The cover for issue 89 of Oncotarget features Figure 4, "Fluorescence images after the probe is sprayed on the extracted tumor," by Mochida, et al.
While a variety of fluorescent probes have been proposed for this task, small molecule enzyme-activatable fluorescent probes are ideal for this application.
In this study two enzyme activatable probes, γ-glutamylhydroxymethyl rhodamine green that reacted with γ-glutamyltransferase and SPiDER-βGal that reacted with β-galactosidase, were employed to determine the effects of temperature on fluorescence signal kinetics in both fresh and frozen and then thawed ex vivo experimental ovarian cancer tissue samples.
The researchers demonstrate that temperature regulation and tissue handling of resected tissue are two pitfalls that may influence ex vivo imaging signals with enzyme-activatable fluorescent probes.
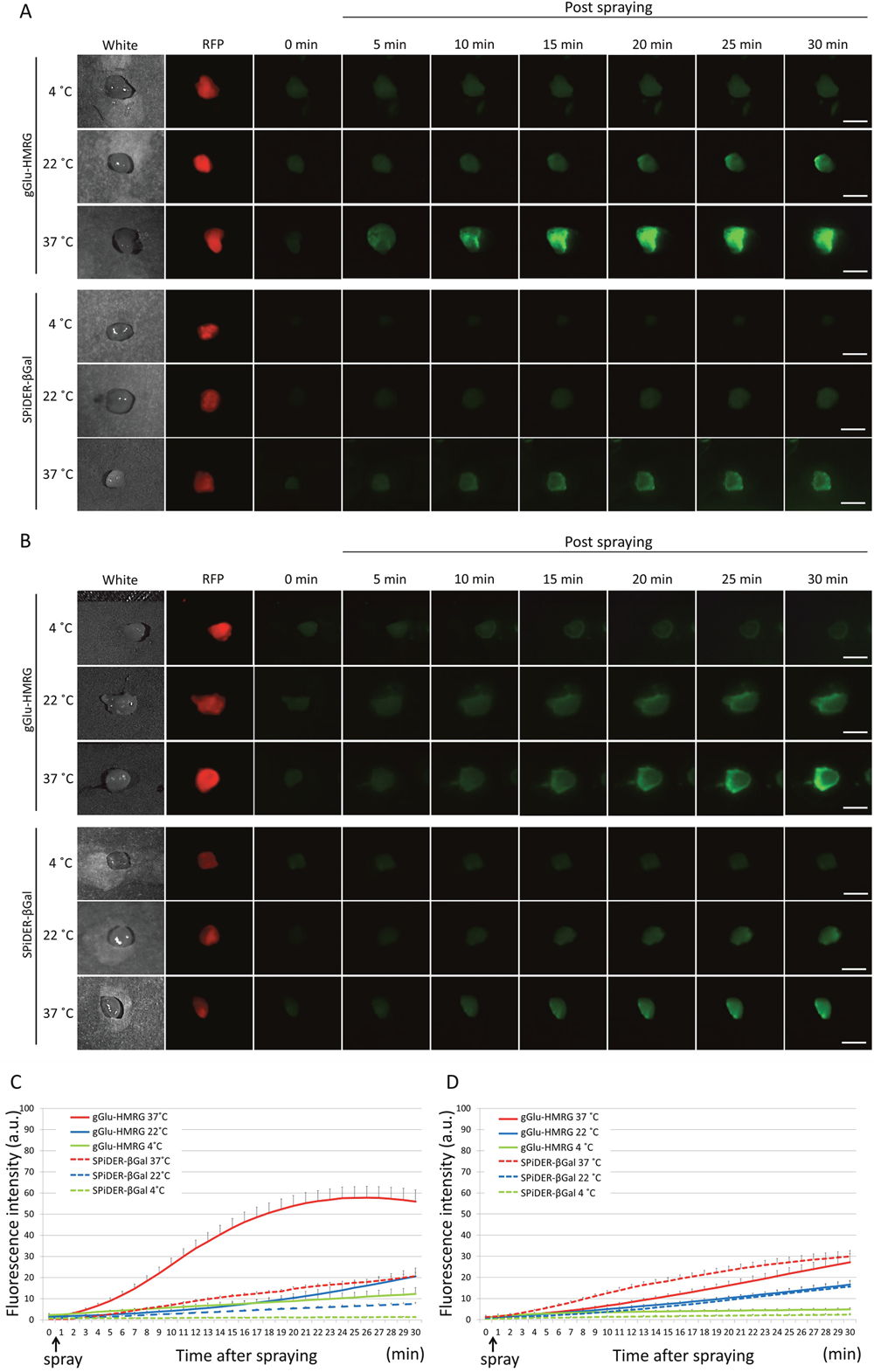
Figure 4: Fluorescence images after the probe is sprayed on the extracted tumor. Sequential fluorescence for 30 min, starting from just after spraying with 10 μM gGlu-HMRG and 50 μM SPiDER-βGal at 4°C, 22°C, and 37°C in fresh specimens (A) and frozen specimens (B). Time fluorescence intensity of the extracted tumors after spraying for fresh tumors (C) and frozen then thawed tumors (D) are shown. Data are mean fluorescence intensities ± SEM of tumors at different time points (n = 9 in each group). Bars are 5 mm.
