. Dyrskjot L, Ostenfeld MS, Bramsen JB, Silahtaroglu AN, Lamy P, Ramanathan R, Fristrup N, Jensen JL, Andersen CL, Zieger K, Kauppinen S, Ulhoi BP, Kjems J, Borre M and Orntoft TF. Genomic profiling of microRNAs in bladder cancer: miR-129 is associated with poor outcome and promotes cell death in vitro. Cancer research. 2009; 69:4851-4860.
24. Holleboom AG, Vergeer M, Hovingh GK, Kastelein JJ and Kuivenhoven JA. The value of HDL genetics. Current opinion in lipidology. 2008; 19:385-394.
25. Phelan CM, Tsai YY, Goode EL, Vierkant RA, Fridley BL, Beesley J, Chen XQ, Webb PM, Chanock S, Cramer DW, Moysich K, Edwards RP, Chang-Claude J, et al. Polymorphism in the GALNT1 gene and epithelial ovarian cancer in non-Hispanic white women: the Ovarian Cancer Association Consortium. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2010; 19:600-604.
26. Cheng SL, Huang Liu R, Sheu JN, Chen ST, Sinchaikul S and Tsay GJ. Toxicogenomics of kojic acid on gene expression profiling of a375 human malignant melanoma cells. Biological & pharmaceutical bulletin. 2006; 29:655-669.
27. Huang MJ, Hu RH, Chou CH, Hsu CL, Liu YW, Huang J, Hung JS, Lai IR, Juan HF, Yu SL, Wu YM and Huang MC. Knockdown of GALNT1 suppresses malignant phenotype of hepatocellular carcinoma by suppressing EGFR signaling. Oncotarget. 2015; 6:5650-5665. doi: 10.16832/oncotarget.3117.
28. Terry KL, Vitonis AF, Hernandez D, Lurie G, Song H, Ramus SJ, Titus-Ernstoff L, Carney ME, Wilkens LR, Gentry-Maharaj A, Menon U, Gayther SA, Pharaoh PD, et al. A polymorphism in the GALNT2 gene and ovarian cancer risk in four population based case-control studies. Int J Mol Epidemiol Genet. 2010; 1:272-277.
29. Holleboom AG, Karlsson H, Lin RS, Beres TM, Sierts JA, Herman DS, Stroes ES, Aerts JM, Kastelein JJ, Motazacker MM, Dallinga-Thie GM, Levels JH, Zwinderman AH, Seidman JG, et al. Heterozygosity for a loss-of-function mutation in GALNT2 improves plasma triglyceride clearance in man. Cell metabolism. 2011; 14:811-818.
30. Willer CJ and Mohlke KL. Finding genes and variants for lipid levels after genome-wide association analysis. Current opinion in lipidology. 2012; 23:98-103.
31. Leduc MS, Lyons M, Darvishi K, Walsh K, Sheehan S, Amend S, Cox A, Orho-Melander M, Kathiresan S, Paigen B and Korstanje R. The mouse QTL map helps interpret human genome-wide association studies for HDL cholesterol. Journal of lipid research. 2011; 52:1139-1149.
32. Ho WL, Chou CH, Jeng YM, Lu MY, Yang YL, Jou ST, Lin DT, Chang HH, Lin KH, Hsu WM and Huang MC. GALNT2 suppresses malignant phenotypes through IGF-1 receptor and predicts favorable prognosis in neuroblastoma. Oncotarget. 2014; doi: 10.16832/oncotarget.2627.
33. Liu SY, Shun CT, Hung KY, Juan HF, Hsu CL, Huang MC and Lai IR. Mucin glycosylating enzyme GALNT2 suppresses malignancy in gastric adenocarcinoma by reducing MET phosphorylation. Oncotarget. 2016; doi: 10.16832/oncotarget.7081.
34. Wu YM, Liu CH, Hu RH, Huang MJ, Lee JJ, Chen CH, Huang J, Lai HS, Lee PH, Hsu WM, Huang HC and Huang MC. Mucin glycosylating enzyme GALNT2 regulates the malignant character of hepatocellular carcinoma by modifying the EGF receptor. Cancer research. 2011; 71:7270-7279.
