Tumor secreted ANGPTL2 facilitates recruitment of neutrophils to the lung to promote lung pre-metastatic niche formation and targeting ANGPTL2 signaling affects metastatic disease
2020-02-05
Oncotarget Volume 11, Issue 5: The authors determined that tumor-derived ANGPTL2 stimulates lung epithelial cells, which is essential for primary tumor-induced neutrophil recruitment in lung and subsequent pre-metastatic niche formation.
Lastly, they identified that a p63 isoform, Np63, drives a high level of ANGPTL2 secretion and pharmaceutical inhibition of ANGPTL2 signaling by a non-RGD-based integrin-binding peptide diminished metastatic load in lungs likely due to reduction of the lung pre-metastatic niche formation.
Dr. Hakan Cam from the Center for Childhood Cancer and Blood Diseases at Nationwide Children's Hospital as well as the Department of Pediatrics at The Ohio State University in Columbus Ohio USA said in their Oncotarget paper, "Primary tumors selectively and actively modify potential sites of metastasis, even prior to dissemination"
For instance, neutrophils have been identified as facilitators of breast cancer metastasis and of lung cancer metastasis after UV-induced inflammation through tumor-secreted exosomal RNAs.
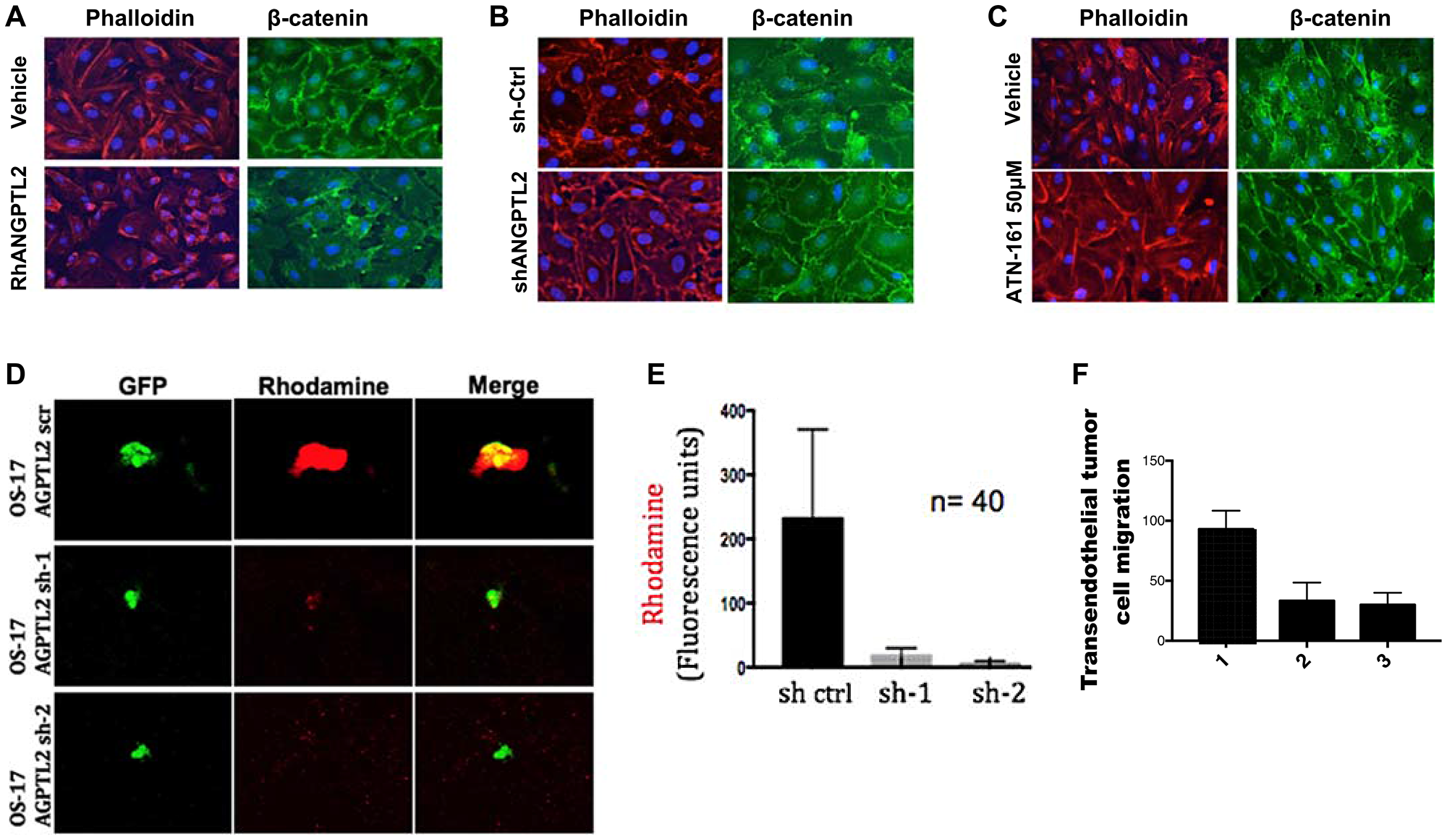
Figure 4: Tumor-secreted ANGPTL2 promotes osteosarcoma metastasis by mediating endothelial monolayer disruption, lung capillary permeability, and trans-endothelial tumor cell migration. (A) HUVEC monolayers were grown to confluence on fibronectin-coated slides and then treated with either vehicle (PBS) or human recombinant ANGPTL2 protein (rhANGPTL2) for 24h. Slides were subsequently fluorescently stained with anti-phalloidin, and anti-β-catenin antibody. (B) HUVEC monolayers were treated for 24 h with media conditioned by control OS17 osteosarcoma cells that express ANGPTL2 (ctrl) or knockdown OS17-shANGPTL2 (sh) cells. Samples were stained for β-catenin and phalloidin. (C) Administration of ANGPTL2 receptor inhibitor, ATN-161, prevents disruption of vascular endothelial cell-cell junctions. HUVEC monolayers were treated for 24 h with media conditioned by OS-17 cells in the presence of drug. Subsequently, samples were stained for β-catenin and phalloidin. ((A–B) Quantitative analysis of paracellular gap formation is shown in Supplementary Figure 7) (D) GFP-labeled indicated OS17 