Oncotarget: PBMCs generate TEM/TCM for adoptive cell therapy in multiple myeloma
2021-09-28
Oncotarget published "Multipeptide stimulated PBMCs generate TEM/TCM for adoptive cell therapy in multiple myeloma" which reported that Multiple Myeloma patients suffer disease relapse due to the development of therapeutic resistance. Increasing evidence suggests that immunotherapeutic strategies can provide durable responses.
The authors designed peptides from antigens including cancer testis antigens that are over expressed in MM. They exposed PBMCs from different healthy donors to single peptides. These peptide sequences were used to compile four different peptide cocktails. Naïve T cells from PBMCs from MM patients or HDs recognized the cognate Ag in all four peptide cocktails, leading to generation of multiclonal Ag-specific CD4+ and CD8+ effector and central memory T cells which produced interferon-gamma, granzyme B and perforin on secondary restimulation.
Dr. Sandra J. Gendler from The Mayo Clinic said, "Multiple Myeloma (MM) is a plasma cell malignancy mostly affecting elderly people."
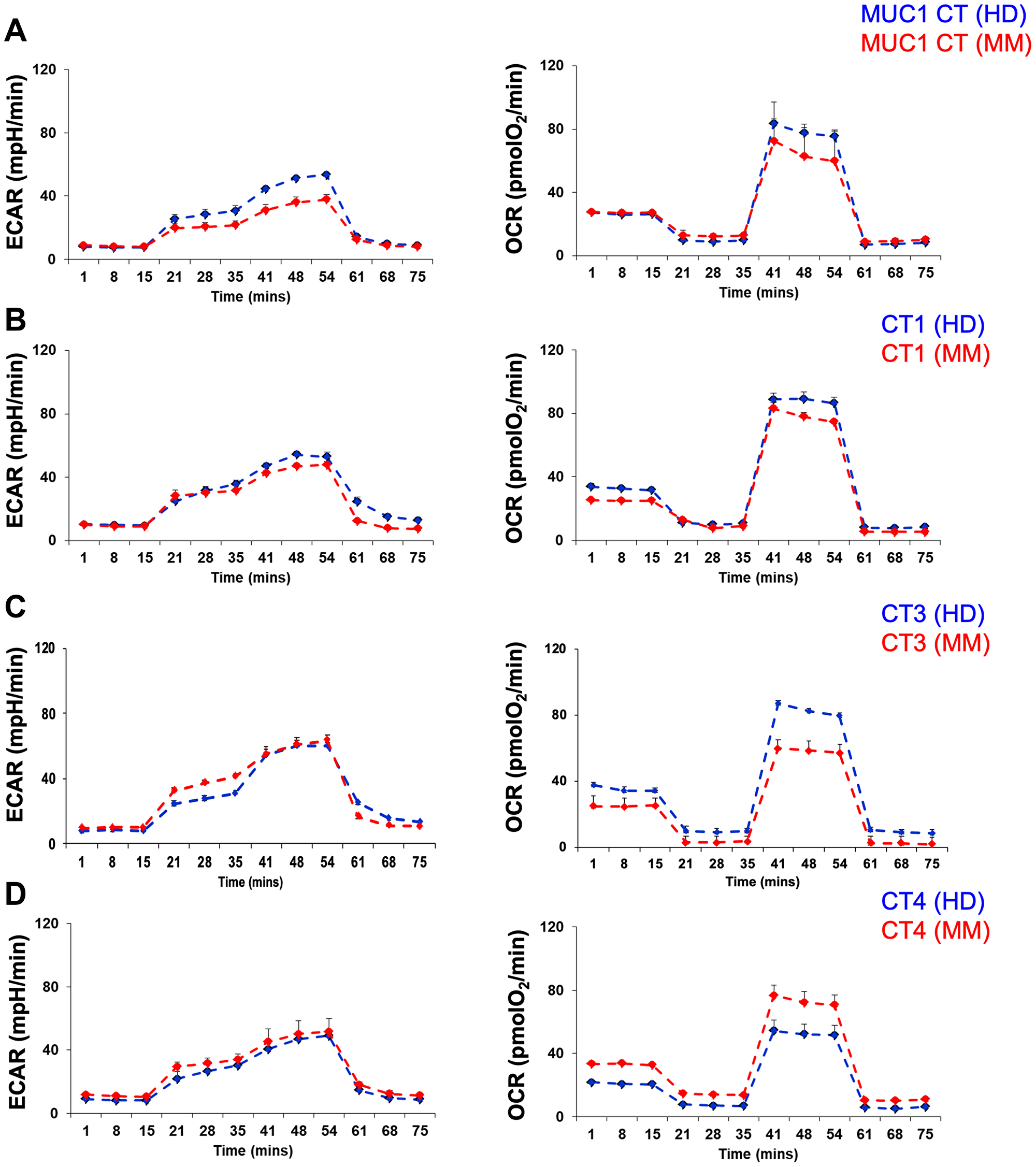
Figure 6: Metabolic profile of healthy donor or multiple myeloma patient's memory T cell population varies depending upon the peptide cocktail used for stimulation. Glycolysis stress test was conducted to examine the extracellular acidification rate (ECAR) in response to glucose, oligomycin, and 2-deoxy-D-glucose (2DG). The Mito-cell stress test assessed the oxygen consumption rate (OCR) following treatment with oligomycin, carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP) and rotenone/antimycin. The histograms for ECAR (left panel) and OCR (right panel) are depicted for T cells generated following exposure of PBMCs from healthy donor (blue histogram) or multiple myeloma patient (red histogram) to MUC1 cocktail (A), cocktail 1 (B), cocktail 3 (C) and cocktail 4 (D). Representative data are shown and no statistically significant differences were observed among the six samples analyzed (Student's t-test).
It is characterized by clonal proliferation of terminally differentiated antibody-producing plasma cells in the bone marrow, leading to osteolytic bone lesions. It is the second most common malignancy among hematological cancers with an incidence rate of 4.5–6 per 100,000 individuals per year. The global rate of incidence and death has increased by 126% and 94%, respectively, from 1990–2016. Despite the availability of various therapeutic regimens, MM remains an incurable disease and patients succumb to it mainly due to development of resistance attributed to the constantly evolving genomic landscape in the plasma cells/tumor microenvironment and the cross-talk between them.
