Oncotarget Volume 11 Issue 8 features Figure 8, "MLN4924 treatment induces DNA damage by stabilizing CDT1 and accumulates the cells in S phase which are enhanced by MLN4924/cisplatin co-treatment," by Misra, et al.
Moreover, adding MLN4924 to the standard TNBC chemotherapeutic agent cisplatin increased the DNA damage level, further enhancing the sensitivity.
In vivo, MLN4924 reduced tumor growth in a NOD-SCID mouse xenograft model by inducing DNA damage which was further augmented with the MLN4924 and cisplatin cotreatment.
Taken together, we demonstrated the mechanism of TNBC sensitization by the MLN4924 and MLN4924/cisplatin treatments irrespective of BRCA1 status, provided a strong justification for using MLN4924 alone or in combination with cisplatin, and identified a genetic background in which this combination will be particularly effective.
Dr. Alo Ray from the Department of Pathology and the Comprehensive Cancer Center at the Ohio State University in Columbus Ohio said, "Triple-negative breast cancer (TNBC) comprises 15–20% of breast cancer."
"Triple-negative breast cancer (TNBC) comprises 15–20% of breast cancer."
- Dr. Alo Ray, Center for Radiological Research, Department of Pathology and the Comprehensive Cancer Center at the Ohio State University
However, whether MLN4924 sensitizes breast tumors in vivo, is cytotoxic to BRCA1-mutated TNBC, and sensitizes TNBC to standard TNBC chemotherapeutics have not been investigated.
Here, we evaluated the efficacy of MLN4924 as a therapeutic agent in BRCA1-wild type and -mutant cells and examined if MLN4924 in combination with cisplatin, a standard platinum-based TNBC chemotherapeutic, enhances cytotoxicity.
Our results demonstrated that TNBC cell lines show a higher sensitivity to MLN4924 compared to cell lines representing other breast cancer subtypes due to the overexpression of NAE1 in TNBC compared to non-TNBC subtypes.
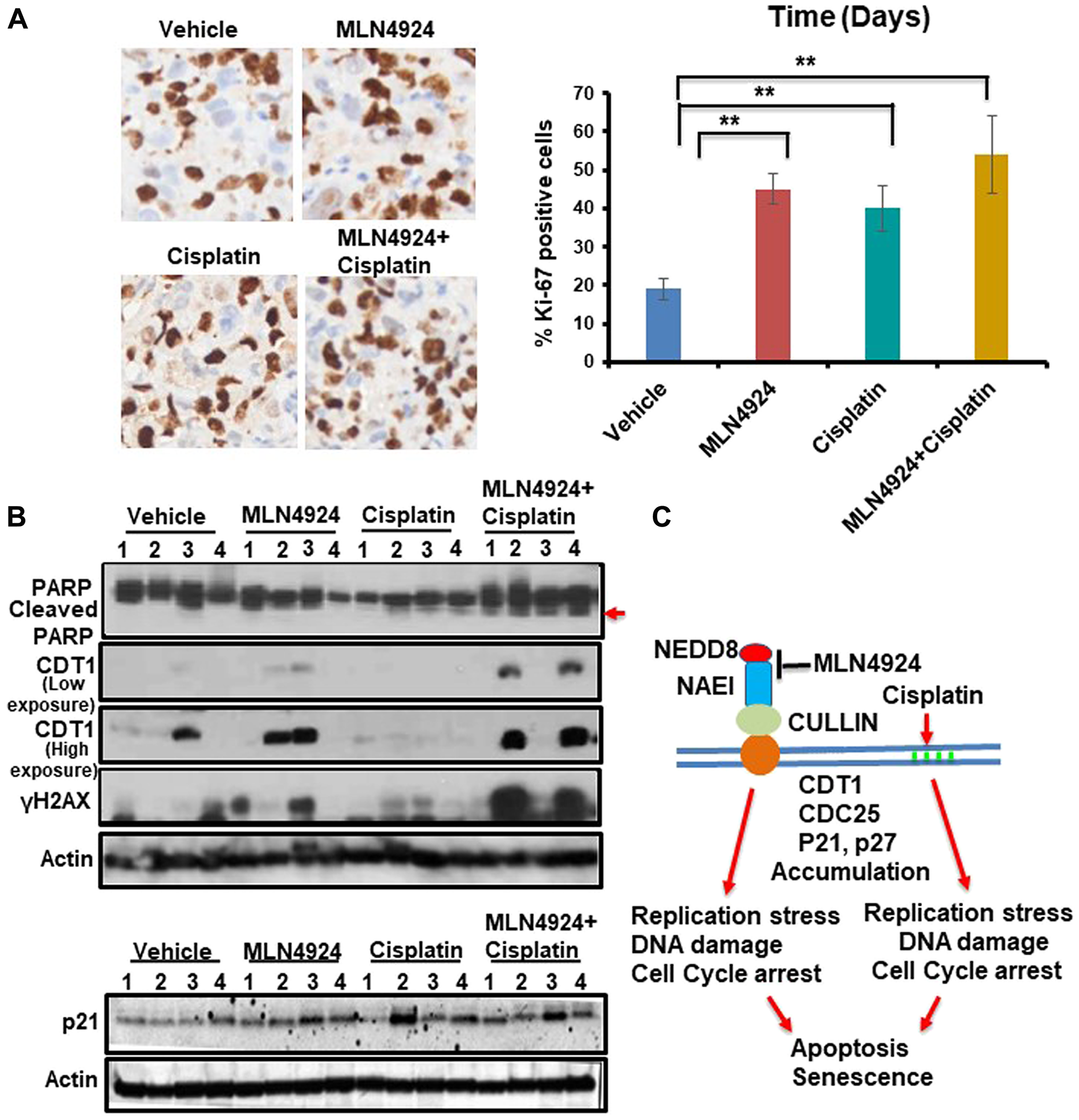