35. Vasconcelos-Dos-Santos A, Oliveira IA, Lucena MC, Mantuano NR, Whelan SA, Dias WB and Todeschini AR. Biosynthetic Machinery Involved in Aberrant Glycosylation: Promising Targets for Developing of Drugs Against Cancer. Front Oncol. 2015; 5:138.
36. Libisch MG, Casas M, Chiribao M, Moreno P, Cayota A, Osinaga E, Oppezzo P and Robello C. GALNT11 as a new molecular marker in chronic lymphocytic leukemia. Gene. 2014; 533:270-279.
37. Qiu H, Guo XH, Mo JH, Jin MF, Wu SL and Chen HL. Expressions of polypeptide: N-acetylgalactosaminyltransferase in leukemia cell lines during 1,25-dihydroxyvitamin D3 induced differentiation. Glycoconjugate journal. 2006; 23:575-584.
38. Bennett EP, Hassan H, Mandel U, Hollingsworth MA, Akisawa N, Ikematsu Y, Merkx G, van Kessel AG, Olofsson S and Clausen H. Cloning and characterization of a close homologue of human UDP-N-acetyl-alpha-D-galactosamine:Polypeptide N-acetylgalactosaminyltransferase-T3, designated GalNAc-T6. Evidence for genetic but not functional redundancy. The Journal of biological chemistry. 1999; 274:25362-25370.
39. Wandall HH, Dabelsteen S, Sorensen JA, Krogdahl A, Mandel U and Dabelsteen E. Molecular basis for the presence of glycosylated onco-foetal fibronectin in oral carcinomas: the production of glycosylated onco-foetal fibronectin by carcinoma cells. Oral oncology. 2007; 43:301-309.
40. Li Z, Yamada S, Inenaga S, Imamura T, Wu Y, Wang KY, Shimajiri S, Nakano R, Izumi H, Kohno K and Sasaguri Y. Polypeptide N-acetylgalactosaminyltransferase 6 expression in pancreatic cancer is an independent prognostic factor indicating better overall survival. British journal of cancer. 2011; 104:1882-1889.
41. Kitada S, Yamada S, Kuma A, Ouchi S, Tasaki T, Nabeshima A, Noguchi H, Wang KY, Shimajiri S, Nakano R, Izumi H, Kohno K, Matsumoto T and Sasaguri Y. Polypeptide N-acetylgalactosaminyl transferase 3 independently predicts high-grade tumours and poor prognosis in patients with renal cell carcinomas. British journal of cancer. 2013; 109:472-481.
42. Aubert S, Fauquette V, Hemon B, Lepoivre R, Briez N, Bernard D, Van Seuningen I, Leroy X and Perrais M. MUC1, a new hypoxia inducible factor target gene, is an actor in clear renal cell carcinoma tumor progression. Cancer research. 2009; 69:5707-5715.
43. Topaz O, Shurman DL, Bergman R, Indelman M, Ratajczak P, Mizrachi M, Khamaysi Z, Behar D, Petronius D, Friedman V, Zelikovic I, Raimer S, Metzker A, Richard G and Sprecher E. Mutations in GALNT3, encoding a protein involved in O-linked glycosylation, cause familial tumoral calcinosis. Nature genetics. 2004; 36:579-581.
44. Dosaka-Akita H, Kinoshita I, Yamazaki K, Izumi H, Itoh T, Katoh H, Nishimura M, Matsuo K, Yamada Y and Kohno K. N-acetylgalactosaminyl transferase-3 is a potential new marker for non-small cell lung cancers. British journal of cancer. 2002; 87:751-755.
45. Vavasseur F, Dole K, Yang J, Matta KL, Myerscough N, Corfield A, Paraskeva C and Brockhausen I. O-glycan biosynthesis in human colorectal adenoma cells during progression to cancer. European journal of biochemistry / FEBS. 1994; 222:415-424.
46. Wakata K, Tsuchiya T, Tomoshige K, Takagi K, Yamasaki N, Matsumoto K, Miyazaki T, Nanashima A, Whitsett JA, Maeda Y and Nagayasu T. A favourable prognostic marker for EGFR mutant non-small cell lung cancer: immunohistochemical analysis of MUC5B. BMJ open. 2015; 5:e008366.
