Delayed adverse events in phase I trials of molecularly targeted and cytotoxic agents
2019-12-20
Mean AE totals for CCs or MTAs respectively was 4.7 versus 3.0 in cycle 1, 3.8 versus 2.8 in cycles 2-6.Patients on CCs had higher mean AEs in six cycles compared to those on MTAs. For MTAs, more frequent incidence of first grade 3/4 AEs after cycle 1 supports incorporation of delayed AEs into recommendations for Phase 2 dosing.
Dr. Debashis Sarker from the School of Cancer and Pharmaceutical Sciences, King's College London, London, UK and Guy's and St Thomas' NHS Foundation Trust, Guy's Hospital, London, UK said "Adverse events in early phase oncology trials play a key role in dose escalation , with dose-limiting toxicities being particularly important."
Cytotoxic agents generally exhibit more severe and higher grades of toxicity in earlier cycles, whereas molecularly targeted agents are often administered chronically until progression, making consideration of lower grade AEs and those in later cycles highly relevant.
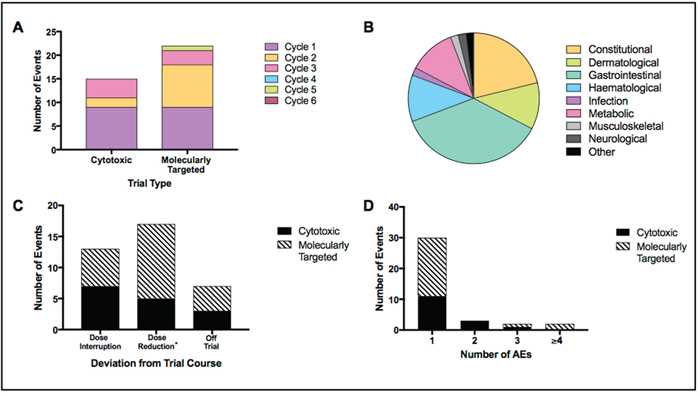
Figure 4: Treatment interruptions and dose modifications. Display of several variables relating to treatment interruptions and dose modifications. Events included dose interruptions, dose reductions or removal of patient from trial. Results displayed as; (A) events by trial type – cytotoxic combination or molecularly targeted single agent; (B) AEs contributing to all events split by category; (C) events by type – dose interruption, dose reduction (*including dose interruptions that resulted in dose reductions) and removal of patient from trial; (D) events by number of contributing AEs.
The primary objective of this study was to analyse AEs from patients enrolled on relevant phase I dose escalation trials at a single UK centre to determine the incidence of AEs between drug classes, and between cycle 1 versus cycles 2-6.
Previous work has compared single agent cytotoxic and MTA dermatological AEs and found a substantial number of AEs occurring outside the DLT window with variation in AE type based on agent studied.
The Debashis Sarker research team concluded, "The results of this study provide an evaluation of drug toxicities between patients treated in cytotoxic combination and MTA trials."
Full text - https://doi.org/10.18632/oncotarget.26104
Correspondence to - Debashis Sarker - [email protected]
Keywords - adverse events, phase I trials, molecularly targeted agents, cytotoxic agents
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