Peptides were designed from numerous Ags based on their level of expression and role in MM pathogenesis: B Cell Maturation Antigen, Mucin1, Fc Receptor Like 5, Myeloid Cell Leukemia 1, Receptor for Hyaluronan-Mediated Mobility, Self-Ligand Receptor of the Signaling Lymphocytic Activation Molecule Family 7, spliced isoform of X-Box Binding Protein 1 XBP, Cancer Testis Antigen, Melanoma Antigen Family 3/6, New York Esophageal Squamous Cell Carcinoma 1, SEPTIN9 and Wilms Tumor 1.
The significantly enhanced expression of BCMA on MM has been shown to be crucial for its growth and survival which makes it an attractive target. It promotes malignancy by regulating cell morphology, adherence, and migration and could be a potential immunotherapeutic target. MAGEA3 promotes MM growth by preventing apoptosis. NY-ESO-1, an immunogenic CTA, induces spontaneous immune responses, and its expression correlates with poor prognosis in MM. SEPT9, a gene that encodes SEPTIN9, is hypermethylated in colorectal cancer and is being proposed for non-invasive screening purposes. Preliminary evidence suggests that SEPT9 is downregulated in MM. The increase in WT1 transcripts correlates with the worsening of clinical factors and stage, and Azuma et al. have shown that MM cells are highly sensitive to the granule exocytosis pathway mediated by WT1-specific cytotoxic T lymphocytes.
MM cells are highly sensitive to the granule exocytosis pathway mediated by WT1-specific cytotoxic T lymphocytes.
The Gendler Research Team concluded in their Oncotarget Research Output that these results provide the basis for an additional novel immunotherapy, ACT, for MM patients, all of whom relapse from available therapies. Generation of memory cells adds an important dimension to the effector cells generated by CAR-T cell therapy. Expression of granzyme B and perforin in > 90% of the IFN-+ T cells suggests cytolytic activity.
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.28067
Full text - https://www.oncotarget.com/article/28067/text/
Correspondence to - Sandra J. Gendler - [email protected]
Keywords - multiple myeloma, cellular therapy, CD4/CD8 T cells, peptides, immunotherapy
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