Figure 8: MLN4924 treatment induces DNA damage by stabilizing CDT1 and accumulates the cells in S phase which are enhanced by MLN4924/cisplatin co-treatment. (A) Representative images of Ki-67 staining (40× magnification) and quantitation of Ki-67 IHC. The results were from 4 tumors and data were presented as mean ± SD. **p < 0.001 indicates a significant difference. (B) WB showing the levels of cleaved PARP, CDT1, γH2AX and p21 in vehicle, MLN4924, Cisplatin, and MLN4924/Cisplatin treated tumors. (C) A model showing the pathway leading to apoptosis and senescence upon NAE1 inhibition by MLN4924 and synthetic lethality upon MLN4924/cisplatin cotreatment.
MLN4924 treatment resulted in >4N DNA content due to the re-replication of DNA leading to the accumulation of DNA damage, apoptosis, and senescence.
In vivo, MLN4924 significantly inhibited the growth of a TNBC xenograft model by inducing DNA damage, and MLN4924/cisplatin combination further reduced tumor growth by enhancing DNA damage.
The Ray Research Team concluded in their Oncotarget Research Paper that Additionally, the presence of p53 mutation in TNBC cells may promote re-replication upon MLN4924 treatment since activation of p53 by ATM/ATR/Chk2 regulates the re-replication through the induction of p21 supporting that p53 status is prognostic of outcome and MLN4924 treatment response of TNBC. Collectively, our results established the molecular mechanism by which MLN4924 induces TNBC cell death and enhances cisplatin sensitivity, provided the rationale of combining MLN4924 with cisplatin in both BRCA1-wild type and mutant TNBCs, and identified a cancer genetic background where this combination will be more effective.
Sign up for free Altmetric alerts about this article
Full text - https://doi.org/10.18632/oncotarget.27485
Correspondence to - Alo Ray - [email protected]
Keywords - triple-negative breast cancer, MLN4924, cisplatin, cell cycle, DNA damage
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit https://www.impactjournals.com/ or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105