Hisataka Kobayashi from the Molecular Imaging Program, Center for Cancer Research, at the National Cancer Institute, National Institutes of Health, in Bethesda, Maryland, United States of America said, "Intraoperative optical fluorescence molecular imaging provides real-time image guidance to surgeons to identify precise tumor margins and detect tiny tumor foci both of which result in improved resections with less residual disease, decreasing the risk of recurrence."
"Intraoperative optical fluorescence molecular imaging provides real-time image guidance to surgeons to identify precise tumor margins and detect tiny tumor foci both of which result in improved resections with less residual disease, decreasing the risk of recurrence."
- Hisataka Kobayashi, the Molecular Imaging Program, Center for Cancer Research, at the National Cancer Institute, National Institutes of Health
Conventional imaging probes, called always-on probes, continuously emit signals similar to conventional contrast agents used in computed tomography, magnetic resonance imaging, and angiography and the images depend on the biodistribution of the probe, resulting in relatively high background signals and requiring time to wash out the background signal from the body.
They are turned on only after binding to a specific target found on tumors and, as a result, maximize the target signal while minimizing the background signal, resulting in a high target to background ratio.
In a previous report, the researchers described the fluorescence signal and the kinetics generated by HMRef-βGal and SPiDER-βGal both activated by β-galactosidase and gGlu-HMRG which is activated by GGT in ovarian cancer cell lines at 37 C.
HMRef-βGal, the original probe, behaved with similar kinetics to gGlu-HMRG probe at this temperature although the fluorescence signal of HMRef-βGal was much lower than that of gGlu-HMRG. Therefore, the authors investigated fluorescence signal kinetics of gGlu-HMRG and SPiDER-βGal at different temperatures in vitro and ex vivo imaging using cancer cell lines expressing the target enzyme.
The Hisataka Kobayashi research team concluded in their Oncotarget Research Paper, "although imaging of fresh samples at 37 C is the best condition for both probes, successful imaging with gGlu-HMRG can be achieved even at room temperature with thawed samples."
Sign up for free Altmetric alerts about this article
DOI - https://doi.org/10.18632/oncotarget.26320
Full text - https://www.oncotarget.com/article/26320/text/
Correspondence to - Hisataka Kobayashi - [email protected]
Keywords - fluorescence-guided surgery, activatable probe, enzyme, ex vivo imaging, temperature
About Oncotarget
Oncotarget is a biweekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https://www.oncotarget.com or connect with:
SoundCloud - https://soundcloud.com/oncotarget
Facebook - https://www.facebook.com/Oncotarget/
Twitter - https://twitter.com/oncotarget
LinkedIn - https://www.linkedin.com/company/oncotarget
Pinterest - https://www.pinterest.com/oncotarget/
Reddit - https://www.reddit.com/user/Oncotarget/
Oncotarget is published by Impact Journals, LLC please visit http://www.ImpactJournals.com or connect with @ImpactJrnls
Media Contact
[email protected]
18009220957x105



