Characterization of cancer-associated IDH2 mutations that differ in tumorigenicity, chemosensitivity and 2-hydroxyglutarate production
2019-10-30
With respect to IDH2, it is unclear whether all IDH2 mutations display the same or differ in tumorigenic properties and degrees of chemosensitivity.
Interestingly, while we found that -2-HG stimulated the growth of non-transformed cells, -2-HG also displayed antitumor activity by suppressing the growth of tumors harboring wild type IDH2.
Thus, the researcher's findings show that despite their shared -2-HG production, IDH2 mutations are not alike and differ in shaping tumor cell behavior and response to chemotherapeutic agents.
Dr. Ana M. Gamero from the Department of Medical Genetics and Molecular Biochemistry, at the Lewis Katz School of Medicine at Temple University in Philadelphia, PA, USA said "Isocitrate dehydrogenase (IDH) enzymes are key components of the tricarboxylic acid (TCA) cycle. NADP+-dependent cytoplasmic IDH1 and mitochondrial IDH2, as well as NAD+-dependent mitochondrial IDH3, catalyze the oxidative decarboxylation of isocitrate to ?-ketoglutarate (?-KG) and produce NADPH or NADH, respectively"
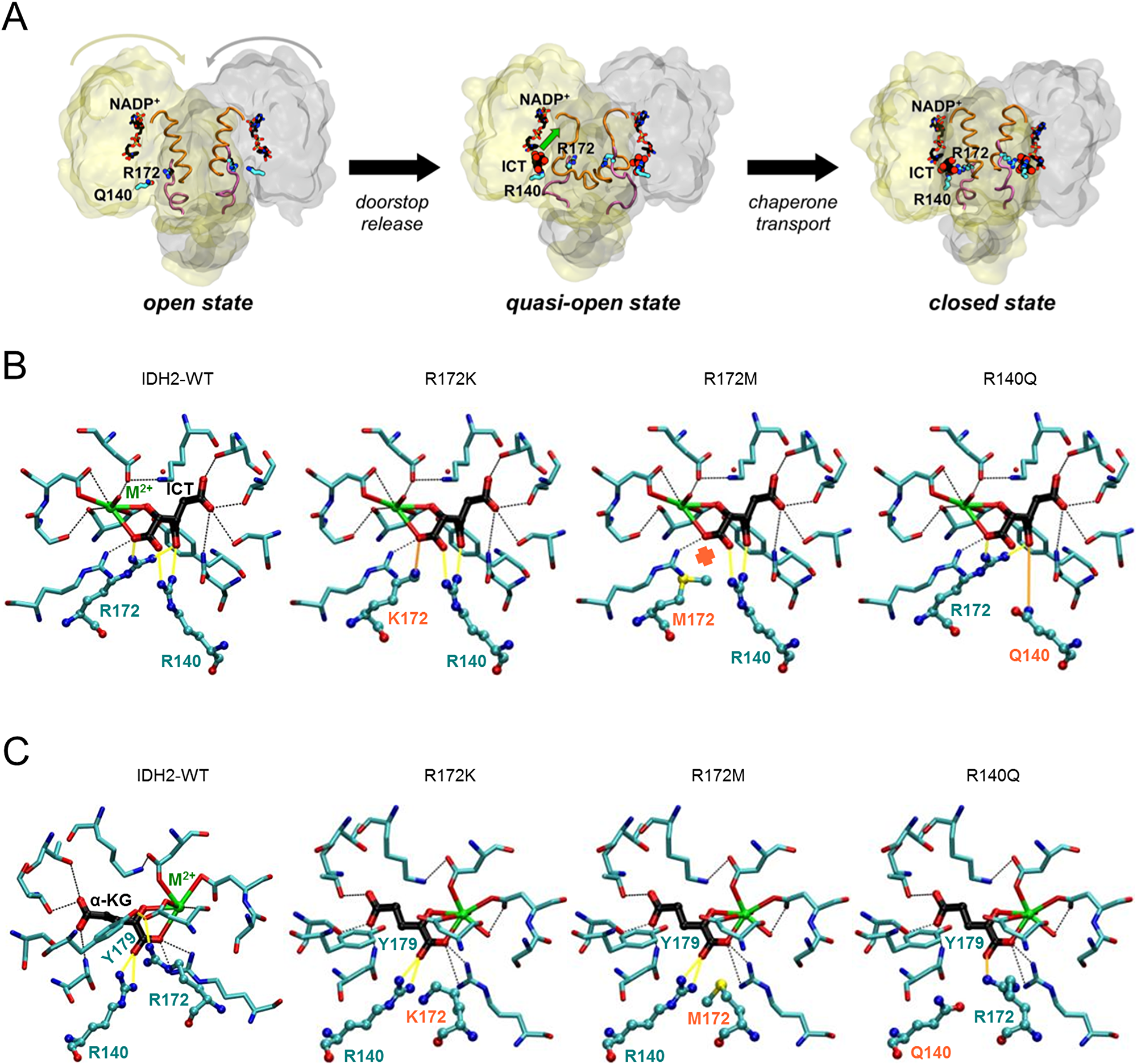
Mutated IDH enzymes utilize -KG, the product of the wild type reaction, as a substrate that is converted to -2-HG. IDH mutations and -2-HG production are also known to antagonize prolyl-hydroxylases, which are regulators of HIF-1 , promote cell growth , alter cell morphology , increase metastatic potential , and are associated with overall longer survival in glioma patients .
Additionally, positive temozolomide chemosensitivity in glioblastoma patients and overall survival were found to be associated with IDH1/2 mutations, but whether this was linked to a specific IDH mutation was not described.
It is presumed that all IDH mutations display similar biological activity due to their shared production of -2-HG, however, this premise has not been carefully investigated.
Thus, our studies demonstrate that in addition to -2-HG production, knowing the inherent nature of a specific IDH2 somatic mutation may be important in predicting the outcome of response to chemotherapeutic agents.
The Gamero research team concluded, "clinical trials have begun after preclinical studies demonstrated the efficacy of small molecule inhibitors against mutant forms of IDH1 and IDH2 accompanied by a reduction of -2-HG levels, which resulted in a block of de-differentiation and growth suppression of AML tumor cells.
Though AML and gliomas are similar in that they harbor IDH mutations, in glioblastoma cells it is a favorable marker for patient survival while in AML it is correlated with a worse prognosis.
Therefore, careful consideration must be given to the role of IDH mutations within the progression and maintenance of each of these diseases: the nature of the IDH mutation, the effects of -2-HG concentrations, and the stage of disease when IDH inhibitors are administered."
Full text - https://doi.org/10.18632/oncotarget.26848
Correspondence to - Ana M. Gamero - [email protected]
Keywords - glioblastoma, IDH2, tumorigenesis, chemotherapy, biomarker
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