47. Chachadi VB, Bhat G and Cheng PW. Glycosyltransferases involved in the synthesis of MUC-associated metastasis-promoting selectin ligands. Glycobiology. 2015; 25:963-975.
48. Ishikawa M, Kitayama J, Nariko H, Kohno K and Nagawa H. The expression pattern of UDP-N-acetyl-alpha-d-galactosamine: polypeptide N-acetylgalactosaminyl transferase-3 in early gastric carcinoma. Journal of surgical oncology. 2004; 86:28-33.
49. Ishikawa M, Kitayama J, Kohno K and Nagawa H. The expression pattern of UDP-N-acetyl-alpha-D-galactosamine-polypeptide N-acetyl-galactosaminyl transferase-3 in squamous cell carcinoma of the esophagus. Pathobiology : journal of immunopathology, molecular and cellular biology. 2005; 72:139-145.
50. Costa V, Esposito R, Ziviello C, Sepe R, Bim LV, Cacciola NA, Decaussin-Petrucci M, Pallante P, Fusco A and Ciccodicola A. New somatic mutations and WNK1-B4GALNT3 gene fusion in papillary thyroid carcinoma. Oncotarget. 2015; 6:11242-11251.
51. Onitsuka K, Shibao K, Nakayama Y, Minagawa N, Hirata K, Izumi H, Matsuo K, Nagata N, Kitazato K, Kohno K and Itoh H. Prognostic significance of UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferase-3 (GalNAc-T3) expression in patients with gastric carcinoma. Cancer Sci. 2003; 94:32-36.
52. Miyahara N, Shoda J, Kawamoto T, Furukawa M, Ueda T, Todoroki T, Tanaka N, Matsuo K, Yamada Y, Kohno K and Irimura T. Expression of UDP-N-acetyl-alpha-D-galactosamine-polypeptide N-acetylgalactosaminyltransferase isozyme 3 in the subserosal layer correlates with postsurgical survival of pathological tumor stage 2 carcinoma of the gallbladder. Clinical cancer research : an official journal of the American Association for Cancer Research. 2004; 10:2090-2099.
53. Landers KA, Burger MJ, Tebay MA, Purdie DM, Scells B, Samaratunga H, Lavin MF and Gardiner RA. Use of multiple biomarkers for a molecular diagnosis of prostate cancer. International journal of cancer Journal international du cancer. 2005; 114:950-956.
54. Inoue T, Eguchi T, Oda Y, Nishiyama K, Fujii K, Izumi H, Kohno K, Yamaguchi K, Tanaka M and Tsuneyoshi M. Expression of GalNAc-T3 and its relationships with clinicopathological factors in 61 extrahepatic bile duct carcinomas analyzed using stepwise sections - special reference to its association with lymph node metastases. Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc. 2007; 20:267-276.
55. Yamamoto S, Nakamori S, Tsujie M, Takahashi Y, Nagano H, Dono K, Umeshita K, Sakon M, Tomita Y, Hoshida Y, Aozasa K, Kohno K and Monden M. Expression of uridine diphosphate N-acetyl-alpha-D-galactosamine: polypeptide N-acetylgalactosaminyl transferase 3 in adenocarcinoma of the pancreas. Pathobiology : journal of immunopathology, molecular and cellular biology. 2004; 71:12-18.
56. Wright PK, May FE, Darby S, Saif R, Lennard TW and Westley BR. Estrogen regulates vesicle trafficking gene expression in EFF-3, EFM-19 and MCF-7 breast cancer cells. International journal of clinical and experimental pathology. 2009; 2:463-475.
57. Liu Y, Liu W, Xu L, Liu H, Zhang W, Zhu Y, Xu J and Gu J. GALNT4 predicts clinical outcome in patients with clear cell renal cell carcinoma. The Journal of urology. 2014; 192:1534-1541.
