The cover for issue 78 of Oncotarget features Figure 4, "Effect of FKBP51 KO on glucocorticoid response and Akt/mTOR in HaCaT human keratinocytes," by Baida, et al.
Here, the researchers used FKBP51 knockout mice to determine FKBP51s role in the major adverse effect of topical glucocorticoids, skin atrophy. Furthermore, despite the absence of inhibitory FKBP51, the basal level of expression and glucocorticoid activation of GR target genes were not increased in FKBP51 KO skin or CRISPR/Cas9-edited FKBP51 KO Ha Ca T human keratinocytes.
Dr. Irina Budunova from the Department of Dermatology, at Northwestern University, in Chicago, IL, USA said, "Glucocorticoid hormones are steroid hormones mostly produced in the adrenal cortex."
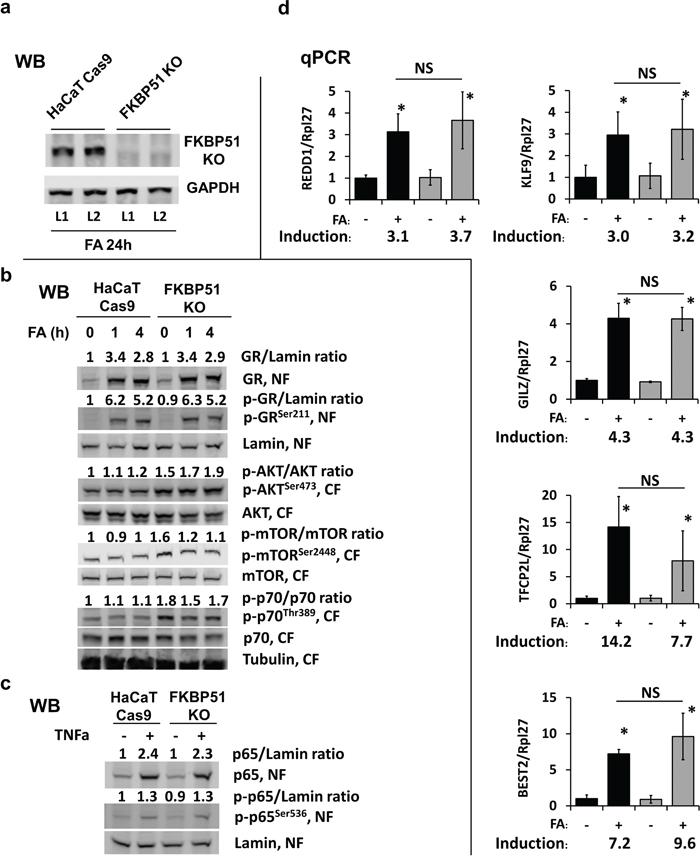
Figure 4: Effect of FKBP51 KO on glucocorticoid response and Akt/mTOR in HaCaT human keratinocytes. FKBP51 KO and Cas9 (control) HaCaT human keratinocytes were treated with glucocorticoid FA (10-6 M) or vehicle as indicated. (a) Verification of FKBP51 knockout by Western blotting (WB), two independent FKBP51 KO clones (L1, L2) tested. GAPDH, loading control. (b, c) Western blot analyses for expression, phosphorylation, and subcellular localization of GR, Akt, mTOR, p70/S6K1, and NF-kB p65 in cells treated with FA (10-6 M x 0 (untreated), 1 h, and 4 h) (b); or with TNF-a (50 μg/ml x 20 min) (c). Nuclear (NF) and cytosol (CF) fractions were isolated as in Materials and Methods. Lamin and tubulin were used as nuclear and cytoplasmic loading controls, respectively. ImageJ was used for densitometry. For phosphorylated and total GR and p65, the intensity of bands was normalized to the corresponding loading controls and expressed as folds of change to vehicle-treated Cas9-HaCaT. For AKT, mTOR, and p70, the ratios of phosphorylation are expressed as folds of change to vehicle-treated Cas9-HaCaT. (d) qPCR analysis of GR target genes in cells treated with FA (10-6 M) for 24 h. The results (normalized to Rpl27) are the means ± SD calculated for three individual RNA samples/condition. Gene induction is fold of change in expression after FA (+) versus basal (-) expression. * P < 0.05 (unpaired two-tailed t-test) for changes compared to corresponding control. NS - nonsignificant difference in FA-induction between Cas9- and FKBP51 KO HaCaT cells.
In unstimulated cells, GR resides in cytoplasm bound to chaperones including heat shock proteins and immunophilins, such as FKBP51, which control GR activation and nuclear import. This research team and others found that FKBP51 is a major GR target gene in skin, as it was strongly up-regulated in keratinocytes of skin-specific keratin5 -GR transgenics and down-regulated in GR knockout mice.
Here, the research group confirmed FKBP51 induction by glucocorticoids in mouse and human skin, and used FKBP51 KO mice to determine the role of FKBP51 in glucocorticoid-induced skin atrophy. As FKBP51 is a major negative GR regulator, the possible outcomes in FKBP51 KO animals could be elevated GR activity and enhanced anti-proliferative effects of endogenous/exogenous glucocorticoids. They also found that Akt and mTOR were constitutively activated in FKBP51 KO skin and in Ha Ca T human keratinocytes lacking FKBP51.
The Irina Budunova research team concluded, "taking into consideration the potential anti-cancer effect of FKBP51 inhibition in skin and similarity between atrophic processes in aged skin and skin chronically treated with glucocorticoids , the same strategy could be relevant to a safe skin protection against aging. Importantly, FKBP51 belongs to the group of the genes whose expression in skin depends on age."
Full text - https://doi.org/10.18632/oncotarget.26194
Correspondence to - Irina Budunova - [email protected]
Keywords - glucocorticoid, glucocorticoid receptor, skin atrophy, FKBP51, Akt


