Oncotarget | Genetic biomarkers predict response to dual BCL-2 and MCL-1 targeting in acute myeloid leukaemia cells
2020-03-04
The cover for issue 102 of Oncotarget features Figure 9, "Apoptotic resistance to Venetoclax in FLT3 mutated primary samples can be reversed when combined with S63845," by Grundy, et al.
Acute myeloid leukaemia cells often up-regulate pro-survival members of the BCL-2 protein family, such as BCL-2 and MCL-1, to avoid apoptosis.
The research team treated MV4-11 cells and primary AML samples for 4 hours with Venetoclax, S63845 or the combination.
The combination of Venetoclax and S63845 produced a synergistic apoptotic response in MV4-11 cells and primary samples, including the leukaemia re-populating leukaemic stem cell population, in 92% of the samples.
Dr. Martin Grundy from the Clinical Haematology Department, at Nottingham University Hospitals, in Nottingham, United Kingdom said "Primary AML cells are fragile in vitro and the majority will die spontaneously in culture fairly rapidly. By focusing on a same-day functional assay, with intact cells, we aim to overcome this obstacle."
"Primary AML cells are fragile in vitro and the majority will die spontaneously in culture fairly rapidly. By focusing on a same-day functional assay, with intact cells, we aim to overcome this obstacle."
- Dr. Martin Grundy, Clinical Haematology Department, at Nottingham University Hospitals
The apoptotic fate of a cell depends on a fine balance of interactions between pro-survival molecules such as BCL-2, MCL-1 and BCL-XL and BH3-only protein sensitizer molecules such as BAD and NOXA.
The most promising BCL-2 inhibitor to date is the BH3 mimetic Venetoclax, which has demonstrated clinical promise in AML, particularly when used in combination with cytarabine or hypomethylating agents.
The research team has previously reported that a number of agents that indirectly inhibit MCL-1 expression produce a synergistic apoptotic response when used in combination with Venetoclax in AML cells.
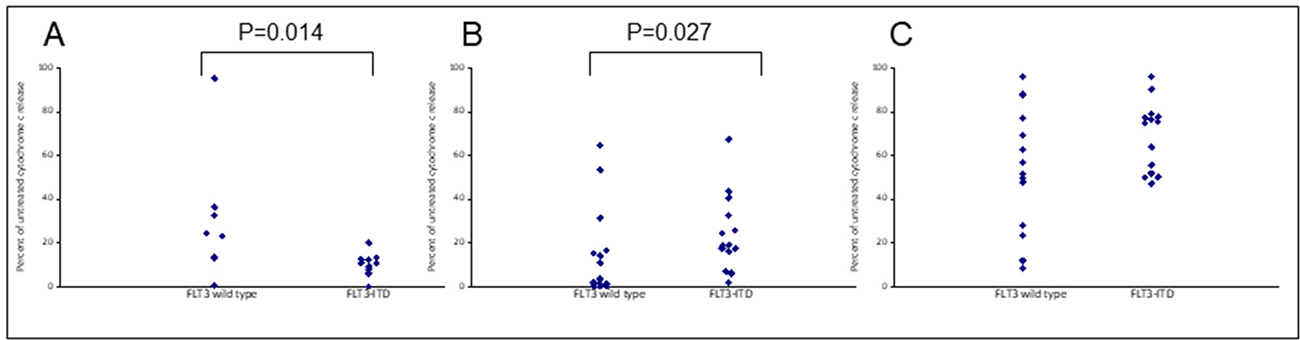
Figure 9: Apoptotic resistance to Venetoclax in FLT3 mutated primary samples can be reversed when combined with S63845. Primary AML cells were treated with 100 nM Venetoclax (A), 50 nM S63845 (B) or a combination of both (C) for 4 hours. Cells were then fixed and processed for cytochrome C release. Samples are grouped as either wild-type or mutant according to FLT3 mutational status.
Resistance to Venetoclax monotherapy is associated with released BIM being sequestered by MCL-1 and others have reported that this can be reversed by combining Venetoclax with cytarabine or daunorubicin.
Using a short-term functional assay, they demonstrate that co-operative targeting of BCL-2 and MCL-1 with the BH3-mimetics Venetoclax and S63845 induces an enhanced apoptotic response in bulk and LSC populations of primary AML samples.
The research team concluded in their Oncotarget Research Paper, "We report a real-time cytochrome C release assay which confirms known biomarkers of response to drugs and identifies others. The assay has the potential to determine which patient subgroups might best respond to novel agents."
Full text - https://doi.org/10.18632/oncotarget.26540
Correspondence to - Martin Grundy - [email protected]
Keywords - AML, BCL-2, MCL-1, Venetoclax, S63845
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.impactjournals.com/ or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