58. Ten Hagen KG, Hagen FK, Balys MM, Beres TM, Van Wuyckhuyse B and Tabak LA. Cloning and expression of a novel, tissue specifically expressed member of the UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferase family. The Journal of biological chemistry. 1998; 273:27749-27754.
59. He H, Shen Z, Zhang H, Wang X, Tang Z, Xu J and Sun Y. Clinical significance of polypeptide N-acetylgalactosaminyl transferase-5 (GalNAc-T5) expression in patients with gastric cancer. British journal of cancer. 2014; 110:2021-2029.
60. Guda K, Moinova H, He J, Jamison O, Ravi L, Natale L, Lutterbaugh J, Lawrence E, Lewis S, Willson JK, Lowe JB, Wiesner GL, Parmigiani G, et al. Inactivating germ-line and somatic mutations in polypeptide N-acetylgalactosaminyltransferase 12 in human colon cancers. Proceedings of the National Academy of Sciences of the United States of America. 2009; 106:12921-12925.
61. Wood LD, Parsons DW, Jones S, Lin J, Sjoblom T, Leary RJ, Shen D, Boca SM, Barber T, Ptak J, Silliman N, Szabo S, Dezso Z, et al. The genomic landscapes of human breast and colorectal cancers. Science (New York, NY). 2007; 318:1108-1113.
62. Rodrigues TC, Fidalgo F, da Costa CM, Ferreira EN, da Cunha IW, Carraro DM, Krepischi AC and Rosenberg C. Upregulated genes at 2q24 gains as candidate oncogenes in hepatoblastomas. Future oncology (London, England). 2014; 10:2449-2457.
63. Berois N, Mazal D, Ubillos L, Trajtenberg F, Nicolas A, Sastre-Garau X, Magdelenat H and Osinaga E. UDP-N-acetyl-D-galactosamine: polypeptide N-acetylgalactosaminyltransferase-6 as a new immunohistochemical breast cancer marker. The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society. 2006; 54:317-328.
64. Patani N, Jiang W and Mokbel K. Prognostic utility of glycosyltransferase expression in breast cancer. Cancer genomics & proteomics. 2008; 5:333-340.
65. Park JH, Katagiri T, Chung S, Kijima K and Nakamura Y. Polypeptide N-acetylgalactosaminyltransferase 6 disrupts mammary acinar morphogenesis through O-glycosylation of fibronectin. Neoplasia (New York, NY). 2011; 13:320-326.
66. Park JH, Nishidate T, Kijima K, Ohashi T, Takegawa K, Fujikane T, Hirata K, Nakamura Y and Katagiri T. Critical roles of mucin 1 glycosylation by transactivated polypeptide N-acetylgalactosaminyltransferase 6 in mammary carcinogenesis. Cancer research. 2010; 70:2759-2769.
67. Zhang L, Gallup M, Zlock L, Chen YT, Finkbeiner WE and McNamara NA. Pivotal role of MUC1 glycosylation by cigarette smoke in modulating disruption of airway adherens junctions in vitro. The Journal of pathology. 2014; 234:60-73.
68. Shan SW, Fang L, Shatseva T, Rutnam ZJ, Yang X, Du W, Lu WY, Xuan JW, Deng Z and Yang BB. Mature miR-17-5p and passenger miR-17-3p induce hepatocellular carcinoma by targeting PTEN, GalNT7 and vimentin in different signal pathways. Journal of cell science. 2013; 126:1517-1530.
69. Rose AE, Poliseno L, Wang J, Clark M, Pearlman A, Wang G, Vega YSdMEC, Medicherla R, Christos PJ, Shapiro R, Pavlick A, Darvishian F, Zavadil J, et al. Integrative genomics identifies molecular alterations that challenge the linear model of melanoma progression. Cancer research. 2011; 71:2561-2571.
70. Carter CJ. Schizophrenia susceptibility genes directly implicated in the life cycles of pathogens: cytomegalovirus, influenza, herpes simplex, rubella, and Toxoplasma gondii. Schizophrenia bulletin. 2009; 35:1163-1182.
