Interview with Dr. Katherine M. Bever from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins
Dr. Katherine Bever from Johns Hopkins University discusses a 2020 research paper she co-authored that was published by Oncotarget, entitled, "An exploratory study of metformin with or without rapamycin as maintenance therapy after induction chemotherapy in patients with metastatic pancreatic adenocarcinoma."
Behind the Study is a series of transcribed videos from researchers elaborating on their oncology-focused studies published by Oncotarget. Visit the Oncotarget YouTube channel for more insights from outstanding authors.
—
My name is Katherine Bever. I'm an instructor in medical oncology at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University in Baltimore, Maryland. I'm going to be discussing our paper entitled "Exploratory study of metformin with Or without rapamycin as maintenance therapy after induction chemotherapy in patients With metastatic pancreatic adenocarcinoma."
The background to this work is that, as we know, metastatic pancreatic cancer is a highly aggressive disease with limited prognosis in most patients. And although we now have multi-aging chemotherapy regimens that can induce deep responses in many patients, the optimal management of patients who do respond is not known. So-called maintenance therapy is of increasing interest as a way to maintain responses with lower toxicity, using either reduced intensity chemotherapy or non-chemotherapeutic approaches. In particular, targeted agents which are ineffective in the progressive disease setting, may be more efficacious in the stable or defaulted disease state.
For example, we've recently seen the approval of the PARP inhibitor, Olaparib, for maintenance therapy in pancreatic cancer associated with germline mutations in BRCA. Our interest going into this study was in the role of mTOR inhibition in pancreatic cancer, and particularly dual inhibition of the mTOR pathway with rapamycin, which blocks mTOR complex 1 directly and metformin, the commonly used antidiabetic drug, which inhibits mTOR through M kinase activation. This combination showed potential synergy in suppressing tumor cell proliferation in preclinical models. So we hypothesized a role for these agents in the maintenance setting for pancreatic cancer.
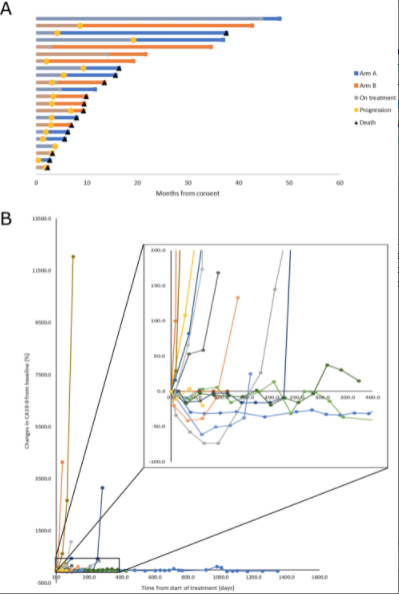
This regimen in the trial demonstrated good safety and tolerability with grade three and four treatment-related adverse events seen only with the combination in all being asymptomatic laboratory abnormalities. Somewhat surprisingly, we observed several exceptionally long survivors in both groups, with a two-year revival rate of 37% overall, which is quite impressive for metastatic pancreatic cancer.
This confirmed for us that there may be a biologically distinct group of patients entering maintenance therapy with exceptional prognosis, which has not been previously well described in the literature. In our analysis low neutrophil to lymphocyte ratio and low FDG avidity on baseline PET scan were associated with better prognosis and may be a useful way to define this subgroup in subsequent maintenance studies.
One challenge we encountered with this study, which really applies broadly to studies in the maintenance setting, is our ability to measure the clinical activity of the therapies under study. Specifically in these patients who have been de bulked by chemotherapy, there may not be a reasonable expectation of further reduction in tumor size, according to the conventional resist criteria for response. We sought to explore the use of PET scans and changes in FDG uptake according to pre-specified response criteria that were developed at Hopkins called Persist.
And although this analysis was also limited by lack of accessible disease at baseline in many patients, we did observe are measurable decreases in activity in two patients who were among the longer survivors on this study. Additionally, we explored the biologic activity of the agents through a recorrelative analysis and in particular, analysis of circulating immune cells. As expected metformin plus rapamycin was associated with decreased mTOR activity in several immune subsets and lower metabolic fitness overall, in circulating immune cells compared to metformin alone. However importantly, these observations did not appear to correlate with outcomes.
We used high-dimensional flow cytometric analysis to identify changes in immune cell subsets on therapy. And while most immune subsets did not change significantly, we observed an expansion of dendritic cells in both arms, which was more pronounced in long-term survivors. Finally, we reported on a unique population of CD14 positive cells, which were significantly more abundant in the peripheral blood of short-term survivors. And so we proposed that these may represent an important inhibitory population of monocytes that deserves further study for its significance, both prognostically and therapeutically.
In summary, this paper, we believe, contributes important biologic clues in a group which has previously been poorly described in the literature, patients with metastatic pancreatic cancer, entering the maintenance setting. We show that this may be a biologically distinct and heterogeneous group, some of whom may have exceptional prognosis. And further studies are needed to more clearly define who may benefit from maintenance-targeted therapies, such as mTOR inhibition versus can of chemotherapy oration alone.
Click here to read the full study published by Oncotarget.
YOU MAY ALSO LIKE: More Oncotarget Videos on LabTube
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues, and other researchers, far and wide.
For media inquiries, please contact [email protected].

Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLCß

