Oncotarget: Enhancing anti-tumor activity in triple negative breast cancer
2021-10-29
Oncotarget published "Selinexor, a selective inhibitor of nuclear export, enhances the anti-tumor activity of olaparib in triple negative breast cancer regardless of BRCA1 mutation status" which reported that the authors examined the effects of co-treatment with selinexor and olaparib in TNBC cell lines.
BRCA1 wildtype and BRCA1 mutant TNBC cell lines were treated with selinexor and/or olaparib and effects on cell viability and cell cycle were evaluated. The effects of treatment were also evaluated in mouse xenograft models generated with BRCA1-wt and BRCA1-mut TNBC cell lines. Treatment with selinexor inhibited cell proliferation and survival of all TNBC cell lines tested in vitro.
Dr. Hélène Marijon from The University of California as well as The Franco-British Hospital said, "Breast cancer is one of the three most common cancers (together with lung and colorectal cancers) in the United States, accounting for 30% of female cancers."
Approximately 15–20% of breast cancers are triple-negative breast cancer, which usually corresponds to basal breast cancer and is characterized by the absence of HER2, estrogen, and progesterone receptors.
Patients with TNBC have poor prognosis with the worst disease-free and overall survival rates of all breast cancer types.
Patients with TNBC have poor prognosis with the worst disease-free and overall survival rates of all breast cancer types
Preclinical and clinical evidence have demonstrated that PARPi also affects cancer cells with other DNA repair mechanism defects. Although 35% of all TNBC cases exhibit homologous recombination repair deficiency, in clinical practice, olaparib does not benefit TNBC patients without BRCA1/2 mutations. This inhibition causes accumulation of tumor suppressor proteins in the nucleus of malignant cells and blocks protein translation of oncogenes that drive cell proliferation, leading to cell cycle arrest and apoptosis of malignant cells.
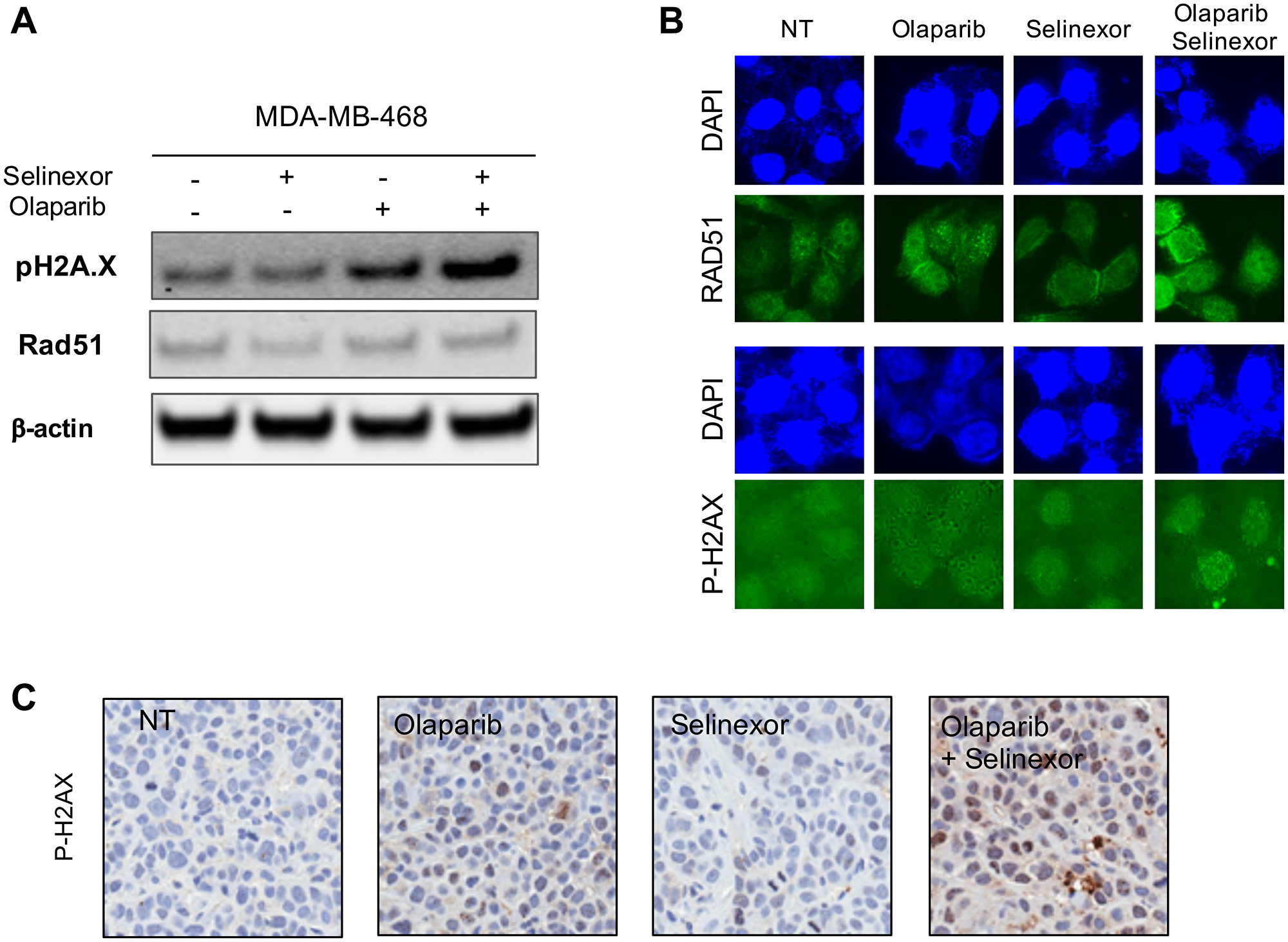
Figure 1: Flowchart of trial screening and inclusion. Of 1877 trials identified on https://clinicaltrials.gov/ (February 20, 2020), 841 were excluded, for a final total of 1036 phase III randomized clinical trials assessing therapeutic interventions in patients with cancer. Of those, 790 trials had peer-reviewed manuscripts of primary study endpoint, and were included in primary analysis for this manuscript.
The Marijon Research Team concluded in their Oncotarget Research Output, "combination of selinexor and olaparib induces robust anti-tumor activity in vitro and in vivo in TNBC cell lines with or without a BRCA1 mutation. Our pre-clinical data support further investigation of the mechanism of action affecting this combination therapy in TNBC."
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.28047
Full text - https://www.oncotarget.com/article/28047/text/
Correspondence to - Hélène Marijon - [email protected]
Keywords - selinexor, XPO1, olaparib, triple negative breast cancer, BRCA1
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