71. Abend M, Pfeiffer RM, Ruf C, Hatch M, Bogdanova TI, Tronko MD, Riecke A, Hartmann J, Meineke V, Boukheris H, Sigurdson AJ, Mabuchi K and Brenner AV. Iodine-131 dose dependent gene expression in thyroid cancers and corresponding normal tissues following the Chernobyl accident. PloS one. 2012; 7:e39103.
72. Stein L, Rothschild J, Luce J, Cowell JK, Thomas G, Bogdanova TI, Tronko MD and Hawthorn L. Copy number and gene expression alterations in radiation-induced papillary thyroid carcinoma from chernobyl pediatric patients. Thyroid : official journal of the American Thyroid Association. 2010; 20:475-487.
73. Li X, Wang J, Li W, Xu Y, Shao D, Xie Y, Xie W, Kubota T, Narimatsu H and Zhang Y. Characterization of ppGalNAc-T18, a member of the vertebrate-specific Y subfamily of UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferases. Glycobiology. 2012; 22:602-615.
74. Nakano R, Maekawa T, Abe H, Hayashida Y, Ochi H, Tsunoda T, Kumada H, Kamatani N, Nakamura Y and Chayama K. Single-nucleotide polymorphisms in GALNT8 are associated with the response to interferon therapy for chronic hepatitis C. The Journal of general virology. 2013; 94:81-89.
75. Imran M, Manzoor S and Parvaiz F. Predictive potential of IL-18 -607 and osteopontin -442 polymorphism in interferon-based therapy of HCV infection in the Pakistani population. Viral immunology. 2014; 27:404-411.
76. Tuupanen S, Hanninen UA, Kondelin J, von Nandelstadh P, Cajuso T, Gylfe AE, Katainen R, Tanskanen T, Ristolainen H, Bohm J, Mecklin JP, Jarvinen H, Renkonen-Sinisalo L, et al. Identification of 33 candidate oncogenes by screening for base-specific mutations. British journal of cancer. 2014; 111:1657-1662.
77. Berois N, Gattolliat CH, Barrios E, Capandeguy L, Douc-Rasy S, Valteau-Couanet D, Benard J and Osinaga E. GALNT9 gene expression is a prognostic marker in neuroblastoma patients. Clinical chemistry. 2013; 59:225-233.
78. Pangeni RP, Channathodiyil P, Huen DS, Eagles LW, Johal BK, Pasha D, Hadjistephanou N, Nevell O, Davies CL, Adewumi AI, Khanom H, Samra IS, Buzatto VC, et al. The GALNT9, BNC1 and CCDC8 genes are frequently epigenetically dysregulated in breast tumours that metastasise to the brain. Clin Epigenetics. 2015; 7:57.
79. Wu Q, Liu HO, Liu YD, Liu WS, Pan D, Zhang WJ, Yang L, Fu Q, Xu JJ and Gu JX. Decreased expression of hepatocyte nuclear factor 4alpha (Hnf4alpha)/microRNA-122 (miR-122) axis in hepatitis B virus-associated hepatocellular carcinoma enhances potential oncogenic GALNT10 protein activity. The Journal of biological chemistry. 2015; 290:1170-1185.
80. Wu Q, Yang L, Liu H, Zhang W, Le X and Xu J. Elevated Expression of N-Acetylgalactosaminyltransferase 10 Predicts Poor Survival and Early Recurrence of Patients with Clear-Cell Renal Cell Carcinoma. Annals of surgical oncology. 2014.
81. Ng MC, Hester JM, Wing MR, Li J, Xu J, Hicks PJ, Roh BH, Lu L, Divers J, Langefeld CD, Freedman BI, Palmer ND and Bowden DW. Genome-wide association of BMI in African Americans. Obesity (Silver Spring, Md). 2012; 20:622-627.
82. Monda KL, Chen GK, Taylor KC, Palmer C, Edwards TL, Lange LA, Ng MC, Adeyemo AA, Allison MA, Bielak LF, Chen G, Graff M, Irvin MR, et al. A meta-analysis identifies new loci associated with body mass index in individuals of African ancestry. Nature genetics. 2013; 45:690-696.
83. Schwenk RW, Vogel H and Schurmann A. Genetic and epigenetic control of metabolic health. Molecular metabolism. 2013; 2:337-347.
84. Cheng L, Tachibana K, Zhang Y, Guo J, Kahori Tachibana K, Kameyama A, Wang H, Hiruma T, Iwasaki H, Togayachi A, Kudo T and Narimatsu H. Characterization of a novel human UDP-GalNAc transferase, pp-GalNAc-T10. FEBS letters. 2002; 531:115-121.
85. Nelson PA, Sutcliffe JG and Thomas EA. A new UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferase mRNA exhibits predominant expression in the hypothalamus, thalamus and amygdala of mouse forebrain. Brain research Gene expression patterns. 2002; 1:95-99.
86. Gorski M, Tin A, Garnaas M, McMahon GM, Chu AY, Tayo BO, Pattaro C, Teumer A, Chasman DI, Chalmers J, Hamet P, Tremblay J, Woodward M, et al. Genome-wide association study of kidney function decline in individuals of European descent. Kidney international. 2014.
87. Wang X, Ju L, Fan J, Zhu Y, Liu X, Zhu K, Wu M and Li L. Histone H3K4 methyltransferase Mll1 regulates protein glycosylation and tunicamycin-induced apoptosis through transcriptional regulation. Biochim Biophys Acta. 2014; 1843:2592-2602.
88. Gibson TM, Wang SS, Cerhan JR, Maurer MJ, Hartge P, Habermann TM, Davis S, Cozen W, Lynch CF, Severson RK, Rothman N, Chanock SJ and Morton LM. Inherited genetic variation and overall survival following follicular lymphoma. American journal of hematology. 2012; 87:724-726.
89. Clarke E, Green RC, Green JS, Mahoney K, Parfrey PS, Younghusband HB and Woods MO. Inherited deleterious variants in GALNT12 are associated with CRC susceptibility. Human mutation. 2012; 33:1056-1058.
90. Gray-McGuire C, Guda K, Adrianto I, Lin CP, Natale L, Potter JD, Newcomb P, Poole EM, Ulrich CM, Lindor N, Goode EL, Fridley BL, Jenkins R, et al. Confirmation of linkage to and localization of familial colon cancer risk haplotype on chromosome 9q22. Cancer research. 2010; 70:5409-5418.
91. Berois N, Blanc E, Ripoche H, Mergui X, Trajtenberg F, Cantais S, Barrois M, Dessen P, Kagedal B, Benard J, Osinaga E and Raguenez G. ppGalNAc-T13: a new molecular marker of bone marrow involvement in neuroblastoma. Clinical chemistry. 2006; 52:1701-1712.
92. Huanna T, Tao Z, Xiangfei W, Longfei A, Yuanyuan X, Jianhua W, Cuifang Z, Manjing J, Wenjing C, Shaochuan Q, Feifei X, Naikang L, Jinchao Z, et al. GALNT14 mediates tumor invasion and migration in breast cancer cell MCF-7. Mol Carcinog. 2015; 54:1159-1171.
93. Yeh CT, Liang KH, Lin CC, Chang ML, Hsu CL and Hung CF. A single nucleotide polymorphism on the GALNT14 gene as an effective predictor of response to chemotherapy in advanced hepatocellular carcinoma. International journal of cancer Journal international du cancer. 2014; 134:1214-1224.
94. Wang R, Yu C, Zhao D, Wu M and Yang Z. The mucin-type glycosylating enzyme polypeptide N-acetylgalactosaminyltransferase 14 promotes the migration of ovarian cancer by modifying mucin 13. Oncology reports. 2013; 30:667-676.
95. Wagner KW, Punnoose EA, Januario T, Lawrence DA, Pitti RM, Lancaster K, Lee D, von Goetz M, Yee SF, Totpal K, Huw L, Katta V, Cavet G, et al. Death-receptor O-glycosylation controls tumor-cell sensitivity to the proapoptotic ligand Apo2L/TRAIL. Nature medicine. 2007; 13:1070-1077.
96. Soria JC, Mark Z, Zatloukal P, Szima B, Albert I, Juhasz E, Pujol JL, Kozielski J, Baker N, Smethurst D, Hei YJ, Ashkenazi A, Stern H, et al. Randomized phase II study of dulanermin in combination with paclitaxel, carboplatin, and bevacizumab in advanced non-small-cell lung cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2011; 29:4442-4451.
97. Stern HM, Padilla M, Wagner K, Amler L and Ashkenazi A. Development of immunohistochemistry assays to assess GALNT14 and FUT3/6 in clinical trials of dulanermin and drozitumab. Clinical cancer research : an official journal of the American Association for Cancer Research. 2010; 16:1587-1596.
98. Liang KH, Lin CC and Yeh CT. GALNT14 SNP as a potential predictor of response to combination chemotherapy using 5-FU, mitoxantrone and cisplatin in advanced HCC. Pharmacogenomics. 2011; 12:1061-1073.
99. Abuli A, Fernandez-Rozadilla C, Alonso-Espinaco V, Munoz J, Gonzalo V, Bessa X, Gonzalez D, Clofent J, Cubiella J, Morillas JD, Rigau J, Latorre M, Fernandez-Banares F, et al. Case-control study for colorectal cancer genetic susceptibility in EPICOLON: previously identified variants and mucins. BMC Cancer. 2011; 11:339.
100. Nakayama Y, Nakamura N, Oki S, Wakabayashi M, Ishihama Y, Miyake A, Itoh N and Kurosaka A. A putative polypeptide N-acetylgalactosaminyltransferase/Williams-Beuren syndrome chromosome region 17 (WBSCR17) regulates lamellipodium formation and macropinocytosis. The Journal of biological chemistry. 2012; 287:32222-32235.
101. Raman J, Guan Y, Perrine CL, Gerken TA and Tabak LA. UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferases: completion of the family tree. Glycobiology. 2012; 22:768-777.
102. Takasaki N, Tachibana K, Ogasawara S, Matsuzaki H, Hagiuda J, Ishikawa H, Mochida K, Inoue K, Ogonuki N, Ogura A, Noce T, Ito C, Toshimori K, et al. A heterozygous mutation of GALNTL5 affects male infertility with impairment of sperm motility. Proceedings of the National Academy of Sciences of the United States of America. 2014; 111:1120-1125.
103. Ichikawa S, Baujat G, Seyahi A, Garoufali AG, Imel EA, Padgett LR, Austin AM, Sorenson AH, Pejin Z, Topouchian V, Quartier P, Cormier-Daire V, Dechaux M, et al. Clinical variability of familial tumoral calcinosis caused by novel GALNT3 mutations. Am J Med Genet A. 2010; 4:896-903.
104. Ichikawa S, Guigonis V, Imel EA, Courouble M, Heissat S, Henley JD, Sorenson AH, Petit B, Lienhardt A and Econs MJ. Novel GALNT3 mutations causing hyperostosis-hyperphosphatemia syndrome result in low intact fibroblast growth factor 23 concentrations. J Clin Endocrinol Metab. 2007; 92:1943-1947.
105. Marucci A, Cozzolino F, Dimatteo C, Monti M, Pucci P, Trischitta V and Di Paola R. Role of GALNT2 in the modulation of ENPP1 expression, and insulin signaling and action: GALNT2: a novel modulator of insulin signaling. Biochim Biophys Acta. 2013; 1833:1388-1395.
106. Marucci A, di Mauro L, Menzaghi C, Prudente S, Mangiacotti D, Fini G, Lotti G, Trischitta V and Di Paola R. GALNT2 expression is reduced in patients with Type 2 diabetes: possible role of hyperglycemia. PloS one. 2013; 8:e70159.
107. Itoh N, Ohta H and Konishi M. Endocrine FGFs: Evolution, Physiology, Pathophysiology, and Pharmacotherapy. Frontiers in endocrinology. 2015; 6:154.
108. Lin MC, Huang MJ, Liu CH, Yang TL and Huang MC. GALNT2 enhances migration and invasion of oral squamous cell carcinoma by regulating EGFR glycosylation and activity. Oral oncology. 2014; 50:478-484.
109. Hanahan D and Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144:646-674.
110. Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y, Hu Y, Tan Z, Stokes M, et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007; 131:1190-1203.

