Repurposed quinacrine synergizes with cisplatin, reducing the effective dose required for treatment of head and neck squamous cell carcinoma
2019-10-05
The authors have developed the Accelera TED platform to repurpose drugs for HNSCC treatment; using in vitro assays and in vivo models.
Screening a library of clinically-approved drugs identified the anti-malarial agent quinacrine as a candidate, which significantly reduced viability in a concentration dependent manner in five HNSCC cell lines and in six primary HNSCC samples.
In vivo, daily treatment with 100 mg/kg oral quinacrine plus cisplatin significantly inhibited tumor outgrowth, extending median time to reach maximum tumor volume from 20 to 32 days versus control, and from 28 to 32 days versus 2 mg/kg cisplatin alone.
Dr. Hisham Mehanna and Dr. Nikolaos Batis said, "Head and neck squamous cell carcinoma is a debilitating disease comprising 600,000 cases per year worldwide"
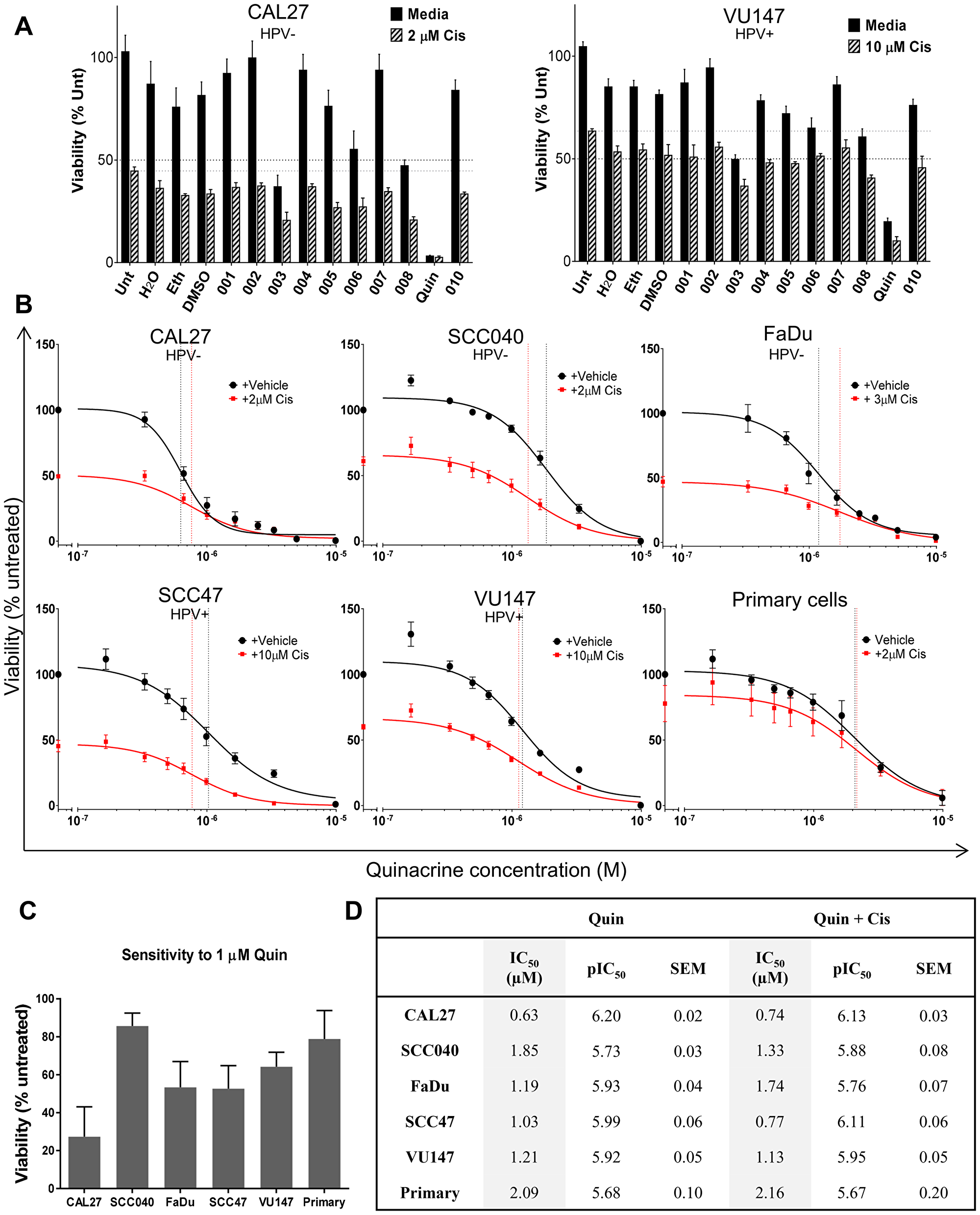
Due to the high morbidity of current treatments, quality of life is severely impaired, with evident unmet need necessitating more effective therapies with lower toxicity; especially as recent studies examining cetuximab as a less toxic alternative for low risk human papillomavirus positive HNSCC have shown similar toxicity, and lower efficacy compared to cisplatin.
On initial screening against HNSCC cell lines, quinacrine was identified as a potential hit.
Quinacrine is a potent late-stage autophagy inhibitor and has been shown to prime cells to the effects of cisplatin via apoptosis in cervical and endometrial cancer.
Further mechanistic insights have been demonstrated in HNSCC whereby quinacrine was able to restore the function of the tumor suppressive protein, tumor protein 53, leading to enhanced capabilities of initiating apoptotic cell death following DNA damage with cisplatin chemotherapy; Moreover, quinacrine treatment has been shown to suppress phosphoinositide 3-kinase, protein kinase B, mechanistic target of rapamycin and nuclear factor kappa-light-chain-enhancer of activated B cells pathways.
The Mehanna/Batis Research team concluded, "Notably, our study is the first to demonstrate that quinacrine is able to reduce tumor burden in mice bearing HNSCC xenograft tumors, using concentrations that are clinically relevant."
Full text - https://www.oncotarget.com/article/27156/text/
Correspondence to - Hisham Mehanna - [email protected] and Nikolaos Batis - [email protected]
Keywords - head and neck cancer, drug repurposing, drug repositioning, quinacrine, mepacrine
Copyright © 2026 Rapamycin Press LLC dba Impact Journals
Oncotarget ® is a registered trademark of Rapamycin Press LLC
Impact Journals ® is a registered trademark of Rapamycin Press LLC
RAPAMYCIN PRESS ® is a registered trademark of Rapamycin Press LLC




