Bladder cancer detection by urinary extracellular vesicle mRNA analysis
2020-01-16
Biomarker candidates were identified through RNA-seq analysis of urinary EV from patients with non-muscle invasive bladder cancer, advanced urothelial cancer, no residual tumor after TURBT, and healthy and disease controls. These genes allowed detection of non-muscle invasive bladder cancer as well as pT2 and higher muscle invasive bladder cancer.
Dr. Taku Murakami from the Hitachi Chemical Co. America, Ltd., Irvine, CA, USA said, "National Cancer Institute estimated that there will be approximately 74,000 new bladder cancer cases and 14,000 deaths in the United States alone."
About 75% of bladder cancer is non-muscle-invasive cancer and about 25% is muscle-invasive cancer. Since the recurrence and progression rate is 50 to 70% for the non-muscle-invasive cancers, the patients with bladder cancer history require lifelong monitoring of recurrence, which makes bladder cancer the most expensive cancer from diagnosis to treatment in the US.
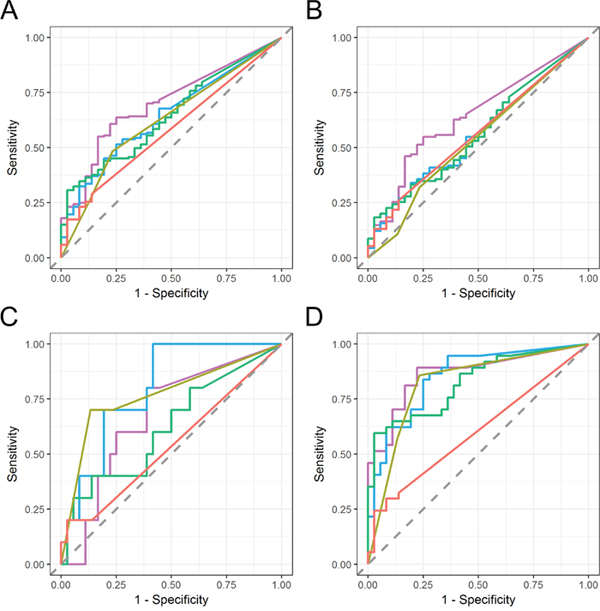
Figure 4: ROC curve analysis of urinary EV mRNA markers in various stages of bladder cancer. Diagnostic performance of urinary EV SLC2A1 (purple), GPRC5A (blue) and KRT17 (green) was evaluated against that of urine cytology (ocher) and BTA ELISA assay (red) for the detection of bladder cancer at various stages. (A) all stage bladder cancer (pTa, pTis, pT1 and > pT2), (B) pTa bladder cancer, (C) pTis bladder cancer, and (D) pT1 bladder cancer. Area under the curve (AUC) of ROC curve are shown in Table 3.
Other urothelial cancers located in ureters and renal pelvises are rare compared to bladder cancer, however 20% to 50% of the patients will have bladder cancer in the future. Since urothelial cancers are located on the urothelium and directly in contact with urine, it is highly possible that EV originating from urothelial cancers are released into urine, suggesting that urinary EV could be a source of urothelial cancer biomarkers.
In this retrospective feasibility study for bladder cancer, the research team's objectives were to conduct a biomarker screening of bladder cancer using urine samples from urothelial cancer patients with various grades and stages and to evaluate diagnostic accuracy of the marker candidates compared to the conventional urine cytology and other assays.
The Taku Murakami research team concluded, "In this study, three urinary EV mRNA were discovered to be elevated in bladder cancer and used to determine diagnostic performance in non-muscle and muscle invasive bladder cancer."
Full text - https://doi.org/10.18632/oncotarget.25998
Correspondence to - Taku Murakami - [email protected]
Keywords - bladder cancer, biomarker, extracellular vesicles, exosome, mRNA
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC



