PD1 is expressed on exhausted T cells as well as virus specific memory CD8+ T cells in the bone marrow of myeloma patients
2020-01-09
Here, the researchers found similar proportions of PD1+ CD8+ T cells and similar levels of PD1 expression on CD8+ T cells in the bone marrow of myeloma patients and healthy controls. PD1 expression on CD8+ T cells did not correlate with tumor load suggesting that at least some of the PD1+ CD8+ T cells were specific for non-myeloma antigens.
Dr. Anne-Marit Sponaas from the Department of Clinical and Molecular Medicine, Myeloma Research Center, Norwegian University of Science and Technology, Trondheim, Norway and the Centre of Molecular Inflammation Research, Centre of Molecular Immune Regulation, Norwegian University of Science and Technology, Trondheim, Norway said, "Anti-PD1 treatment has been effective in clinical trials of several advanced hematological and solid cancers."
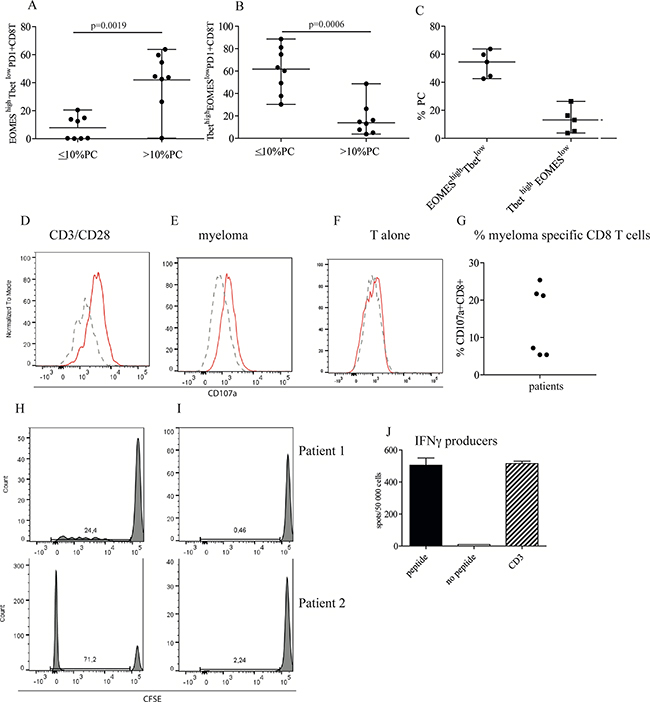
Figure 3: High tumor load is associated with increased number of CD8+EOMEShigh T betlow cells. (A, B, C) Frequency of EOMEShigh Tbetlow of PD1+CD8+ T cells. Bone marrow cells were surface stained with anti CD3, CD4, CD8 and PD1 followed by intranuclear staining with anti-Tbet and anti-EOMES, or isotype controls (Supplementary Figure 4A). Gates were set on isotype controls. Figure shows % EOMEShighTbetlow (A) and TbethighEOMESlow (B) of PD1+CD8+ T cells in patients with low plasma cell percentage (≤ 10%) or high plasma cell percentage (>10 % PC). (C) % EOMEShighTbetlow and Tbethigh EOMESlowof PD1 expressing CD8 T cells in patients with high tumor load of 40% and above. (D-G) Cytotoxic activity against autologous myeloma cells. Purified CD8+ T cells from the bone marrow of patients were co-cultured with anti CD3/CD28 beads (D) or purified, autologous myeloma cells (E) or the T cells alone (F). For the last 4 hours of the culture period, anti CD8, PD1 and CD107a or isotype controls were added as described in the Materials and Method section. Figures show the staining of isotype control (dashed lines) and CD107a (solid lines) on one representative sample of three positive (Supplementary Figure 4C). (G) Pooled data for cytotoxic activity (% CD107a expression) of CD8+ T cells co-cultured with autologous myeloma cells. (H, I) Proliferation of CD8+PD1+ T cells in response to autologous APCs. CFSE labeled CD3+PD1+ cells from the bone marrow of two myeloma patients (1,2) were cultured with (H) or without (I) autologous, adherent cells in the presence of recombinant IL2 as described. (J) PD1+ EBV specific CD8+ T cells in the bone marrow of myeloma patients. PD1+ cells enriched from the bone marrow was cultured with autologous, adherent cells as described in Materials and Methods in the presence of EBV specific peptides. After 10 days of culture the cells were seeded onto IFNγ ELIspot strips together with differentiated DC and EBV peptides (filled bar), without peptides (open bar) or with anti CD3 antibody (striped bar). Spots were developed after 24 hrs. Figure shows representative data from one of the 6 EBV positive patients tested. Data from all the 6 patients are shown in Table 1.
PD1 expression on T cells at the tumor site has been proposed to be a prerequisite for successful treatment, as lack of PD1 expression on tumor infiltrating T cells were associated with reduced response to checkpoint therapy. Although PD1-treatment has been successful in an animal model on myeloma and anti-PDL1 antibodies have reinvigorated exhausted T cells from a myeloma patient to kill myeloma cells in vitro, it is not clear whether anti-PD1/PDL1 treatment induce anti-tumor activity by reinvigorating myeloma-specific exhausted T cells in myeloma patients.
PD1 is not only expressed on dysfunctional T cells, such as anergic and exhausted T cells, but also on terminal effector T cells and memory T cells. In this study we tested whether PD1 expression on CD8+ T cells from bone marrow correlated with tumor load and investigated whether these T cells could respond to autologous myeloma cells in vitro. However, these PD1+ CD8+ T cells failed to degranulate in the presence of autologous myeloma cells and PD1 antibody, suggesting specificity to non- tumor antigens.
The Anne-Marit Sponaas research team concluded, "Our data showing the presence of PD1intermediate EOMEShigh Tbetlow, as well as PD1+ memory cells in patients with tumor load above 10% bone marrow plasma cells could give an indication that checkpoint inhibition would be beneficial if given at the correct time when there are enough tumor responsive CD8+ T cells around."
Full text - https://doi.org/10.18632/oncotarget.25882
Correspondence to - Anne-Marit Sponaas - [email protected]
Keywords - myeloma, bone marrow, checkpoint molecules, CD8 T cells, exhaustion
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC



