Anetumab ravtansine inhibits tumor growth and shows additive effect in combination with targeted agents and chemotherapy in mesothelin-expressing human ovarian cancer models
2019-12-30
Herein, the research team investigated the antitumor efficacy of the mesothelin-targeting antibody-drug conjugate anetumab ravtansine as a novel treatment option for ovarian cancer in monotherapy and in combination with the antitumor agents pegylated liposomal doxorubicin, carboplatin, copanlisib and bevacizumab.
The combination of anetumab ravtansine with the PI3K/inhibitor copanlisib was additive in the OVCAR-3 and OVCAR-8 cell lines in vitro, showing increased apoptosis in response to the combination treatment. Taken together, these data support the development of anetumab ravtansine for ovarian cancer treatment and highlight its suitability for combination therapy with PLD, carboplatin, copanlisib, or bevacizumab.
Dr. Christoph A. Schatz from Bayer AG Preclinical Research, Pharmaceuticals, in Berlin 13353, Germany said "With 239,000 new cases per year, ovarian cancer is the seventh most frequent cancer in women in the world."
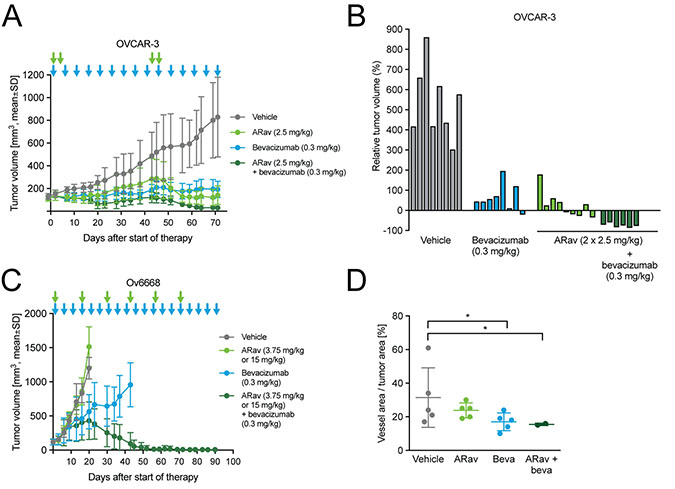
Figure 7: Antitumor efficacy of anetumab ravtansine in combination with bevacizumab in human ovarian cancer xenograft models in mice. Anetumab ravtansine (i.v.) and/or bevacizumab (i.p.) were administered as indicated by arrows. (A) Tumor growth in the OVCAR-3 ovarian cancer model (n = 9). Treatments were initiated 43 days after tumor cell inoculation. (B) Changes in the relative volume of OVCAR-3 tumors in panel A on day 71 after start of therapy, represented as a percentage of the initial tumor volume in each individual mouse. (C) Tumor growth in the Ov6668 ovarian cancer PDX model (n = 9). Treatments were initiated 21 days after tumor inoculation. (D) CD31-positive vessel area as a percentage of tumor area in OVCAR-3 mice described in panel A. ARav, anetumab ravtansine; Beva, bevacizumab.
The vascular endothelial growth factor inhibitor bevacizumab has been approved by the European Medicines Agency and the U.S. Food & Drug Administration for the treatment of advanced ovarian cancer patients in combination with chemotherapy, and the poly polymerase inhibitor olaparib has been approved for BRCA1/BRCA2-mutated high-grade serous ovarian cancer.
Mesothelin is highly expressed on the surface of tumor cells in various cancers, including ovarian cancer, whereas in normal tissue mesothelin shows limited expression. Mesothelin is frequently co-expressed with and binds to CA125, a well-established ovarian cancer biomarker, and may be involved in the peritoneal spread of ovarian cancer. Anetumab ravtansine has demonstrated potent antitumor activity and good tolerability as single agent in preclinical models including mesothelioma, pancreatic cancer and ovarian cancer.
Therefore, targeted agents for the treatment of ovarian cancer either in monotherapy or in combination with chemotherapy should be explored as they may result in increased treatment benefit with more favorable tolerability.
The Christoph A. Schatz research team concluded, "Overall, this work supports the development of anetumab ravtansine as monotherapy or in combination with various targeted agents and chemotherapy in the treatment of ovarian cancer."
Full text - https://doi.org/10.18632/oncotarget.26135
Correspondence to - Christoph A. Schatz - [email protected]
Keywords - mesothelin, anetumab ravtansine, antibody-drug conjugate, copanlisib, ovarian cancer
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




