Oncotarget: Clinician's perspective on the 21-gene assay in early breast cancer
2021-12-21
Oncotarget published "The clinician's perspective on the 21-gene assay in early breast cancer" which reported that Most patients with early HR and HER2- breast cancer receive a hormone therapy; the clinical question still open is how to identify patients who can really benefit from adjuvant chemotherapy.
The accurate identification of these patients is essential to avoid an over-treatment, increasing the risk of an unnecessary toxicity; on the contrary, the omission of chemotherapy can deprive high risk patients of a potential life-saving treatment.
The different available tests can be classified according to their clinical utility based on their prognostic and predictive value.
When the aim of the test is to drive the treatment decisions, the predictive component, and therefore the ability to accurately identify which patients could benefit from chemotherapy, is essential.
This review summarizes the clinical evidences of the Oncotype DX® test supporting its clinical utility.
Dr. Francesco Cognetti from The University La Sapienza said, "The breast cancer represents 24% of all tumors and it is globally responsible for 15% of cancer death."
The breast cancer represents 24% of all tumors and it is globally responsible for 15% of cancer death
Breast cancer is the most frequent cancer in women and the first cause of death for cancer in women.
In Italy 53.000 new cases of breast cancer have been estimated in 2019. The breast cancer is the most diagnosed cancer in women, with a different rate depending on age: 40% in patients aged 0–50 years; 35% in patients aged 50–69 years and 22% in patients aged >70 years.
In patients undergoing surgery for early stage hormone-responsive breast cancers, the standard therapeutic approach for the prevention of local disease recurrence and metastases mainly consist of hormone therapy.
An overview performed by the Early Breast Cancer Trialists' Collaborative Group in 2012, showed a limited benefit from CT and, mostly, the inability to identify a subgroup of patients who could significantly benefit from CT, based on clinical and pathological characteristics only.
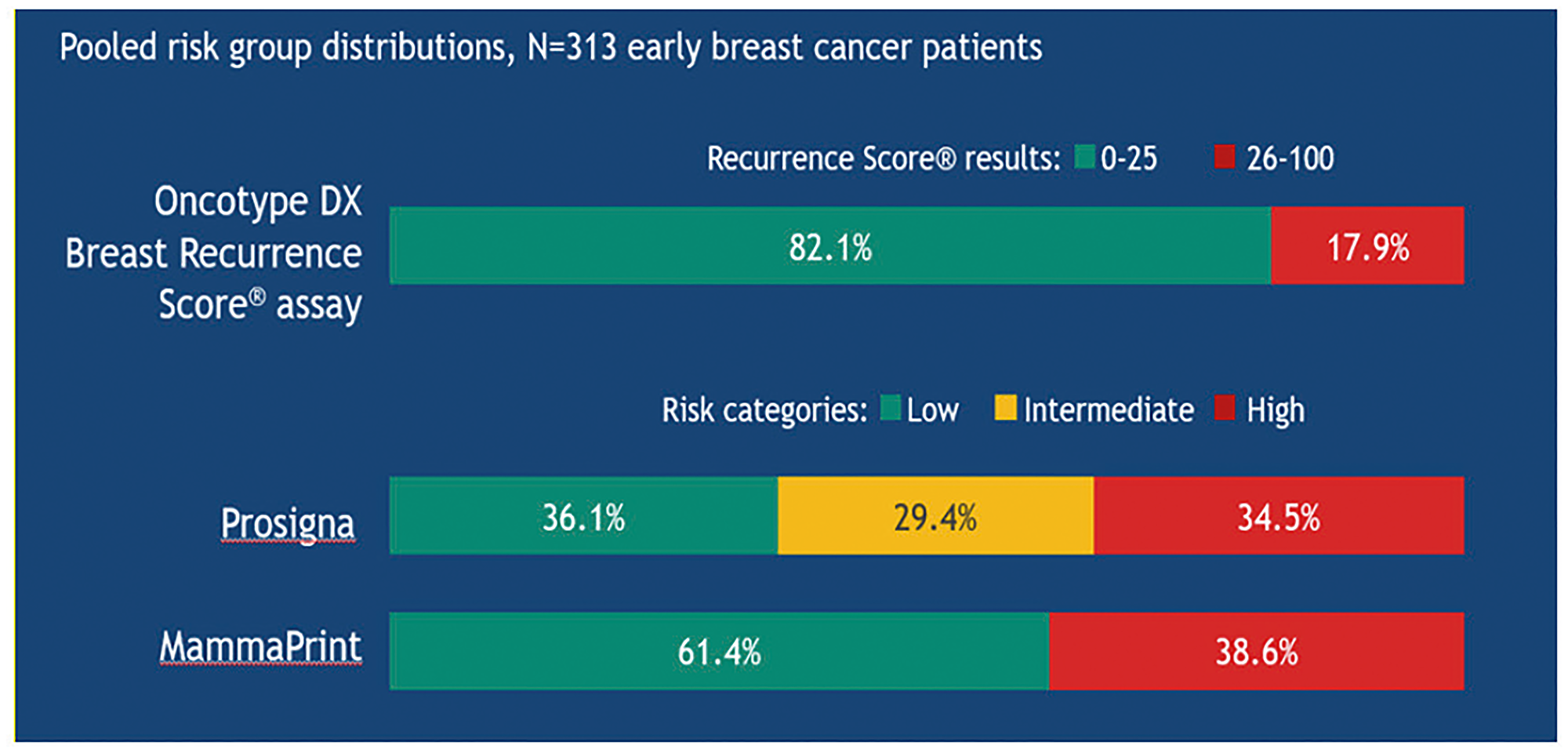
Figure 14: Risk stratification in the OPTIMA trial using different diagnostic tests in the same patients with early breast cancer.
The Research Team concluded in their Oncotarget Research Output that Since many patients with a HR, HER2- early breast cancer do not benefit from an adjuvant CT, there is a need to have a molecular rationale for identifying patients who can benefit from CT, as well those for whom CT would not add significant clinical benefit.
All multigene assays have a prognostic value, but only the Oncotype DX test has been shown to be able to identify which may benefit from adjuvant CT.
Studies comparing different genomic tests showed that they are not interchangeable, because they provide different results: MammaPrint, Prosigna and EndoPredict can identify a higher rate of “high risk” patients than the Oncotype DX test, with the risk of an absolutely unjustified over-treatment without any clinical benefit.
To date, the 21-gene assay is the only test developed and validated, with a level of evidence 1A, to be clinically useful, i.e. able to predict the benefit from an adjuvant CT and therefore suitable to guide the therapeutic choice based on RS results.
Clinical evidences from randomised controlled trials show that RS results 26–100 can predict a significant absolute benefit from the adjuvant CT, while RS results 0–25 indicate a minimal or totally absent benefit The scientific evidence supporting the Oncotype DX test shows that the adjuvant CT can be avoided in most HR /HER2- patients.
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.28148
Correspondence to - Francesco Cognetti - [email protected]
Keywords - Oncotype DX test, recurrence score results, multigene assays, genomic tests, personalized medicine
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