cells were injected via the tail vein and allowed to lodge in the lungs. One day post injection, a rhodamine-dextran dye was injected into circulation. Three hours after dye injection, lungs were extracted and frozen sections were obtained. Representative confocal images are shown here of cells with and without accumulation of dye in the lung parenchyma. (E) Images were obtained as described in (D) with control or ANGPTL2 knockdown OS17 cells. A region of interest was drawn around the GFP-labeled cells and the amount of dextran dye was quantified based on rhodamine emissions. n = 40 cells; error bars indicate s. e. m; p-values calculated using the one-tailed unpaired t-test. (F) OS17-shCtr (1), OS17-sh1ANGPTL2 (2) or OS17-sh2ANGPTL2 (3) were seeded into trans-well inserts that were previously covered with a HUVEC monolayer. Cells that migrated cross the endothelial layer into the bottom side of the transwell membrane were quantified with Volocity software. n = 15, error bars indicate s. e. m; p-values calculated using the one-tailed unpaired t-test.
Here, the scientists set out to determine how tumor-derived factors might affect the activation of lung epithelial cells in ways that elicit pro-metastatic inflammatory responses and facilitate the formation of the pre-metastatic following the recruitment of neutrophils.
It is also well established that the induction of inflammation-related genes results in the activation of neutrophils and as described above a body of evidence suggesting that the recruitment of neutrophils promotes cancer metastasis.
Collectively, by using spontaneous metastatic models, they investigated whether tumor secreted ANGPTL2 induces inflammation on lung epithelial cells by activating alpha5beta1 receptor and recruiting neutrophils to the pre-metastatic niche.
The Cam Research Team concluded in their Oncotarget article that they investigated whether lung epithelial cells might be essential for primary tumor-induced neutrophil recruitment to lung and the role that these infiltrating cells play in osteosarcoma PMN formation using spontaneous metastatic models.
Full text - https://doi.org/10.18632/oncotarget.27433
Correspondence to - Hakan Cam - [email protected]
Keywords - pre-metastatic niche formation, osteosarcoma, ANGPTL2, tumor microenvironment, neutrophils
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




